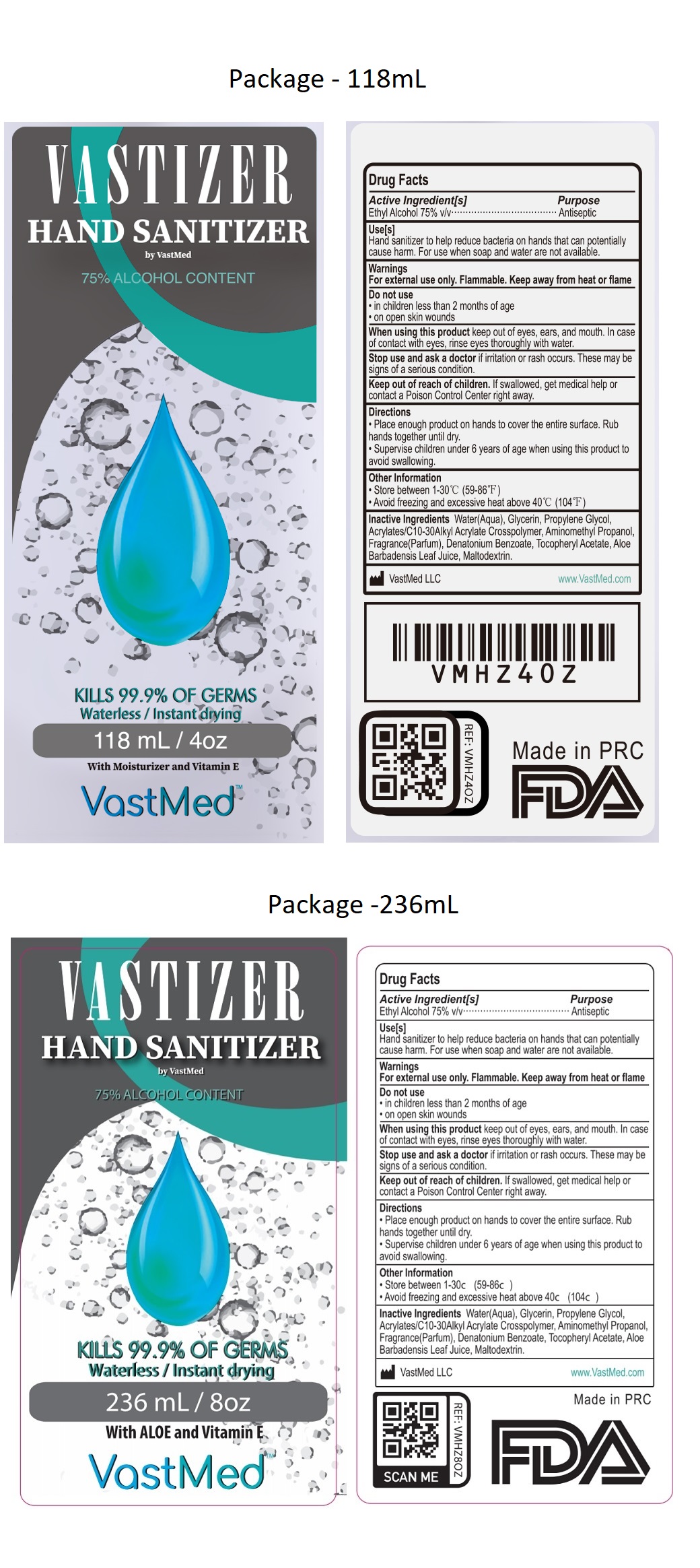
Vastizer Hand Sanitizer
18adf861-b1a5-40e5-a2bf-7e7cf132cb99
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl Alcohol 75% v/v
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Ethyl Alcohol 75% v/v
Use[s]
Hand sanitizer to help reduce bacteria on hands that can potentially cause harm. For use when soap and water are not available.
Section 42229-5
by VastMed
75% ALCOHOL CONTENT
KILLS 99.9% OF GERMS
Waterless / Instant drying
With ALOE and Vitamin E
VastMed LLC www.VastMed.com
Made in PRC
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only. Flammable. Keep away from heat or flame.
Do not use
• in children less than 2 months of age
• on open skin wounds
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Packaging
Directions
• Place enough product on hands to cover the entire surface. Rub hands together until dry.
• Supervise children under 6 years of age when using this product to avoid swallowing.
Drug Facts
Other Information
• Store between 1-30c (59-86c)
• Avoid freezing and excessive heat above 40c (104c)
Active Ingredient[s]
Ethyl Alcohol 75% v/v
Inactive Ingredients
Water(Aqua), Glycerin, Propylene Glycol, Acrylates/C10-30Alkyl Acrylate Crosspolymer, Aminomethyl Propanol, Fragrance(Parfum), Denatonium Benzoate, Tocopheryl Acetate, Aloe Barbadensis Leaf Juice, Maltodextrin.
Structured Label Content
Use[s]
Hand sanitizer to help reduce bacteria on hands that can potentially cause harm. For use when soap and water are not available.
Section 42229-5 (42229-5)
by VastMed
75% ALCOHOL CONTENT
KILLS 99.9% OF GERMS
Waterless / Instant drying
With ALOE and Vitamin E
VastMed LLC www.VastMed.com
Made in PRC
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Antiseptic
Warnings
For external use only. Flammable. Keep away from heat or flame.
Do not use
• in children less than 2 months of age
• on open skin wounds
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Packaging
Directions
• Place enough product on hands to cover the entire surface. Rub hands together until dry.
• Supervise children under 6 years of age when using this product to avoid swallowing.
Drug Facts
Other Information
• Store between 1-30c (59-86c)
• Avoid freezing and excessive heat above 40c (104c)
Active Ingredient[s]
Ethyl Alcohol 75% v/v
Inactive Ingredients
Water(Aqua), Glycerin, Propylene Glycol, Acrylates/C10-30Alkyl Acrylate Crosspolymer, Aminomethyl Propanol, Fragrance(Parfum), Denatonium Benzoate, Tocopheryl Acetate, Aloe Barbadensis Leaf Juice, Maltodextrin.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:12.673944 · Updated: 2026-03-14T23:00:34.965652