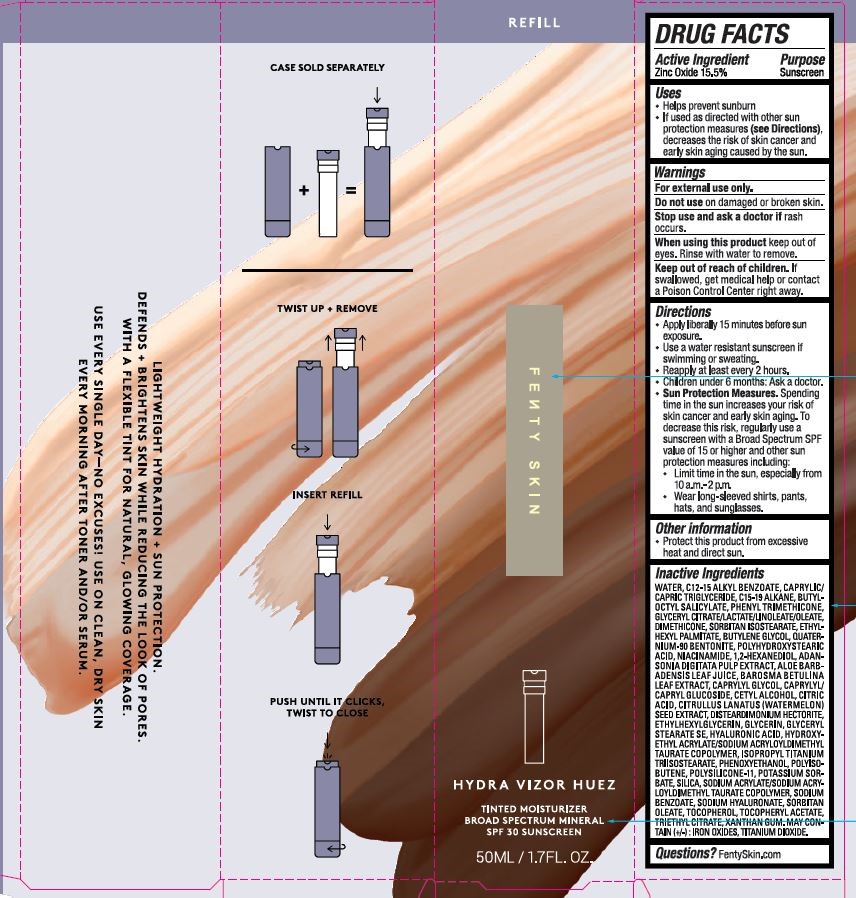
Kendo - Hydra Vizor Huez Tinted Moisturizer Broad Spectrum Spf 30
18fb7472-746f-339a-e063-6294a90a267d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ZINC OXIDE 15.5%
Purpose
SUNSCREEN
Medication Information
Purpose
SUNSCREEN
Description
ZINC OXIDE 15.5%
Uses
HELPS PREVENT SUNBURN
IF USED AS DIRECTED WITH OTHER SUN PROTECTION MEASURES (SEE DIRECTIONS), DECREASES THE RISK OF SKIN CANCER AND EARLY SKIN AGING CAUSED BY THE SUN.
Section 50565-1
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Section 51945-4
Warnings
FOR EXTERNAL USE ONLY.
DO NOT USE ON DAMAGED OR BROKEN SKIN.
STOP USE AND ASK A DOCTOR IF RASH OCCURS.
WHEN USING THIS PRODUCT KEEP OUT OF EYES. RINSE WITH WATER TO REMOVE.
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Directions
DIRECTIONS
APPLY LIBERALLY 15 MINUTES BEFORE SUN EXPOSURE.
USE A WATER RESISTANT SUNSCREEN IF SWIMMING OR SWEATING.
REAPPLY AT LEAST EVERY 2 HOURS.
CHILDREN UNDER 6 MONTHS: ASK A DOCTOR.
SUN PROTECTION MEASURES. SPENDING TIME IN THE SUN INCREASES YOUR RISK OF SKIN CANCER AND EARLY SKIN AGING. TO DECREASE THIS RISK, REGULARLY USE A SUNCREEN WITH A BROAD SPECTRUM SPF VALUE OF 15 OR HIGHER AND OTHER SUN PROTECTION MEASURES INCLUDING:
LIMIT TIME IN THE SUN, ESPECIALLY FROM 10 A.M. - 2 P.M.
WEAR LONG=SLEEVED SHIRTS, PANTS, HATS, AND SUNGLASSES.
Questions?
FENTYSKIN.COM
Active Ingredient
ZINC OXIDE 15.5%
Other Information
PROTECT THIS PRODUCT FROM EXCESSIVE HEAT AND DIRECT SUN.
