These Highlights Do Not Include All The Information Needed To Use Eribulin Mesylate Injection Safely And Effectively. See Full Prescribing Information For Eribulin Mesylate Injection.
1a1a9cff-d5da-4232-b98f-26f55208dfc4
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
None
Indications and Usage
Eribulin Mesylate Injection is a microtubule inhibitor indicated for the treatment of patients with: Metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting. ( 1.1 ) Unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen. ( 1.2 )
Dosage and Administration
Administer 1.4 mg/m 2 intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle. ( 2.1 ) Reduce dose in patients with hepatic impairment or with moderate or severe renal impairment. ( 2.1 ) Do not mix with other drugs or administer with dextrose-containing solutions. ( 2.3 )
Warnings and Precautions
Neutropenia: Monitor peripheral blood cell counts and adjust dose as appropriate. ( 5.1 ) Peripheral Neuropathy : Monitor for signs of neuropathy. Manage with dose delay and adjustment. ( 5.2 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. ( 5.3 , 8.1 , 8.3 ) QT Prolongation : Monitor for prolonged QT intervals in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, and electrolyte abnormalities. Avoid in patients with congenital long QT syndrome. ( 5.4 )
Contraindications
None.
Adverse Reactions
The most common adverse reactions (≥25%) in metastatic breast cancer were neutropenia, anemia, asthenia/fatigue, alopecia, peripheral neuropathy, nausea, and constipation. ( 6.1 ) The most common adverse reactions (≥25%) in liposarcoma and leiomyosarcoma were fatigue, nausea, alopecia, constipation, peripheral neuropathy, abdominal pain, and pyrexia. The most common (≥5%) Grade 3-4 laboratory abnormalities in liposarcoma and leiomyosarcoma were neutropenia, hypokalemia, and hypocalcemia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Long Grove Pharmaceuticals, LLC at 1-855-642-2594 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Storage and Handling
Eribulin mesylate injection is supplied as a clear colorless solution in a clear glass vial, essentially free of visible foreign particulate matter. NDC 68001-656-41 Injection: 1 mg/2 mL, in a single-dose vial. One vial per carton. Store at 25°C (77°F); excursions permitted to 15° to 30° C (59° to 86° F). Do not freeze or refrigerate. Store the vials in their original cartons. Eribulin mesylate injection is a cytotoxic drug. Follow applicable special handling and disposal procedures.¹
How Supplied
Eribulin mesylate injection is supplied as a clear colorless solution in a clear glass vial, essentially free of visible foreign particulate matter. NDC 68001-656-41 Injection: 1 mg/2 mL, in a single-dose vial. One vial per carton. Store at 25°C (77°F); excursions permitted to 15° to 30° C (59° to 86° F). Do not freeze or refrigerate. Store the vials in their original cartons. Eribulin mesylate injection is a cytotoxic drug. Follow applicable special handling and disposal procedures.¹
Medication Information
Warnings and Precautions
Neutropenia: Monitor peripheral blood cell counts and adjust dose as appropriate. ( 5.1 ) Peripheral Neuropathy : Monitor for signs of neuropathy. Manage with dose delay and adjustment. ( 5.2 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. ( 5.3 , 8.1 , 8.3 ) QT Prolongation : Monitor for prolonged QT intervals in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, and electrolyte abnormalities. Avoid in patients with congenital long QT syndrome. ( 5.4 )
Indications and Usage
Eribulin Mesylate Injection is a microtubule inhibitor indicated for the treatment of patients with: Metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting. ( 1.1 ) Unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen. ( 1.2 )
Dosage and Administration
Administer 1.4 mg/m 2 intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle. ( 2.1 ) Reduce dose in patients with hepatic impairment or with moderate or severe renal impairment. ( 2.1 ) Do not mix with other drugs or administer with dextrose-containing solutions. ( 2.3 )
Contraindications
None.
Adverse Reactions
The most common adverse reactions (≥25%) in metastatic breast cancer were neutropenia, anemia, asthenia/fatigue, alopecia, peripheral neuropathy, nausea, and constipation. ( 6.1 ) The most common adverse reactions (≥25%) in liposarcoma and leiomyosarcoma were fatigue, nausea, alopecia, constipation, peripheral neuropathy, abdominal pain, and pyrexia. The most common (≥5%) Grade 3-4 laboratory abnormalities in liposarcoma and leiomyosarcoma were neutropenia, hypokalemia, and hypocalcemia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Long Grove Pharmaceuticals, LLC at 1-855-642-2594 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Storage and Handling
Eribulin mesylate injection is supplied as a clear colorless solution in a clear glass vial, essentially free of visible foreign particulate matter. NDC 68001-656-41 Injection: 1 mg/2 mL, in a single-dose vial. One vial per carton. Store at 25°C (77°F); excursions permitted to 15° to 30° C (59° to 86° F). Do not freeze or refrigerate. Store the vials in their original cartons. Eribulin mesylate injection is a cytotoxic drug. Follow applicable special handling and disposal procedures.¹
How Supplied
Eribulin mesylate injection is supplied as a clear colorless solution in a clear glass vial, essentially free of visible foreign particulate matter. NDC 68001-656-41 Injection: 1 mg/2 mL, in a single-dose vial. One vial per carton. Store at 25°C (77°F); excursions permitted to 15° to 30° C (59° to 86° F). Do not freeze or refrigerate. Store the vials in their original cartons. Eribulin mesylate injection is a cytotoxic drug. Follow applicable special handling and disposal procedures.¹
Description
None
Section 42229-5
Metastatic Breast Cancer
The most common adverse reactions (≥25%) reported in patients receiving eribulin mesylate injection were neutropenia, anemia, asthenia/fatigue, alopecia, peripheral neuropathy, nausea, and constipation. The most common serious adverse reactions reported in patients receiving eribulin mesylate injection were febrile neutropenia (4%) and neutropenia (2%). The most common adverse reaction resulting in discontinuation of eribulin mesylate injection was peripheral neuropathy (5%).
The adverse reactions described in Table 2were identified in 750 patients treated in Study 1 [see Clinical Studies ( 14.1)] . In Study 1, patients were randomized (2:1) to receive either eribulin mesylate injection (1.4 mg/m 2on Days 1 and 8 of a 21-day cycle) or single agent treatment chosen by their physician (control group). A total of 503 patients received eribulin mesylate injection and 247 patients in the control group received therapy consisting of chemotherapy [total 97% (anthracyclines 10%, capecitabine 18%, gemcitabine 19%, taxanes 15%, vinorelbine 25%, other chemotherapies 10%)] or hormonal therapy (3%). The median duration of exposure was 118 days for patients receiving eribulin mesylate injection and 63 days for patients receiving control therapy. Table 2reports the most common adverse reactions occurring in at least 10% of patients in either group.
| aadverse reactions were graded per National Cancer Institute Criteria for Adverse Events version 4.0. | ||||
| bbased upon laboratory data. | ||||
| cincludes peripheral neuropathy, peripheral sensorimotor neuropathy, peripheral motor neuropathy, polyneuropathy, peripheral sensory neuropathy, and paraesthesia. | ||||
| dnot applicable; (grading system does not specify > Grade 2 for alopecia). | ||||
|
Adverse Reactions |
Eribulin Mesylate Injection
|
Control Group
|
||
|
All Grades |
≥ Grade 3 |
All Grades |
≥ Grade 3 |
|
|
Blood and lymphatic system disorders b |
||||
|
Neutropenia |
82% |
57% |
53% |
23% |
|
Anemia |
58% |
2% |
55% |
4% |
|
Nervous system disorders |
||||
|
Peripheral neuropathy c |
35% |
8% |
16% |
2% |
|
Headache |
19% |
<1% |
12% |
<1% |
|
General disorders |
||||
|
Asthenia/Fatigue |
54% |
10% |
40% |
11% |
|
Pyrexia |
21% |
<1% |
13% |
<1% |
|
Mucosal inflammation |
9% |
1% |
10% |
2% |
|
Gastrointestinal disorders |
||||
|
Nausea |
35% |
1% |
28% |
3% |
|
Constipation |
25% |
1% |
21% |
1% |
|
Vomiting |
18% |
1% |
18% |
1% |
|
Diarrhea |
18% |
0 |
18% |
0 |
|
Musculoskeletal and connective tissue disorders |
||||
|
Arthralgia/Myalgia |
22% |
<1% |
12% |
1% |
|
Back pain |
16% |
1% |
7% |
2% |
|
Bone pain |
12% |
2% |
9% |
2% |
|
Pain in extremity |
11% |
1% |
10% |
1% |
|
Metabolism and nutrition disorders |
||||
|
Decreased weight |
21% |
1% |
14% |
<1% |
|
Anorexia |
20% |
1% |
13% |
1% |
|
Respiratory, thoracic, and mediastinal disorders |
||||
|
Dyspnea |
16% |
4% |
13% |
4% |
|
Cough |
14% |
0 |
9% |
0 |
|
Skin and subcutaneous tissue disorders |
||||
|
Alopecia |
45% |
NA d |
10% |
NA d |
|
Infections |
||||
|
Urinary Tract Infection |
10% |
1% |
5% |
0 |
Section 42230-3
|
PATIENT INFORMATION
|
|
|
What is the most important information I should know about Eribulin Mesylate Injection?
