Perjeta
17f85d17-ab71-4f5b-9fe3-0b8c822f69ff
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
PERJETA is a HER2/neu receptor antagonist indicated for: Use in combination with trastuzumab and docetaxel for treatment of adults with HER2-positive metastatic breast cancer (MBC) who have not received prior anti-HER2 therapy or chemotherapy for metastatic disease. ( 1.1 ) Use in combination with trastuzumab and chemotherapy as neoadjuvant treatment of adults with HER2-positive, locally advanced, inflammatory, or early stage breast cancer (either greater than 2 cm in diameter or node positive) as part of a complete treatment regimen for early breast cancer. ( 1.2 , 2.2 , 14.2 ) adjuvant treatment of adults with HER2-positive early breast cancer at high risk of recurrence ( 1.2 , 2.2 , 14.3 )
Dosage and Administration
For intravenous infusion only. Do not administer as an intravenous push or bolus. ( 2.5 ) HER2 testing: Perform using FDA-approved tests by laboratories with demonstrated proficiency. ( 2.1 ) The initial PERJETA dose is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks thereafter by 420 mg administered as a 30 to 60 minute intravenous infusion. ( 2.3 ) MBC: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and docetaxel every 3 weeks. ( 2.3 ) Neoadjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy preoperatively every 3 weeks for 3 to 6 cycles. ( 2.3 ) Adjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy postoperatively every 3 weeks for a total of 1 year (up to 18 cycles). ( 2.3 )
Contraindications
PERJETA is contraindicated in patients with known hypersensitivity to pertuzumab or to any of its excipients [see Warnings and Precautions (5.4)].
Warnings and Precautions
Infusion-Related Reactions: PERJETA can cause serious infusion reactions, including fatal events: Monitor for signs and symptoms. If a significant infusion-associated reaction occurs, slow or interrupt the infusion and administer appropriate medical therapies. ( 5.3 ) Hypersensitivity Reactions/Anaphylaxis: PERJETA can cause hypersensitivity reactions, including anaphylaxis: Monitor for signs and symptoms, including angioedema. If a severe hypersensitivity reaction/anaphylaxis occurs, discontinue the infusion immediately and administer appropriate medical therapies. ( 5.4 )
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label: Left Ventricular Dysfunction [see Warnings and Precautions (5.1) ] Embryo-Fetal Toxicity [see Warnings and Precautions (5.2) ] Infusion-Related Reactions [see Warnings and Precautions (5.3) ] Hypersensitivity Reactions/Anaphylaxis [see Warnings and Precautions (5.4) ]
Description
Left Ventricular Dysfunction: PERJETA can cause subclinical and clinical cardiac failure manifesting as decreased LVEF and CHF. Evaluate cardiac function prior to and during treatment. Discontinue PERJETA treatment for a confirmed clinically significant decrease in left ventricular function [see Dosage and Administration (2.3) , Warnings and Precautions (5.1) and Adverse Reactions (6.1) ]. Embryo-fetal Toxicity: Exposure to PERJETA can cause embryo-fetal death and birth defects. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1) (8.3) ].
