Cyclosporine, Modified - Cyclosporine Capsule, Liquid Filled
1952d4c7-a40e-4924-b669-c41400774cb9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING Only physicians experienced in management of systemic immunosuppressive therapy for the indicated disease should prescribe cyclosporine capsules, (modified). At doses used in solid organ transplantation, only physicians experienced in immunosuppressive therapy and management of organ transplant recipients should prescribe cyclosporine capsules, (modified). Patients receiving the drug should be managed in facilities equipped and staffed with adequate laboratory and supportive medical resources. The physician responsible for maintenance therapy should have complete information requisite for the follow-up of the patient. Cyclosporine capsules, (modified), a systemic immunosuppressant, may increase the susceptibility to infection and the development of neoplasia. In kidney, liver, and heart transplant patients cyclosporine capsules, (modified) may be administered with other immunosuppressive agents. Increased susceptibility to infection and the possible development of lymphoma and other neoplasms may result from the increase in the degree of immunosuppression in transplant patients. Cyclosporine capsules, (modified) have increased bioavailability in comparison to Sandimmune ® Soft Gelatin Capsules (cyclosporine capsules, USP) [NON-MODIFIED]. Cyclosporine capsules, (modified) and Sandimmune ® are not bioequivalent and cannot be used interchangeably without physician supervision. For a given trough concentration, cyclosporine exposure will be greater with cyclosporine capsules, (modified) than with Sandimmune ® . If a patient who is receiving exceptionally high doses of Sandimmune ® is converted to cyclosporine capsules, (modified), particular caution should be exercised. Cyclosporine blood concentrations should be monitored in transplant and rheumatoid arthritis patients taking cyclosporine capsules, (modified) to avoid toxicity due to high concentrations. Dose adjustments should be made in transplant patients to minimize possible organ rejection due to low concentrations. Comparison of blood concentrations in the published literature with blood concentrations obtained using current assays must be done with detailed knowledge of the assay methods employed (see Dosage and Administration ).
Dosage and Administration
Cyclosporine capsules (modified) has increased bioavailability in comparison to Sandimmune ® . Cyclosporine capsules (modified) and Sandimmune ® are not bioequivalent and cannot be used interchangeably without physician supervision. The daily dose of cyclosporine capsules (modified) should always be given in two divided doses (BID). It is recommended that cyclosporine capsules (modified) be administered on a consistent schedule with regard to time of day and relation to meals. Grapefruit and grapefruit juice affect metabolism, increasing blood concentration of cyclosporine, thus should be avoided. Specific Populations Renal Impairment in Kidney, Liver, and Heart Transplantation Cyclosporine undergoes minimal renal elimination and its pharmacokinetics do not appear to be significantly altered in patients with end-stage renal disease who receive routine hemodialysis treatments (see CLINICAL PHARMACOLOGY ). However, due to its nephrotoxic potential (see WARNINGS ), careful monitoring of renal function is recommended; cyclosporine dosage should be reduced if indicated (see WARNINGS and PRECAUTIONS ). Renal Impairment in Rheumatoid Arthritis and Psoriasis Patients with impaired renal function should not receive cyclosporine (see CONTRAINDICATIONS, WARNINGS and PRECAUTIONS ). Hepatic Impairment The clearance of cyclosporine may be significantly reduced in severe liver disease patients ( See CLINICAL PHARMACOLOGY ). Dose reduction may be necessary in patients with severe liver impairment to maintain blood concentrations within the recommended target range (see WARNINGS and PRECAUTIONS ).
Drug Interactions
(See PRECAUTIONS, Drug Interactions ) When diclofenac or methotrexate was coadministered with cyclosporine in rheumatoid arthritis patients, the AUC of diclofenac and methotrexate, each was significantly increased (see PRECAUTIONS, Drug Interactions ). No clinically significant pharmacokinetic interactions occurred between cyclosporine and aspirin, ketoprofen, piroxicam, or indomethacin. Specific Populations Renal Impairment In a study performed in 4 subjects with end-stage renal disease (creatinine clearance < 5 mL/min), an intravenous infusion of 3.5 mg/kg of cyclosporine over 4 hours administered at the end of a hemodialysis session resulted in a mean volume of distribution (Vdss) of 3.49 L/kg and systemic clearance (CL) of 0.369 L/hr/kg. This systemic CL (0.369 L/hr/kg) was approximately two thirds of the mean systemic CL (0.56 L/hr/kg) of cyclosporine in historical control subjects with normal renal function. In 5 liver transplant patients, the mean clearance of cyclosporine on and off hemodialysis was 463 mL/min and 398 mL/min, respectively. Less than 1% of the dose of cyclosporine was recovered in the dialysate. Hepatic Impairment Cyclosporine is extensively metabolized by the liver. Since severe hepatic impairment may result in significantly increased cyclosporine exposures, the dosage of cyclosporine may need to be reduced in these patients. Pediatric Population Pharmacokinetic data from pediatric patients administered cyclosporine capsules (modified) or Sandimmune ® are very limited. In 15 renal transplant patients aged 3 to 16 years, cyclosporine whole blood clearance after IV administration of Sandimmune ® was 10.6 ± 3.7 mL/min/kg (assay: Cyclo-trac specific RIA). In a study of 7 renal transplant patients aged 2 to 16, the cyclosporine clearance ranged from 9.8 to 15.5 mL/min/kg. In 9 liver transplant patients aged 0.6 to 5.6 years, clearance was 9.3 ± 5.4 mL/min/kg (assay: HPLC). In the pediatric population, cyclosporine capsules (modified) also demonstrates an increased bioavailability as compared to Sandimmune ® . In 7 liver de novo transplant patients aged 1.4 to 10 years, the absolute bioavailability of cyclosporine capsules (modified) was 43% (range 30% to 68%) and for Sandimmune ® in the same individuals absolute bioavailability was 28% (range 17% to 42%). Pediatric Pharmacokinetic Parameters (mean ± SD) Dose/day Dose/weight AUC 1 C max CL/F CL/F Patient Population (mg/d) (mg/kg/d) (ng·hr/mL) (ng/mL) (mL/min) (mL/min/kg) Stable liver transplant 2 Age 2 to 8, Dosed TID (N = 9) 101 ± 25 5.95 ± 1.32 2163 ± 801 629 ± 219 285 ± 94 16.6 ± 4.3 Age 8 to 15, Dosed BID (N = 8) 188 ± 55 4.96 ± 2.09 4272 ± 1462 975 ± 281 378 ± 80 10.2 ± 4.0 Stable liver transplant 3 Age 3, Dosed BID (N = 1) 120 8.33 5832 1050 171 11.9 Age 8 to 15, Dosed BID (N = 5) 158 ± 55 5.51 ± 1.91 4452 ± 2475 1013 ± 635 328 ± 121 11.0 ± 1.9 Stable renal transplant 3 Age 7 to 15, Dosed BID (N = 5) 328 ± 83 7.37 ± 4.11 6922 ± 1988 1827 ± 487 418 ± 143 8.7 ± 2.9 1 AUC was measured over one dosing interval. 2 Assay: Cyclo-trac specific monoclonal radioimmunoassay. 3 Assay: TDx specific monoclonal fluorescence polarization immunoassay.
How Supplied
Cyclosporine capsules, USP (modified) (Soft Gelatin Capsules) 25 mg Blue gray colored opaque oval shaped soft gelatin capsule imprinted as 'S 25' in red color ink, containing clear to cloudy pale yellow colored oily liquid. Packages of 30 unit-dose blisters (NDC 23155-837-30). 50 mg White colored opaque oblong shaped soft gelatin capsule imprinted as 'S 50' in red color ink, containing clear to cloudy pale yellow colored oily liquid Packages of 30 unit-dose blisters (NDC 23155-838-30). 100 mg Blue gray colored opaque oblong shaped soft gelatin capsule imprinted as 'S 100' in red color ink, containing clear to cloudy pale yellow colored oily liquid. Packages of 30 unit-dose blisters (NDC 23155-839-30). Store and Dispense In the original unit-dose container at controlled room temperature 68°F to 77°F (20°C to 25°C). Cyclosporine Capsules, USP (MODIFIED) (Soft Gelatin Capsules) All registered trademarks in this document are the property of their respective owners. Manufactured by: Strides Pharma Science Limited. Bengaluru – 562106, India Manufactured for: Avet Pharmaceuticals Inc. East Brunswick, NJ 08816 1.866.901.DRUG (3784) Revised: 09/2022
Medication Information
Recent Major Changes
WARNING
Only physicians experienced in management of systemic immunosuppressive therapy for the indicated disease should prescribe cyclosporine capsules, (modified). At doses used in solid organ transplantation, only physicians experienced in immunosuppressive therapy and management of organ transplant recipients should prescribe cyclosporine capsules, (modified). Patients receiving the drug should be managed in facilities equipped and staffed with adequate laboratory and supportive medical resources. The physician responsible for maintenance therapy should have complete information requisite for the follow-up of the patient.
Cyclosporine capsules, (modified), a systemic immunosuppressant, may increase the susceptibility to infection and the development of neoplasia. In kidney, liver, and heart transplant patients cyclosporine capsules, (modified) may be administered with other immunosuppressive agents. Increased susceptibility to infection and the possible development of lymphoma and other neoplasms may result from the increase in the degree of immunosuppression in transplant patients.
Cyclosporine capsules, (modified) have increased bioavailability in comparison to Sandimmune®Soft Gelatin Capsules (cyclosporine capsules, USP) [NON-MODIFIED]. Cyclosporine capsules, (modified) and Sandimmune®are not bioequivalent and cannot be used interchangeably without physician supervision. For a given trough concentration, cyclosporine exposure will be greater with cyclosporine capsules, (modified) than with Sandimmune®. If a patient who is receiving exceptionally high doses of Sandimmune® is converted to cyclosporine capsules, (modified), particular caution should be exercised. Cyclosporine blood concentrations should be monitored in transplant and rheumatoid arthritis patients taking cyclosporine capsules, (modified) to avoid toxicity due to high concentrations. Dose adjustments should be made in transplant patients to minimize possible organ rejection due to low concentrations. Comparison of blood concentrations in the published literature with blood concentrations obtained using current assays must be done with detailed knowledge of the assay methods employed (see Dosage and Administration).
Dosage and Administration
Cyclosporine capsules (modified) has increased bioavailability in comparison to Sandimmune ® . Cyclosporine capsules (modified) and Sandimmune ® are not bioequivalent and cannot be used interchangeably without physician supervision.
The daily dose of cyclosporine capsules (modified) should always be given in two divided doses (BID). It is recommended that cyclosporine capsules (modified) be administered on a consistent schedule with regard to time of day and relation to meals. Grapefruit and grapefruit juice affect metabolism, increasing blood concentration of cyclosporine, thus should be avoided.
Specific Populations
Renal Impairment in Kidney, Liver, and Heart Transplantation
Cyclosporine undergoes minimal renal elimination and its pharmacokinetics do not appear to be significantly altered in patients with end-stage renal disease who receive routine hemodialysis treatments (see CLINICAL PHARMACOLOGY). However, due to its nephrotoxic potential (see WARNINGS), careful monitoring of renal function is recommended; cyclosporine dosage should be reduced if indicated (see WARNINGS and PRECAUTIONS).
Renal Impairment in Rheumatoid Arthritis and Psoriasis
Patients with impaired renal function should not receive cyclosporine (see CONTRAINDICATIONS, WARNINGS and PRECAUTIONS).
Hepatic Impairment
The clearance of cyclosporine may be significantly reduced in severe liver disease patients (See CLINICAL PHARMACOLOGY). Dose reduction may be necessary in patients with severe liver impairment to maintain blood concentrations within the recommended target range (see WARNINGS and PRECAUTIONS).
Drug Interactions
(See PRECAUTIONS, Drug Interactions) When diclofenac or methotrexate was coadministered with cyclosporine in rheumatoid arthritis patients, the AUC of diclofenac and methotrexate, each was significantly increased (see PRECAUTIONS, Drug Interactions). No clinically significant pharmacokinetic interactions occurred between cyclosporine and aspirin, ketoprofen, piroxicam, or indomethacin.
Specific Populations
Renal Impairment
In a study performed in 4 subjects with end-stage renal disease (creatinine clearance < 5 mL/min), an intravenous infusion of 3.5 mg/kg of cyclosporine over 4 hours administered at the end of a hemodialysis session resulted in a mean volume of distribution (Vdss) of 3.49 L/kg and systemic clearance (CL) of 0.369 L/hr/kg. This systemic CL (0.369 L/hr/kg) was approximately two thirds of the mean systemic CL (0.56 L/hr/kg) of cyclosporine in historical control subjects with normal renal function. In 5 liver transplant patients, the mean clearance of cyclosporine on and off hemodialysis was 463 mL/min and 398 mL/min, respectively. Less than 1% of the dose of cyclosporine was recovered in the dialysate.
Hepatic Impairment
Cyclosporine is extensively metabolized by the liver. Since severe hepatic impairment may result in significantly increased cyclosporine exposures, the dosage of cyclosporine may need to be reduced in these patients.
Pediatric Population
Pharmacokinetic data from pediatric patients administered cyclosporine capsules (modified) or Sandimmune® are very limited. In 15 renal transplant patients aged 3 to 16 years, cyclosporine whole blood clearance after IV administration of Sandimmune® was 10.6 ± 3.7 mL/min/kg (assay: Cyclo-trac specific RIA). In a study of 7 renal transplant patients aged 2 to 16, the cyclosporine clearance ranged from 9.8 to 15.5 mL/min/kg. In 9 liver transplant patients aged 0.6 to 5.6 years, clearance was 9.3 ± 5.4 mL/min/kg (assay: HPLC).