|
|
|
See “ What are possible side effects of eribulin mesylate injection?” for more information about side effects. |
|
|
What is eribulin mesylate injection?
|
|
|
It is not known if eribulin mesylate injection is safe and effective in children under 18 years of age. |
|
|
Before you receive eribulin mesylate injection, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
|
|
How will I receive eribulin mesylate injection?
|
|
|
What are the possible side effects of eribulin mesylate injection?
|
|
|
The most common side effects of eribulin mesylate injection in people with breast cancer include: |
|
|
|
|
The most common side effects of eribulin mesylate injection in people with liposarcoma include: |
|
|
|
|
Your healthcare provider will do blood tests before and during treatment while you are taking eribulin mesylate injection. The most common changes to blood tests in people with liposarcoma include:
|
|
|
Tell your healthcare provider about any side effect that bothers you or that does not go away. |
|
|
These are not all the possible side effects of eribulin mesylate injection. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about eribulin mesylate injection |
|
|
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about eribulin mesylate injection that is written for health professionals. |
|
|
What are the ingredients in eribulin mesylate injection? |
|
|
Active Ingredient: eribulin mesylate |
|
|
Inactive Ingredients:dehydrated alcohol, water for injection, and sodium hydroxide or hydrochloric acid may be used for pH adjustment. |
|
|
Manufactured by: Cenexi - Laboratoires Thissen Sa Rue de la Papyrée, 2-4-6, Braine l’Alleud - 1420, Belgium |
|
|
For BluePoint Laboratories |
|
|
1-855-642-2594 |
|
|
This Patient Information has been approved by the U.S. Food and Drug Administration.
|
Section 43683-2
None
Section 51945-4
Principal Display Panel - 0.5 mg/2 mL Carton Label
NDC 68001-656-41
Rx only
Eribulin Mesylate Injection
1 mg/2 mL (0.5 mg/mL)
For Intravenous Use
STERILE SOLUTION
CAUTION: Cytotoxic Agent
SINGLE -DOSE VIAL - discard unused portion.
DOSAGE AND USE:See accompanying Prescribing Information.
BluePoint Laboratories
Principal Display Panel - 0.5 mg/2 mL Vial Label
NDC 68001-656-41
Rx only
Eribulin Mesylate Injection
1 mg/2 mL (0.5 mg/mL)
For Intravenous Use
STERILE SOLUTION
CAUTION: Cytotoxic Agent
SINGLE -DOSE VIAL - discard unused portion.
DOSAGE AND USE:See accompanying Prescribing Information.
BluePoint Laboratories
10 Overdosage
Overdosage of eribulin mesylate injection has been reported at approximately 4 times the recommended dose, which resulted in Grade 3 neutropenia lasting seven days and a Grade 3 hypersensitivity reaction lasting one day.
There is no known antidote for eribulin mesylate injection overdose.
15 References
- OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description
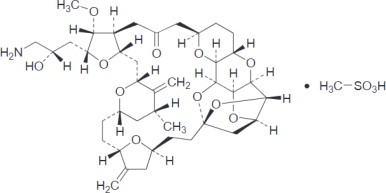
Eribulin mesylate injection contains eribulin mesylate, a microtubule dynamics inhibitor. Eribulin mesylate is a synthetic analogue of halichondrin B, a product isolated from the marine sponge Halichondria okadai. The chemical name for eribulin mesylate is 11,15:18,21:24,28-Triepoxy-7,9-ethano-12,15-methano-9 H,15 H-furo[3,2- i]furo[2',3':5,6]pyrano[4,3- b][1,4]dioxacyclopentacosin-5(4 H)-one, 2-[(2 S)-3-amino-2-hydroxypropyl]hexacosahydro-3-methoxy-26-methyl-20,27-bis(methylene)-,(2 R,3 R,3a S,7 R,8a S,9 S,10a R,11 S,12 R,13a R, 13b S,15 S,18 S,21 S,24 S,26 R,28 R,29a S)-, methanesulfonate (salt). It has a molecular weight of 826.0 (729.9 for free base). The empirical formula is C 40H 59NO 11•CH 4O 3S. Eribulin mesylate has the following structural formula:
Eribulin mesylate injection is a clear, colorless, sterile solution for intravenous administration. Each single-dose vial contains 1 mg of eribulin mesylate in 2 mL of solution. Each mL of solution contains 0.5 mg of eribulin mesylate (equivalent to 0.44 mg eribulin) in dehydrated alcohol (5% v/v) and water for injection (95% v/v). Sodium hydroxide or hydrochloric acid may be used for pH adjustment.
1.2 Liposarcoma
Eribulin mesylate injection is indicated for the treatment of patients with unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen [ see Clinical Studies ( 14.2) ].
5.1 Neutropenia
In Study 1, severe neutropenia (ANC < 500/mm 3) lasting more than one week occurred in 12% (62/503) of patients with metastatic breast cancer, leading to discontinuation in <1% of patients. Febrile neutropenia (fever ≥38.5°C with Grade 3 or 4 neutropenia) occurred in 5% (23/503) of patients; two patients (0.4%) died from complications of febrile neutropenia [ see Adverse Reactions ( 6.1)] .
In Study 1, patients with alanine aminotransferase (ALT) or aspartate aminotransferase (AST) > 3 × ULN (upper limit of normal) experienced a higher incidence of Grade 4 neutropenia and febrile neutropenia than patients with normal aminotransferase levels. Patients with bilirubin > 1.5 × ULN also had a higher incidence of Grade 4 neutropenia and febrile neutropenia.
In Study 2, severe neutropenia (ANC < 500/mm 3) lasting more than one week occurred in 12% (26/222) of patients with liposarcoma or leiomyosarcoma. Febrile neutropenia occurred in 0.9% of patients treated with eribulin mesylate injection and fatal neutropenic sepsis in 0.9% [ see Adverse Reactions ( 6.1) ].
Monitor complete blood counts prior to each dose; increase the frequency of monitoring in patients who develop Grade 3 or 4 cytopenias. Delay administration of eribulin mesylate injection and reduce subsequent doses in patients who experience febrile neutropenia or Grade 4 neutropenia lasting longer than 7 days [ see Dosage and Administration ( 2.2) ]. Clinical studies of eribulin mesylate injection did not include patients with baseline neutrophil counts below 1,500/mm 3.
14.2 Liposarcoma
The efficacy and safety of eribulin mesylate injection were evaluated in Study 2, an open-label, randomized (1:1), multicenter, active-controlled trial. Eligible patients were required to have unresectable, locally advanced or metastatic liposarcoma or leiomyosarcoma, at least two prior systemic chemotherapies (one of which must have included an anthracycline), and disease progression within 6 months of the most recent chemotherapy regimen. Patients were randomized to eribulin mesylate injection 1.4 mg/m 2administered intravenously on Days 1 and 8 of a 21-day cycle or to dacarbazine at a dose of 850 mg/m 2, 1000 mg/m 2, or 1200 mg/m 2administered intravenously every 21 days (dacarbazine dose was selected by the investigator prior to randomization). Treatment continued until disease progression or unacceptable toxicity. Randomization was stratified by histology (liposarcoma or leiomyosarcoma), number of prior therapies (2 vs. > 2), and geographic region (U.S. and Canada vs. Western Europe, Australia, and Israel vs. Eastern Europe, Latin America, and Asia). The major efficacy outcome measure was overall survival (OS). Additional efficacy outcome measures were progression-free survival (PFS) and confirmed objective response rate (ORR) as assessed by the investigator according to Response Evaluation Criteria in Solid Tumors (RECIST v1.1). Patients in the dacarbazine arm were not offered eribulin mesylate injection at the time of disease progression.
A total of 446 patients were randomized, 225 to the eribulin mesylate injection arm and 221 to the dacarbazine arm. The median age was 56 years (range: 24 to 83); 33% were male; 73% were White; 44% had ECOG performance status (PS) 0 and 53% had ECOG PS 1; 68% had leiomyosarcoma and 32% had liposarcoma; 39% were enrolled in U.S. and Canada (Region 1) and 46% were enrolled in Western Europe, Australia, and Israel (Region 2); and 47% received more than two prior systemic chemotherapies. The most common (>40%) prior systemic chemotherapies were doxorubicin (90%), ifosfamide (62%), gemcitabine (59%), trabectedin (50%), and docetaxel (48%).
Of the 143 patients with liposarcoma, the median age was 55 years (range: 32 to 83); 62% were male, 72% were White; 41% had ECOG PS of 0 and 53% had ECOG PS of 1; 35% were enrolled in Region 1 and 51% were enrolled in Region 2; and 44% received more than two prior systemic chemotherapies. The distribution of subtypes of liposarcoma, based on local histologic assessment, were 45% dedifferentiated, 37% myxoid/round cell, and 18% pleomorphic.