Medication Information
Warnings and Precautions
Infusion-Related Reactions: PERJETA can cause serious infusion reactions, including fatal events: Monitor for signs and symptoms. If a significant infusion-associated reaction occurs, slow or interrupt the infusion and administer appropriate medical therapies. ( 5.3 ) Hypersensitivity Reactions/Anaphylaxis: PERJETA can cause hypersensitivity reactions, including anaphylaxis: Monitor for signs and symptoms, including angioedema. If a severe hypersensitivity reaction/anaphylaxis occurs, discontinue the infusion immediately and administer appropriate medical therapies. ( 5.4 )
Indications and Usage
PERJETA is a HER2/neu receptor antagonist indicated for: Use in combination with trastuzumab and docetaxel for treatment of adults with HER2-positive metastatic breast cancer (MBC) who have not received prior anti-HER2 therapy or chemotherapy for metastatic disease. ( 1.1 ) Use in combination with trastuzumab and chemotherapy as neoadjuvant treatment of adults with HER2-positive, locally advanced, inflammatory, or early stage breast cancer (either greater than 2 cm in diameter or node positive) as part of a complete treatment regimen for early breast cancer. ( 1.2 , 2.2 , 14.2 ) adjuvant treatment of adults with HER2-positive early breast cancer at high risk of recurrence ( 1.2 , 2.2 , 14.3 )
Dosage and Administration
For intravenous infusion only. Do not administer as an intravenous push or bolus. ( 2.5 ) HER2 testing: Perform using FDA-approved tests by laboratories with demonstrated proficiency. ( 2.1 ) The initial PERJETA dose is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks thereafter by 420 mg administered as a 30 to 60 minute intravenous infusion. ( 2.3 ) MBC: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and docetaxel every 3 weeks. ( 2.3 ) Neoadjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy preoperatively every 3 weeks for 3 to 6 cycles. ( 2.3 ) Adjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy postoperatively every 3 weeks for a total of 1 year (up to 18 cycles). ( 2.3 )
Contraindications
PERJETA is contraindicated in patients with known hypersensitivity to pertuzumab or to any of its excipients [see Warnings and Precautions (5.4)].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label: Left Ventricular Dysfunction [see Warnings and Precautions (5.1) ] Embryo-Fetal Toxicity [see Warnings and Precautions (5.2) ] Infusion-Related Reactions [see Warnings and Precautions (5.3) ] Hypersensitivity Reactions/Anaphylaxis [see Warnings and Precautions (5.4) ]
Description
Left Ventricular Dysfunction: PERJETA can cause subclinical and clinical cardiac failure manifesting as decreased LVEF and CHF. Evaluate cardiac function prior to and during treatment. Discontinue PERJETA treatment for a confirmed clinically significant decrease in left ventricular function [see Dosage and Administration (2.3) , Warnings and Precautions (5.1) and Adverse Reactions (6.1) ]. Embryo-fetal Toxicity: Exposure to PERJETA can cause embryo-fetal death and birth defects. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1) (8.3) ].
Section 42229-5
Metastatic Breast Cancer (MBC)
When administered with PERJETA, the recommended initial dose of docetaxel is 75 mg/m2 administered as an intravenous infusion. The dose may be escalated to 100 mg/m2 administered every 3 weeks if the initial dose is well tolerated.
Section 44425-7
Storage and Handling
Store vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton until time of use to protect from light.
Do not freeze. Do not shake.
DO NOT FREEZE. DO NOT SHAKE.
11 Description
Pertuzumab is a recombinant humanized monoclonal antibody and HER2/neu receptor antagonist that targets the extracellular dimerization domain (Subdomain II) of the human epidermal growth factor receptor 2 protein (HER2). Pertuzumab is produced by recombinant DNA technology in a mammalian cell (Chinese Hamster Ovary) culture. Pertuzumab has an approximate molecular weight of 148 kDa.
PERJETA (pertuzumab) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brown solution for intravenous infusion. Each single-dose vial contains 420 mg of pertuzumab,glacial acetic acid (9.2 mg), histidine (43.5 mg), polysorbate 20 (2.8 mg), and sucrose (575.1 mg) with a pH of 6.
8.4 Pediatric Use
The safety and effectiveness of PERJETA have not been established in pediatric patients.
8.5 Geriatric Use
In CLEOPATRA, NeoSphere, TRYPHAENA, BERENICE, and APHINITY, 464 patients who received PERJETA were ≥ 65 years of age and 47 were ≥ 75 years of age.
The incidence of adverse reactions was increased in patients aged ≥ 65 years of age compared to patients aged < 65 years of age for decreased appetite, anemia, decreased weight, asthenia, dysgeusia, peripheral neuropathy, and hypomagnesemia.