In the pediatric population, cyclosporine capsules (modified) also demonstrates an increased bioavailability as compared to Sandimmune®. In 7 liver de novo transplant patients aged 1.4 to 10 years, the absolute bioavailability of cyclosporine capsules (modified) was 43% (range 30% to 68%) and for Sandimmune® in the same individuals absolute bioavailability was 28% (range 17% to 42%).
|
Pediatric Pharmacokinetic Parameters (mean ± SD)
|
||||||
|
Dose/day
|
Dose/weight
|
AUC1
|
Cmax
|
CL/F
|
CL/F
|
|
|
Patient Population
|
(mg/d)
|
(mg/kg/d)
|
(ng·hr/mL)
|
(ng/mL)
|
(mL/min)
|
(mL/min/kg)
|
| Stable liver transplant2
|
||||||
| Age 2 to 8, Dosed TID (N = 9) |
101 ± 25 |
5.95 ± 1.32 |
2163 ± 801 |
629 ± 219 |
285 ± 94 |
16.6 ± 4.3 |
| Age 8 to 15, Dosed BID (N = 8) |
188 ± 55 |
4.96 ± 2.09 |
4272 ± 1462 |
975 ± 281 |
378 ± 80 |
10.2 ± 4.0 |
| Stable liver transplant3
|
||||||
| Age 3, Dosed BID (N = 1) |
120 |
8.33 |
5832 |
1050 |
171 |
11.9 |
| Age 8 to 15, Dosed BID (N = 5) |
158 ± 55 |
5.51 ± 1.91 |
4452 ± 2475 |
1013 ± 635 |
328 ± 121 |
11.0 ± 1.9 |
| Stable renal transplant3
|
||||||
| Age 7 to 15, Dosed BID (N = 5) |
328 ± 83 |
7.37 ± 4.11 |
6922 ± 1988 |
1827 ± 487 |
418 ± 143 |
8.7 ± 2.9 |
|
1AUC was measured over one dosing interval. 2Assay: Cyclo-trac specific monoclonal radioimmunoassay. 3Assay: TDx specific monoclonal fluorescence polarization immunoassay. |
How Supplied
Cyclosporine capsules, USP (modified) (Soft Gelatin Capsules)
25 mg
Blue gray colored opaque oval shaped soft gelatin capsule imprinted as 'S 25' in red color ink, containing clear to cloudy pale yellow colored oily liquid.
Packages of 30 unit-dose blisters (NDC 23155-837-30).
50 mg
White colored opaque oblong shaped soft gelatin capsule imprinted as 'S 50' in red color ink, containing clear to cloudy pale yellow colored oily liquid
Packages of 30 unit-dose blisters (NDC 23155-838-30).
100 mg
Blue gray colored opaque oblong shaped soft gelatin capsule imprinted as 'S 100' in red color ink, containing clear to cloudy pale yellow colored oily liquid.
Packages of 30 unit-dose blisters (NDC 23155-839-30).
Store and Dispense
In the original unit-dose container at controlled room temperature 68°F to 77°F (20°C to 25°C).
Cyclosporine Capsules, USP (MODIFIED) (Soft Gelatin Capsules)
All registered trademarks in this document are the property of their respective owners.
Manufactured by:
Strides Pharma Science Limited.
Bengaluru – 562106, India
Manufactured for:
Avet Pharmaceuticals Inc.
East Brunswick, NJ 08816
1.866.901.DRUG (3784)
Revised: 09/2022
Description
Cyclosporine capsules, USP (modified) is an oral formulation of cyclosporine that immediately forms a microemulsion in an aqueous environment.
Cyclosporine, USP the active principle in cyclosporine capsules, USP (modified), is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.
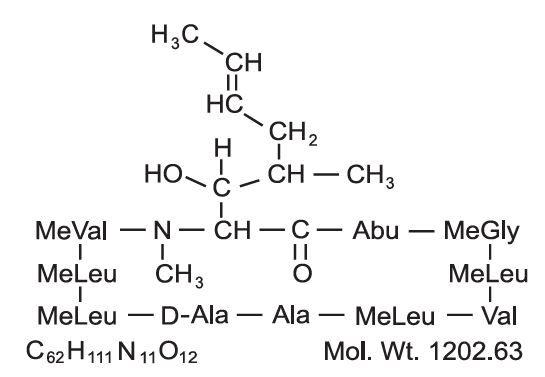
Chemically, cyclosporine, USP is designated as [R-[R*,R*-(E)]]-cyclic-(L-alanyl-D-alanyl-N-methyl-L-leucyl-N-methyl-L-leucyl-N-methyl-L-valyl-3-hydroxy-N,4-dimethyl-L-2-amino-6-octenoyl-L-α -amino-butyryl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucyl).
Cyclosporine capsules, USP (modified) (Soft Gelatin Capsules) are available in 25 mg, 50 mg and 100 mg strengths.
Each 25 mg capsule contains:
Cyclosporine, USP………………………………………………………………………………25 mg
Dehydrated alcohol................................................. (9.5% w/v or 12.0% v/v)
Each 50 mg capsule contains:
Cyclosporine, USP……………………………………………………………………………...50 mg
Dehydrated alcohol................................................... (9.5% w/v or 12.0% v/v)
Each 100 mg capsule contains:
Cyclosporine, USP…………………………………………………………………………...100 mg
Dehydrated alcohol.................................................. (9.5% w/v or 12.0% v/v)
Inactive Ingredients: gelatin, glycerin, propylene glycol, titanium dioxide, ferric oxide black [25 mg and 100 mg], glyceryl monolinoleate, polyoxyl 40 hydrogenated castor oil, all-rac-alpha tocopherol [vitamin E synthetic], Ink contains- ammonium hydroxide 28%, iron oxide red, polyethylene glycol, polyvinyl acetate phthalate, propylene glycol.
The chemical structure of cyclosporine (also known as cyclosporin A) is:
General
Cyclosporine capsules (modified) is contraindicated in patients with a hypersensitivity to cyclosporine or to any of the ingredients of the formulation.
Warnings
(See also BOXED WARNING)
Excretion
Only 0.1% of a cyclosporine dose is excreted unchanged in the urine. Elimination is primarily biliary with only 6% of the dose (parent drug and metabolites) excreted in the urine. Neither dialysis nor renal failure alters cyclosporine clearance significantly.
Pregnancy
Risk Summary
Available data from published literature, including the Transplant Pregnancy Registry International, observational cohort studies, case-controlled studies, meta-analysis, case series, and case reports, over decades of use with cyclosporine in pregnancy have not identified a drug associated risk of major birth defects, or miscarriage. Adverse maternal or fetal outcomes including hypertension, preeclampsia, preterm birth, and low birth weight are increased in patients treated with cyclosporine. However, patients receiving cyclosporine during pregnancy have underlying medical conditions and may be treated with concomitant medications that limit the interpretability of these findings (see Data).
Embryo-fetal developmental (EFD) studies in rats and rabbits with cyclosporine have shown embryo-fetal toxicity at dose levels below the maximum recommended human dose (MRHD) based on body surface area (BSA).
The alcohol content of cyclosporine capsules, (modified) should be taken into account when given to pregnant women (see WARNINGS, Special Excipients).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
Available data from the National Transplantation Pregnancy Registry (NTPR) including 622 pregnancies in renal, liver, and heart transplant recipients exposed to cyclosporine during pregnancy found that the overall rate of major birth defects, live birth rates, and miscarriage rates were comparable to the general population. Maternal and fetal adverse outcomes, including the rate of hypertension, preeclampsia, premature births, and low birth weight infants appear to be increased in transplant recipients treated with cyclosporine compared to the general population. However, these patients have underlying medical conditions that confound the above findings.
Animal Data
Animal studies have shown reproductive toxicity in rats and rabbits.
Three EFD studies (two oral and one intravenous) are available in rats. In two EFD studies, pregnant rats were orally administered with cyclosporine either at doses of 10, 17, 30, 100 and 300 mg/kg/day or 4, 10 and 25 mg/kg/day from gestation day (GD) 6 to 15 or from GD 7 to 17, respectively. Maternal toxicity characterized by mortality, clinical signs of toxicity and impaired body weight gain were observed at 30 mg/kg/day and above. Cyclosporine was embryo- and fetotoxic as indicated by increased embryonic mortality and reduced fetal weight together with skeletal retardations in rats at 25 mg/kg/day and above. In addition, ventricular septal defect was observed at 25 mg/kg/day in fetuses. In the first study, the oral no observed effect level (NOEL) for both dams and fetuses was 17 mg/kg/day (0.2 times the MRHD based on BSA). In the other oral study, the NOEL for dams and fetuses were 10 and 4 mg/kg/day (0.13 and 0.05 times the MRHD based on BSA), respectively. In the IV EFD study, rats were administered with 3, 6 and 12 mg/kg/day of cyclosporine from GD 7 to 17. An increase in post implantation loss was observed at 12 mg/kg/day; ventricular septal defect was observed at ≥ 6 mg/kg/day in fetuses. The IV NOEL for dams and fetus were 6 and 3 mg/kg/day (0.08 and 0.04 times the MRHD, respectively, based on BSA), respectively, after IV administration.
In rabbits, cyclosporine was orally administered at dose levels of 10, 30, 100 or 300 mg/kg/day from GD 6 to 18. At 100 mg/kg/day and above, reduction in body weight gain of dams and at 300 mg/kg/day abortions were observed. Maternal toxicity, embryo-fetotoxicity as indicated by increased pre- and postnatal mortality, reduced fetal weight together with skeletal retardations were observed at 100 mg/kg/day and above. The NOEL for dams and fetuses was 30 mg/kg/day (1 times the MRHD based on BSA).
In two published research studies, rabbits exposed to cyclosporine in utero (10 mg/kg/day subcutaneously) demonstrated reduced numbers of nephrons, renal hypertrophy, systemic hypertension and progressive renal insufficiency up to 35 weeks of age. These findings have not been demonstrated in other species and their relevance for humans is unknown.
In a peri- and postnatal development study in rats, pregnant rats were orally administered with cyclosporine (5, 15 or 45 mg/kg/day) from GD 15 until end of lactation. At 45 mg/kg/day (0.5 times the MRHD based on BSA), increased pre and postnatal mortality of offspring and reduced body weight gain of surviving pups were observed. Cyclosporine up to 15 mg/kg/day (0.2 times the MRHD based on BSA) had no effect on pregnancy, pre and postnatal development of offspring.
Psoriasis
Cyclosporine capsules (modified) is indicated for the treatment of adult, nonimmunocompromised patients with severe (i.e., extensive and/or disabling), recalcitrant, plaque psoriasis who have failed to respond to at least one systemic therapy (e.g., PUVA, retinoids, or methotrexate) or in patients for whom other systemic therapies are contraindicated, or cannot be tolerated.
While rebound rarely occurs, most patients will experience relapse with cyclosporine capsules (modified) as with other therapies upon cessation of treatment.
Absorption
Cyclosporine capsules (modified) has increased bioavailability compared to Sandimmune®. The absolute bioavailability of cyclosporine administered as Sandimmune® is dependent on the patient population, estimated to be less than 10% in liver transplant patients and as great as 89% in some renal transplant patients. The absolute bioavailability of cyclosporine administered as cyclosporine capsules (modified) has not been determined in adults. In studies of renal transplant, rheumatoid arthritis and psoriasis patients, the mean cyclosporine AUC was approximately 20% to 50% greater and the peak blood cyclosporine concentration (Cmax) was approximately 40% to 106% greater following administration of cyclosporine capsules (modified) compared to following administration of Sandimmune®. The dose normalized AUC in de novo liver transplant patients administered cyclosporine capsules (modified) 28 days after transplantation was 50% greater and Cmax was 90% greater than in those patients administered Sandimmune®. AUC and Cmax are also increased (cyclosporine capsules (modified) relative to Sandimmune®) in heart transplant patients, but data are very limited. Although the AUC and Cmax values are higher on cyclosporine capsules (modified) relative to Sandimmune®, the predose trough concentrations (dose-normalized) are similar for the two formulations.
Following oral administration of cyclosporine capsules, (modified), the time to peak blood cyclosporine concentrations (Tmax) ranged from 1.5 to 2.0 hours. The administration of food with cyclosporine capsules (modified) decreases the cyclosporine AUC and Cmax. A high fat meal (669 kcal, 45 grams fat) consumed within one-half hour before cyclosporine capsules (modified) administration decreased the AUC by 13% and Cmax by 33%. The effects of a low-fat meal (667 kcal, 15 grams fat) were similar.
The effect of T-tube diversion of bile on the absorption of cyclosporine from cyclosporine capsules (modified) was investigated in eleven de novo liver transplant patients. When the patients were administered cyclosporine capsules (modified) with and without T-tube diversion of bile, very little difference in absorption was observed, as measured by the change in maximal cyclosporine blood concentrations from pre-dose values with the T-tube closed relative to when it was open: 6.9 ± 41% (range 55% to 68%).
|
Pharmacokinetic Parameters (mean ± SD)
|
|||||||
|
Dose/day1
|
Dose/weight
|
AUC2
|
Cmax
|
Trough3
|
CL/F
|
CL/F
|
|
|
Patient Population
|
(mg/d)
|
(mg/kg/d)
|
(ng·hr/mL)
|
(ng/mL)
|
(ng/mL)
|
(mL/min)
|
(mL/min/kg)
|
| De novo renal transplant4
|
597 ± 174 |
7.95 ± 2.81 |
8772 ± 2089 |
1802 ± 428 |
361 ± 129 |
593 ± 204 |
7.8 ± 2.9 |
| Week 4 (N = 37) |
|||||||
| Stable renal transplant4
|
344 ± 122 |
4.10 ± 1.58 |
6035 ± 2194 |
1333 ± 469 |
251 ± 116 |
492 ± 140 |
5.9 ± 2.1 |
| (N = 55) |
|||||||
| De novo liver transplant5
|
458 ± 190 |
6.89 ± 3.68 |
7187 ± 2816 |
1555 ± 740 |
268 ± 101 |
577 ± 309 |
8.6 ± 5.7 |
| Week 4 (N = 18) |
|||||||
| De novo rheumatoid arthritis6
|
182 ± 55.6 |
2.37 ± 0.36 |
2641 ± 877 |
728 ± 263 |
96.4 ± 37.7 |
613 ± 196 |
8.3 ± 2.8 |
| (N = 23) |
|||||||
| De novo psoriasis6
|
189 ± 69.8 |
2.48 ± 0.65 |
2324 ± 1048 |
655 ± 186 |
74.9 ± 46.7 |
723 ± 186 |
10.2 ± 3.9 |
| Week 4 (N = 18) |
|||||||
|
1Total daily dose was divided into two doses administered every 12 hours. 2AUC was measured over one dosing interval. 3Trough concentration was measured just prior to the morning cyclosporine capsules (Modified) dose, approximately 12 hours after the previous dose. 4Assay: TDx specific monoclonal fluorescence polarization immunoassay. 5Assay: Cyclo-trac specific monoclonal radioimmunoassay. 6Assay: INCSTAR specific monoclonal radioimmunoassay. |
Metabolism
Cyclosporine is extensively metabolized by the cytochrome P-450 3A enzyme system in the liver, and to a lesser degree in the gastrointestinal tract, and the kidney. The metabolism of cyclosporine can be altered by the coadministration of a variety of agents (see PRECAUTIONS, Drug Interactions). At least 25 metabolites have been identified from human bile, feces, blood, and urine. The biological activity of the metabolites and their contributions to toxicity are considerably less than those of the parent compound. The major metabolites (M1, M9, and M4N) result from oxidation at the 1-beta, 9-gamma, and 4-N-demethylated positions, respectively. At steady state following the oral administration of Sandimmune®, the mean AUCs for blood concentrations of M1, M9, and M4N are about 70%, 21%, and 7.5% of the AUC for blood cyclosporine concentrations, respectively. Based on blood concentration data from stable renal transplant patients (13 patients administered cyclosporine capsules (modified) and Sandimmune® in a crossover study), and bile concentration data from de novo liver transplant patients (4 administered cyclosporine capsules, (modified), 3 administered Sandimmune®), the percentage of dose present as M1, M9, and M4N metabolites is similar when either cyclosporine capsules (modified) or Sandimmune® is administered.