Study 2 demonstrated a statistically significant improvement in OS in patients randomized to eribulin mesylate injection compared with dacarbazine (see Table 6). There was no significant difference in progression-free survival in the overall population. Treatment effects of eribulin mesylate injection were limited to patients with liposarcoma based on pre-planned, exploratory subgroup analyses of OS and PFS (see Tables 6and 7and Figure 2). There was no evidence of efficacy of eribulin mesylate injection in patients with advanced or metastatic leiomyosarcoma in Study 2 (see Table 7).
| aEfficacy data from one study site enrolling six patients were excluded. | ||||
| *All patients = liposarcoma and leiomyosarcoma. | ||||
| †N/A = not applicable | ||||
|
Liposarcoma
|
All Patients* |
|||
|
Eribulin Mesylate Injection (n=71) |
Dacarbazine (n=72) |
Eribulin Mesylate Injection (n=225) |
Dacarbazine (n=221) |
|
|
Overall survival |
||||
|
Deaths, n (%) |
52 (73) |
63 (88) |
173 (77) |
179 (81) |
|
Median, months (95% CI) |
15.6
|
8.4
|
13.5
|
11.3
|
|
Hazard ratio (HR)
|
0.51
|
0.75
|
||
|
Stratified log-rank p value |
N/A † |
0.011 |
||
|
Progression-free survival |
||||
|
Events, n (%) |
57 (80) |
59 (82) |
194 (86) |
185 (84) |
|
Disease progression |
53 |
52 |
180 |
170 |
|
Death |
4 |
7 |
14 |
15 |
|
Median, months
|
2.9
|
1.7
|
2.6
|
2.6
|
|
HR
|
0.52
|
0.86
|
||
|
Objective response rate |
||||
|
Objective response rate (%)
|
1.4
|
0
|
4.0
|
5.0
|
Figure 2: Kaplan-Meier Curves of Overall Survival in the Liposarcoma Stratum in Study 2
| aEfficacy data from one study site enrolling six patients were excluded. | ||
|
Leiomyosarcoma
|
||
|
Eribulin Mesylate Injection (n=154) |
Dacarbazine
|
|
|
Overall survival |
||
|
Deaths, n (%) |
121 (79) |
116 (78) |
|
Median, months
|
12.8
|
12.3
|
|
HR (95% CI) |
0.90 (0.69, 1.18) |
|
|
Progression-free survival |
||
|
Events, n (%) |
137 (89) |
126 (85) |
|
Disease progression |
127 |
118 |
|
Death |
10 |
8 |
|
Median, months
|
2.2
|
2.6
|
|
HR (95% CI) |
1.05 (0.81, 1.35) |
|
|
Objective response rate (%)
|
5.2
|
7.4
|
8.4 Pediatric Use
The safety and effectiveness of eribulin mesylate injection in pediatric patients have not been established.
Pediatric use information describing clinical studies in which efficacy was not demonstrated is approved for Eisai Inc's HALAVEN ®(eribulin mesylate) injection. However, due to Eisai Inc's marketing exclusivity rights, this drug product is not labeled with that information.
8.5 Geriatric Use
Study 1 did not include sufficient numbers of subjects with metastatic breast cancer aged 65 years and older to determine whether they respond differently from younger subjects. Of the 827 subjects who received the recommended dose and schedule of eribulin mesylate injection in clinical studies with advanced breast cancer, 15% (121/827) were 65 and older, and 2% (17/827) patients were 75 and older. No overall differences in safety were observed between these subjects and younger subjects.
Clinical studies of eribulin mesylate injection did not include a sufficient number of subjects in Study 2 aged 65 years and older to determine whether they respond differently from younger subjects.
4 Contraindications
None.
5.4 Qt Prolongation
In an uncontrolled open-label ECG study in 26 patients, QT prolongation was observed on Day 8, independent of eribulin concentration, with no QT prolongation observed on Day 1. ECG monitoring is recommended if therapy is initiated in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, including Class Ia and III antiarrhythmics, and electrolyte abnormalities. Correct hypokalemia or hypomagnesemia prior to initiating eribulin mesylate injection and monitor these electrolytes periodically during therapy. Avoid eribulin mesylate injection in patients with congenital long QT syndrome.
6 Adverse Reactions
The most common adverse reactions (≥25%) in metastatic breast cancer were neutropenia, anemia, asthenia/fatigue, alopecia, peripheral neuropathy, nausea, and constipation. ( 6.1)
The most common adverse reactions (≥25%) in liposarcoma and leiomyosarcoma were fatigue, nausea, alopecia, constipation, peripheral neuropathy, abdominal pain, and pyrexia. The most common (≥5%) Grade 3-4 laboratory abnormalities in liposarcoma and leiomyosarcoma were neutropenia, hypokalemia, and hypocalcemia. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Long Grove Pharmaceuticals, LLC at 1-855-642-2594 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
2.1 Recommended Dose
The recommended dose of eribulin mesylate injection is 1.4 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle.
The recommended dose of eribulin mesylate injection in patients with mild hepatic impairment (Child-Pugh A) is 1.1 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle [see Use in Specific Populations ( 8.6)] .
The recommended dose of eribulin mesylate injection in patients with moderate hepatic impairment (Child-Pugh B) is 0.7 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle [see Use in Specific Populations ( 8.6)] .
The recommended dose of eribulin mesylate injection in patients with moderate or severe renal impairment (creatinine clearance (CLcr) 15-49 mL/min) is 1.1 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle [see Use in Specific Populations (8.7) ].
8.7 Renal Impairment
12.3 Pharmacokinetics
The pharmacokinetics (PK) of eribulin is linear with a mean elimination half-life of approximately 40 hours, a mean volume of distribution of 43 L/m 2to 114 L/m 2and mean clearance of 1.16 L/hr/m 2to 2.42 L/hr/m 2over the dose range of 0.25 mg/m 2to 4.0 mg/m 2. The human plasma protein binding of eribulin at concentrations of 100 ng/mL to 1,000 ng/mL ranges from 49% to 65%. Eribulin exposure after multiple dosing is comparable to that following a single dose. No accumulation of eribulin is observed with weekly administration.
2.2 Dose Modification
Assess for peripheral neuropathy and obtain complete blood cell counts prior to each dose.
Recommended dose delays
- Do not administer eribulin mesylate injection on Day 1 or Day 8 for any of the following
- ANC < 1,000/mm 3
- Platelets < 75,000/mm 3
- Grade 3 or 4 non-hematological toxicities.
- The Day 8 dose may be delayed for a maximum of 1 week.
- If toxicities do not resolve or improve to ≤ Grade 2 severity by Day 15, omit the dose.
- If toxicities resolve or improve to ≤ Grade 2 severity by Day 15, administer eribulin mesylate injection at a reduced dose and initiate the next cycle no sooner than 2 weeks later.
Recommended dose reductions
- If a dose has been delayed for toxicity and toxicities have recovered to Grade 2 severity or less, resume eribulin mesylate injection at a reduced dose as set out in Table 1.
Do not re-escalate eribulin mesylate injection dose after it has been reduced.
|
Event Description |
Recommended Eribulin Mesylate Injection Dose |
|
Permanently reduce the 1.4 mg/m 2eribulin mesylate injection dose for any of the following: |
1.1 mg/m 2 |
|
ANC <500/mm 3for >7 days |
|
|
ANC <1,000 /mm 3with fever or infection |
|
|
Platelets <25,000/mm 3 |
|
|
Platelets <50,000/mm 3requiring transfusion |
|
|
Non-hematologic Grade 3 or 4 toxicities |
|
|
Omission or delay of Day 8 eribulin mesylate injection dose in previous cycle for toxicity |
|
|
Occurrenceof any event requiring permanent dose reduction while receiving 1.1 mg/m 2 |
0.7 mg/m 2 |
|
Occurrenceof any event requiring permanent dose reduction while receiving 0.7 mg/m 2 |
Discontinue Eribulin Mesylate Injection |
|
ANC = absolute neutrophil count.
|
8.6 Hepatic Impairment
Administration of eribulin mesylate injection at a dose of 1.1 mg/m 2to patients with mild hepatic impairment and 0.7 mg/m 2to patients with moderate hepatic impairment resulted in similar exposure to eribulin as a dose of 1.4 mg/m 2to patients with normal hepatic function. Therefore, a lower starting dose of 1.1 mg/m 2is recommended for patients with mild hepatic impairment (Child-Pugh A) and of 0.7 mg/m 2is recommended for patients with moderate hepatic impairment (Child-Pugh B). Eribulin mesylate injection was not studied in patients with severe hepatic impairment (Child-Pugh C) [see Dosage and Administration ( 2.1), Clinical Pharmacology ( 12.3)].