No overall differences in efficacy of PERJETA were observed in patients aged ≥ 65 and <65 years of age. Clinical studies did not include sufficient numbers of patients aged ≥ 75 years to determine if these patients respond differently than younger patients.
Based on a population pharmacokinetic analysis, no significant difference was observed in the pharmacokinetics of pertuzumab between patients < 65 years (n=306) and patients ≥ 65 years (n=175).
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of PERJETA or of other pertuzumab products.
Patients in CLEOPATRA were tested at multiple time-points for anti-pertuzumab antibodies. 3% (13/389) of patients in the PERJETA-treated group and 7% (25/372) of patients in the placebo-treated group tested positive for anti-pertuzumab antibodies. Of these 38 patients, none experienced anaphylactic/hypersensitivity reactions that were clearly related to anti-pertuzumab antibodies. The presence of pertuzumab in patient serum at the levels expected at the time of ADA sampling can interfere with the ability of this assay to detect anti-pertuzumab antibodies. In addition, the assay may be detecting antibodies to trastuzumab. As a result, data may not accurately reflect the true incidence of anti-pertuzumab antibody development.
In the neoadjuvant period of BERENICE, 0.3% (1/383) of patients treated with PERJETA tested positive for anti-pertuzumab antibodies. This patient did not experience any anaphylactic/hypersensitivity reactions.
Because of limited immunogenicity data, the clinical impact of anti-pertuzumab antibodies is unknown. There was no identified clinically significant effect of anti-pertuzumab antibodies on the safety of PERJETA.
4 Contraindications
PERJETA is contraindicated in patients with known hypersensitivity to pertuzumab or to any of its excipients [see Warnings and Precautions (5.4)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label:
- Left Ventricular Dysfunction [see Warnings and Precautions (5.1)]
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.2)]
- Infusion-Related Reactions [see Warnings and Precautions (5.3)]
- Hypersensitivity Reactions/Anaphylaxis [see Warnings and Precautions (5.4)]
8.6 Renal Impairment
Dose adjustments of PERJETA are not needed in patients with mild (creatinine clearance [CLcr] 60 to 90 mL/min) or moderate (CLcr 30 to 60 mL/min) renal impairment. No dose adjustment can be recommended for patients with severe renal impairment (CLcr less than 30 mL/min) because of the limited pharmacokinetic data available [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of PERJETA have not been fully characterized.
12.3 Pharmacokinetics
Based on a population PK analysis that included 481 patients, pertuzumab demonstrated linear pharmacokinetics at a dose range of 2 – 25 mg/kg.
With an initial dose of 840 mg followed by a maintenance dose of 420 mg every three weeks thereafter, the steady-state concentration of pertuzumab was reached after the first maintenance dose.
2.2 Patient Selection
Select patients based on HER2 protein overexpression or HER2 gene amplification in tumor specimens [see Indications and Usage (1) and Clinical Studies (14)]. Assessment of HER2 protein overexpression and HER2 gene amplification should be performed using FDA-approved tests specific for breast cancer by laboratories with demonstrated proficiency.
Information on the FDA-approved tests for the detection of HER2 protein overexpression and HER2 gene amplification is available at: http://www.fda.gov/CompanionDiagnostics.
Improper assay performance, including use of suboptimally fixed tissue, failure to utilize specified reagents, deviation from specific assay instructions, and failure to include appropriate controls for assay validation, can lead to unreliable results.