Overdosage
There is a minimal experience with cyclosporine overdosage. Forced emesis and gastric lavage can be of value up to 2 hours after administration of cyclosporine capsules, (modified). Transient hepatotoxicity and nephrotoxicity may occur which should resolve following drug withdrawal. Oral doses of cyclosporine up to 10 g (about 150 mg/kg) have been tolerated with relatively minor clinical consequences, such as vomiting, drowsiness, headache, tachycardia and, in a few patients, moderately severe, reversible impairment of renal function. However, serious symptoms of intoxication have been reported following accidental parenteral overdosage with cyclosporine in premature neonates. General supportive measures and symptomatic treatment should be followed in all cases of overdosage. Cyclosporine is not dialyzable to any great extent, nor is it cleared well by charcoal hemoperfusion. The oral dosage at which half of experimental animals are estimated to die is 31 times, 39 times, and > 54 times the human maintenance dose for transplant patients (6mg/kg; corrections based on body surface area) in mice, rats, and rabbits.
All Patients
Cyclosporine, the active ingredient of cyclosporine capsules, (modified), can cause nephrotoxicity and hepatotoxicity. The risk increases with increasing doses of cyclosporine. Renal dysfunction including structural kidney damage is a potential consequence of cyclosporine capsules (modified) and therefore renal function must be monitored during therapy. Care should be taken in using cyclosporine with nephrotoxic drugs (see PRECAUTIONS).
Patients receiving cyclosporine capsules (modified) require frequent monitoring of serum creatinine (see Special Monitoring under DOSAGE AND ADMINISTRATION). Elderly patients should be monitored with particular care, since decreases in renal function also occur with age. If patients are not properly monitored and doses are not properly adjusted, cyclosporine therapy can be associated with the occurrence of structural kidney damage and persistent renal dysfunction.
An increase in serum creatinine and BUN may occur during cyclosporine capsules (modified) therapy and reflect a reduction in the glomerular filtration rate. Impaired renal function at any time requires close monitoring, and frequent dosage adjustment may be indicated. The frequency and severity of serum creatinine elevations increase with dose and duration of cyclosporine therapy. These elevations are likely to become more pronounced without dose reduction or discontinuation.
Because cyclosporine capsules (modified) is not bioequivalent to Sandimmune®, conversion from cyclosporine capsules (modified) to Sandimmune® using a 1:1 ratio (mg/kg/day) may result in lower cyclosporine blood concentrations. Conversion from cyclosporine capsules (modified) to Sandimmune® should be made with increased monitoring to avoid the potential of underdosing.
Distribution
Cyclosporine is distributed largely outside the blood volume. The steady state volume of distribution during intravenous dosing has been reported as 3 to 5 L/kg in solid organ transplant recipients. In blood, the distribution is concentration dependent. Approximately 33% to 47% is in plasma, 4% to 9% in lymphocytes, 5% to 12% in granulocytes, and 41% to 58% in erythrocytes. At high concentrations, the binding capacity of leukocytes and erythrocytes becomes saturated. In plasma, approximately 90% is bound to proteins, primarily lipoproteins. Cyclosporine is excreted in human milk (see PRECAUTIONS, Nursing Mothers).
Geriatric Use
In rheumatoid arthritis clinical trials with cyclosporine, 17.5% of patients were age 65 or older. These patients were more likely to develop systolic hypertension on therapy, and more likely to show serum creatinine rises ≥ 50% above the baseline after 3 to 4 months of therapy.
Clinical studies of cyclosporine capsules (modified) in transplant and psoriasis patients did not include a sufficient number of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experiences have not identified differences in response between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Pediatric Use
Although no adequate and well-controlled studies have been completed in children, transplant recipients as young as one year of age have received cyclosporine capsules (modified) with no unusual adverse effects. The safety and efficacy of cyclosporine capsules (modified) treatment in children with juvenile rheumatoid arthritis or psoriasis below the age of 18 have not been established.
Nursing Mothers
Risk Summary
Cyclosporine and its metabolites are present in human milk following oral and intravenous administration. Adverse effects on the breastfed infant have not been reported. There are no data on the effects of the drug on milk production. The alcohol content of cyclosporine capsules, (modified) should be taken into account when given to lactating women (see WARNINGS, Special Excipients). Lactating women are encouraged to avoid additional alcohol intake during treatment. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for cyclosporine capsules, (modified) and any potential adverse effects on the breastfed infant from cyclosporine capsules, (modified) or from the underlying maternal condition.
Laboratory Tests
In all patients treated with cyclosporine, renal and liver functions should be assessed repeatedly by measurement of serum creatinine, BUN, serum bilirubin, and liver enzymes. Serum lipids, magnesium, and potassium should also be monitored. Cyclosporine blood concentrations should be routinely monitored in transplant patients (see DOSAGE AND ADMINISTRATION, Blood Concentration Monitoring in Transplant Patients), and periodically monitored in rheumatoid arthritis patients.
Pharmacokinetics
The immunosuppressive activity of cyclosporine is primarily due to parent drug. Following oral administration, absorption of cyclosporine is incomplete. The extent of absorption of cyclosporine is dependent on the individual patient, the patient population, and the formulation. Elimination of cyclosporine is primarily biliary with only 6% of the dose (parent drug and metabolites) excreted in urine. The disposition of cyclosporine from blood is generally biphasic, with a terminal half-life of approximately 8.4 hours (range 5 to 18 hours). Following intravenous administration, the blood clearance of cyclosporine (assay: HPLC) is approximately 5 to 7 mL/min/kg in adult recipients of renal or liver allografts. Blood cyclosporine clearance appears to be slightly slower in cardiac transplant patients.
Cyclosporine capsules (MODIFIED) and cyclosporine oral solution (MODIFIED) are bioequivalent.
The relationship between administered dose and exposure (area under the concentration versus time curve, AUC) is linear within the therapeutic dose range. The intersubject variability (total, %CV) of cyclosporine exposure (AUC) when cyclosporine capsules, (modified) or Sandimmune® is administered ranges from approximately 20% to 50% in renal transplant patients. This intersubject variability contributes to the need for individualization of the dosing regimen for optimal therapy (see DOSAGE AND ADMINISTRATION). Intrasubject variability of AUC in renal transplant recipients (%CV) was 9% to 21% for cyclosporine capsules, (modified) and 19% to 26% for Sandimmune®. In the same studies, intrasubject variability of trough concentrations (%CV) was 17% to 30% for cyclosporine capsules, (modified) and 16% to 38% for Sandimmune®.
Geriatric Population
Comparison of single dose data from both normal elderly volunteers (N = 18, mean age 69 years) and elderly rheumatoid arthritis patients (N = 16, mean age 68 years) to single dose data in young adult volunteers (N = 16, mean age 26 years) showed no significant difference in the pharmacokinetic parameters.
Rheumatoid Arthritis
The effectiveness of Sandimmune® and cyclosporine capsules (modified) in the treatment of severe rheumatoid arthritis was evaluated in 5 clinical studies involving a total of 728 cyclosporine treated patients and 273 placebo treated patients.
A summary of the results is presented for the "responder" rates per treatment group, with a responder being defined as a patient having completed the trial with a 20% improvement in the tender and the swollen joint count and a 20% improvement in 2 of 4 of investigator global, patient global, disability, and erythrocyte sedimentation rates (ESR) for the Studies 651 and 652 and 3 of 5 of investigator global, patient global, disability, visual analog pain, and ESR for Studies 2008, 654 and 302.
Study 651 enrolled 264 patients with active rheumatoid arthritis with at least 20 involved joints, who had failed at least one major RA drug, using a 3:3:2 randomization to one of the following three groups: (1) cyclosporine dosed at 2.5 to 5 mg/kg/day, (2) methotrexate at 7.5 to 15 mg/week, or (3) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 3.1 mg/kg/day. See Graph below.
Study 652 enrolled 250 patients with active RA with > 6 active painful or tender joints who had failed at least one major RA drug. Patients were randomized using a 3:3:2 randomization to 1 of 3 treatment arms: (1) 1.5 to 5 mg/kg/day of cyclosporine, (2) 2.5 to 5 mg/kg/day of cyclosporine, and (3) placebo. Treatment duration was 16 weeks. The mean cyclosporine dose for group 2 at the last visit was 2.92 mg/kg/day. See Graph below.
Study 2008 enrolled 144 patients with active RA and > 6 active joints who had unsuccessful treatment courses of aspirin and gold or Penicillamine. Patients were randomized to 1 of 2 treatment groups (1) cyclosporine 2.5 to 5 mg/kg/day with adjustments after the first month to achieve a target trough level and (2) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 3.63 mg/kg/day. See Graph below.
Study 654 enrolled 148 patients who remained with active joint counts of 6 or more despite treatment with maximally tolerated methotrexate doses for at least three months. Patients continued to take their current dose of methotrexate and were randomized to receive, in addition, one of the following medications: (1) cyclosporine 2.5 mg/kg/day with dose increases of 0.5 mg/kg/day at weeks 2 and 4 if there was no evidence of toxicity and further increases of 0.5 mg/kg/day at weeks 8 and 16 if a < 30% decrease in active joint count occurred without any significant toxicity; dose decreases could be made at any time for toxicity or (2) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 2.8 mg/kg/day (range: 1.3 to 4.1). See Graph below.
Study 302 enrolled 299 patients with severe active RA, 99% of whom were unresponsive or intolerant to at least one prior major RA drug. Patients were randomized to 1 of 2 treatment groups (1) cyclosporine capsules (modified) and (2) cyclosporine, both of which were started at 2.5 mg/kg/day and increased after 4 weeks for inefficacy in increments of 0.5 mg/kg/day to a maximum of 5 mg/kg/day and decreased at any time for toxicity. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 2.91 mg/kg/day (range: 0.72 to 5.17) for cyclosporine capsules (modified) and 3.27 mg/kg/day (range: 0.73 to 5.68) for cyclosporine. See Graph below.
Clinical Pharmacology
Cyclosporine is a potent immunosuppressive agent that in animals prolongs survival of allogeneic transplants involving skin, kidney, liver, heart, pancreas, bone marrow, small intestine, and lung. Cyclosporine has been demonstrated to suppress some humoral immunity and to a greater extent, cell-mediated immune reactions such as allograft rejection, delayed hypersensitivity, experimental allergic encephalomyelitis, Freund's adjuvant arthritis, and graft versus host disease in many animal species for a variety of organs.
The effectiveness of cyclosporine results from specific and reversible inhibition of immunocompetent lymphocytes in the G0- and G1-phase of the cell cycle. T-lymphocytes are preferentially inhibited. The T-helper cell is the main target, although the T-suppressor cell may also be suppressed. Cyclosporine also inhibits lymphokine production and release including interleukin-2.
No effects on phagocytic function (changes in enzyme secretions, chemotactic migration of granulocytes, macrophage migration, carbon clearance in vivo) have been detected in animals. Cyclosporine does not cause bone marrow suppression in animal models or man.
Newly Transplanted Patients
The initial oral dose of cyclosporine capsules (modified) can be given 4 to 12 hours prior to transplantation or be given postoperatively. The initial dose of cyclosporine capsules (modified) varies depending on the transplanted organ and the other immunosuppressive agents included in the immunosuppressive protocol. In newly transplanted patients, the initial oral dose of cyclosporine capsules (modified) is the same as the initial oral dose of Sandimmune®. Suggested initial doses are available from the results of a 1994 survey of the use of Sandimmune® in US transplant centers. The mean ± SD initial doses were 9 ± 3 mg/kg/day for renal transplant patients (75 centers), 8 ± 4 mg/kg/day for liver transplant patients (30 centers), and 7 ± 3 mg/kg/day for heart transplant patients (24 centers). Total daily doses were divided into two equal daily doses. The cyclosporine capsules (modified) dose is subsequently adjusted to achieve a pre-defined cyclosporine blood concentration (see Blood Concentration Monitoring in Transplant Patients, below). If cyclosporine trough blood concentrations are used, the target range is the same for cyclosporine capsules (modified) as for Sandimmune®. Using the same trough concentration target range for cyclosporine capsules (modified) as for Sandimmune® results in greater cyclosporine exposure when cyclosporine capsules (modified) is administered (see Pharmacokinetics, Absorption). Dosing should be titrated based on clinical assessments of rejection and tolerability. Lower cyclosporine capsules (modified) doses may be sufficient as maintenance therapy.
Adjunct therapy with adrenal corticosteroids is recommended initially. Different tapering dosage schedules of prednisone appear to achieve similar results. A representative dosage schedule based on the patient's weight started with 2.0 mg/kg/day for the first 4 days tapered to 1.0 mg/kg/day by 1 week, 0.6 mg/kg/day by 2 weeks, 0.3 mg/kg/day by 1 month, and 0.15 mg/kg/day by 2 months and thereafter as a maintenance dose. Steroid doses may be further tapered on an individualized basis depending on status of patient and function of graft. Adjustments in dosage of prednisone must be made according to the clinical situation.