1 Indications and Usage
Eribulin Mesylate Injection is a microtubule inhibitor indicated for the treatment of patients with:
- Metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting. ( 1.1)
- Unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen. ( 1.2)
12.1 Mechanism of Action
Eribulin inhibits the growth phase of microtubules without affecting the shortening phase and sequesters tubulin into nonproductive aggregates. Eribulin exerts its effects via a tubulin-based antimitotic mechanism leading to G 2/M cell-cycle block, disruption of mitotic spindles, and, ultimately, apoptotic cell death after prolonged mitotic blockage.
In addition, eribulin treatment of human breast cancer cells caused changes in morphology and gene expression as well as decreased migration and invasiveness in vitro. In mouse xenograft models of human breast cancer, eribulin treatment was associated with increased vascular perfusion and permeability in the tumor cores, resulting in reduced tumor hypoxia, and changes in the expression of genes in tumor specimens associated with a change in phenotype.
5.2 Peripheral Neuropathy
In Study 1, Grade 3 peripheral neuropathy occurred in 8% (40/503) of patients, and Grade 4 in 0.4% (2/503) of patients with metastatic breast cancer (MBC). Peripheral neuropathy was the most common toxicity leading to discontinuation of eribulin mesylate injection (5% of patients; 24/503) in Study 1. Neuropathy lasting more than one year occurred in 5% (26/503) of patients. Twenty-two percent (109/503) of patients developed a new or worsening neuropathy that had not recovered within a median follow-up duration of 269 days (range 25-662 days).
In Study 2, Grade 3 peripheral neuropathy occurred in 3.1% (7/223) of eribulin mesylate injection-treated patients. Peripheral neuropathy led to discontinuation of eribulin mesylate injection in 0.9% of patients. The median time to first occurrence of peripheral neuropathy of any severity was 5 months (range: 3.5 months to 9 months). Neuropathy lasting more than 60 days occurred in 58% (38/65) of patients. Sixty three percent (41/65) had not recovered within a median follow-up duration of 6.4 months (range: 27 days to 29 months).
Monitor patients closely for signs of peripheral motor and sensory neuropathy. Withhold eribulin mesylate injection in patients who experience Grade 3 or 4 peripheral neuropathy, until resolution to Grade 2 or less [see Dosage and Administration ( 2.2)] .
5.3 Embryo Fetal Toxicity
Based on findings from an animal reproduction study and its mechanism of action, eribulin mesylate injection can cause fetal harm when administered to a pregnant woman. There are no adequate and well-controlled studies of eribulin mesylate injection in pregnant women. In animal reproduction studies, eribulin mesylate caused embryo-fetal toxicity when administered to pregnant rats during organogenesis at doses below the recommended human dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with eribulin mesylate injection and for at least 2 weeks following the final dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with eribulin mesylate injection and for 3.5 months following the final dose [ see Use in Specific Populations ( 8.1) ].
5 Warnings and Precautions
- Neutropenia:Monitor peripheral blood cell counts and adjust dose as appropriate. ( 5.1)
- Peripheral Neuropathy: Monitor for signs of neuropathy. Manage with dose delay and adjustment. ( 5.2)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. ( 5.3, 8.1, 8.3)
- QT Prolongation: Monitor for prolonged QT intervals in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, and electrolyte abnormalities. Avoid in patients with congenital long QT syndrome. ( 5.4)
2 Dosage and Administration
1.1 Metastatic Breast Cancer
Eribulin mesylate injection is indicated for the treatment of patients with metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting [see Clinical Studies ( 14.1)] .
3 Dosage Forms and Strengths
Injection: 1 mg/2 mL (0.5 mg/mL) eribulin mesylate is a clear, colorless, sterile solution in a single-dose vial.
6.2 Postmarketing Experience
The following adverse drug reactions have been identified during post-approval use of eribulin mesylate injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Blood and Lymphatic System Disorders:lymphopenia
- Gastrointestinal Disorders:pancreatitis
- Hepatobiliary Disorders:hepatotoxicity
- Immune System Disorders:drug hypersensitivity
- Infections and Infestations:pneumonia, sepsis/neutropenic sepsis
- Metabolism and Nutrition Disorders:hypomagnesemia, dehydration
- Respiratory, thoracic and mediastinal disorders:interstitial lung disease
- Skin and Subcutaneous Tissue Disorders:pruritus, Stevens-Johnson syndrome, toxic epidermal necrolysis
14.1 Metastatic Breast Cancer
Study 1 was an open-label, randomized, multicenter trial of 762 patients with metastatic breast cancer who received at least two chemotherapeutic regimens for the treatment of metastatic disease and experienced disease progression within 6 months of their last chemotherapeutic regimen. Patients were required to receive prior anthracycline- and taxane-based chemotherapy for adjuvant or metastatic disease. Patients were randomized (2:1) to receive eribulin mesylate injection (n=508) or a single agent therapy selected prior to randomization (control arm, n=254). Randomization was stratified by geographic region, HER2/ neustatus, and prior capecitabine exposure. Eribulin mesylate injection was administered at a dose of 1.4 mg/m 2on Days 1 and 8 of a 21-day cycle. Eribulin mesylate injection-treated patients received a median of 5 cycles (range: 1 to 23 cycles) of therapy. Control arm therapy consisted of 97% chemotherapy (26% vinorelbine, 18% gemcitabine, 18% capecitabine, 16% taxane, 9% anthracycline, 10% other chemotherapy), and 3% hormonal therapy. The main efficacy outcome was overall survival.
Patient demographic and baseline characteristics were comparable between the treatment arms. The median age was 55 (range: 27 to 85 years) and 92% were White. Sixty-four percent of patients were enrolled in North America/Western Europe/Australia, 25% in Eastern Europe/Russia, and 11% in Latin America/South Africa. Ninety-one percent of patients had a baseline ECOG performance status of 0 or 1. Tumor prognostic characteristics, including estrogen receptor status (positive: 67%, negative: 28%), progesterone receptor status (positive: 49%, negative: 39%), HER2/ neureceptor status (positive: 16%, negative: 74%), triple negative status (ER-, PR-, HER2/ neu-: 19%), presence of visceral disease (82%, including 60% liver and 38% lung) and bone disease (61%), and number of sites of metastases (greater than two: 50%), were also similar in the eribulin mesylate injection and control arms. Patients received a median of four prior chemotherapy regimens in both arms.
In Study 1, a statistically significant improvement in overall survival was observed in patients randomized to the eribulin mesylate injection arm compared to the control arm (see Table 5). An updated, unplanned survival analysis, conducted when 77% of events had been observed (see Figure 1), was consistent with the primary analysis. In patients randomized to eribulin mesylate injection, the objective response rate by the RECIST criteria was 11% (95% CI: 8.6%, 14.3%) and the median response duration was 4.2 months (95% CI: 3.8, 5.0 months).
| CI = confidence interval | ||
| aBased on Cox proportional hazards model stratified by geographic region, HER2 status, and prior capecitabine therapy. | ||
| bBased on a log-rank test stratified by geographic region, HER2 status, and prior capecitabine therapy. | ||
|
Overall Survival |
Eribulin Mesylate Injection
|
Control Arm (n=254) |
|
Primary survival analysis |
||
|
Number of deaths |
274 |
148 |
|
Median, months (95% CI) |
13.1 (11.8, 14.3) |
10.6 (9.3, 12.5) |
|
Hazard Ratio (95% CI) a |
0.81 (0.66, 0.99) |
|
|
Pvalue b |
0.041 |
|
|
Updated survival analysis |
||
|
Number of deaths |
386 |
203 |
|
Median, months (95% CI) |
13.2 (12.1, 14.4) |
10.6 (9.2, 12.0) |
Figure 1: Updated Overall Survival Analysis for Study 1
8 Use in Specific Populations
- Lactation: Do not breastfeed. ( 8.2)
- Hepatic Impairment: A lower starting dose is recommended for patients with mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment. Patients with severe hepatic impairment (Child-Pugh C) were not studied. ( 8.6)
- Renal Impairment: A lower starting dose is recommended for patients with moderate (CLcr 30-49 mL/min) or severe (CLcr 15-29 mL/min) renal impairment. ( 8.7)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in clinical practice.
The following adverse reactions are discussed in detail in other sections of the labeling:
- Neutropenia [see Warnings and Precautions ( 5.1)]
- Peripheral neuropathy [see Warnings and Precautions ( 5.2)]
- QT prolongation [see Warnings and Precautions ( 5.4)]
In clinical trials, eribulin mesylate injection has been administered to 1963 patients including 467 patients exposed to eribulin mesylate injection for 6 months or longer. The majority of the 1963 patients were women (92%) with a median age of 55 years (range: 17 to 85 years). The racial and ethnic distribution was White (72%), Black (4%), Asian (9%), and other (3%).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Eribulin mesylate injection is supplied as a clear colorless solution in a clear glass vial, essentially free of visible foreign particulate matter.
NDC 68001-656-41
Injection: 1 mg/2 mL, in a single-dose vial. One vial per carton.
Store at 25°C (77°F); excursions permitted to 15° to 30° C (59° to 86° F). Do not freeze or refrigerate. Store the vials in their original cartons.