1 Indications and Usage
PERJETA is a HER2/neu receptor antagonist indicated for:
- Use in combination with trastuzumab and docetaxel for treatment of adults with HER2-positive metastatic breast cancer (MBC) who have not received prior anti-HER2 therapy or chemotherapy for metastatic disease. (1.1)
- Use in combination with trastuzumab and chemotherapy as
- neoadjuvant treatment of adults with HER2-positive, locally advanced, inflammatory, or early stage breast cancer (either greater than 2 cm in diameter or node positive) as part of a complete treatment regimen for early breast cancer. (1.2, 2.2, 14.2)
- adjuvant treatment of adults with HER2-positive early breast cancer at high risk of recurrence (1.2, 2.2, 14.3)
12.1 Mechanism of Action
Pertuzumab targets the extracellular dimerization domain (Subdomain II) of the human epidermal growth factor receptor 2 protein (HER2) and, thereby, blocks ligand-dependent heterodimerization of HER2 with other HER family members, including EGFR, HER3, and HER4. As a result, pertuzumab inhibits ligand-initiated intracellular signaling through two major signal pathways, mitogen-activated protein (MAP) kinase, and phosphoinositide 3-kinase (PI3K). Inhibition of these signaling pathways can result in cell growth arrest and apoptosis, respectively. In addition, pertuzumab mediates antibody-dependent cell-mediated cytotoxicity (ADCC).
While pertuzumab alone inhibited the proliferation of human tumor cells, the combination of pertuzumab and trastuzumab augmented anti-tumor activity in HER2-overexpressing xenograft models.
5.2 Embryo Fetal Toxicity
Based on its mechanism of action and findings in animal studies, PERJETA can cause fetal harm when administered to a pregnant woman. PERJETA is a HER2/neu receptor antagonist. Cases of oligohydramnios and oligohydramnios sequence manifesting as pulmonary hypoplasia, skeletal abnormalities, and neonatal death have been reported with use of another HER2/neu receptor antagonist (trastuzumab) during pregnancy. In an animal reproduction study, administration of pertuzumab to pregnant cynomolgus monkeys during the period of organogenesis resulted in oligohydramnios, delayed fetal kidney development, and embryo-fetal death at exposures 2.5 to 20 times the exposure in humans at the recommended dose, based on Cmax.
Verify the pregnancy status of females of reproductive potential prior to the initiation of PERJETA. Advise pregnant women and females of reproductive potential that exposure to PERJETA in combination with trastuzumab during pregnancy or within 7 months prior to conception can result in fetal harm, including embryo-fetal death or birth defects. Advise females of reproductive potential to use effective contraception during treatment and for 7 months following the last dose of PERJETA in combination with trastuzumab [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
- Infusion-Related Reactions: PERJETA can cause serious infusion reactions, including fatal events: Monitor for signs and symptoms. If a significant infusion-associated reaction occurs, slow or interrupt the infusion and administer appropriate medical therapies. (5.3)
- Hypersensitivity Reactions/Anaphylaxis: PERJETA can cause hypersensitivity reactions, including anaphylaxis: Monitor for signs and symptoms, including angioedema. If a severe hypersensitivity reaction/anaphylaxis occurs, discontinue the infusion immediately and administer appropriate medical therapies. (5.4)
2 Dosage and Administration
- For intravenous infusion only. Do not administer as an intravenous push or bolus. (2.5)
- HER2 testing: Perform using FDA-approved tests by laboratories with demonstrated proficiency. (2.1)
- The initial PERJETA dose is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks thereafter by 420 mg administered as a 30 to 60 minute intravenous infusion. (2.3)
- MBC: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and docetaxel every 3 weeks. (2.3)
- Neoadjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy preoperatively every 3 weeks for 3 to 6 cycles. (2.3)
- Adjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy postoperatively every 3 weeks for a total of 1 year (up to 18 cycles). (2.3)
3 Dosage Forms and Strengths
Injection: 420 mg/14 mL (30 mg/mL) clear to slightly opalescent and colorless to pale brown solution in a single-dose vial
1.2 Early Breast Cancer (ebc)
PERJETA is indicated for use in combination with trastuzumab and chemotherapy for
- the neoadjuvant treatment of adults with HER2-positive, locally advanced, inflammatory, or early stage breast cancer (either greater than 2 cm in diameter or node positive) as part of a complete treatment regimen for early breast cancer [see Dosage and Administration (2.2) and Clinical Studies (14.2)].