Kidney, Liver, and Heart Transplant
Nephrotoxicity
Cyclosporine, the active ingredient of cyclosporine capsules, (modified), can cause nephrotoxicity and hepatotoxicity when used in high doses. It is not unusual for serum creatinine and BUN levels to be elevated during cyclosporine therapy. These elevations in renal transplant patients do not necessarily indicate rejection, and each patient must be fully evaluated before dosage adjustment is initiated.
Based on the historical Sandimmune ® experience with oral solution, nephrotoxicity associated with cyclosporine had been noted in 25% of cases of renal transplantation, 38% of cases of cardiac transplantation, and 37% of cases of liver transplantation. Mild nephrotoxicity was generally noted 2 to 3 months after renal transplant and consisted of an arrest in the fall of the pre-operative elevations of BUN and creatinine at a range of 35 to 45 mg/dL and 2.0 to 2.5 mg/dL, respectively. These elevations were often responsive to cyclosporine dosage reduction.
More overt nephrotoxicity was seen early after transplantation and was characterized by a rapidly rising BUN and creatinine. Since these events are similar to renal rejection episodes, care must be taken to differentiate between them. This form of nephrotoxicity is usually responsive to cyclosporine dosage reduction.
Although specific diagnostic criteria which reliably differentiate renal graft rejection from drug toxicity have not been found, a number of parameters have been significantly associated with one or the other. It should be noted however, that up to 20% of patients may have simultaneous nephrotoxicity and rejection.
|
Nephrotoxicity vs. Rejection
|
||
|
Parameter
|
Nephrotoxicity
|
Rejection
|
| History |
Donor >50 years old or hypotensive Prolonged kidney preservation Prolonged anastomosis time Concomitant nephrotoxic drugs |
Anti-donor immune response Retransplant patient |
| Clinical |
Often >6 weeks postopb
Prolonged initial nonfunction (acute tubular necrosis) |
Often < 4 weeks postopb
Fever > 37.5°C Weight gain > 0.5 kg Graft swelling and tenderness Decrease in daily urine volume > 500 mL (or 50%) |
| Laboratory |
CyA serum trough level > 200 ng/mL Gradual rise in Cr (<0.15 mg/dL/day)a Cr plateau < 25% above baseline BUN/Cr ≥ 20 |
CyA serum trough level < 150 ng/mL Rapid rise in Cr (> 0.3 mg/dL/day)a Cr > 25% above baseline BUN/Cr < 20 |
| Biopsy |
Arteriolopathy (medial hypertrophy a, hyalinosis, nodular deposits, intimal thickening, endothelial vacuolization, progressive scarring) Tubular atrophy, isometric vacuolization, isolated calcifications Minimal edema Mild focal infiltratesc |
Endovasculitisc (proliferationa, intimal arteritisb, necrosis, sclerosis) Tubulitis with RBCb and WBCb casts, some irregular vacuolization Interstitial edemac and hemorrhageb Diffuse moderate to severe mononuclear infiltratesd |
| Aspiration Cytology |
Diffuse interstitial fibrosis, often striped form CyA deposits in tubular and endothelial cells Fine isometric vacuolization of tubular cells |
Glomerulitis (mononuclear cells)c
Inflammatory infiltrate with mononuclear phagocytes, macrophages, lymphoblastoid cells, and activated T-cells These strongly express HLA-DR antigens |
| Urine Cytology |
Tubular cells with vacuolization and granularization |
Degenerative tubular cells, plasma cells, and lymphocyturia > 20% of sediment |
| Manometry Ultrasonography |
Intracapsular pressure < 40 mm Hgb
Unchanged graft cross sectional area |
Intracapsular pressure > 40 mm Hgb
Increase in graft cross sectional area AP diameter ≥ Transverse diameter |
| Magnetic Resonance Imagery |
Normal appearance |
Loss of distinct corticomedullary junction, swelling image intensity of parachyma approaching that of psoas, loss of hilar fat |
| Radionuclide Scan |
Normal or generally decreased perfusion Decrease in tubular function (131 I-hippuran) > decrease in perfusion (99m Tc DTPA) |
Patchy arterial flow Decrease in perfusion > decrease in tubular function Increased uptake of Indium 111 labeled platelets or Tc-99m in colloid |
| Therapy |
Responds to decreased cyclosporine |
Responds to increased steroids or antilymphocyte globulin |
|
ap < 0.05, bp < 0.01, cp < 0.001, dp < 0.0001 |
A form of a cyclosporine-associated nephropathy is characterized by serial deterioration in renal function and morphologic changes in the kidneys. From 5% to 15% of transplant recipients who have received cyclosporine will fail to show a reduction in rising serum creatinine despite a decrease or discontinuation of cyclosporine therapy. Renal biopsies from these patients will demonstrate one or several of the following alterations: tubular vacuolization, tubular microcalcifications, peritubular capillary congestion, arteriolopathy, and a striped form of interstitial fibrosis with tubular atrophy. Though none of these morphologic changes is entirely specific, a diagnosis of cyclosporine-associated structural nephrotoxicity requires evidence of these findings.
When considering the development of cyclosporine-associated nephropathy, it is noteworthy that several authors have reported an association between the appearance of interstitial fibrosis and higher cumulative doses or persistently high circulating trough concentrations of cyclosporine. This is particularly true during the first 6 post-transplant months when the dosage tends to be highest and when, in kidney recipients, the organ appears to be most vulnerable to the toxic effects of cyclosporine. Among other contributing factors to the development of interstitial fibrosis in these patients are prolonged perfusion time, warm ischemia time, as well as episodes of acute toxicity, and acute and chronic rejection. The reversibility of interstitial fibrosis and its correlation to renal function have not yet been determined. Reversibility of arteriolopathy has been reported after stopping cyclosporine or lowering the dosage.
Impaired renal function at any time requires close monitoring, and frequent dosage adjustment may be indicated.
In the event of severe and unremitting rejection, when rescue therapy with pulse steroids and monoclonal antibodies fail to reverse the rejection episode, it may be preferable to switch to alternative immunosuppressive therapy rather than increase the cyclosporine capsules (modified) dose to excessive blood concentrations.
Due to the potential for additive or synergistic impairment of renal function, caution should be exercised when coadministering cyclosporine capsules (modified) with other drugs that may impair renal function (see PRECAUTIONS, Drug Interactions).
Thrombotic Microangiopathy
Occasionally patients have developed a syndrome of thrombocytopenia and microangiopathic hemolytic anemia which may result in graft failure. The vasculopathy can occur in the absence of rejection and is accompanied by avid platelet consumption within the graft as demonstrated by Indium 111 labeled platelet studies. Neither the pathogenesis nor the management of this syndrome is clear. Though resolution has occurred after reduction or discontinuation of cyclosporine and 1) administration of streptokinase and heparin or 2) plasmapheresis, this appears to depend upon early detection with Indium 111 labeled platelet scans (see ADVERSE REACTIONS).
Hyperkalemia
Significant hyperkalemia (sometimes associated with hyperchloremic metabolic acidosis) and hyperuricemia have been seen occasionally in individual patients.
Hepatotoxicity
Cases of hepatotoxicity and liver injury including cholestasis, jaundice, hepatitis, and liver failure, have been reported in patients treated with cyclosporine. Most reports included patients with significant co-morbidities, underlying conditions and other confounding factors including infectious complications and comedications with hepatotoxic potential. In some cases, mainly in transplant patients, fatal outcomes have been reported (see ADVERSE REACTIONS, Postmarketing Experience, Kidney, Liver and Heart Transplantation).
Hepatotoxicity, usually manifested by elevations in hepatic enzymes and bilirubin, was reported in patients treated with cyclosporine in clinical trials: 4% in renal transplantation, 7% in cardiac transplantation, and 4% in liver transplantation. This was usually noted during the first month of therapy when high doses of cyclosporine were used. The chemistry elevations usually decreased with a reduction in dosage.
Malignancies
As in patients receiving other immunosuppressants, those patients receiving cyclosporine are at increased risk for development of lymphomas and other malignancies, particularly those of the skin. Patients taking cyclosporine should be warned to avoid excess ultraviolet light exposure. The increased risk appears related to the intensity and duration of immunosuppression rather than to the use of specific agents. Because of the danger of oversuppression of the immune system resulting in increased risk of infection or malignancy, a treatment regimen containing multiple immunosuppressants should be used with caution. Some malignancies may be fatal. Transplant patients receiving cyclosporine are at increased risk for serious infection with fatal outcome.
Serious Infections
Patients receiving immunosuppressants, including cyclosporine capsules, (modified), are at increased risk of developing bacterial, viral, fungal, and protozoal infections, including opportunistic infections. These infections may lead to serious, including fatal, outcomes (see BOXED WARNING, and ADVERSE REACTIONS).
Polyoma Virus Infections
Patients receiving immunosuppressants, including cyclosporine capsules, (modified), are at increased risk for opportunistic infections, including polyoma virus infections. Polyoma virus infections in transplant patients may have serious, and sometimes, fatal outcomes. These include cases of JC virus-associated progressive multifocal leukoencephalopathy (PML), and polyoma virus-associated nephropathy (PVAN), especially due to BK virus infection, which have been observed in patients receiving cyclosporine. PVAN is associated with serious outcomes, including deteriorating renal function and renal graft loss, (see ADVERSE REACTIONS, Postmarketing Experience, Kidney, Liver and Heart Transplantation).
Patient monitoring may help detect patients at risk for PVAN.
Cases of PML have been reported in patients treated with cyclosporine capsules, (modified). PML, which is sometimes fatal, commonly presents with hemiparesis, apathy, confusion, cognitive deficiencies and ataxia. Risk factors for PML include treatment with immunosuppressant therapies and impairment of immune function. In immunosuppressed patients, physicians should consider PML in the differential diagnosis in patients reporting neurological symptoms and consultation with a neurologist should be considered as clinically indicated.
Consideration should be given to reducing the total immunosuppression in transplant patients who develop PML or PVAN. However, reduced immunosuppression may place the graft at risk.
Neurotoxicity
There have been reports of convulsions in adult and pediatric patients receiving cyclosporine, particularly in combination with high dose methylprednisolone.
Encephalopathy, including Posterior Reversible Encephalopathy Syndrome (PRES), has been described both in post-marketing reports and in the literature. Manifestations include impaired consciousness, convulsions, visual disturbances (including blindness), loss of motor function, movement disorders and psychiatric disturbances. In many cases, changes in the white matter have been detected using imaging techniques and pathologic specimens. Predisposing factors such as hypertension, hypomagnesemia, hypocholesterolemia, high-dose corticosteroids, high cyclosporine blood concentrations, and graft-versus-host disease have been noted in many but not all of the reported cases. The changes in most cases have been reversible upon discontinuation of cyclosporine, and in some cases improvement was noted after reduction of dose. It appears that patients receiving liver transplant are more susceptible to encephalopathy than those receiving kidney transplant. Another rare manifestation of cyclosporine-induced neurotoxicity, occurring in transplant patients more frequently than in other indications, is optic disc edema including papilloedema, with possible visual impairment, secondary to benign intracranial hypertension.
Care should be taken in using cyclosporine with nephrotoxic drugs (see PRECAUTIONS).
Package Label.principal Display Panel
20 mg
50 mg
100 mg
Kidney, Liver, and Heart Transplantation
Cyclosporine capsules (modified) is indicated for the prophylaxis of organ rejection in kidney, liver, and heart allogeneic transplants. Cyclosporine capsules (modified) has been used in combination with azathioprine and corticosteroids.
Blood Concentration Monitoring in Transplant Patients
Transplant centers have found blood concentration monitoring of cyclosporine to be an essential component of patient management. Of importance to blood concentration analysis are the type of assay used, the transplanted organ, and other immunosuppressant agents being administered. While no fixed relationship has been established, blood concentration monitoring may assist in the clinical evaluation of rejection and toxicity, dose adjustments, and the assessment of compliance.
Various assays have been used to measure blood concentrations of cyclosporine. Older studies using a nonspecific assay often cited concentrations that were roughly twice those of the specific assays. Therefore, comparison between concentrations in the published literature and an individual patient concentration using current assays must be made with detailed knowledge of the assay methods employed. Current assay results are also not interchangeable and their use should be guided by their approved labeling. A discussion of the different assay methods is contained in Annals of Clinical Biochemistry 1994; 31:420 to 446. While several assays and assay matrices are available, there is a consensus that parent-compound-specific assays correlate best with clinical events. Of these, HPLC is the standard reference, but the monoclonal antibody RIAs and the monoclonal antibody FPIA offer sensitivity, reproducibility, and convenience. Most clinicians base their monitoring on trough cyclosporine concentrations. Applied Pharmacokinetics, Principles of Therapeutic Drug Monitoring (1992) contains a broad discussion of cyclosporine pharmacokinetics and drug monitoring techniques. Blood concentration monitoring is not a replacement for renal function monitoring or tissue biopsies.
Transplant Patients With Poor Absorption of Sandimmune
Patients with lower than expected cyclosporine blood trough concentrations in relation to the oral dose of Sandimmune may have poor or inconsistent absorption of cyclosporine from Sandimmune. After conversion to cyclosporine capsules, (modified), patients tend to have higher cyclosporine concentrations. Due to the increase in bioavailability of cyclosporine following conversion to cyclosporine capsules, (modified), the cyclosporine blood trough concentration may exceed the target range. Particular caution should be exercised when converting patients to cyclosporine capsules (modified) at doses greater than 10 mg/kg/day. The dose of cyclosporine capsules (modified) should be titrated individually based on cyclosporine trough concentrations, tolerability, and clinical response. In this population the cyclosporine blood trough concentration should be measured more frequently, at least twice a week (daily, if initial dose exceeds 10 mg/kg/day) until the concentration stabilizes within the desired range.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Carcinogenicity studies were carried out in male and female rats and mice. In the 78-week mouse study, evidence of a statistically significant trend was found for lymphocytic lymphomas in females, and the incidence of hepatocellular carcinomas in mid-dose (0.03 times the maximum recommended human dose (MRHD) based on body surface area (BSA) males significantly exceeded the control value. In the 24-month rat study, pancreatic islet cell adenomas significantly exceeded the control rate in the low dose level (0.006 times the MRHD based on BSA). The hepatocellular carcinomas and pancreatic islet cell adenomas were not dose related. Published reports indicate that co-treatment of hairless mice with UV irradiation and cyclosporine or other immunosuppressive agents shorten the time to skin tumor formation compared to UV irradiation alone.
