Povidone Iodine Topical Solution Usp
181a6363-2249-45f0-a28b-79f2c5ffad59
34390-5
HUMAN OTC DRUG LABEL
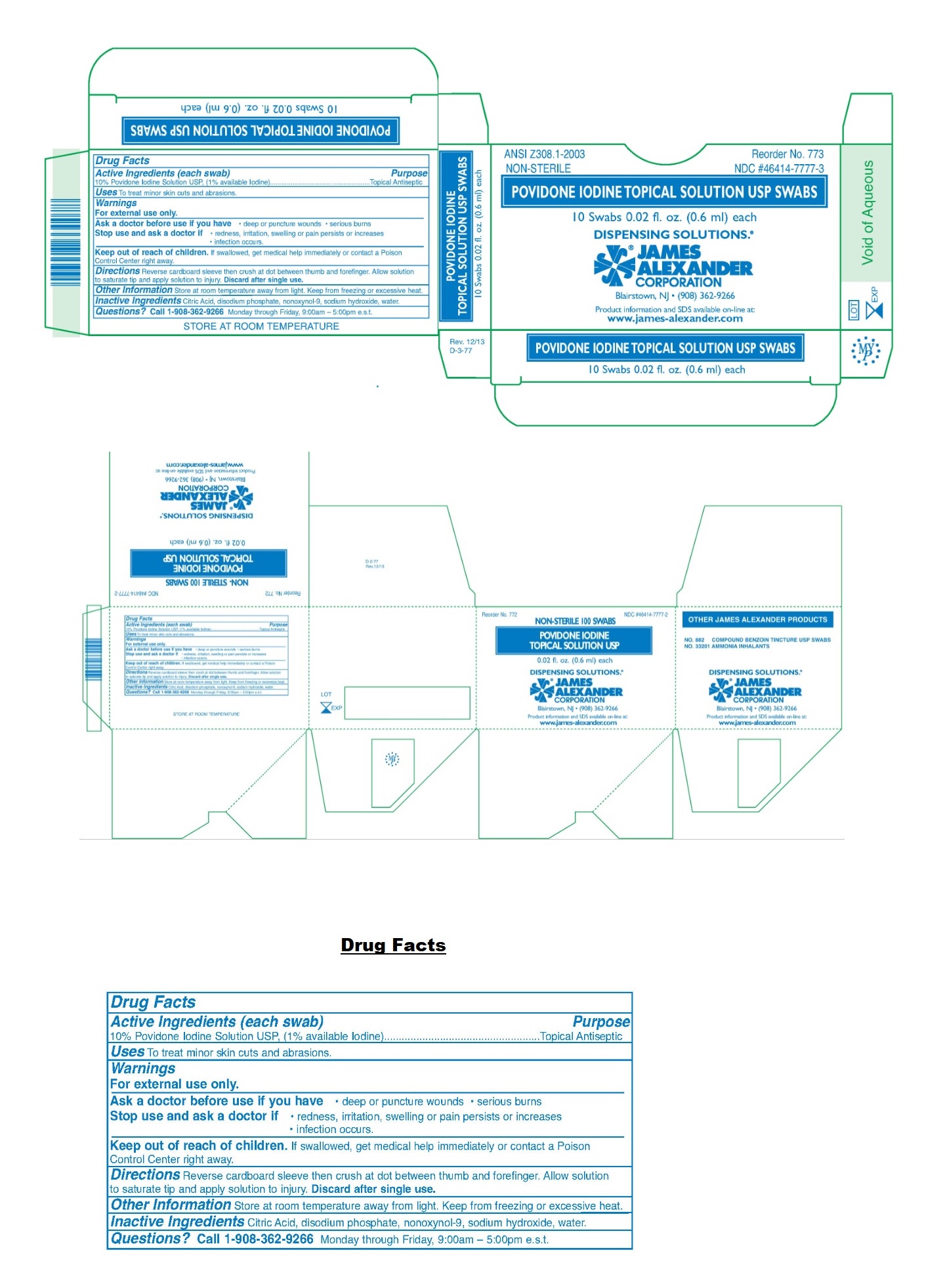
Drug Facts
Composition & Product
Identifiers & Packaging
Description
10% Povidone Iodine Solution USP, (1% available Iodine)
Purpose
Topical Antiseptic
Medication Information
Purpose
Topical Antiseptic
Description
10% Povidone Iodine Solution USP, (1% available Iodine)
Uses
To treat minor skin cuts and abrasions.
Section 42229-5
DISPENSING SOLUTIONS®
JAMES ALEXANDER CORPORATION
Blairstown, NJ• (908) 362-9266
Product information and MSDS available on-line at:
www.james-alexander.com
Void of Aqueous
Section 50565-1
Keep out of reach of children. If swallowed, get medical help immediately or contact a Poison Control Center right away.
Warnings
For external use only.
Ask a doctor before use if you have • deep or puncture wounds • serious burns
Stop use and ask a doctor if • redness, irritation, swelling or pain persists or increases
• infection occurs.
Packaging
Directions
Reverse cardboard sleeve then crush at dot between thumb and forefinger. Allow solution to saturate tip and apply solution to injury. Discard after single use.
Questions?
Call 1-908-362-9266 Monday through Friday, 9:00am - 5:00pm e.s.t.
Other Information
Store at room temperature away from light. Keep from freezing or excessive heat.
Inactive Ingredients
Citric acid, disodium phosphate, nonoxynol-9, sodium hydroxide, water.
Active Ingredients (each Swab)
10% Povidone Iodine Solution USP, (1% available Iodine)
Structured Label Content
Uses
To treat minor skin cuts and abrasions.
Section 42229-5 (42229-5)
DISPENSING SOLUTIONS®
JAMES ALEXANDER CORPORATION
Blairstown, NJ• (908) 362-9266
Product information and MSDS available on-line at:
www.james-alexander.com
Void of Aqueous
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help immediately or contact a Poison Control Center right away.
Purpose
Topical Antiseptic
Warnings
For external use only.
Ask a doctor before use if you have • deep or puncture wounds • serious burns
Stop use and ask a doctor if • redness, irritation, swelling or pain persists or increases
• infection occurs.
Packaging
Directions
Reverse cardboard sleeve then crush at dot between thumb and forefinger. Allow solution to saturate tip and apply solution to injury. Discard after single use.
Questions?
Call 1-908-362-9266 Monday through Friday, 9:00am - 5:00pm e.s.t.
Other Information
Store at room temperature away from light. Keep from freezing or excessive heat.
Inactive Ingredients
Citric acid, disodium phosphate, nonoxynol-9, sodium hydroxide, water.
Active Ingredients (each Swab) (Active Ingredients (each swab))
10% Povidone Iodine Solution USP, (1% available Iodine)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:50.536005 · Updated: 2026-03-14T23:13:07.898715