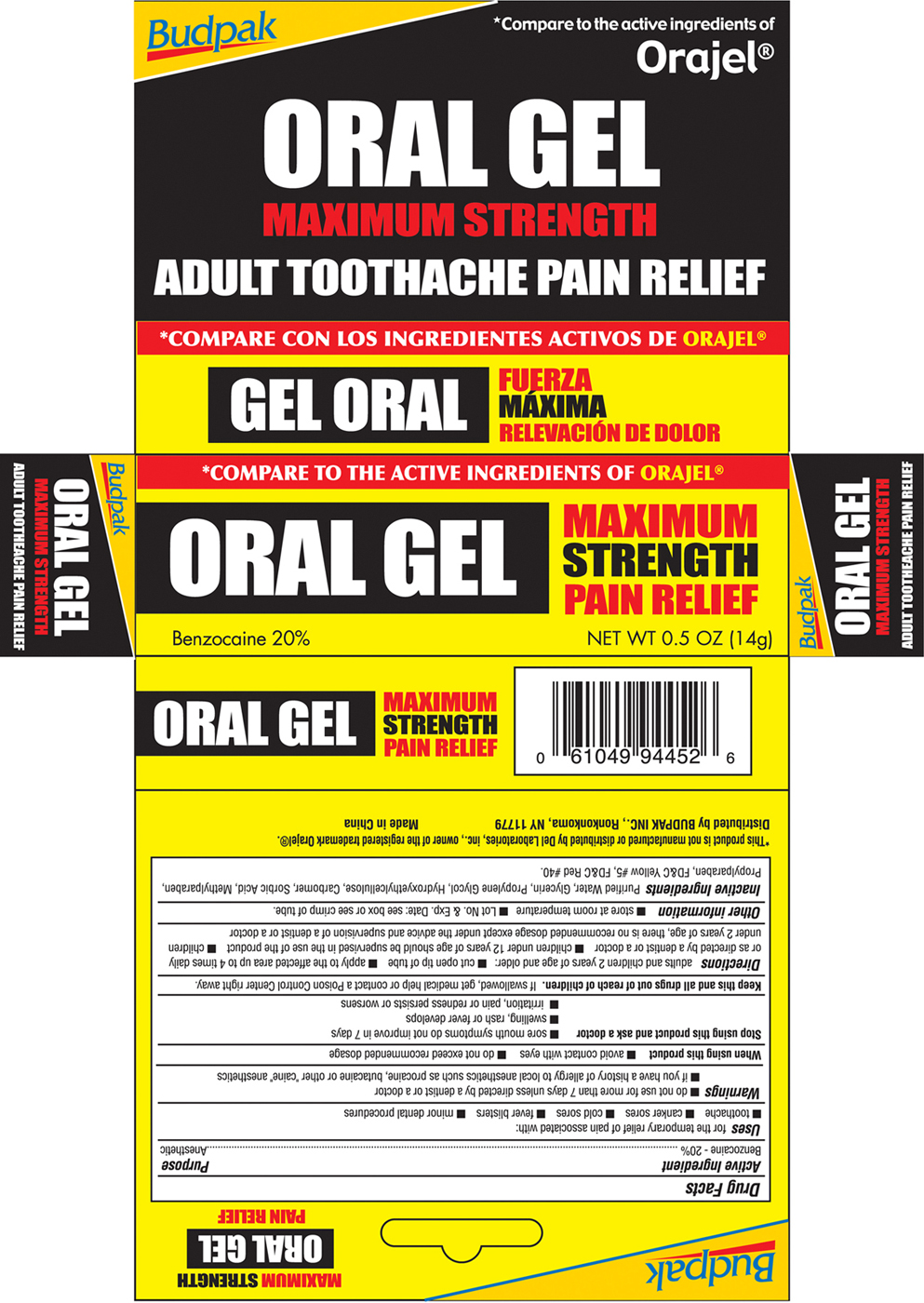
Drug Facts
194150a3-7492-4d7d-84e2-5b4297000eae
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient Benzocaine 20%
Medication Information
Warnings and Precautions
- do not use for more than 7 days unless directed by a dentist or a doctor
- if you have a history of allergy to local anesthetics such as procaine, butacaine or other "caine" anesthetics
Indications and Usage
- toothache
- canker sores
- cold sores
- fever blisters
- minor dental procedures
Dosage and Administration
- cut open tip of tube
- apply to the affected area up to 4 times daily or as directed by a dentist or doctor
- children under 12 years of age should be supervised in the use of the product
- children under 2 years of age, there is no recommended dosage except under the advice and supervision of a dentist or a doctor
Description
Active Ingredient Benzocaine 20%
Section 42229-5
- store at room temperature
- Lot No. and Exp. Date: see box or see crimp of tube
Section 50565-1
Section 50566-9
- sore mouth symptoms do not improve in 7 days
- swelling, rash or fever develops
- irritation, pain or redness persists or worsens
Section 50567-7
- avoid contact with the eyes
- do not exceed recommended dosage
Section 51727-6
Section 51945-4
Section 55105-1
Anesthetic
Section 55106-9
Benzocaine 20%
Structured Label Content
Indications and Usage (34067-9)
- toothache
- canker sores
- cold sores
- fever blisters
- minor dental procedures
Dosage and Administration (34068-7)
- cut open tip of tube
- apply to the affected area up to 4 times daily or as directed by a dentist or doctor
- children under 12 years of age should be supervised in the use of the product
- children under 2 years of age, there is no recommended dosage except under the advice and supervision of a dentist or a doctor
Warnings and Precautions (34071-1)
- do not use for more than 7 days unless directed by a dentist or a doctor
- if you have a history of allergy to local anesthetics such as procaine, butacaine or other "caine" anesthetics
Section 42229-5 (42229-5)
- store at room temperature
- Lot No. and Exp. Date: see box or see crimp of tube
Section 50565-1 (50565-1)
Section 50566-9 (50566-9)
- sore mouth symptoms do not improve in 7 days
- swelling, rash or fever develops
- irritation, pain or redness persists or worsens
Section 50567-7 (50567-7)
- avoid contact with the eyes
- do not exceed recommended dosage
Section 51727-6 (51727-6)
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Anesthetic
Section 55106-9 (55106-9)
Benzocaine 20%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:00.080680 · Updated: 2026-03-14T22:52:17.029964