- the adjuvant treatment of adults with HER2-positive early breast cancer at high risk of recurrence [see Dosage and Administration (2.2) and Clinical Studies (14.3)].
14.1 Metastatic Breast Cancer
CLEOPATRA (NCT00567190) was a multicenter, double-blind, placebo-controlled trial of 808 patients with HER2-positive metastatic breast cancer. HER2 overexpression was defined as a score of 3+ IHC or FISH amplification ratio of 2.0 or greater as determined by a central laboratory. Patients were randomly allocated 1:1 to receive placebo plus trastuzumab and docetaxel or PERJETA plus trastuzumab and docetaxel. Randomization was stratified by prior treatment (prior or no prior adjuvant/neoadjuvant anti-HER2 therapy or chemotherapy) and geographic region (Europe, North America, South America, and Asia). Patients with prior adjuvant or neoadjuvant therapy were required to have a disease-free interval of greater than 12 months before trial enrollment.
PERJETA was given intravenously at an initial dose of 840 mg, followed by 420 mg every 3 weeks thereafter. Trastuzumab was given intravenously at an initial dose of 8 mg/kg, followed by 6 mg/kg every 3 weeks thereafter. Patients were treated with PERJETA and trastuzumab until progression of disease, withdrawal of consent, or unacceptable toxicity. Docetaxel was given as an initial dose of 75 mg/m2 by intravenous infusion every 3 weeks for at least 6 cycles. The docetaxel dose could be escalated to 100 mg/m2 at the investigator's discretion if the initial dose was well tolerated. At the time of the primary analysis, the mean number of cycles of study treatment administered was 16.2 in the placebo-treated group and 19.9 in the PERJETA-treated group.
The major efficacy outcome measure of CLEOPATRA was progression-free survival (PFS) as assessed by an independent review facility (IRF). PFS was defined as the time from the date of randomization to the date of disease progression or death (from any cause) if the death occurred within 18 weeks of the last tumor assessment. Additional endpoints included overall survival (OS), PFS (investigator-assessed), objective response rate (ORR), and duration of response.
Patient demographic and baseline characteristics were balanced between the treatment arms. The median age was 54 (range 22 to 89 years), 59% were White, 32% were Asian, and 4% were Black. All were female with the exception of 2 patients (0.2%). Seventeen percent (17%) of patients were enrolled in North America, 14% in South America, 38% in Europe, and 31% in Asia. Tumor prognostic characteristics, including hormone receptor status (positive 48%, negative 50%), presence of visceral disease (78%) and non-visceral disease only (22%) were similar in the study arms. Approximately half of the patients received prior adjuvant or neoadjuvant anti-HER2 therapy or chemotherapy (PERJETA 46%, placebo 47%). Among patients with hormone receptor positive tumors, 45% received prior adjuvant hormonal therapy and 11% received hormonal therapy for metastatic disease. Eleven percent of patients received prior adjuvant or neoadjuvant trastuzumab.