Inactive Ingredients
WATER, C12-15 ALKYL BENZOATE, CAPRYLIC/CAPRIC TRIGLYCERIDE, C15-19 ALKANE, BUTYLOCTYL SALICYLATE, PHENYL TRIMETHICONE, GLYCERYL CITRATE/LACTATE/LINOLEATE/OLEATE, DIMETHICONE, SORBITAN ISOSTEARATE, ETHYLHEXYL PALMITATE, BUTYLENE GLYCOL, QUATERNIUM-90 BENTONITE, POLYHYDROXYSTEARIC ACID, NIACINAMIDE, 1,2-HEXANEDIOL, ADANSONIA DIGITATA PULP EXTRACT, ALOE BARBADENSIS LEAF JUICE, BAROSMA BETULINA LEAF EXTRACT, CAPRYLYL GLYCOL, CAPRYLYL/CAPRYL GLUCOSIDE, CETYL ALCOHOL, CITRIC ACID, CITRULLUS LANATUS (WATERMELON) SEED EXTRACT, DISTEARDIMONIUM HECTORITE, ETHYLHEXYLGLYCERIN, GLYCERIN, GLYCERYL STEARATE SE, HYALURONIC ACID, HYDROXYETHYL ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER, ISOPROPYL TITANIUM TRIISOSTEARATE, PHENOXYETHANOL, POLYISOBUTENE, POLYSILICONE-11, POTASSIUM SORBATE, SILICA, SODIUM ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER, SODIUM BENZOATE, SODIUM HYALURONATE, SORBITAN OLEATE, TOCOPHEROL, TOCOPHERYL ACETATE, TRIETHYL CITRATE, XANTHAN GUM. MAY CONTAIN (+/-): IRON OXIDES,TITANIUM DIOXIDE.
Structured Label Content
Uses (USES)
HELPS PREVENT SUNBURN
IF USED AS DIRECTED WITH OTHER SUN PROTECTION MEASURES (SEE DIRECTIONS), DECREASES THE RISK OF SKIN CANCER AND EARLY SKIN AGING CAUSED BY THE SUN.
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Section 51945-4 (51945-4)
Purpose (PURPOSE)
SUNSCREEN
Warnings (WARNINGS)
FOR EXTERNAL USE ONLY.
DO NOT USE ON DAMAGED OR BROKEN SKIN.
STOP USE AND ASK A DOCTOR IF RASH OCCURS.
WHEN USING THIS PRODUCT KEEP OUT OF EYES. RINSE WITH WATER TO REMOVE.
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Directions (DIRECTIONS)
DIRECTIONS
APPLY LIBERALLY 15 MINUTES BEFORE SUN EXPOSURE.
USE A WATER RESISTANT SUNSCREEN IF SWIMMING OR SWEATING.
REAPPLY AT LEAST EVERY 2 HOURS.
CHILDREN UNDER 6 MONTHS: ASK A DOCTOR.
SUN PROTECTION MEASURES. SPENDING TIME IN THE SUN INCREASES YOUR RISK OF SKIN CANCER AND EARLY SKIN AGING. TO DECREASE THIS RISK, REGULARLY USE A SUNCREEN WITH A BROAD SPECTRUM SPF VALUE OF 15 OR HIGHER AND OTHER SUN PROTECTION MEASURES INCLUDING:
LIMIT TIME IN THE SUN, ESPECIALLY FROM 10 A.M. - 2 P.M.
WEAR LONG=SLEEVED SHIRTS, PANTS, HATS, AND SUNGLASSES.
Questions? (QUESTIONS?)
FENTYSKIN.COM
Active Ingredient (ACTIVE INGREDIENT)
ZINC OXIDE 15.5%
Other Information (OTHER INFORMATION)
PROTECT THIS PRODUCT FROM EXCESSIVE HEAT AND DIRECT SUN.
Inactive Ingredients (INACTIVE INGREDIENTS)
WATER, C12-15 ALKYL BENZOATE, CAPRYLIC/CAPRIC TRIGLYCERIDE, C15-19 ALKANE, BUTYLOCTYL SALICYLATE, PHENYL TRIMETHICONE, GLYCERYL CITRATE/LACTATE/LINOLEATE/OLEATE, DIMETHICONE, SORBITAN ISOSTEARATE, ETHYLHEXYL PALMITATE, BUTYLENE GLYCOL, QUATERNIUM-90 BENTONITE, POLYHYDROXYSTEARIC ACID, NIACINAMIDE, 1,2-HEXANEDIOL, ADANSONIA DIGITATA PULP EXTRACT, ALOE BARBADENSIS LEAF JUICE, BAROSMA BETULINA LEAF EXTRACT, CAPRYLYL GLYCOL, CAPRYLYL/CAPRYL GLUCOSIDE, CETYL ALCOHOL, CITRIC ACID, CITRULLUS LANATUS (WATERMELON) SEED EXTRACT, DISTEARDIMONIUM HECTORITE, ETHYLHEXYLGLYCERIN, GLYCERIN, GLYCERYL STEARATE SE, HYALURONIC ACID, HYDROXYETHYL ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER, ISOPROPYL TITANIUM TRIISOSTEARATE, PHENOXYETHANOL, POLYISOBUTENE, POLYSILICONE-11, POTASSIUM SORBATE, SILICA, SODIUM ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER, SODIUM BENZOATE, SODIUM HYALURONATE, SORBITAN OLEATE, TOCOPHEROL, TOCOPHERYL ACETATE, TRIETHYL CITRATE, XANTHAN GUM. MAY CONTAIN (+/-): IRON OXIDES,TITANIUM DIOXIDE.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:16.518453 · Updated: 2026-03-14T23:12:26.991151