Cyclosporine has not been found to be mutagenic/genotoxic in the Ames Test, the V79-HGPRT Test, the micronucleus test in mice and Chinese hamsters, the chromosome-aberration tests in Chinese hamster bone-marrow, the mouse dominant lethal assay, and the DNA-repair test in sperm from treated mice. A recent study analyzing sister chromatid exchange (SCE) induction by cyclosporine using human lymphocytes in vitro gave indication of a positive effect (i.e., induction of SCE), at high concentrations in this system.
In a fertility study in rats, increased perinatal mortality and impaired postnatal development of F1 pups were observed at 15 mg/kg/day (0.2 times the MRHD based on BSA). No adverse effects on fertility and reproduction were observed upto 5 mg/kg/day (0.06 times the MRHD based on BSA) in male and female rats.
Widely distributed papillomatosis of the skin was observed after chronic treatment of dogs with cyclosporine at 9 times the human initial psoriasis treatment dose of 2.5 mg/kg, where doses are expressed on a body surface area basis. This papillomatosis showed a spontaneous regression upon discontinuation of cyclosporine.
An increased incidence of malignancy is a recognized complication of immunosuppression in recipients of organ transplants and patients with rheumatoid arthritis and psoriasis. The most common forms of neoplasms are non-Hodgkin's lymphoma and carcinomas of the skin. The risk of malignancies in cyclosporine recipients is higher than in the normal, healthy population but similar to that in patients receiving other immunosuppressive therapies. Reduction or discontinuance of immunosuppression may cause the lesions to regress.
In psoriasis patients on cyclosporine, development of malignancies, especially those of the skin has been reported (see WARNINGS). Skin lesions not typical for psoriasis should be biopsied before starting cyclosporine treatment. Patients with malignant or premalignant changes of the skin should be treated with cyclosporine only after appropriate treatment of such lesions and if no other treatment option exists.
Postmarketing Experience, Kidney, Liver and Heart Transplantation
Hepatotoxicity
Cases of hepatotoxicity and liver injury including cholestasis, jaundice, hepatitis and liver failure; serious and/or fatal outcomes have been reported (see WARNINGS, Hepatotoxicity).
Increased Risk of Infections
Cases of JC virus-associated progressive multifocal leukoencephalopathy (PML), sometimes fatal; and polyoma virus-associated nephropathy (PVAN), especially BK virus resulting in graft loss have been reported (see WARNINGS, Polyoma Virus Infection)
Headache, including Migraine
Cases of migraine have been reported. In some cases, patients have been unable to continue cyclosporine, however, the final decision on treatment discontinuation should be made by the treating physician following the careful assessment of benefits versus risks.
Pain of lower extremities
Isolated cases of pain of lower extremities have been reported in association with cyclosporine. Pain of lower extremities has also been noted as part of Calcineurin-Inhibitor Induced Pain Syndrome (CIPS) as described in the literature.
Conversion From Sandimmune® to Cyclosporine Capsules (modified) in Transplant Patients
In transplanted patients who are considered for conversion to cyclosporine capsules (modified) from Sandimmune®, cyclosporine capsules (modified) should be started with the same daily dose as was previously used with Sandimmune® (1:1 dose conversion). The cyclosporine capsules (modified) dose should subsequently be adjusted to attain the pre-conversion cyclosporine blood trough concentration. Using the same trough concentration target range for cyclosporine capsules (modified) as for Sandimmune® results in greater cyclosporine exposure when cyclosporine capsules (modified) is administered (see Pharmacokinetics, Absorption). Patients with suspected poor absorption of Sandimmune® require different dosing strategies (see Transplant Patients with Poor absorption of Sandimmune ® , below). In some patients, the increase in blood trough concentration is more pronounced and may be of clinical significance.
Until the blood trough concentration attains the pre-conversion value, it is strongly recommended that the cyclosporine blood trough concentration be monitored every 4 to 7 days after conversion to cyclosporine capsules, (modified). In addition, clinical safety parameters such as serum creatinine and blood pressure should be monitored every two weeks during the first two months after conversion. If the blood trough concentrations are outside the desired range and/or if the clinical safety parameters worsen, the dosage of cyclosporine capsules (modified) must be adjusted accordingly.
Structured Label Content
Recent Major Changes (34066-1)
WARNING
Only physicians experienced in management of systemic immunosuppressive therapy for the indicated disease should prescribe cyclosporine capsules, (modified). At doses used in solid organ transplantation, only physicians experienced in immunosuppressive therapy and management of organ transplant recipients should prescribe cyclosporine capsules, (modified). Patients receiving the drug should be managed in facilities equipped and staffed with adequate laboratory and supportive medical resources. The physician responsible for maintenance therapy should have complete information requisite for the follow-up of the patient.
Cyclosporine capsules, (modified), a systemic immunosuppressant, may increase the susceptibility to infection and the development of neoplasia. In kidney, liver, and heart transplant patients cyclosporine capsules, (modified) may be administered with other immunosuppressive agents. Increased susceptibility to infection and the possible development of lymphoma and other neoplasms may result from the increase in the degree of immunosuppression in transplant patients.
Cyclosporine capsules, (modified) have increased bioavailability in comparison to Sandimmune®Soft Gelatin Capsules (cyclosporine capsules, USP) [NON-MODIFIED]. Cyclosporine capsules, (modified) and Sandimmune®are not bioequivalent and cannot be used interchangeably without physician supervision. For a given trough concentration, cyclosporine exposure will be greater with cyclosporine capsules, (modified) than with Sandimmune®. If a patient who is receiving exceptionally high doses of Sandimmune® is converted to cyclosporine capsules, (modified), particular caution should be exercised. Cyclosporine blood concentrations should be monitored in transplant and rheumatoid arthritis patients taking cyclosporine capsules, (modified) to avoid toxicity due to high concentrations. Dose adjustments should be made in transplant patients to minimize possible organ rejection due to low concentrations. Comparison of blood concentrations in the published literature with blood concentrations obtained using current assays must be done with detailed knowledge of the assay methods employed (see Dosage and Administration).
General
Cyclosporine capsules (modified) is contraindicated in patients with a hypersensitivity to cyclosporine or to any of the ingredients of the formulation.
Warnings (WARNINGS)
(See also BOXED WARNING)
Excretion
Only 0.1% of a cyclosporine dose is excreted unchanged in the urine. Elimination is primarily biliary with only 6% of the dose (parent drug and metabolites) excreted in the urine. Neither dialysis nor renal failure alters cyclosporine clearance significantly.
Pregnancy
Risk Summary
Available data from published literature, including the Transplant Pregnancy Registry International, observational cohort studies, case-controlled studies, meta-analysis, case series, and case reports, over decades of use with cyclosporine in pregnancy have not identified a drug associated risk of major birth defects, or miscarriage. Adverse maternal or fetal outcomes including hypertension, preeclampsia, preterm birth, and low birth weight are increased in patients treated with cyclosporine. However, patients receiving cyclosporine during pregnancy have underlying medical conditions and may be treated with concomitant medications that limit the interpretability of these findings (see Data).
Embryo-fetal developmental (EFD) studies in rats and rabbits with cyclosporine have shown embryo-fetal toxicity at dose levels below the maximum recommended human dose (MRHD) based on body surface area (BSA).
The alcohol content of cyclosporine capsules, (modified) should be taken into account when given to pregnant women (see WARNINGS, Special Excipients).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
Available data from the National Transplantation Pregnancy Registry (NTPR) including 622 pregnancies in renal, liver, and heart transplant recipients exposed to cyclosporine during pregnancy found that the overall rate of major birth defects, live birth rates, and miscarriage rates were comparable to the general population. Maternal and fetal adverse outcomes, including the rate of hypertension, preeclampsia, premature births, and low birth weight infants appear to be increased in transplant recipients treated with cyclosporine compared to the general population. However, these patients have underlying medical conditions that confound the above findings.
Animal Data
Animal studies have shown reproductive toxicity in rats and rabbits.
Three EFD studies (two oral and one intravenous) are available in rats. In two EFD studies, pregnant rats were orally administered with cyclosporine either at doses of 10, 17, 30, 100 and 300 mg/kg/day or 4, 10 and 25 mg/kg/day from gestation day (GD) 6 to 15 or from GD 7 to 17, respectively. Maternal toxicity characterized by mortality, clinical signs of toxicity and impaired body weight gain were observed at 30 mg/kg/day and above. Cyclosporine was embryo- and fetotoxic as indicated by increased embryonic mortality and reduced fetal weight together with skeletal retardations in rats at 25 mg/kg/day and above. In addition, ventricular septal defect was observed at 25 mg/kg/day in fetuses. In the first study, the oral no observed effect level (NOEL) for both dams and fetuses was 17 mg/kg/day (0.2 times the MRHD based on BSA). In the other oral study, the NOEL for dams and fetuses were 10 and 4 mg/kg/day (0.13 and 0.05 times the MRHD based on BSA), respectively. In the IV EFD study, rats were administered with 3, 6 and 12 mg/kg/day of cyclosporine from GD 7 to 17. An increase in post implantation loss was observed at 12 mg/kg/day; ventricular septal defect was observed at ≥ 6 mg/kg/day in fetuses. The IV NOEL for dams and fetus were 6 and 3 mg/kg/day (0.08 and 0.04 times the MRHD, respectively, based on BSA), respectively, after IV administration.
In rabbits, cyclosporine was orally administered at dose levels of 10, 30, 100 or 300 mg/kg/day from GD 6 to 18. At 100 mg/kg/day and above, reduction in body weight gain of dams and at 300 mg/kg/day abortions were observed. Maternal toxicity, embryo-fetotoxicity as indicated by increased pre- and postnatal mortality, reduced fetal weight together with skeletal retardations were observed at 100 mg/kg/day and above. The NOEL for dams and fetuses was 30 mg/kg/day (1 times the MRHD based on BSA).
In two published research studies, rabbits exposed to cyclosporine in utero (10 mg/kg/day subcutaneously) demonstrated reduced numbers of nephrons, renal hypertrophy, systemic hypertension and progressive renal insufficiency up to 35 weeks of age. These findings have not been demonstrated in other species and their relevance for humans is unknown.
In a peri- and postnatal development study in rats, pregnant rats were orally administered with cyclosporine (5, 15 or 45 mg/kg/day) from GD 15 until end of lactation. At 45 mg/kg/day (0.5 times the MRHD based on BSA), increased pre and postnatal mortality of offspring and reduced body weight gain of surviving pups were observed. Cyclosporine up to 15 mg/kg/day (0.2 times the MRHD based on BSA) had no effect on pregnancy, pre and postnatal development of offspring.
Psoriasis
Cyclosporine capsules (modified) is indicated for the treatment of adult, nonimmunocompromised patients with severe (i.e., extensive and/or disabling), recalcitrant, plaque psoriasis who have failed to respond to at least one systemic therapy (e.g., PUVA, retinoids, or methotrexate) or in patients for whom other systemic therapies are contraindicated, or cannot be tolerated.
While rebound rarely occurs, most patients will experience relapse with cyclosporine capsules (modified) as with other therapies upon cessation of treatment.
Absorption
Cyclosporine capsules (modified) has increased bioavailability compared to Sandimmune®. The absolute bioavailability of cyclosporine administered as Sandimmune® is dependent on the patient population, estimated to be less than 10% in liver transplant patients and as great as 89% in some renal transplant patients. The absolute bioavailability of cyclosporine administered as cyclosporine capsules (modified) has not been determined in adults. In studies of renal transplant, rheumatoid arthritis and psoriasis patients, the mean cyclosporine AUC was approximately 20% to 50% greater and the peak blood cyclosporine concentration (Cmax) was approximately 40% to 106% greater following administration of cyclosporine capsules (modified) compared to following administration of Sandimmune®. The dose normalized AUC in de novo liver transplant patients administered cyclosporine capsules (modified) 28 days after transplantation was 50% greater and Cmax was 90% greater than in those patients administered Sandimmune®. AUC and Cmax are also increased (cyclosporine capsules (modified) relative to Sandimmune®) in heart transplant patients, but data are very limited. Although the AUC and Cmax values are higher on cyclosporine capsules (modified) relative to Sandimmune®, the predose trough concentrations (dose-normalized) are similar for the two formulations.
Following oral administration of cyclosporine capsules, (modified), the time to peak blood cyclosporine concentrations (Tmax) ranged from 1.5 to 2.0 hours. The administration of food with cyclosporine capsules (modified) decreases the cyclosporine AUC and Cmax. A high fat meal (669 kcal, 45 grams fat) consumed within one-half hour before cyclosporine capsules (modified) administration decreased the AUC by 13% and Cmax by 33%. The effects of a low-fat meal (667 kcal, 15 grams fat) were similar.
