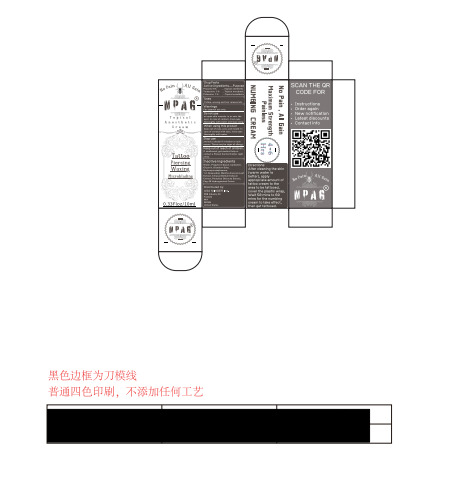
Npag Tattoo Numbing Cream
1929a6cc-75f0-4d20-e063-6394a90a0892
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
PROCAINE 5% TETRACAINE 1% PRILOCAINE 1%
Purpose
Topical anesthetic
Medication Information
Purpose
Topical anesthetic
Description
PROCAINE 5% TETRACAINE 1% PRILOCAINE 1%
Use
Tattoo, waxing and hair removal etc.
Section 50565-1
If swallowed, get medical help or contact a Poison C ontrol Center right
away.
Section 50566-9
and ask a doctor if irritation or rash occurs. These may be signs of allergy.
Section 50567-7
keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes
thoroughly with water.
Warnings
For external use only.
Directions
After cleaning the skin [warm water is better), apply appropriate amount of
tattoo cream to the area to be tattooed, cover the plastic wrap,
Wait 50 mins to 60 mins for the numbing cream to take effect,
then get tattooed.
Do Not Use
on open skin wounds. in or near the eyes. In case of contact, rinse eyes
thoroughly with water and stop using.
Other Information
No data available
Active Ingredient(s)
PROCAINE 5%
TETRACAINE 1%
PRILOCAINE 1%
Inactive Ingredients
Water, Propylene Glycol, Carbomer, Glycerin, Disodium Edta,
Hydroxyacetophenone, 1 ,2- Hexanediol, Mentha Arvensis Leaf
Extract, Chrysanthellum Indicum Extract, Portulaca Oleracea Extract,
Peg- 40 Hydrogenated Castor.
Package Label Principal Display Panel
Structured Label Content
Use
Tattoo, waxing and hair removal etc.
Section 50565-1 (50565-1)
If swallowed, get medical help or contact a Poison C ontrol Center right
away.
Section 50566-9 (50566-9)
and ask a doctor if irritation or rash occurs. These may be signs of allergy.
Section 50567-7 (50567-7)
keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes
thoroughly with water.
Purpose
Topical anesthetic
Warnings
For external use only.
Directions
After cleaning the skin [warm water is better), apply appropriate amount of
tattoo cream to the area to be tattooed, cover the plastic wrap,
Wait 50 mins to 60 mins for the numbing cream to take effect,
then get tattooed.
Do Not Use (Do not use)
on open skin wounds. in or near the eyes. In case of contact, rinse eyes
thoroughly with water and stop using.
Other Information (Other information)
No data available
Active Ingredient(s)
PROCAINE 5%
TETRACAINE 1%
PRILOCAINE 1%
Inactive Ingredients (Inactive ingredients)
Water, Propylene Glycol, Carbomer, Glycerin, Disodium Edta,
Hydroxyacetophenone, 1 ,2- Hexanediol, Mentha Arvensis Leaf
Extract, Chrysanthellum Indicum Extract, Portulaca Oleracea Extract,
Peg- 40 Hydrogenated Castor.
Package Label Principal Display Panel (Package Label - Principal Display Panel)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:35.344097 · Updated: 2026-03-14T23:07:36.685051