Eribulin mesylate injection is a cytotoxic drug. Follow applicable special handling and disposal procedures.¹
2.3 Instructions for Preparation and Administration
Aseptically withdraw the required amount of eribulin mesylate injection from the single-dose vial and administer undiluted or diluted in 100 mL of 0.9% Sodium Chloride Injection, USP.
Do not dilute in or administer through an intravenous line containing solutions with dextrose.Do not administer in the same intravenous line concurrent with the other medicinal products.
Store undiluted eribulin mesylate injection in the syringe for up to 4 hours at room temperature or for up to 24 hours under refrigeration at 4°C (40°F). Store diluted solutions of eribulin mesylate injection for up to 4 hours at room temperature or up to 24 hours under refrigeration at 4°C (40°F).
Discard unused portions of the vial.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with eribulin mesylate. Eribulin mesylate was not mutagenic in in vitrobacterial reverse mutation assays (Ames test). Eribulin mesylate was positive in mouse lymphoma mutagenesis assays, and was clastogenic in an in vivorat bone marrow micronucleus assay.
Fertility studies have not been conducted with eribulin mesylate in humans or animals; however, nonclinical findings in repeat-dose dog and rat toxicology studies suggest that male fertility may be compromised by treatment with eribulin mesylate. Rats exhibited testicular toxicity (hypocellularity of seminiferous epithelium with hypospermia/aspermia) following dosing with eribulin mesylate at or above 0.43 times the recommended human dose (based on body surface area) given once weekly for 3 weeks, or at or above 0.21 times the recommended human dose (based on body surface area) given once weekly for 3 out of 5 weeks, repeated for 6 cycles. Testicular toxicity was also observed in dogs given 0.64 times the recommended human dose (based on body surface area) weekly for 3 out of 5 weeks, repeated for 6 cycles.
7.1 Effects of Other Drugs On Eribulin Mesylate Injection
No drug-drug interactions are expected with CYP3A4 inhibitors, CYP3A4 inducers or P-glycoprotein (P-gp) inhibitors. Clinically meaningful differences in exposure (AUC) were not observed in patients with advanced solid tumors when eribulin mesylate injection was administered with or without ketoconazole (a strong inhibitor of CYP3A4 and a P-gp inhibitor) and when eribulin mesylate injection was administered with or without rifampin (a CYP3A4 inducer) [see Clinical Pharmacology ( 12.3)].
7.2 Effects of Eribulin Mesylate Injection On Other Drugs
Eribulin does not inhibit CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP2E1 or CYP3A4 enzymes or induce CYP1A2, CYP2C9, CYP2C19 or CYP3A4 enzymes at relevant clinical concentrations. Eribulin is not expected to alter the plasma concentrations of drugs that are substrates of these enzymes [see Clinical Pharmacology ( 12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Metastatic Breast Cancer
The most common adverse reactions (≥25%) reported in patients receiving eribulin mesylate injection were neutropenia, anemia, asthenia/fatigue, alopecia, peripheral neuropathy, nausea, and constipation. The most common serious adverse reactions reported in patients receiving eribulin mesylate injection were febrile neutropenia (4%) and neutropenia (2%). The most common adverse reaction resulting in discontinuation of eribulin mesylate injection was peripheral neuropathy (5%).
The adverse reactions described in Table 2were identified in 750 patients treated in Study 1 [see Clinical Studies ( 14.1)] . In Study 1, patients were randomized (2:1) to receive either eribulin mesylate injection (1.4 mg/m 2on Days 1 and 8 of a 21-day cycle) or single agent treatment chosen by their physician (control group). A total of 503 patients received eribulin mesylate injection and 247 patients in the control group received therapy consisting of chemotherapy [total 97% (anthracyclines 10%, capecitabine 18%, gemcitabine 19%, taxanes 15%, vinorelbine 25%, other chemotherapies 10%)] or hormonal therapy (3%). The median duration of exposure was 118 days for patients receiving eribulin mesylate injection and 63 days for patients receiving control therapy. Table 2reports the most common adverse reactions occurring in at least 10% of patients in either group.
| aadverse reactions were graded per National Cancer Institute Criteria for Adverse Events version 4.0. | ||||
| bbased upon laboratory data. | ||||
| cincludes peripheral neuropathy, peripheral sensorimotor neuropathy, peripheral motor neuropathy, polyneuropathy, peripheral sensory neuropathy, and paraesthesia. | ||||
| dnot applicable; (grading system does not specify > Grade 2 for alopecia). | ||||
|
Adverse Reactions |
Eribulin Mesylate Injection
|
Control Group
|
||
|
All Grades |
≥ Grade 3 |
All Grades |
≥ Grade 3 |
|
|
Blood and lymphatic system disorders b |
||||
|
Neutropenia |
82% |
57% |
53% |
23% |
|
Anemia |
58% |
2% |
55% |
4% |
|
Nervous system disorders |
||||
|
Peripheral neuropathy c |
35% |
8% |
16% |
2% |
|
Headache |
19% |
<1% |
12% |
<1% |
|
General disorders |
||||
|
Asthenia/Fatigue |
54% |
10% |
40% |
11% |
|
Pyrexia |
21% |
<1% |
13% |
<1% |
|
Mucosal inflammation |
9% |
1% |
10% |
2% |
|
Gastrointestinal disorders |
||||
|
Nausea |
35% |
1% |
28% |
3% |
|
Constipation |
25% |
1% |
21% |
1% |
|
Vomiting |
18% |
1% |
18% |
1% |
|
Diarrhea |
18% |
0 |
18% |
0 |
|
Musculoskeletal and connective tissue disorders |
||||
|
Arthralgia/Myalgia |
22% |
<1% |
12% |
1% |
|
Back pain |
16% |
1% |
7% |
2% |
|
Bone pain |
12% |
2% |
9% |
2% |
|
Pain in extremity |
11% |
1% |
10% |
1% |
|
Metabolism and nutrition disorders |
||||
|
Decreased weight |
21% |
1% |
14% |
<1% |
|
Anorexia |
20% |
1% |
13% |
1% |
|
Respiratory, thoracic, and mediastinal disorders |
||||
|
Dyspnea |
16% |
4% |
13% |
4% |
|
Cough |
14% |
0 |
9% |
0 |
|
Skin and subcutaneous tissue disorders |
||||
|
Alopecia |
45% |
NA d |
10% |
NA d |
|
Infections |
||||
|
Urinary Tract Infection |
10% |
1% |
5% |
0 |
Section 42230-3 (42230-3)
|
PATIENT INFORMATION
|
|
|
What is the most important information I should know about Eribulin Mesylate Injection?
|
|
|
See “ What are possible side effects of eribulin mesylate injection?” for more information about side effects. |
|
|
What is eribulin mesylate injection?
|
|
|
It is not known if eribulin mesylate injection is safe and effective in children under 18 years of age. |
|
|
Before you receive eribulin mesylate injection, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
|
|
How will I receive eribulin mesylate injection?
|
|
|
What are the possible side effects of eribulin mesylate injection?
|
|
|
The most common side effects of eribulin mesylate injection in people with breast cancer include: |
|
|
|
|
The most common side effects of eribulin mesylate injection in people with liposarcoma include: |
|
|
|
|
Your healthcare provider will do blood tests before and during treatment while you are taking eribulin mesylate injection. The most common changes to blood tests in people with liposarcoma include:
|
|
|
Tell your healthcare provider about any side effect that bothers you or that does not go away. |
|
|
These are not all the possible side effects of eribulin mesylate injection. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about eribulin mesylate injection |
|
|
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about eribulin mesylate injection that is written for health professionals. |
|
|
What are the ingredients in eribulin mesylate injection? |
|
|
Active Ingredient: eribulin mesylate |
|
|
Inactive Ingredients:dehydrated alcohol, water for injection, and sodium hydroxide or hydrochloric acid may be used for pH adjustment. |
|
|
Manufactured by: Cenexi - Laboratoires Thissen Sa Rue de la Papyrée, 2-4-6, Braine l’Alleud - 1420, Belgium |
|
|
For BluePoint Laboratories |
|
|
1-855-642-2594 |
|
|
This Patient Information has been approved by the U.S. Food and Drug Administration.
|
Section 43683-2 (43683-2)
None
Section 51945-4 (51945-4)
Principal Display Panel - 0.5 mg/2 mL Carton Label
NDC 68001-656-41
Rx only
Eribulin Mesylate Injection
1 mg/2 mL (0.5 mg/mL)
For Intravenous Use
STERILE SOLUTION
CAUTION: Cytotoxic Agent
SINGLE -DOSE VIAL - discard unused portion.
DOSAGE AND USE:See accompanying Prescribing Information.
BluePoint Laboratories
Principal Display Panel - 0.5 mg/2 mL Vial Label
NDC 68001-656-41
Rx only
Eribulin Mesylate Injection
1 mg/2 mL (0.5 mg/mL)
For Intravenous Use
STERILE SOLUTION
CAUTION: Cytotoxic Agent
SINGLE -DOSE VIAL - discard unused portion.