The effect of T-tube diversion of bile on the absorption of cyclosporine from cyclosporine capsules (modified) was investigated in eleven de novo liver transplant patients. When the patients were administered cyclosporine capsules (modified) with and without T-tube diversion of bile, very little difference in absorption was observed, as measured by the change in maximal cyclosporine blood concentrations from pre-dose values with the T-tube closed relative to when it was open: 6.9 ± 41% (range 55% to 68%).
|
Pharmacokinetic Parameters (mean ± SD)
|
|||||||
|
Dose/day1
|
Dose/weight
|
AUC2
|
Cmax
|
Trough3
|
CL/F
|
CL/F
|
|
|
Patient Population
|
(mg/d)
|
(mg/kg/d)
|
(ng·hr/mL)
|
(ng/mL)
|
(ng/mL)
|
(mL/min)
|
(mL/min/kg)
|
| De novo renal transplant4
|
597 ± 174 |
7.95 ± 2.81 |
8772 ± 2089 |
1802 ± 428 |
361 ± 129 |
593 ± 204 |
7.8 ± 2.9 |
| Week 4 (N = 37) |
|||||||
| Stable renal transplant4
|
344 ± 122 |
4.10 ± 1.58 |
6035 ± 2194 |
1333 ± 469 |
251 ± 116 |
492 ± 140 |
5.9 ± 2.1 |
| (N = 55) |
|||||||
| De novo liver transplant5
|
458 ± 190 |
6.89 ± 3.68 |
7187 ± 2816 |
1555 ± 740 |
268 ± 101 |
577 ± 309 |
8.6 ± 5.7 |
| Week 4 (N = 18) |
|||||||
| De novo rheumatoid arthritis6
|
182 ± 55.6 |
2.37 ± 0.36 |
2641 ± 877 |
728 ± 263 |
96.4 ± 37.7 |
613 ± 196 |
8.3 ± 2.8 |
| (N = 23) |
|||||||
| De novo psoriasis6
|
189 ± 69.8 |
2.48 ± 0.65 |
2324 ± 1048 |
655 ± 186 |
74.9 ± 46.7 |
723 ± 186 |
10.2 ± 3.9 |
| Week 4 (N = 18) |
|||||||
|
1Total daily dose was divided into two doses administered every 12 hours. 2AUC was measured over one dosing interval. 3Trough concentration was measured just prior to the morning cyclosporine capsules (Modified) dose, approximately 12 hours after the previous dose. 4Assay: TDx specific monoclonal fluorescence polarization immunoassay. 5Assay: Cyclo-trac specific monoclonal radioimmunoassay. 6Assay: INCSTAR specific monoclonal radioimmunoassay. |
Metabolism
Cyclosporine is extensively metabolized by the cytochrome P-450 3A enzyme system in the liver, and to a lesser degree in the gastrointestinal tract, and the kidney. The metabolism of cyclosporine can be altered by the coadministration of a variety of agents (see PRECAUTIONS, Drug Interactions). At least 25 metabolites have been identified from human bile, feces, blood, and urine. The biological activity of the metabolites and their contributions to toxicity are considerably less than those of the parent compound. The major metabolites (M1, M9, and M4N) result from oxidation at the 1-beta, 9-gamma, and 4-N-demethylated positions, respectively. At steady state following the oral administration of Sandimmune®, the mean AUCs for blood concentrations of M1, M9, and M4N are about 70%, 21%, and 7.5% of the AUC for blood cyclosporine concentrations, respectively. Based on blood concentration data from stable renal transplant patients (13 patients administered cyclosporine capsules (modified) and Sandimmune® in a crossover study), and bile concentration data from de novo liver transplant patients (4 administered cyclosporine capsules, (modified), 3 administered Sandimmune®), the percentage of dose present as M1, M9, and M4N metabolites is similar when either cyclosporine capsules (modified) or Sandimmune® is administered.
Overdosage (OVERDOSAGE)
There is a minimal experience with cyclosporine overdosage. Forced emesis and gastric lavage can be of value up to 2 hours after administration of cyclosporine capsules, (modified). Transient hepatotoxicity and nephrotoxicity may occur which should resolve following drug withdrawal. Oral doses of cyclosporine up to 10 g (about 150 mg/kg) have been tolerated with relatively minor clinical consequences, such as vomiting, drowsiness, headache, tachycardia and, in a few patients, moderately severe, reversible impairment of renal function. However, serious symptoms of intoxication have been reported following accidental parenteral overdosage with cyclosporine in premature neonates. General supportive measures and symptomatic treatment should be followed in all cases of overdosage. Cyclosporine is not dialyzable to any great extent, nor is it cleared well by charcoal hemoperfusion. The oral dosage at which half of experimental animals are estimated to die is 31 times, 39 times, and > 54 times the human maintenance dose for transplant patients (6mg/kg; corrections based on body surface area) in mice, rats, and rabbits.
Description (DESCRIPTION)
Cyclosporine capsules, USP (modified) is an oral formulation of cyclosporine that immediately forms a microemulsion in an aqueous environment.
Cyclosporine, USP the active principle in cyclosporine capsules, USP (modified), is a cyclic polypeptide immunosuppressant agent consisting of 11 amino acids. It is produced as a metabolite by the fungus species Beauveria nivea.
Chemically, cyclosporine, USP is designated as [R-[R*,R*-(E)]]-cyclic-(L-alanyl-D-alanyl-N-methyl-L-leucyl-N-methyl-L-leucyl-N-methyl-L-valyl-3-hydroxy-N,4-dimethyl-L-2-amino-6-octenoyl-L-α -amino-butyryl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucyl).
Cyclosporine capsules, USP (modified) (Soft Gelatin Capsules) are available in 25 mg, 50 mg and 100 mg strengths.
Each 25 mg capsule contains:
Cyclosporine, USP………………………………………………………………………………25 mg
Dehydrated alcohol................................................. (9.5% w/v or 12.0% v/v)
Each 50 mg capsule contains:
Cyclosporine, USP……………………………………………………………………………...50 mg
Dehydrated alcohol................................................... (9.5% w/v or 12.0% v/v)
Each 100 mg capsule contains:
Cyclosporine, USP…………………………………………………………………………...100 mg
Dehydrated alcohol.................................................. (9.5% w/v or 12.0% v/v)
Inactive Ingredients: gelatin, glycerin, propylene glycol, titanium dioxide, ferric oxide black [25 mg and 100 mg], glyceryl monolinoleate, polyoxyl 40 hydrogenated castor oil, all-rac-alpha tocopherol [vitamin E synthetic], Ink contains- ammonium hydroxide 28%, iron oxide red, polyethylene glycol, polyvinyl acetate phthalate, propylene glycol.
The chemical structure of cyclosporine (also known as cyclosporin A) is:
All Patients
Cyclosporine, the active ingredient of cyclosporine capsules, (modified), can cause nephrotoxicity and hepatotoxicity. The risk increases with increasing doses of cyclosporine. Renal dysfunction including structural kidney damage is a potential consequence of cyclosporine capsules (modified) and therefore renal function must be monitored during therapy. Care should be taken in using cyclosporine with nephrotoxic drugs (see PRECAUTIONS).
Patients receiving cyclosporine capsules (modified) require frequent monitoring of serum creatinine (see Special Monitoring under DOSAGE AND ADMINISTRATION). Elderly patients should be monitored with particular care, since decreases in renal function also occur with age. If patients are not properly monitored and doses are not properly adjusted, cyclosporine therapy can be associated with the occurrence of structural kidney damage and persistent renal dysfunction.
An increase in serum creatinine and BUN may occur during cyclosporine capsules (modified) therapy and reflect a reduction in the glomerular filtration rate. Impaired renal function at any time requires close monitoring, and frequent dosage adjustment may be indicated. The frequency and severity of serum creatinine elevations increase with dose and duration of cyclosporine therapy. These elevations are likely to become more pronounced without dose reduction or discontinuation.
Because cyclosporine capsules (modified) is not bioequivalent to Sandimmune®, conversion from cyclosporine capsules (modified) to Sandimmune® using a 1:1 ratio (mg/kg/day) may result in lower cyclosporine blood concentrations. Conversion from cyclosporine capsules (modified) to Sandimmune® should be made with increased monitoring to avoid the potential of underdosing.
Distribution
Cyclosporine is distributed largely outside the blood volume. The steady state volume of distribution during intravenous dosing has been reported as 3 to 5 L/kg in solid organ transplant recipients. In blood, the distribution is concentration dependent. Approximately 33% to 47% is in plasma, 4% to 9% in lymphocytes, 5% to 12% in granulocytes, and 41% to 58% in erythrocytes. At high concentrations, the binding capacity of leukocytes and erythrocytes becomes saturated. In plasma, approximately 90% is bound to proteins, primarily lipoproteins. Cyclosporine is excreted in human milk (see PRECAUTIONS, Nursing Mothers).
How Supplied (HOW SUPPLIED)
Cyclosporine capsules, USP (modified) (Soft Gelatin Capsules)
25 mg
Blue gray colored opaque oval shaped soft gelatin capsule imprinted as 'S 25' in red color ink, containing clear to cloudy pale yellow colored oily liquid.
Packages of 30 unit-dose blisters (NDC 23155-837-30).
50 mg
White colored opaque oblong shaped soft gelatin capsule imprinted as 'S 50' in red color ink, containing clear to cloudy pale yellow colored oily liquid
Packages of 30 unit-dose blisters (NDC 23155-838-30).
100 mg
Blue gray colored opaque oblong shaped soft gelatin capsule imprinted as 'S 100' in red color ink, containing clear to cloudy pale yellow colored oily liquid.
Packages of 30 unit-dose blisters (NDC 23155-839-30).
Store and Dispense
In the original unit-dose container at controlled room temperature 68°F to 77°F (20°C to 25°C).
Cyclosporine Capsules, USP (MODIFIED) (Soft Gelatin Capsules)
All registered trademarks in this document are the property of their respective owners.
Manufactured by:
Strides Pharma Science Limited.
Bengaluru – 562106, India
Manufactured for:
Avet Pharmaceuticals Inc.
East Brunswick, NJ 08816
1.866.901.DRUG (3784)
Revised: 09/2022
Geriatric Use
In rheumatoid arthritis clinical trials with cyclosporine, 17.5% of patients were age 65 or older. These patients were more likely to develop systolic hypertension on therapy, and more likely to show serum creatinine rises ≥ 50% above the baseline after 3 to 4 months of therapy.
Clinical studies of cyclosporine capsules (modified) in transplant and psoriasis patients did not include a sufficient number of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experiences have not identified differences in response between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Pediatric Use
Although no adequate and well-controlled studies have been completed in children, transplant recipients as young as one year of age have received cyclosporine capsules (modified) with no unusual adverse effects. The safety and efficacy of cyclosporine capsules (modified) treatment in children with juvenile rheumatoid arthritis or psoriasis below the age of 18 have not been established.
Nursing Mothers
Risk Summary
Cyclosporine and its metabolites are present in human milk following oral and intravenous administration. Adverse effects on the breastfed infant have not been reported. There are no data on the effects of the drug on milk production. The alcohol content of cyclosporine capsules, (modified) should be taken into account when given to lactating women (see WARNINGS, Special Excipients). Lactating women are encouraged to avoid additional alcohol intake during treatment. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for cyclosporine capsules, (modified) and any potential adverse effects on the breastfed infant from cyclosporine capsules, (modified) or from the underlying maternal condition.
Laboratory Tests
In all patients treated with cyclosporine, renal and liver functions should be assessed repeatedly by measurement of serum creatinine, BUN, serum bilirubin, and liver enzymes. Serum lipids, magnesium, and potassium should also be monitored. Cyclosporine blood concentrations should be routinely monitored in transplant patients (see DOSAGE AND ADMINISTRATION, Blood Concentration Monitoring in Transplant Patients), and periodically monitored in rheumatoid arthritis patients.
Pharmacokinetics
The immunosuppressive activity of cyclosporine is primarily due to parent drug. Following oral administration, absorption of cyclosporine is incomplete. The extent of absorption of cyclosporine is dependent on the individual patient, the patient population, and the formulation. Elimination of cyclosporine is primarily biliary with only 6% of the dose (parent drug and metabolites) excreted in urine. The disposition of cyclosporine from blood is generally biphasic, with a terminal half-life of approximately 8.4 hours (range 5 to 18 hours). Following intravenous administration, the blood clearance of cyclosporine (assay: HPLC) is approximately 5 to 7 mL/min/kg in adult recipients of renal or liver allografts. Blood cyclosporine clearance appears to be slightly slower in cardiac transplant patients.
Cyclosporine capsules (MODIFIED) and cyclosporine oral solution (MODIFIED) are bioequivalent.
The relationship between administered dose and exposure (area under the concentration versus time curve, AUC) is linear within the therapeutic dose range. The intersubject variability (total, %CV) of cyclosporine exposure (AUC) when cyclosporine capsules, (modified) or Sandimmune® is administered ranges from approximately 20% to 50% in renal transplant patients. This intersubject variability contributes to the need for individualization of the dosing regimen for optimal therapy (see DOSAGE AND ADMINISTRATION). Intrasubject variability of AUC in renal transplant recipients (%CV) was 9% to 21% for cyclosporine capsules, (modified) and 19% to 26% for Sandimmune®. In the same studies, intrasubject variability of trough concentrations (%CV) was 17% to 30% for cyclosporine capsules, (modified) and 16% to 38% for Sandimmune®.
Drug Interactions
(See PRECAUTIONS, Drug Interactions) When diclofenac or methotrexate was coadministered with cyclosporine in rheumatoid arthritis patients, the AUC of diclofenac and methotrexate, each was significantly increased (see PRECAUTIONS, Drug Interactions). No clinically significant pharmacokinetic interactions occurred between cyclosporine and aspirin, ketoprofen, piroxicam, or indomethacin.
Specific Populations
Renal Impairment
In a study performed in 4 subjects with end-stage renal disease (creatinine clearance < 5 mL/min), an intravenous infusion of 3.5 mg/kg of cyclosporine over 4 hours administered at the end of a hemodialysis session resulted in a mean volume of distribution (Vdss) of 3.49 L/kg and systemic clearance (CL) of 0.369 L/hr/kg. This systemic CL (0.369 L/hr/kg) was approximately two thirds of the mean systemic CL (0.56 L/hr/kg) of cyclosporine in historical control subjects with normal renal function. In 5 liver transplant patients, the mean clearance of cyclosporine on and off hemodialysis was 463 mL/min and 398 mL/min, respectively. Less than 1% of the dose of cyclosporine was recovered in the dialysate.
Hepatic Impairment
Cyclosporine is extensively metabolized by the liver. Since severe hepatic impairment may result in significantly increased cyclosporine exposures, the dosage of cyclosporine may need to be reduced in these patients.
Pediatric Population
Pharmacokinetic data from pediatric patients administered cyclosporine capsules (modified) or Sandimmune® are very limited. In 15 renal transplant patients aged 3 to 16 years, cyclosporine whole blood clearance after IV administration of Sandimmune® was 10.6 ± 3.7 mL/min/kg (assay: Cyclo-trac specific RIA). In a study of 7 renal transplant patients aged 2 to 16, the cyclosporine clearance ranged from 9.8 to 15.5 mL/min/kg. In 9 liver transplant patients aged 0.6 to 5.6 years, clearance was 9.3 ± 5.4 mL/min/kg (assay: HPLC).
