Lidocaine 4% Pain Relief Patch
19c69983-4e78-8ec5-e063-6394a90a84be
34390-5
HUMAN OTC DRUG LABEL
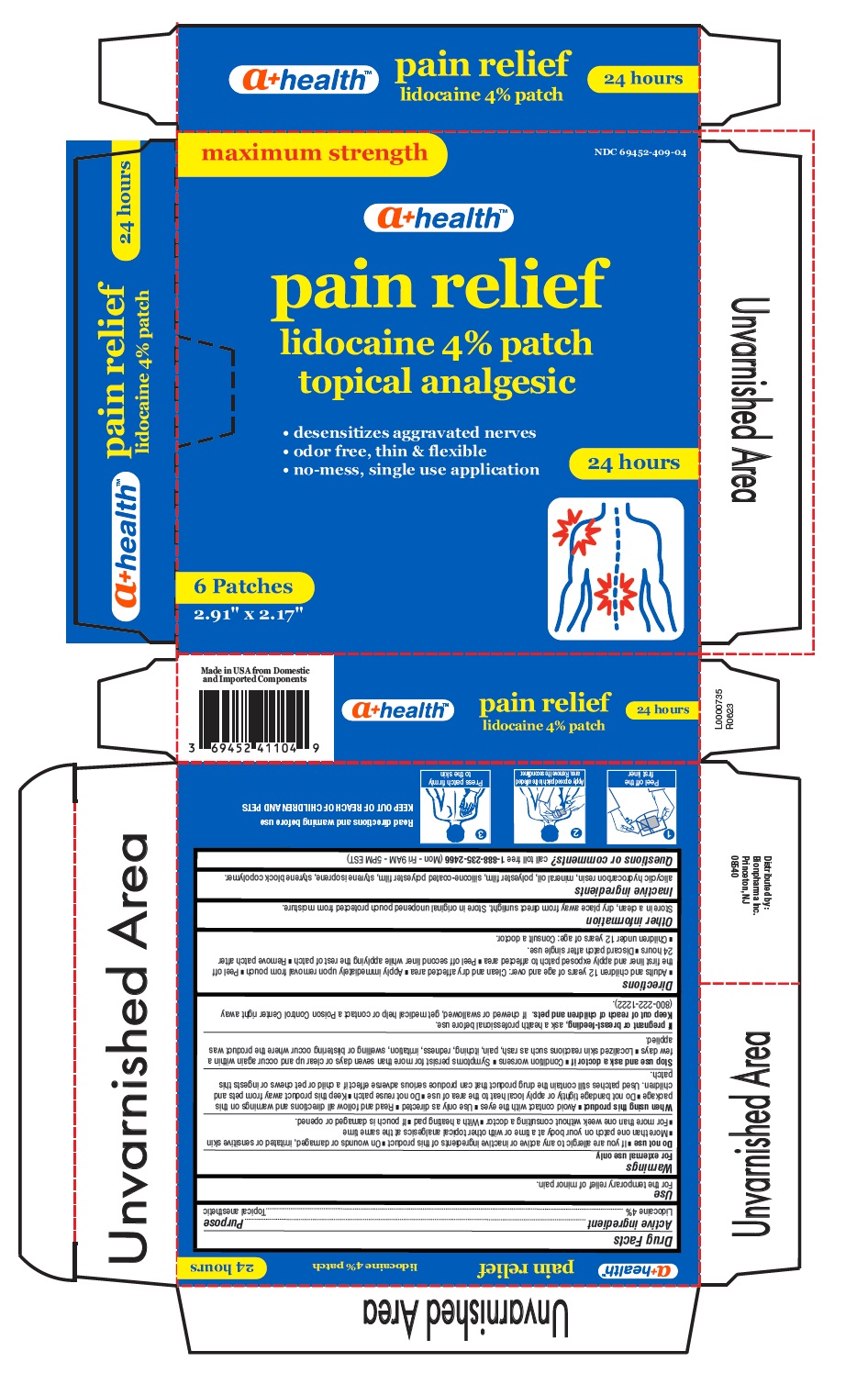
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%
Purpose
Topical anesthetic
Medication Information
Purpose
Topical anesthetic
Description
Lidocaine 4%
Uses
For the temporary relief of minor pain.
Section 42229-5
Read directions and warning before use
KEEP OUT OF REACH OF CHILDREN AND PETS
Made in USA from Domestic and Imported Components
Distributed by:
Bionpharma Inc.
Princeton, NJ
08540
L0000735
R0623
Warnings
For external use only.