DOSAGE AND USE:See accompanying Prescribing Information.
BluePoint Laboratories
10 Overdosage (10 OVERDOSAGE)
Overdosage of eribulin mesylate injection has been reported at approximately 4 times the recommended dose, which resulted in Grade 3 neutropenia lasting seven days and a Grade 3 hypersensitivity reaction lasting one day.
There is no known antidote for eribulin mesylate injection overdose.
15 References (15 REFERENCES)
- OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
Eribulin mesylate injection contains eribulin mesylate, a microtubule dynamics inhibitor. Eribulin mesylate is a synthetic analogue of halichondrin B, a product isolated from the marine sponge Halichondria okadai. The chemical name for eribulin mesylate is 11,15:18,21:24,28-Triepoxy-7,9-ethano-12,15-methano-9 H,15 H-furo[3,2- i]furo[2',3':5,6]pyrano[4,3- b][1,4]dioxacyclopentacosin-5(4 H)-one, 2-[(2 S)-3-amino-2-hydroxypropyl]hexacosahydro-3-methoxy-26-methyl-20,27-bis(methylene)-,(2 R,3 R,3a S,7 R,8a S,9 S,10a R,11 S,12 R,13a R, 13b S,15 S,18 S,21 S,24 S,26 R,28 R,29a S)-, methanesulfonate (salt). It has a molecular weight of 826.0 (729.9 for free base). The empirical formula is C 40H 59NO 11•CH 4O 3S. Eribulin mesylate has the following structural formula:
Eribulin mesylate injection is a clear, colorless, sterile solution for intravenous administration. Each single-dose vial contains 1 mg of eribulin mesylate in 2 mL of solution. Each mL of solution contains 0.5 mg of eribulin mesylate (equivalent to 0.44 mg eribulin) in dehydrated alcohol (5% v/v) and water for injection (95% v/v). Sodium hydroxide or hydrochloric acid may be used for pH adjustment.
1.2 Liposarcoma
Eribulin mesylate injection is indicated for the treatment of patients with unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen [ see Clinical Studies ( 14.2) ].
5.1 Neutropenia
In Study 1, severe neutropenia (ANC < 500/mm 3) lasting more than one week occurred in 12% (62/503) of patients with metastatic breast cancer, leading to discontinuation in <1% of patients. Febrile neutropenia (fever ≥38.5°C with Grade 3 or 4 neutropenia) occurred in 5% (23/503) of patients; two patients (0.4%) died from complications of febrile neutropenia [ see Adverse Reactions ( 6.1)] .
In Study 1, patients with alanine aminotransferase (ALT) or aspartate aminotransferase (AST) > 3 × ULN (upper limit of normal) experienced a higher incidence of Grade 4 neutropenia and febrile neutropenia than patients with normal aminotransferase levels. Patients with bilirubin > 1.5 × ULN also had a higher incidence of Grade 4 neutropenia and febrile neutropenia.
In Study 2, severe neutropenia (ANC < 500/mm 3) lasting more than one week occurred in 12% (26/222) of patients with liposarcoma or leiomyosarcoma. Febrile neutropenia occurred in 0.9% of patients treated with eribulin mesylate injection and fatal neutropenic sepsis in 0.9% [ see Adverse Reactions ( 6.1) ].
Monitor complete blood counts prior to each dose; increase the frequency of monitoring in patients who develop Grade 3 or 4 cytopenias. Delay administration of eribulin mesylate injection and reduce subsequent doses in patients who experience febrile neutropenia or Grade 4 neutropenia lasting longer than 7 days [ see Dosage and Administration ( 2.2) ]. Clinical studies of eribulin mesylate injection did not include patients with baseline neutrophil counts below 1,500/mm 3.
14.2 Liposarcoma
The efficacy and safety of eribulin mesylate injection were evaluated in Study 2, an open-label, randomized (1:1), multicenter, active-controlled trial. Eligible patients were required to have unresectable, locally advanced or metastatic liposarcoma or leiomyosarcoma, at least two prior systemic chemotherapies (one of which must have included an anthracycline), and disease progression within 6 months of the most recent chemotherapy regimen. Patients were randomized to eribulin mesylate injection 1.4 mg/m 2administered intravenously on Days 1 and 8 of a 21-day cycle or to dacarbazine at a dose of 850 mg/m 2, 1000 mg/m 2, or 1200 mg/m 2administered intravenously every 21 days (dacarbazine dose was selected by the investigator prior to randomization). Treatment continued until disease progression or unacceptable toxicity. Randomization was stratified by histology (liposarcoma or leiomyosarcoma), number of prior therapies (2 vs. > 2), and geographic region (U.S. and Canada vs. Western Europe, Australia, and Israel vs. Eastern Europe, Latin America, and Asia). The major efficacy outcome measure was overall survival (OS). Additional efficacy outcome measures were progression-free survival (PFS) and confirmed objective response rate (ORR) as assessed by the investigator according to Response Evaluation Criteria in Solid Tumors (RECIST v1.1). Patients in the dacarbazine arm were not offered eribulin mesylate injection at the time of disease progression.
A total of 446 patients were randomized, 225 to the eribulin mesylate injection arm and 221 to the dacarbazine arm. The median age was 56 years (range: 24 to 83); 33% were male; 73% were White; 44% had ECOG performance status (PS) 0 and 53% had ECOG PS 1; 68% had leiomyosarcoma and 32% had liposarcoma; 39% were enrolled in U.S. and Canada (Region 1) and 46% were enrolled in Western Europe, Australia, and Israel (Region 2); and 47% received more than two prior systemic chemotherapies. The most common (>40%) prior systemic chemotherapies were doxorubicin (90%), ifosfamide (62%), gemcitabine (59%), trabectedin (50%), and docetaxel (48%).
Of the 143 patients with liposarcoma, the median age was 55 years (range: 32 to 83); 62% were male, 72% were White; 41% had ECOG PS of 0 and 53% had ECOG PS of 1; 35% were enrolled in Region 1 and 51% were enrolled in Region 2; and 44% received more than two prior systemic chemotherapies. The distribution of subtypes of liposarcoma, based on local histologic assessment, were 45% dedifferentiated, 37% myxoid/round cell, and 18% pleomorphic.
Study 2 demonstrated a statistically significant improvement in OS in patients randomized to eribulin mesylate injection compared with dacarbazine (see Table 6). There was no significant difference in progression-free survival in the overall population. Treatment effects of eribulin mesylate injection were limited to patients with liposarcoma based on pre-planned, exploratory subgroup analyses of OS and PFS (see Tables 6and 7and Figure 2). There was no evidence of efficacy of eribulin mesylate injection in patients with advanced or metastatic leiomyosarcoma in Study 2 (see Table 7).
| aEfficacy data from one study site enrolling six patients were excluded. | ||||
| *All patients = liposarcoma and leiomyosarcoma. | ||||
| †N/A = not applicable | ||||
|
Liposarcoma
|
All Patients* |
|||
|
Eribulin Mesylate Injection (n=71) |
Dacarbazine (n=72) |
Eribulin Mesylate Injection (n=225) |
Dacarbazine (n=221) |
|
|
Overall survival |
||||
|
Deaths, n (%) |
52 (73) |
63 (88) |
173 (77) |
179 (81) |
|
Median, months (95% CI) |
15.6
|
8.4
|
13.5
|
11.3
|
|
Hazard ratio (HR)
|
0.51
|
0.75
|
||
|
Stratified log-rank p value |
N/A † |
0.011 |
||
|
Progression-free survival |
||||
|
Events, n (%) |
57 (80) |
59 (82) |
194 (86) |
185 (84) |
|
Disease progression |
53 |
52 |
180 |
170 |
|
Death |
4 |
7 |
14 |
15 |
|
Median, months
|
2.9
|
1.7
|
2.6
|
2.6
|
|
HR
|
0.52
|
0.86
|
||
|
Objective response rate |
||||
|
Objective response rate (%)
|
1.4
|
0
|
4.0
|
5.0
|
Figure 2: Kaplan-Meier Curves of Overall Survival in the Liposarcoma Stratum in Study 2
| aEfficacy data from one study site enrolling six patients were excluded. | ||
|
Leiomyosarcoma
|
||
|
Eribulin Mesylate Injection (n=154) |
Dacarbazine
|
|
|
Overall survival |
||
|
Deaths, n (%) |
121 (79) |
116 (78) |
|
Median, months
|
12.8
|
12.3
|
|
HR (95% CI) |
0.90 (0.69, 1.18) |
|
|
Progression-free survival |
||
|
Events, n (%) |
137 (89) |
126 (85) |
|
Disease progression |
127 |
118 |
|
Death |
10 |
8 |
|
Median, months
|
2.2
|
2.6
|
|
HR (95% CI) |
1.05 (0.81, 1.35) |
|
|
Objective response rate (%)
|
5.2
|
7.4
|
8.4 Pediatric Use
The safety and effectiveness of eribulin mesylate injection in pediatric patients have not been established.