In the pediatric population, cyclosporine capsules (modified) also demonstrates an increased bioavailability as compared to Sandimmune®. In 7 liver de novo transplant patients aged 1.4 to 10 years, the absolute bioavailability of cyclosporine capsules (modified) was 43% (range 30% to 68%) and for Sandimmune® in the same individuals absolute bioavailability was 28% (range 17% to 42%).
|
Pediatric Pharmacokinetic Parameters (mean ± SD)
|
||||||
|
Dose/day
|
Dose/weight
|
AUC1
|
Cmax
|
CL/F
|
CL/F
|
|
|
Patient Population
|
(mg/d)
|
(mg/kg/d)
|
(ng·hr/mL)
|
(ng/mL)
|
(mL/min)
|
(mL/min/kg)
|
| Stable liver transplant2
|
||||||
| Age 2 to 8, Dosed TID (N = 9) |
101 ± 25 |
5.95 ± 1.32 |
2163 ± 801 |
629 ± 219 |
285 ± 94 |
16.6 ± 4.3 |
| Age 8 to 15, Dosed BID (N = 8) |
188 ± 55 |
4.96 ± 2.09 |
4272 ± 1462 |
975 ± 281 |
378 ± 80 |
10.2 ± 4.0 |
| Stable liver transplant3
|
||||||
| Age 3, Dosed BID (N = 1) |
120 |
8.33 |
5832 |
1050 |
171 |
11.9 |
| Age 8 to 15, Dosed BID (N = 5) |
158 ± 55 |
5.51 ± 1.91 |
4452 ± 2475 |
1013 ± 635 |
328 ± 121 |
11.0 ± 1.9 |
| Stable renal transplant3
|
||||||
| Age 7 to 15, Dosed BID (N = 5) |
328 ± 83 |
7.37 ± 4.11 |
6922 ± 1988 |
1827 ± 487 |
418 ± 143 |
8.7 ± 2.9 |
|
1AUC was measured over one dosing interval. 2Assay: Cyclo-trac specific monoclonal radioimmunoassay. 3Assay: TDx specific monoclonal fluorescence polarization immunoassay. |
Geriatric Population
Comparison of single dose data from both normal elderly volunteers (N = 18, mean age 69 years) and elderly rheumatoid arthritis patients (N = 16, mean age 68 years) to single dose data in young adult volunteers (N = 16, mean age 26 years) showed no significant difference in the pharmacokinetic parameters.
Rheumatoid Arthritis
The effectiveness of Sandimmune® and cyclosporine capsules (modified) in the treatment of severe rheumatoid arthritis was evaluated in 5 clinical studies involving a total of 728 cyclosporine treated patients and 273 placebo treated patients.
A summary of the results is presented for the "responder" rates per treatment group, with a responder being defined as a patient having completed the trial with a 20% improvement in the tender and the swollen joint count and a 20% improvement in 2 of 4 of investigator global, patient global, disability, and erythrocyte sedimentation rates (ESR) for the Studies 651 and 652 and 3 of 5 of investigator global, patient global, disability, visual analog pain, and ESR for Studies 2008, 654 and 302.
Study 651 enrolled 264 patients with active rheumatoid arthritis with at least 20 involved joints, who had failed at least one major RA drug, using a 3:3:2 randomization to one of the following three groups: (1) cyclosporine dosed at 2.5 to 5 mg/kg/day, (2) methotrexate at 7.5 to 15 mg/week, or (3) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 3.1 mg/kg/day. See Graph below.
Study 652 enrolled 250 patients with active RA with > 6 active painful or tender joints who had failed at least one major RA drug. Patients were randomized using a 3:3:2 randomization to 1 of 3 treatment arms: (1) 1.5 to 5 mg/kg/day of cyclosporine, (2) 2.5 to 5 mg/kg/day of cyclosporine, and (3) placebo. Treatment duration was 16 weeks. The mean cyclosporine dose for group 2 at the last visit was 2.92 mg/kg/day. See Graph below.
Study 2008 enrolled 144 patients with active RA and > 6 active joints who had unsuccessful treatment courses of aspirin and gold or Penicillamine. Patients were randomized to 1 of 2 treatment groups (1) cyclosporine 2.5 to 5 mg/kg/day with adjustments after the first month to achieve a target trough level and (2) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 3.63 mg/kg/day. See Graph below.
Study 654 enrolled 148 patients who remained with active joint counts of 6 or more despite treatment with maximally tolerated methotrexate doses for at least three months. Patients continued to take their current dose of methotrexate and were randomized to receive, in addition, one of the following medications: (1) cyclosporine 2.5 mg/kg/day with dose increases of 0.5 mg/kg/day at weeks 2 and 4 if there was no evidence of toxicity and further increases of 0.5 mg/kg/day at weeks 8 and 16 if a < 30% decrease in active joint count occurred without any significant toxicity; dose decreases could be made at any time for toxicity or (2) placebo. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 2.8 mg/kg/day (range: 1.3 to 4.1). See Graph below.
Study 302 enrolled 299 patients with severe active RA, 99% of whom were unresponsive or intolerant to at least one prior major RA drug. Patients were randomized to 1 of 2 treatment groups (1) cyclosporine capsules (modified) and (2) cyclosporine, both of which were started at 2.5 mg/kg/day and increased after 4 weeks for inefficacy in increments of 0.5 mg/kg/day to a maximum of 5 mg/kg/day and decreased at any time for toxicity. Treatment duration was 24 weeks. The mean cyclosporine dose at the last visit was 2.91 mg/kg/day (range: 0.72 to 5.17) for cyclosporine capsules (modified) and 3.27 mg/kg/day (range: 0.73 to 5.68) for cyclosporine. See Graph below.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Cyclosporine is a potent immunosuppressive agent that in animals prolongs survival of allogeneic transplants involving skin, kidney, liver, heart, pancreas, bone marrow, small intestine, and lung. Cyclosporine has been demonstrated to suppress some humoral immunity and to a greater extent, cell-mediated immune reactions such as allograft rejection, delayed hypersensitivity, experimental allergic encephalomyelitis, Freund's adjuvant arthritis, and graft versus host disease in many animal species for a variety of organs.
The effectiveness of cyclosporine results from specific and reversible inhibition of immunocompetent lymphocytes in the G0- and G1-phase of the cell cycle. T-lymphocytes are preferentially inhibited. The T-helper cell is the main target, although the T-suppressor cell may also be suppressed. Cyclosporine also inhibits lymphokine production and release including interleukin-2.
No effects on phagocytic function (changes in enzyme secretions, chemotactic migration of granulocytes, macrophage migration, carbon clearance in vivo) have been detected in animals. Cyclosporine does not cause bone marrow suppression in animal models or man.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Cyclosporine capsules (modified) has increased bioavailability in comparison to Sandimmune ® . Cyclosporine capsules (modified) and Sandimmune ® are not bioequivalent and cannot be used interchangeably without physician supervision.
The daily dose of cyclosporine capsules (modified) should always be given in two divided doses (BID). It is recommended that cyclosporine capsules (modified) be administered on a consistent schedule with regard to time of day and relation to meals. Grapefruit and grapefruit juice affect metabolism, increasing blood concentration of cyclosporine, thus should be avoided.
Specific Populations
Renal Impairment in Kidney, Liver, and Heart Transplantation
Cyclosporine undergoes minimal renal elimination and its pharmacokinetics do not appear to be significantly altered in patients with end-stage renal disease who receive routine hemodialysis treatments (see CLINICAL PHARMACOLOGY). However, due to its nephrotoxic potential (see WARNINGS), careful monitoring of renal function is recommended; cyclosporine dosage should be reduced if indicated (see WARNINGS and PRECAUTIONS).
Renal Impairment in Rheumatoid Arthritis and Psoriasis
Patients with impaired renal function should not receive cyclosporine (see CONTRAINDICATIONS, WARNINGS and PRECAUTIONS).
Hepatic Impairment
The clearance of cyclosporine may be significantly reduced in severe liver disease patients (See CLINICAL PHARMACOLOGY). Dose reduction may be necessary in patients with severe liver impairment to maintain blood concentrations within the recommended target range (see WARNINGS and PRECAUTIONS).
Newly Transplanted Patients
The initial oral dose of cyclosporine capsules (modified) can be given 4 to 12 hours prior to transplantation or be given postoperatively. The initial dose of cyclosporine capsules (modified) varies depending on the transplanted organ and the other immunosuppressive agents included in the immunosuppressive protocol. In newly transplanted patients, the initial oral dose of cyclosporine capsules (modified) is the same as the initial oral dose of Sandimmune®. Suggested initial doses are available from the results of a 1994 survey of the use of Sandimmune® in US transplant centers. The mean ± SD initial doses were 9 ± 3 mg/kg/day for renal transplant patients (75 centers), 8 ± 4 mg/kg/day for liver transplant patients (30 centers), and 7 ± 3 mg/kg/day for heart transplant patients (24 centers). Total daily doses were divided into two equal daily doses. The cyclosporine capsules (modified) dose is subsequently adjusted to achieve a pre-defined cyclosporine blood concentration (see Blood Concentration Monitoring in Transplant Patients, below). If cyclosporine trough blood concentrations are used, the target range is the same for cyclosporine capsules (modified) as for Sandimmune®. Using the same trough concentration target range for cyclosporine capsules (modified) as for Sandimmune® results in greater cyclosporine exposure when cyclosporine capsules (modified) is administered (see Pharmacokinetics, Absorption). Dosing should be titrated based on clinical assessments of rejection and tolerability. Lower cyclosporine capsules (modified) doses may be sufficient as maintenance therapy.
Adjunct therapy with adrenal corticosteroids is recommended initially. Different tapering dosage schedules of prednisone appear to achieve similar results. A representative dosage schedule based on the patient's weight started with 2.0 mg/kg/day for the first 4 days tapered to 1.0 mg/kg/day by 1 week, 0.6 mg/kg/day by 2 weeks, 0.3 mg/kg/day by 1 month, and 0.15 mg/kg/day by 2 months and thereafter as a maintenance dose. Steroid doses may be further tapered on an individualized basis depending on status of patient and function of graft. Adjustments in dosage of prednisone must be made according to the clinical situation.
Kidney, Liver, and Heart Transplant
Nephrotoxicity
Cyclosporine, the active ingredient of cyclosporine capsules, (modified), can cause nephrotoxicity and hepatotoxicity when used in high doses. It is not unusual for serum creatinine and BUN levels to be elevated during cyclosporine therapy. These elevations in renal transplant patients do not necessarily indicate rejection, and each patient must be fully evaluated before dosage adjustment is initiated.
Based on the historical Sandimmune ® experience with oral solution, nephrotoxicity associated with cyclosporine had been noted in 25% of cases of renal transplantation, 38% of cases of cardiac transplantation, and 37% of cases of liver transplantation. Mild nephrotoxicity was generally noted 2 to 3 months after renal transplant and consisted of an arrest in the fall of the pre-operative elevations of BUN and creatinine at a range of 35 to 45 mg/dL and 2.0 to 2.5 mg/dL, respectively. These elevations were often responsive to cyclosporine dosage reduction.
More overt nephrotoxicity was seen early after transplantation and was characterized by a rapidly rising BUN and creatinine. Since these events are similar to renal rejection episodes, care must be taken to differentiate between them. This form of nephrotoxicity is usually responsive to cyclosporine dosage reduction.
Although specific diagnostic criteria which reliably differentiate renal graft rejection from drug toxicity have not been found, a number of parameters have been significantly associated with one or the other. It should be noted however, that up to 20% of patients may have simultaneous nephrotoxicity and rejection.
|
Nephrotoxicity vs. Rejection
|
||
|
Parameter
|
Nephrotoxicity
|
Rejection
|
| History |
Donor >50 years old or hypotensive Prolonged kidney preservation Prolonged anastomosis time Concomitant nephrotoxic drugs |
Anti-donor immune response Retransplant patient |
| Clinical |
Often >6 weeks postopb
Prolonged initial nonfunction (acute tubular necrosis) |
Often < 4 weeks postopb
Fever > 37.5°C Weight gain > 0.5 kg Graft swelling and tenderness Decrease in daily urine volume > 500 mL (or 50%) |
| Laboratory |
CyA serum trough level > 200 ng/mL Gradual rise in Cr (<0.15 mg/dL/day)a Cr plateau < 25% above baseline BUN/Cr ≥ 20 |
CyA serum trough level < 150 ng/mL Rapid rise in Cr (> 0.3 mg/dL/day)a Cr > 25% above baseline BUN/Cr < 20 |
| Biopsy |
Arteriolopathy (medial hypertrophy a, hyalinosis, nodular deposits, intimal thickening, endothelial vacuolization, progressive scarring) Tubular atrophy, isometric vacuolization, isolated calcifications Minimal edema Mild focal infiltratesc |
Endovasculitisc (proliferationa, intimal arteritisb, necrosis, sclerosis) Tubulitis with RBCb and WBCb casts, some irregular vacuolization Interstitial edemac and hemorrhageb Diffuse moderate to severe mononuclear infiltratesd |
| Aspiration Cytology |
Diffuse interstitial fibrosis, often striped form CyA deposits in tubular and endothelial cells Fine isometric vacuolization of tubular cells |
Glomerulitis (mononuclear cells)c
Inflammatory infiltrate with mononuclear phagocytes, macrophages, lymphoblastoid cells, and activated T-cells These strongly express HLA-DR antigens |
| Urine Cytology |
Tubular cells with vacuolization and granularization |
Degenerative tubular cells, plasma cells, and lymphocyturia > 20% of sediment |
| Manometry Ultrasonography |
Intracapsular pressure < 40 mm Hgb
Unchanged graft cross sectional area |
Intracapsular pressure > 40 mm Hgb
Increase in graft cross sectional area AP diameter ≥ Transverse diameter |
| Magnetic Resonance Imagery |
Normal appearance |
Loss of distinct corticomedullary junction, swelling image intensity of parachyma approaching that of psoas, loss of hilar fat |
| Radionuclide Scan |
Normal or generally decreased perfusion Decrease in tubular function (131 I-hippuran) > decrease in perfusion (99m Tc DTPA) |
Patchy arterial flow Decrease in perfusion > decrease in tubular function Increased uptake of Indium 111 labeled platelets or Tc-99m in colloid |
| Therapy |
Responds to decreased cyclosporine |
Responds to increased steroids or antilymphocyte globulin |
|
ap < 0.05, bp < 0.01, cp < 0.001, dp < 0.0001 |
A form of a cyclosporine-associated nephropathy is characterized by serial deterioration in renal function and morphologic changes in the kidneys. From 5% to 15% of transplant recipients who have received cyclosporine will fail to show a reduction in rising serum creatinine despite a decrease or discontinuation of cyclosporine therapy. Renal biopsies from these patients will demonstrate one or several of the following alterations: tubular vacuolization, tubular microcalcifications, peritubular capillary congestion, arteriolopathy, and a striped form of interstitial fibrosis with tubular atrophy. Though none of these morphologic changes is entirely specific, a diagnosis of cyclosporine-associated structural nephrotoxicity requires evidence of these findings.
