dermazinc soap
1a3bf938-fd9d-5544-e063-6294a90a9b64
34390-5
HUMAN OTC DRUG LABEL
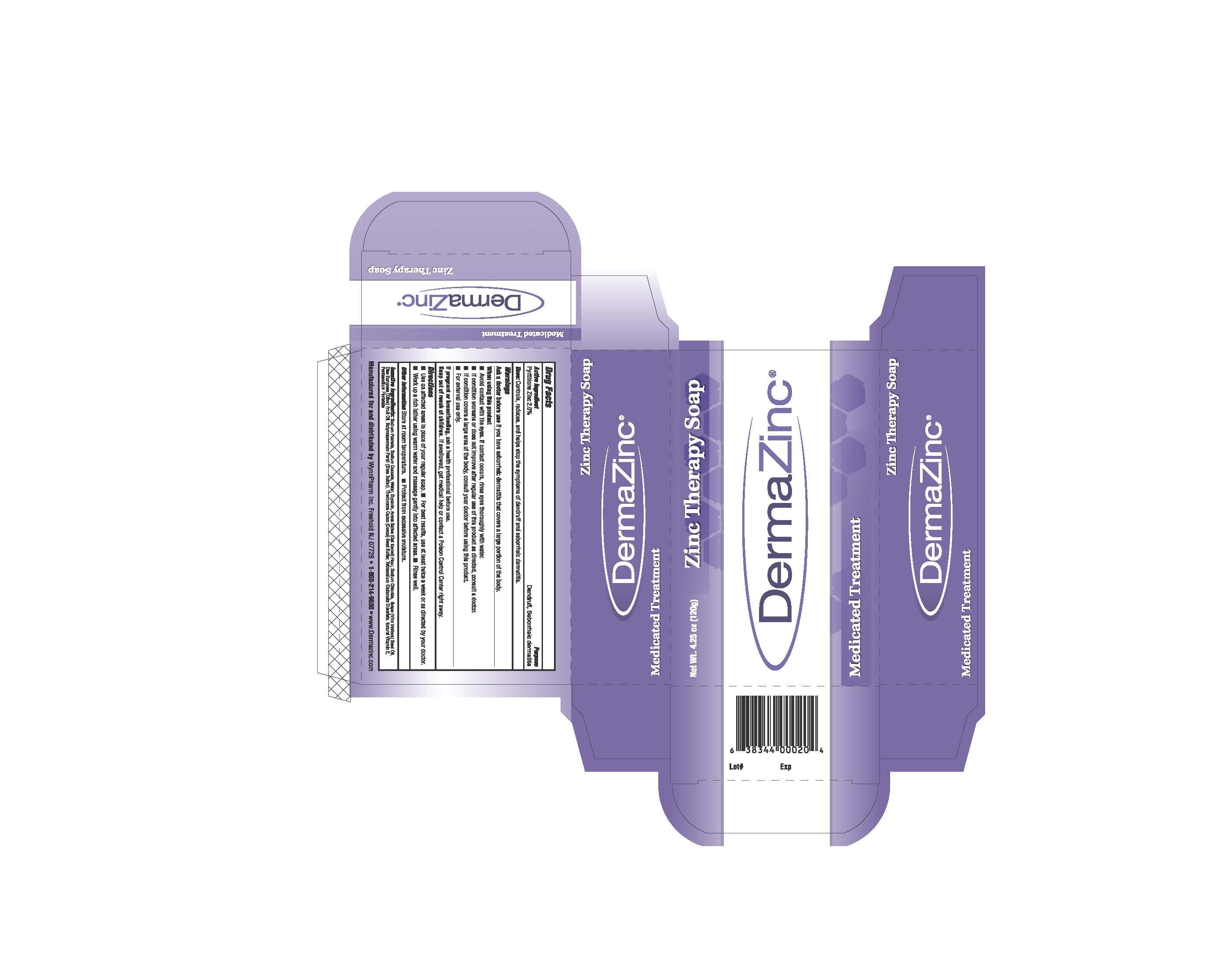
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Pyrithione Zinc 2%
Medication Information
Warnings and Precautions
Warnings
■ Ask a doctor before use if you have a condition that covers a large area of the body.
When using this product avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with
water.
Stop use and ask a doctor if condition worsens or does not improve after regular use of this
product as directed.
If pregnant or breast-feeding, ask a health professional before use. Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
■ For external use only.
Active Ingredient
Pyrithione Zinc 2%
Indications and Usage
Controls, reduces and helps stop the symptoms of dandruff and seborrheic dermatitis.
Dosage and Administration
-Use on affected areas in place of regular soap.
-For best results, use at least twice a week, or as directed by your doctor.
-Work up a rich lather using warm water and massage gently into affected areas.
-Rinse well.
Description
Pyrithione Zinc 2%
Section 50565-1
Keep Out of Reach of Children.
Section 51727-6
Sodium Palmate, Sodium Cocoate, Water, Glycerin, Avena Sativa (Oat Kernel) Flour, Sodium Chloride, Grape Seed Oil, Olea Europaea (Olive) Fruit Oil, Butyrospermum Parkii (Shea Butter), Theobroma Cacao (Cocoa) Seed Butter, Tetrasodium Glutamate Diacetate, Natural Vitamin E, Pentasodium Pentatate.
Section 51945-4
Section 55105-1
Dandruff, Seborrheic Dermatitis treatment
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
■ Ask a doctor before use if you have a condition that covers a large area of the body.
When using this product avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with
water.
Stop use and ask a doctor if condition worsens or does not improve after regular use of this
product as directed.
If pregnant or breast-feeding, ask a health professional before use. Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
■ For external use only.
Active Ingredient (55106-9)
Pyrithione Zinc 2%
Indications and Usage (34067-9)
Controls, reduces and helps stop the symptoms of dandruff and seborrheic dermatitis.
Dosage and Administration (34068-7)
-Use on affected areas in place of regular soap.
-For best results, use at least twice a week, or as directed by your doctor.
-Work up a rich lather using warm water and massage gently into affected areas.
-Rinse well.
Section 50565-1 (50565-1)
Keep Out of Reach of Children.
Section 51727-6 (51727-6)
Sodium Palmate, Sodium Cocoate, Water, Glycerin, Avena Sativa (Oat Kernel) Flour, Sodium Chloride, Grape Seed Oil, Olea Europaea (Olive) Fruit Oil, Butyrospermum Parkii (Shea Butter), Theobroma Cacao (Cocoa) Seed Butter, Tetrasodium Glutamate Diacetate, Natural Vitamin E, Pentasodium Pentatate.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Dandruff, Seborrheic Dermatitis treatment
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:12.476334 · Updated: 2026-03-14T23:10:26.709421