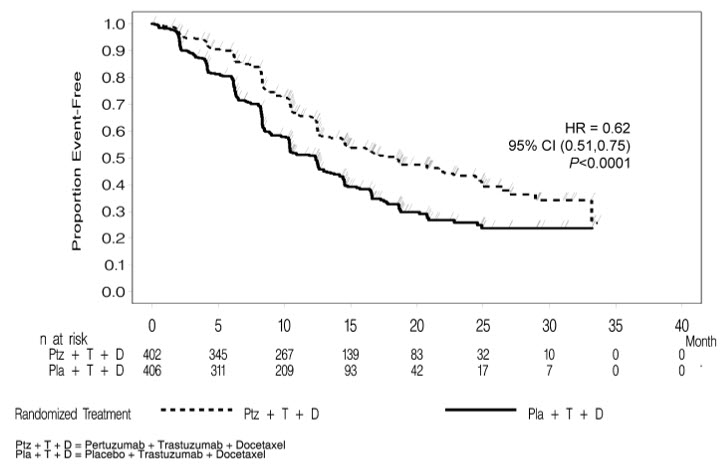
CLEOPATRA demonstrated a statistically significant improvement in IRF-assessed PFS in the PERJETA-treated group compared with the placebo-treated group. The results for investigator-assessed PFS were comparable to those observed for IRF-assessed PFS. A statistically significant OS improvement was demonstrated for the PERJETA-treated group compared with the placebo-treated group) with the final OS analysis. OS results in patient subgroups were consistent with those observed for IRF-assessed PFS with the exception of the subgroup of patients with disease limited to non-visceral metastasis [HR=1.11 (95% CI: 0.66, 1.85)].
| Endpoint | PERJETA + trastuzumab + docetaxel n=402 |
Placebo + trastuzumab + docetaxel n=406 |
|---|---|---|
| CI=Confidence Interval | ||
|
Progression-Free Survival
(independent review) |
||
| Number of events (%) | 191 (47.5%) | 242 (59.6%) |
| Median (months) | 18.5 | 12.4 |
| Hazard Ratio (95% CI) |
0.62 (0.51, 0.75) |
|
| p-value | < 0.0001 | |
|
Overall Survival
Final analysis of overall survival performed when 389 patients had died.
(final) |
||
| Deaths (%) | 168 (41.8%) | 221 (54.4%) |
| Median (months) | 56.5 | 40.8 |
| Hazard Ratio (95% CI) |
0.68 (0.56, 0.84) |
|
| p-value | 0.0002 | |
|
Objective Response Rate
(independent review) |
n = 343 | n = 336 |
| Objective response (CR + PR) | 275 (80.2%) | 233 (69.3%) |
| Complete response (CR) (%) | 19 (5.5%) | 14 (4.2%) |
| Partial Response (PR) (%) | 256 (74.6%) | 219 (65.2%) |
| Difference in ORR (95% CI) |
10.8% (4.2%, 17.5%) |
|
| p-value | 0.0011 | |
|
Duration of Response
Median (months) |
20.2 | 12.5 |
Consistent results were observed across several patient subgroups including age (< 65 or ≥ 65 years), race, geographic region, prior adjuvant/neoadjuvant anti-HER2 therapy or chemotherapy (yes or no), and prior adjuvant/neoadjuvant trastuzumab (yes or no). In the subgroup of patients with hormone receptor-negative disease (n=408), the hazard ratio was 0.55 (95% CI: 0.42, 0.72). In the subgroup of patients with hormone receptor-positive disease (n=388), the hazard ratio was 0.72 (95% CI: 0.55, 0.95). In the subgroup of patients with disease limited to non-visceral metastasis (n=178), the hazard ratio was 0.96 (95% CI: 0.61, 1.52).
Figure 1 Kaplan-Meier Curve of Progression-Free Survival for CLEOPATRA (IRF-Assessed)
Figure 2 Kaplan-Meier Curve of Overall Survival for CLEOPATRA (Final Analysis)
8 Use in Specific Populations
Females and Males of Reproductive Potential: Verify the pregnancy status of females prior to initiation of PERJETA. (8.3)
5.3 Infusion Related Reactions
PERJETA can cause serious infusion reactions, including fatal events [see Adverse Reactions (6.1)].
In CLEOPATRA, on the first day, when only PERJETA was administered, infusion-related reactions occurred in 13% of patients and < 1% were Grade 3 or 4. The most common infusion reactions (≥ 1%) were pyrexia, chills, fatigue, headache, asthenia, hypersensitivity, and vomiting. During the second cycle when all drugs were administered on the same day, the most common infusion reactions in the PERJETA-treated group (≥ 1%) were fatigue, dysgeusia, hypersensitivity, myalgia, and vomiting.