When considering the development of cyclosporine-associated nephropathy, it is noteworthy that several authors have reported an association between the appearance of interstitial fibrosis and higher cumulative doses or persistently high circulating trough concentrations of cyclosporine. This is particularly true during the first 6 post-transplant months when the dosage tends to be highest and when, in kidney recipients, the organ appears to be most vulnerable to the toxic effects of cyclosporine. Among other contributing factors to the development of interstitial fibrosis in these patients are prolonged perfusion time, warm ischemia time, as well as episodes of acute toxicity, and acute and chronic rejection. The reversibility of interstitial fibrosis and its correlation to renal function have not yet been determined. Reversibility of arteriolopathy has been reported after stopping cyclosporine or lowering the dosage.
Impaired renal function at any time requires close monitoring, and frequent dosage adjustment may be indicated.
In the event of severe and unremitting rejection, when rescue therapy with pulse steroids and monoclonal antibodies fail to reverse the rejection episode, it may be preferable to switch to alternative immunosuppressive therapy rather than increase the cyclosporine capsules (modified) dose to excessive blood concentrations.
Due to the potential for additive or synergistic impairment of renal function, caution should be exercised when coadministering cyclosporine capsules (modified) with other drugs that may impair renal function (see PRECAUTIONS, Drug Interactions).
Thrombotic Microangiopathy
Occasionally patients have developed a syndrome of thrombocytopenia and microangiopathic hemolytic anemia which may result in graft failure. The vasculopathy can occur in the absence of rejection and is accompanied by avid platelet consumption within the graft as demonstrated by Indium 111 labeled platelet studies. Neither the pathogenesis nor the management of this syndrome is clear. Though resolution has occurred after reduction or discontinuation of cyclosporine and 1) administration of streptokinase and heparin or 2) plasmapheresis, this appears to depend upon early detection with Indium 111 labeled platelet scans (see ADVERSE REACTIONS).
Hyperkalemia
Significant hyperkalemia (sometimes associated with hyperchloremic metabolic acidosis) and hyperuricemia have been seen occasionally in individual patients.
Hepatotoxicity
Cases of hepatotoxicity and liver injury including cholestasis, jaundice, hepatitis, and liver failure, have been reported in patients treated with cyclosporine. Most reports included patients with significant co-morbidities, underlying conditions and other confounding factors including infectious complications and comedications with hepatotoxic potential. In some cases, mainly in transplant patients, fatal outcomes have been reported (see ADVERSE REACTIONS, Postmarketing Experience, Kidney, Liver and Heart Transplantation).
Hepatotoxicity, usually manifested by elevations in hepatic enzymes and bilirubin, was reported in patients treated with cyclosporine in clinical trials: 4% in renal transplantation, 7% in cardiac transplantation, and 4% in liver transplantation. This was usually noted during the first month of therapy when high doses of cyclosporine were used. The chemistry elevations usually decreased with a reduction in dosage.
Malignancies
As in patients receiving other immunosuppressants, those patients receiving cyclosporine are at increased risk for development of lymphomas and other malignancies, particularly those of the skin. Patients taking cyclosporine should be warned to avoid excess ultraviolet light exposure. The increased risk appears related to the intensity and duration of immunosuppression rather than to the use of specific agents. Because of the danger of oversuppression of the immune system resulting in increased risk of infection or malignancy, a treatment regimen containing multiple immunosuppressants should be used with caution. Some malignancies may be fatal. Transplant patients receiving cyclosporine are at increased risk for serious infection with fatal outcome.
Serious Infections
Patients receiving immunosuppressants, including cyclosporine capsules, (modified), are at increased risk of developing bacterial, viral, fungal, and protozoal infections, including opportunistic infections. These infections may lead to serious, including fatal, outcomes (see BOXED WARNING, and ADVERSE REACTIONS).
Polyoma Virus Infections
Patients receiving immunosuppressants, including cyclosporine capsules, (modified), are at increased risk for opportunistic infections, including polyoma virus infections. Polyoma virus infections in transplant patients may have serious, and sometimes, fatal outcomes. These include cases of JC virus-associated progressive multifocal leukoencephalopathy (PML), and polyoma virus-associated nephropathy (PVAN), especially due to BK virus infection, which have been observed in patients receiving cyclosporine. PVAN is associated with serious outcomes, including deteriorating renal function and renal graft loss, (see ADVERSE REACTIONS, Postmarketing Experience, Kidney, Liver and Heart Transplantation).
Patient monitoring may help detect patients at risk for PVAN.
Cases of PML have been reported in patients treated with cyclosporine capsules, (modified). PML, which is sometimes fatal, commonly presents with hemiparesis, apathy, confusion, cognitive deficiencies and ataxia. Risk factors for PML include treatment with immunosuppressant therapies and impairment of immune function. In immunosuppressed patients, physicians should consider PML in the differential diagnosis in patients reporting neurological symptoms and consultation with a neurologist should be considered as clinically indicated.
Consideration should be given to reducing the total immunosuppression in transplant patients who develop PML or PVAN. However, reduced immunosuppression may place the graft at risk.
Neurotoxicity
There have been reports of convulsions in adult and pediatric patients receiving cyclosporine, particularly in combination with high dose methylprednisolone.
Encephalopathy, including Posterior Reversible Encephalopathy Syndrome (PRES), has been described both in post-marketing reports and in the literature. Manifestations include impaired consciousness, convulsions, visual disturbances (including blindness), loss of motor function, movement disorders and psychiatric disturbances. In many cases, changes in the white matter have been detected using imaging techniques and pathologic specimens. Predisposing factors such as hypertension, hypomagnesemia, hypocholesterolemia, high-dose corticosteroids, high cyclosporine blood concentrations, and graft-versus-host disease have been noted in many but not all of the reported cases. The changes in most cases have been reversible upon discontinuation of cyclosporine, and in some cases improvement was noted after reduction of dose. It appears that patients receiving liver transplant are more susceptible to encephalopathy than those receiving kidney transplant. Another rare manifestation of cyclosporine-induced neurotoxicity, occurring in transplant patients more frequently than in other indications, is optic disc edema including papilloedema, with possible visual impairment, secondary to benign intracranial hypertension.
Care should be taken in using cyclosporine with nephrotoxic drugs (see PRECAUTIONS).
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
20 mg
50 mg
100 mg
Kidney, Liver, and Heart Transplantation
Cyclosporine capsules (modified) is indicated for the prophylaxis of organ rejection in kidney, liver, and heart allogeneic transplants. Cyclosporine capsules (modified) has been used in combination with azathioprine and corticosteroids.
Blood Concentration Monitoring in Transplant Patients
Transplant centers have found blood concentration monitoring of cyclosporine to be an essential component of patient management. Of importance to blood concentration analysis are the type of assay used, the transplanted organ, and other immunosuppressant agents being administered. While no fixed relationship has been established, blood concentration monitoring may assist in the clinical evaluation of rejection and toxicity, dose adjustments, and the assessment of compliance.
Various assays have been used to measure blood concentrations of cyclosporine. Older studies using a nonspecific assay often cited concentrations that were roughly twice those of the specific assays. Therefore, comparison between concentrations in the published literature and an individual patient concentration using current assays must be made with detailed knowledge of the assay methods employed. Current assay results are also not interchangeable and their use should be guided by their approved labeling. A discussion of the different assay methods is contained in Annals of Clinical Biochemistry 1994; 31:420 to 446. While several assays and assay matrices are available, there is a consensus that parent-compound-specific assays correlate best with clinical events. Of these, HPLC is the standard reference, but the monoclonal antibody RIAs and the monoclonal antibody FPIA offer sensitivity, reproducibility, and convenience. Most clinicians base their monitoring on trough cyclosporine concentrations. Applied Pharmacokinetics, Principles of Therapeutic Drug Monitoring (1992) contains a broad discussion of cyclosporine pharmacokinetics and drug monitoring techniques. Blood concentration monitoring is not a replacement for renal function monitoring or tissue biopsies.
Transplant Patients With Poor Absorption of Sandimmune (Transplant Patients with Poor Absorption of Sandimmune)
Patients with lower than expected cyclosporine blood trough concentrations in relation to the oral dose of Sandimmune may have poor or inconsistent absorption of cyclosporine from Sandimmune. After conversion to cyclosporine capsules, (modified), patients tend to have higher cyclosporine concentrations. Due to the increase in bioavailability of cyclosporine following conversion to cyclosporine capsules, (modified), the cyclosporine blood trough concentration may exceed the target range. Particular caution should be exercised when converting patients to cyclosporine capsules (modified) at doses greater than 10 mg/kg/day. The dose of cyclosporine capsules (modified) should be titrated individually based on cyclosporine trough concentrations, tolerability, and clinical response. In this population the cyclosporine blood trough concentration should be measured more frequently, at least twice a week (daily, if initial dose exceeds 10 mg/kg/day) until the concentration stabilizes within the desired range.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Carcinogenicity studies were carried out in male and female rats and mice. In the 78-week mouse study, evidence of a statistically significant trend was found for lymphocytic lymphomas in females, and the incidence of hepatocellular carcinomas in mid-dose (0.03 times the maximum recommended human dose (MRHD) based on body surface area (BSA) males significantly exceeded the control value. In the 24-month rat study, pancreatic islet cell adenomas significantly exceeded the control rate in the low dose level (0.006 times the MRHD based on BSA). The hepatocellular carcinomas and pancreatic islet cell adenomas were not dose related. Published reports indicate that co-treatment of hairless mice with UV irradiation and cyclosporine or other immunosuppressive agents shorten the time to skin tumor formation compared to UV irradiation alone.
Cyclosporine has not been found to be mutagenic/genotoxic in the Ames Test, the V79-HGPRT Test, the micronucleus test in mice and Chinese hamsters, the chromosome-aberration tests in Chinese hamster bone-marrow, the mouse dominant lethal assay, and the DNA-repair test in sperm from treated mice. A recent study analyzing sister chromatid exchange (SCE) induction by cyclosporine using human lymphocytes in vitro gave indication of a positive effect (i.e., induction of SCE), at high concentrations in this system.
In a fertility study in rats, increased perinatal mortality and impaired postnatal development of F1 pups were observed at 15 mg/kg/day (0.2 times the MRHD based on BSA). No adverse effects on fertility and reproduction were observed upto 5 mg/kg/day (0.06 times the MRHD based on BSA) in male and female rats.
Widely distributed papillomatosis of the skin was observed after chronic treatment of dogs with cyclosporine at 9 times the human initial psoriasis treatment dose of 2.5 mg/kg, where doses are expressed on a body surface area basis. This papillomatosis showed a spontaneous regression upon discontinuation of cyclosporine.
An increased incidence of malignancy is a recognized complication of immunosuppression in recipients of organ transplants and patients with rheumatoid arthritis and psoriasis. The most common forms of neoplasms are non-Hodgkin's lymphoma and carcinomas of the skin. The risk of malignancies in cyclosporine recipients is higher than in the normal, healthy population but similar to that in patients receiving other immunosuppressive therapies. Reduction or discontinuance of immunosuppression may cause the lesions to regress.
In psoriasis patients on cyclosporine, development of malignancies, especially those of the skin has been reported (see WARNINGS). Skin lesions not typical for psoriasis should be biopsied before starting cyclosporine treatment. Patients with malignant or premalignant changes of the skin should be treated with cyclosporine only after appropriate treatment of such lesions and if no other treatment option exists.
Postmarketing Experience, Kidney, Liver and Heart Transplantation
Hepatotoxicity
Cases of hepatotoxicity and liver injury including cholestasis, jaundice, hepatitis and liver failure; serious and/or fatal outcomes have been reported (see WARNINGS, Hepatotoxicity).
Increased Risk of Infections
Cases of JC virus-associated progressive multifocal leukoencephalopathy (PML), sometimes fatal; and polyoma virus-associated nephropathy (PVAN), especially BK virus resulting in graft loss have been reported (see WARNINGS, Polyoma Virus Infection)
Headache, including Migraine
Cases of migraine have been reported. In some cases, patients have been unable to continue cyclosporine, however, the final decision on treatment discontinuation should be made by the treating physician following the careful assessment of benefits versus risks.
Pain of lower extremities
Isolated cases of pain of lower extremities have been reported in association with cyclosporine. Pain of lower extremities has also been noted as part of Calcineurin-Inhibitor Induced Pain Syndrome (CIPS) as described in the literature.
Conversion From Sandimmune® to Cyclosporine Capsules (modified) in Transplant Patients (Conversion from Sandimmune® to cyclosporine capsules (modified) in Transplant Patients)
In transplanted patients who are considered for conversion to cyclosporine capsules (modified) from Sandimmune®, cyclosporine capsules (modified) should be started with the same daily dose as was previously used with Sandimmune® (1:1 dose conversion). The cyclosporine capsules (modified) dose should subsequently be adjusted to attain the pre-conversion cyclosporine blood trough concentration. Using the same trough concentration target range for cyclosporine capsules (modified) as for Sandimmune® results in greater cyclosporine exposure when cyclosporine capsules (modified) is administered (see Pharmacokinetics, Absorption). Patients with suspected poor absorption of Sandimmune® require different dosing strategies (see Transplant Patients with Poor absorption of Sandimmune ® , below). In some patients, the increase in blood trough concentration is more pronounced and may be of clinical significance.
Until the blood trough concentration attains the pre-conversion value, it is strongly recommended that the cyclosporine blood trough concentration be monitored every 4 to 7 days after conversion to cyclosporine capsules, (modified). In addition, clinical safety parameters such as serum creatinine and blood pressure should be monitored every two weeks during the first two months after conversion. If the blood trough concentrations are outside the desired range and/or if the clinical safety parameters worsen, the dosage of cyclosporine capsules (modified) must be adjusted accordingly.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:46.381257 · Updated: 2026-03-14T22:38:36.946565