Directions
■ Adults and children 12 years of age and over: Clean and dry affected area ■ Apply immediately upon removal from pouch ■ Peel off
the first liner and apply exposed patch to affected area ■ Peel off second liner while applying the rest of patch ■ Remove patch after
24 hours ■ Discard patch after single use.
■ Children under 12 years of age: Consult a doctor.
Do Not Use
■ If you are allergic to any active or inactive ingredients of this product ■ On wounds or damaged, irritated or sensitive skin
■ More than one patch on your body at a time or with other topical analgesics at the same time
■ For more than one week without consulting a doctor ■ With a heating pad ■ If pouch is damaged or opened.
Carton Label
NDC 69452-409-04
maximum strength
a+ health TM
pain relief
lidocaine 4% patch
topical analgesic
• desensitizes aggravated nerves
• odor free, thin & flexible
• no-mess, single use application
24 hours
6 Patches
2.91" x 2.17"
Active Ingredient
Lidocaine 4%
Other Information
Store in a clean, dry place away from direct sunlight. Store in original unopened pouch protected from moisture.
Inactive Ingredients
alicyclic hydrocarbon resin, mineral oil, polyester film, silicone-coated polyester film, styrene isoprene, styrene block copolymer.
Questions Or Comments?
call toll free 1-888-235-2466 (Mon - Fri 9AM - 5PM EST)
When Using This Product
■ Avoid contact with the eyes ■ Use only as directed ■ Read and follow all directions and warnings on this
package ■ Do not bandage tightly or apply local heat to the area of use ■ Do not reuse patch ■ Keep this product away from pets and
children. Used patches still contain the drug product that can produce serious adverse effect if a child or pet chews or ingests this
patch
Stop Use and Ask A Doctor If
■ Condition worsens ■ Symptoms persist for more than seven days or clear up and occur again within a
few days ■ Localized skin reactions such as rash, pain, itching, redness, irritation, swelling or blistering occur where the product was
applied.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children and Pets
If chewed or swallowed, get medical help or contact a Poison Control Center right away (800-222-1222).
Structured Label Content
Uses
For the temporary relief of minor pain.
Section 42229-5 (42229-5)
Read directions and warning before use
KEEP OUT OF REACH OF CHILDREN AND PETS
Made in USA from Domestic and Imported Components
Distributed by:
Bionpharma Inc.
Princeton, NJ
08540
L0000735
R0623
Purpose
Topical anesthetic
Warnings
For external use only.
Directions
■ Adults and children 12 years of age and over: Clean and dry affected area ■ Apply immediately upon removal from pouch ■ Peel off
the first liner and apply exposed patch to affected area ■ Peel off second liner while applying the rest of patch ■ Remove patch after
24 hours ■ Discard patch after single use.
■ Children under 12 years of age: Consult a doctor.
Do Not Use (Do not use)
■ If you are allergic to any active or inactive ingredients of this product ■ On wounds or damaged, irritated or sensitive skin
■ More than one patch on your body at a time or with other topical analgesics at the same time
■ For more than one week without consulting a doctor ■ With a heating pad ■ If pouch is damaged or opened.
Carton Label
NDC 69452-409-04
maximum strength
a+ health TM
pain relief
lidocaine 4% patch
topical analgesic
• desensitizes aggravated nerves
• odor free, thin & flexible
• no-mess, single use application
24 hours
6 Patches
2.91" x 2.17"
Active Ingredient (Active ingredient)
Lidocaine 4%
Other Information (Other information)
Store in a clean, dry place away from direct sunlight. Store in original unopened pouch protected from moisture.
Inactive Ingredients (Inactive ingredients)
alicyclic hydrocarbon resin, mineral oil, polyester film, silicone-coated polyester film, styrene isoprene, styrene block copolymer.
Questions Or Comments? (Questions or comments?)
call toll free 1-888-235-2466 (Mon - Fri 9AM - 5PM EST)
When Using This Product (When using this product)
■ Avoid contact with the eyes ■ Use only as directed ■ Read and follow all directions and warnings on this
package ■ Do not bandage tightly or apply local heat to the area of use ■ Do not reuse patch ■ Keep this product away from pets and
children. Used patches still contain the drug product that can produce serious adverse effect if a child or pet chews or ingests this
patch
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
■ Condition worsens ■ Symptoms persist for more than seven days or clear up and occur again within a
few days ■ Localized skin reactions such as rash, pain, itching, redness, irritation, swelling or blistering occur where the product was
applied.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children and Pets (Keep out of reach of children and pets)
If chewed or swallowed, get medical help or contact a Poison Control Center right away (800-222-1222).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:39.346360 · Updated: 2026-03-14T23:03:50.462651