In APHINITY, when PERJETA was administered in combination with trastuzumab and chemotherapy on the same day, infusion-related reactions occurred in 21% of patients with <1% of patients experiencing Grade 3-4 events.
Observe patients closely for 60 minutes after the first infusion and for 30 minutes after subsequent infusions of PERJETA. If a significant infusion-related reaction occurs, slow or interrupt the infusion, and administer appropriate medical therapies. Monitor patients carefully until complete resolution of signs and symptoms. Consider permanent discontinuation in patients with severe infusion reactions [see Dosage and Administration (2.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.1 Left Ventricular Dysfunction
PERJETA can cause left ventricular dysfunction, including symptomatic heart failure. Decreases in LVEF have been reported with drugs that block HER2 activity, including PERJETA.
Assess LVEF prior to initiation of PERJETA and at regular intervals during treatment to ensure that LVEF is within normal limits. If the LVEF declines and has not improved, or has declined further at the subsequent assessment, consider permanent discontinuation of PERJETA and trastuzumab [see Dosage Modification for Adverse Reactions (2.5)].
In the PERJETA-treated patients with MBC in CLEOPATRA, left ventricular dysfunction occurred in 4% of patients and symptomatic left ventricular systolic dysfunction (LVSD) (congestive heart failure) occurred in 1% of patients. Patients who received prior anthracyclines or prior radiotherapy to the chest area may be at higher risk of decreased LVEF or left ventricular dysfunction.
In patients receiving PERJETA as a neoadjuvant treatment in combination with trastuzumab and docetaxel in NeoSphere, LVEF decline > 10% and a drop to < 50% occurred in 8% of patients and left ventricular dysfunction occurred in 3% of patients. LVEF recovered to ≥ 50% in all these patients.
In patients receiving neoadjuvant PERJETA in TRYPHAENA, LVEF decline > 10% and a drop to < 50% occurred in 7% of patients treated with PERJETA plus trastuzumab and FEC followed by PERJETA plus trastuzumab and docetaxel, 16% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC, and 11% of patients treated with PERJETA in combination with TCH. Left ventricular dysfunction occurred in 6% of patients treated with PERJETA plus trastuzumab and FEC followed by PERJETA plus trastuzumab and docetaxel, 4% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC, and 3% of patients treated with PERJETA in combination with TCH. Symptomatic LVSD occurred in 4% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC, 1% of patients treated with PERJETA in combination with TCH, and none of the patients treated with PERJETA plus trastuzumab and FEC followed by PERJETA plus trastuzumab and docetaxel. LVEF recovered to ≥ 50% in all but one patient.
In patients receiving neoadjuvant PERJETA in BERENICE, in the neoadjuvant period, LVEF decline ≥ 10% and a drop to < 50% as measured by ECHO/MUGA assessment occurred in 7% of patients treated with PERJETA plus trastuzumab and paclitaxel following ddAC, and 2% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC. Ejection fraction decreased (asymptomatic LVD) occurred in 7% of patients treated with PERJETA plus trastuzumab and paclitaxel following ddAC and 4% of the patients treated with PERJETA plus trastuzumab and docetaxel following FEC in the neoadjuvant period. Symptomatic LVSD (NYHA Class III/IV Congestive Heart Failure) occurred in 2% of patients treated with PERJETA plus trastuzumab and paclitaxel following ddAC and none of the patients treated with PERJETA plus trastuzumab and docetaxel following FEC in the neoadjuvant period.
In patients receiving adjuvant PERJETA in APHINITY, the incidence of symptomatic heart failure (NYHA Class III/IV) with a LVEF decline ≥ 10% and a drop to < 50% was 0.6%. Of the patients who experienced symptomatic heart failure, 47% of PERJETA-treated patients had recovered (defined as 2 consecutive LVEF measurements above 50%) at the data cutoff. The majority of the events (86%) were reported in anthracycline-treated patients. Asymptomatic or mildly symptomatic (NYHA Class II) declines in LVEF ≥ 10% and a drop to < 50% were reported in 3% of PERJETA-treated patients, of whom 80% recovered at the data cutoff.