Pediatric use information describing clinical studies in which efficacy was not demonstrated is approved for Eisai Inc's HALAVEN ®(eribulin mesylate) injection. However, due to Eisai Inc's marketing exclusivity rights, this drug product is not labeled with that information.
8.5 Geriatric Use
Study 1 did not include sufficient numbers of subjects with metastatic breast cancer aged 65 years and older to determine whether they respond differently from younger subjects. Of the 827 subjects who received the recommended dose and schedule of eribulin mesylate injection in clinical studies with advanced breast cancer, 15% (121/827) were 65 and older, and 2% (17/827) patients were 75 and older. No overall differences in safety were observed between these subjects and younger subjects.
Clinical studies of eribulin mesylate injection did not include a sufficient number of subjects in Study 2 aged 65 years and older to determine whether they respond differently from younger subjects.
4 Contraindications (4 CONTRAINDICATIONS)
None.
5.4 Qt Prolongation (5.4 QT Prolongation)
In an uncontrolled open-label ECG study in 26 patients, QT prolongation was observed on Day 8, independent of eribulin concentration, with no QT prolongation observed on Day 1. ECG monitoring is recommended if therapy is initiated in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, including Class Ia and III antiarrhythmics, and electrolyte abnormalities. Correct hypokalemia or hypomagnesemia prior to initiating eribulin mesylate injection and monitor these electrolytes periodically during therapy. Avoid eribulin mesylate injection in patients with congenital long QT syndrome.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (≥25%) in metastatic breast cancer were neutropenia, anemia, asthenia/fatigue, alopecia, peripheral neuropathy, nausea, and constipation. ( 6.1)
The most common adverse reactions (≥25%) in liposarcoma and leiomyosarcoma were fatigue, nausea, alopecia, constipation, peripheral neuropathy, abdominal pain, and pyrexia. The most common (≥5%) Grade 3-4 laboratory abnormalities in liposarcoma and leiomyosarcoma were neutropenia, hypokalemia, and hypocalcemia. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Long Grove Pharmaceuticals, LLC at 1-855-642-2594 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
2.1 Recommended Dose
The recommended dose of eribulin mesylate injection is 1.4 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle.
The recommended dose of eribulin mesylate injection in patients with mild hepatic impairment (Child-Pugh A) is 1.1 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle [see Use in Specific Populations ( 8.6)] .
The recommended dose of eribulin mesylate injection in patients with moderate hepatic impairment (Child-Pugh B) is 0.7 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle [see Use in Specific Populations ( 8.6)] .
The recommended dose of eribulin mesylate injection in patients with moderate or severe renal impairment (creatinine clearance (CLcr) 15-49 mL/min) is 1.1 mg/m 2administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle [see Use in Specific Populations (8.7) ].
8.7 Renal Impairment
12.3 Pharmacokinetics
The pharmacokinetics (PK) of eribulin is linear with a mean elimination half-life of approximately 40 hours, a mean volume of distribution of 43 L/m 2to 114 L/m 2and mean clearance of 1.16 L/hr/m 2to 2.42 L/hr/m 2over the dose range of 0.25 mg/m 2to 4.0 mg/m 2. The human plasma protein binding of eribulin at concentrations of 100 ng/mL to 1,000 ng/mL ranges from 49% to 65%. Eribulin exposure after multiple dosing is comparable to that following a single dose. No accumulation of eribulin is observed with weekly administration.
2.2 Dose Modification
Assess for peripheral neuropathy and obtain complete blood cell counts prior to each dose.
Recommended dose delays
- Do not administer eribulin mesylate injection on Day 1 or Day 8 for any of the following
- ANC < 1,000/mm 3
- Platelets < 75,000/mm 3
- Grade 3 or 4 non-hematological toxicities.
- The Day 8 dose may be delayed for a maximum of 1 week.
- If toxicities do not resolve or improve to ≤ Grade 2 severity by Day 15, omit the dose.
- If toxicities resolve or improve to ≤ Grade 2 severity by Day 15, administer eribulin mesylate injection at a reduced dose and initiate the next cycle no sooner than 2 weeks later.
Recommended dose reductions
- If a dose has been delayed for toxicity and toxicities have recovered to Grade 2 severity or less, resume eribulin mesylate injection at a reduced dose as set out in Table 1.
Do not re-escalate eribulin mesylate injection dose after it has been reduced.
|
Event Description |
Recommended Eribulin Mesylate Injection Dose |
|
Permanently reduce the 1.4 mg/m 2eribulin mesylate injection dose for any of the following: |
1.1 mg/m 2 |
|
ANC <500/mm 3for >7 days |
|
|
ANC <1,000 /mm 3with fever or infection |
|
|
Platelets <25,000/mm 3 |
|
|
Platelets <50,000/mm 3requiring transfusion |
|
|
Non-hematologic Grade 3 or 4 toxicities |
|
|
Omission or delay of Day 8 eribulin mesylate injection dose in previous cycle for toxicity |
|
|
Occurrenceof any event requiring permanent dose reduction while receiving 1.1 mg/m 2 |
0.7 mg/m 2 |
|
Occurrenceof any event requiring permanent dose reduction while receiving 0.7 mg/m 2 |
Discontinue Eribulin Mesylate Injection |
|
ANC = absolute neutrophil count.
|
8.6 Hepatic Impairment
Administration of eribulin mesylate injection at a dose of 1.1 mg/m 2to patients with mild hepatic impairment and 0.7 mg/m 2to patients with moderate hepatic impairment resulted in similar exposure to eribulin as a dose of 1.4 mg/m 2to patients with normal hepatic function. Therefore, a lower starting dose of 1.1 mg/m 2is recommended for patients with mild hepatic impairment (Child-Pugh A) and of 0.7 mg/m 2is recommended for patients with moderate hepatic impairment (Child-Pugh B). Eribulin mesylate injection was not studied in patients with severe hepatic impairment (Child-Pugh C) [see Dosage and Administration ( 2.1), Clinical Pharmacology ( 12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Eribulin Mesylate Injection is a microtubule inhibitor indicated for the treatment of patients with:
- Metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting. ( 1.1)
- Unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen. ( 1.2)
12.1 Mechanism of Action
Eribulin inhibits the growth phase of microtubules without affecting the shortening phase and sequesters tubulin into nonproductive aggregates. Eribulin exerts its effects via a tubulin-based antimitotic mechanism leading to G 2/M cell-cycle block, disruption of mitotic spindles, and, ultimately, apoptotic cell death after prolonged mitotic blockage.
In addition, eribulin treatment of human breast cancer cells caused changes in morphology and gene expression as well as decreased migration and invasiveness in vitro. In mouse xenograft models of human breast cancer, eribulin treatment was associated with increased vascular perfusion and permeability in the tumor cores, resulting in reduced tumor hypoxia, and changes in the expression of genes in tumor specimens associated with a change in phenotype.
5.2 Peripheral Neuropathy
In Study 1, Grade 3 peripheral neuropathy occurred in 8% (40/503) of patients, and Grade 4 in 0.4% (2/503) of patients with metastatic breast cancer (MBC). Peripheral neuropathy was the most common toxicity leading to discontinuation of eribulin mesylate injection (5% of patients; 24/503) in Study 1. Neuropathy lasting more than one year occurred in 5% (26/503) of patients. Twenty-two percent (109/503) of patients developed a new or worsening neuropathy that had not recovered within a median follow-up duration of 269 days (range 25-662 days).
In Study 2, Grade 3 peripheral neuropathy occurred in 3.1% (7/223) of eribulin mesylate injection-treated patients. Peripheral neuropathy led to discontinuation of eribulin mesylate injection in 0.9% of patients. The median time to first occurrence of peripheral neuropathy of any severity was 5 months (range: 3.5 months to 9 months). Neuropathy lasting more than 60 days occurred in 58% (38/65) of patients. Sixty three percent (41/65) had not recovered within a median follow-up duration of 6.4 months (range: 27 days to 29 months).
Monitor patients closely for signs of peripheral motor and sensory neuropathy. Withhold eribulin mesylate injection in patients who experience Grade 3 or 4 peripheral neuropathy, until resolution to Grade 2 or less [see Dosage and Administration ( 2.2)] .
5.3 Embryo Fetal Toxicity (5.3 Embryo-Fetal Toxicity)
Based on findings from an animal reproduction study and its mechanism of action, eribulin mesylate injection can cause fetal harm when administered to a pregnant woman. There are no adequate and well-controlled studies of eribulin mesylate injection in pregnant women. In animal reproduction studies, eribulin mesylate caused embryo-fetal toxicity when administered to pregnant rats during organogenesis at doses below the recommended human dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with eribulin mesylate injection and for at least 2 weeks following the final dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with eribulin mesylate injection and for 3.5 months following the final dose [ see Use in Specific Populations ( 8.1) ].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Neutropenia:Monitor peripheral blood cell counts and adjust dose as appropriate. ( 5.1)
- Peripheral Neuropathy: Monitor for signs of neuropathy. Manage with dose delay and adjustment. ( 5.2)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. ( 5.3, 8.1, 8.3)
- QT Prolongation: Monitor for prolonged QT intervals in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, and electrolyte abnormalities. Avoid in patients with congenital long QT syndrome. ( 5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
1.1 Metastatic Breast Cancer
Eribulin mesylate injection is indicated for the treatment of patients with metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting [see Clinical Studies ( 14.1)] .
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 1 mg/2 mL (0.5 mg/mL) eribulin mesylate is a clear, colorless, sterile solution in a single-dose vial.
6.2 Postmarketing Experience
The following adverse drug reactions have been identified during post-approval use of eribulin mesylate injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Blood and Lymphatic System Disorders:lymphopenia
- Gastrointestinal Disorders:pancreatitis
- Hepatobiliary Disorders:hepatotoxicity
- Immune System Disorders:drug hypersensitivity
- Infections and Infestations:pneumonia, sepsis/neutropenic sepsis
- Metabolism and Nutrition Disorders:hypomagnesemia, dehydration
- Respiratory, thoracic and mediastinal disorders:interstitial lung disease
- Skin and Subcutaneous Tissue Disorders:pruritus, Stevens-Johnson syndrome, toxic epidermal necrolysis
14.1 Metastatic Breast Cancer
Study 1 was an open-label, randomized, multicenter trial of 762 patients with metastatic breast cancer who received at least two chemotherapeutic regimens for the treatment of metastatic disease and experienced disease progression within 6 months of their last chemotherapeutic regimen. Patients were required to receive prior anthracycline- and taxane-based chemotherapy for adjuvant or metastatic disease. Patients were randomized (2:1) to receive eribulin mesylate injection (n=508) or a single agent therapy selected prior to randomization (control arm, n=254). Randomization was stratified by geographic region, HER2/ neustatus, and prior capecitabine exposure. Eribulin mesylate injection was administered at a dose of 1.4 mg/m 2on Days 1 and 8 of a 21-day cycle. Eribulin mesylate injection-treated patients received a median of 5 cycles (range: 1 to 23 cycles) of therapy. Control arm therapy consisted of 97% chemotherapy (26% vinorelbine, 18% gemcitabine, 18% capecitabine, 16% taxane, 9% anthracycline, 10% other chemotherapy), and 3% hormonal therapy. The main efficacy outcome was overall survival.
Patient demographic and baseline characteristics were comparable between the treatment arms. The median age was 55 (range: 27 to 85 years) and 92% were White. Sixty-four percent of patients were enrolled in North America/Western Europe/Australia, 25% in Eastern Europe/Russia, and 11% in Latin America/South Africa. Ninety-one percent of patients had a baseline ECOG performance status of 0 or 1. Tumor prognostic characteristics, including estrogen receptor status (positive: 67%, negative: 28%), progesterone receptor status (positive: 49%, negative: 39%), HER2/ neureceptor status (positive: 16%, negative: 74%), triple negative status (ER-, PR-, HER2/ neu-: 19%), presence of visceral disease (82%, including 60% liver and 38% lung) and bone disease (61%), and number of sites of metastases (greater than two: 50%), were also similar in the eribulin mesylate injection and control arms. Patients received a median of four prior chemotherapy regimens in both arms.
In Study 1, a statistically significant improvement in overall survival was observed in patients randomized to the eribulin mesylate injection arm compared to the control arm (see Table 5). An updated, unplanned survival analysis, conducted when 77% of events had been observed (see Figure 1), was consistent with the primary analysis. In patients randomized to eribulin mesylate injection, the objective response rate by the RECIST criteria was 11% (95% CI: 8.6%, 14.3%) and the median response duration was 4.2 months (95% CI: 3.8, 5.0 months).
| CI = confidence interval | ||
| aBased on Cox proportional hazards model stratified by geographic region, HER2 status, and prior capecitabine therapy. | ||
| bBased on a log-rank test stratified by geographic region, HER2 status, and prior capecitabine therapy. | ||
|
Overall Survival |
Eribulin Mesylate Injection
|
Control Arm (n=254) |
|
Primary survival analysis |
||
|
Number of deaths |
274 |
148 |
|
Median, months (95% CI) |
13.1 (11.8, 14.3) |
10.6 (9.3, 12.5) |
|
Hazard Ratio (95% CI) a |
0.81 (0.66, 0.99) |
|
|
Pvalue b |
0.041 |
|
|
Updated survival analysis |
||
|
Number of deaths |
386 |
203 |
|
Median, months (95% CI) |
13.2 (12.1, 14.4) |
10.6 (9.2, 12.0) |
Figure 1: Updated Overall Survival Analysis for Study 1
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Do not breastfeed. ( 8.2)
- Hepatic Impairment: A lower starting dose is recommended for patients with mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment. Patients with severe hepatic impairment (Child-Pugh C) were not studied. ( 8.6)
- Renal Impairment: A lower starting dose is recommended for patients with moderate (CLcr 30-49 mL/min) or severe (CLcr 15-29 mL/min) renal impairment. ( 8.7)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in clinical practice.
The following adverse reactions are discussed in detail in other sections of the labeling:
- Neutropenia [see Warnings and Precautions ( 5.1)]
- Peripheral neuropathy [see Warnings and Precautions ( 5.2)]
- QT prolongation [see Warnings and Precautions ( 5.4)]
In clinical trials, eribulin mesylate injection has been administered to 1963 patients including 467 patients exposed to eribulin mesylate injection for 6 months or longer. The majority of the 1963 patients were women (92%) with a median age of 55 years (range: 17 to 85 years). The racial and ethnic distribution was White (72%), Black (4%), Asian (9%), and other (3%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Eribulin mesylate injection is supplied as a clear colorless solution in a clear glass vial, essentially free of visible foreign particulate matter.
NDC 68001-656-41
Injection: 1 mg/2 mL, in a single-dose vial. One vial per carton.
Store at 25°C (77°F); excursions permitted to 15° to 30° C (59° to 86° F). Do not freeze or refrigerate. Store the vials in their original cartons.
Eribulin mesylate injection is a cytotoxic drug. Follow applicable special handling and disposal procedures.¹
2.3 Instructions for Preparation and Administration
Aseptically withdraw the required amount of eribulin mesylate injection from the single-dose vial and administer undiluted or diluted in 100 mL of 0.9% Sodium Chloride Injection, USP.
Do not dilute in or administer through an intravenous line containing solutions with dextrose.Do not administer in the same intravenous line concurrent with the other medicinal products.
Store undiluted eribulin mesylate injection in the syringe for up to 4 hours at room temperature or for up to 24 hours under refrigeration at 4°C (40°F). Store diluted solutions of eribulin mesylate injection for up to 4 hours at room temperature or up to 24 hours under refrigeration at 4°C (40°F).
Discard unused portions of the vial.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with eribulin mesylate. Eribulin mesylate was not mutagenic in in vitrobacterial reverse mutation assays (Ames test). Eribulin mesylate was positive in mouse lymphoma mutagenesis assays, and was clastogenic in an in vivorat bone marrow micronucleus assay.
Fertility studies have not been conducted with eribulin mesylate in humans or animals; however, nonclinical findings in repeat-dose dog and rat toxicology studies suggest that male fertility may be compromised by treatment with eribulin mesylate. Rats exhibited testicular toxicity (hypocellularity of seminiferous epithelium with hypospermia/aspermia) following dosing with eribulin mesylate at or above 0.43 times the recommended human dose (based on body surface area) given once weekly for 3 weeks, or at or above 0.21 times the recommended human dose (based on body surface area) given once weekly for 3 out of 5 weeks, repeated for 6 cycles. Testicular toxicity was also observed in dogs given 0.64 times the recommended human dose (based on body surface area) weekly for 3 out of 5 weeks, repeated for 6 cycles.
7.1 Effects of Other Drugs On Eribulin Mesylate Injection (7.1 Effects of Other Drugs on Eribulin Mesylate Injection)
No drug-drug interactions are expected with CYP3A4 inhibitors, CYP3A4 inducers or P-glycoprotein (P-gp) inhibitors. Clinically meaningful differences in exposure (AUC) were not observed in patients with advanced solid tumors when eribulin mesylate injection was administered with or without ketoconazole (a strong inhibitor of CYP3A4 and a P-gp inhibitor) and when eribulin mesylate injection was administered with or without rifampin (a CYP3A4 inducer) [see Clinical Pharmacology ( 12.3)].
7.2 Effects of Eribulin Mesylate Injection On Other Drugs (7.2 Effects of Eribulin Mesylate Injection on Other Drugs)
Eribulin does not inhibit CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP2E1 or CYP3A4 enzymes or induce CYP1A2, CYP2C9, CYP2C19 or CYP3A4 enzymes at relevant clinical concentrations. Eribulin is not expected to alter the plasma concentrations of drugs that are substrates of these enzymes [see Clinical Pharmacology ( 12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:32.037486 · Updated: 2026-03-14T22:33:31.162945