PERJETA has not been studied in patients with a pretreatment LVEF value of < 50%, a prior history of CHF, decreases in LVEF to < 50% during prior trastuzumab therapy, or conditions that could impair left ventricular function such as uncontrolled hypertension, recent myocardial infarction, serious cardiac arrhythmia requiring treatment or a cumulative prior anthracycline exposure to > 360 mg/m2 of doxorubicin or its equivalent.
1.1 Metastatic Breast Cancer (mbc)
PERJETA is indicated for use in combination with trastuzumab and docetaxel for the treatment of adults with HER2-positive metastatic breast cancer who have not received prior anti-HER2 therapy or chemotherapy for metastatic disease [see Dosage and Administration (2.2) and Clinical Studies (14.1)].
2.6 Preparation for Administration
Administer as an intravenous infusion only. Do not administer as an intravenous push or bolus. Do not mix PERJETA with other drugs.
14.3 Adjuvant Treatment of Breast Cancer
APHINITY (NCT01358877) was a multicenter, randomized, double-blind, placebo-controlled study conducted in 4804 patients with HER2-positive early breast cancer who had their primary tumor excised prior to randomization. Patients were then randomized to receive PERJETA or placebo, in combination with adjuvant trastuzumab and chemotherapy. Randomization was stratified by the following factors: region, nodal status, protocol version, central hormone receptor status, and adjuvant chemotherapy regimen.
Investigators selected one of the following anthracycline-based or non-anthracycline-based chemotherapy regimens for individual patients:
- 3 or 4 cycles of FEC (5-FU 500-600 mg/m2, epirubicin 90-120 mg/m2, cyclophosphamide 500-600 mg/m2) or FAC (5-FU 500-600 mg/m2, doxorubicin 50 mg/m2, cyclophosphamide 500-600 mg/m2), followed by 3 or 4 cycles of docetaxel (75 mg/m2 which could be escalated to 100 mg/m2 every 3 weeks) or 12 cycles of weekly paclitaxel (80 mg/m2).
- 4 cycles of AC (doxorubicin 60 mg/m2 and cyclophosphamide 500-600 mg/m2) or EC (epirubicin 90-120 mg/m2 and cyclophosphamide 500-600 mg/m2) either every 3 weeks or every 2 weeks with GCSF support, followed by docetaxel (100 mg/m2 for 3 cycles or 75 mg/m2 for first cycle and 100 mg/m2 for subsequent three cycles, or 75 mg/m2 for four cycles) or 12 cycles of weekly paclitaxel (80 mg/m2).
- 6 cycles of docetaxel (75 mg/m2) in combination with carboplatin (AUC 6)
PERJETA and trastuzumab were administered intravenously every 3 weeks starting on Day 1 of the first taxane-containing cycle, for a total of 52 weeks (up to 18 cycles) or until recurrence, withdrawal of consent, or unmanageable toxicity.
After completion of chemotherapy, patients received radiotherapy and/or hormone therapy as per investigator's discretion.
The major efficacy outcome of the study was invasive disease-free survival (IDFS), defined as the time from randomization to first occurrence of ipsilateral local or regional invasive breast cancer recurrence, distant recurrence, contralateral invasive breast cancer, or death from any cause. Additional efficacy endpoints were IDFS including second primary non-breast cancer, disease-free survival (DFS), and overall survival (OS).
Demographics were balanced between the two treatment arms. The median age was 51 years (range 18-86), 13% of patients were 65 or older, and over 99% of patients were female. Sixty-three percent of patients had node-positive disease, 64% had hormone receptor-positive disease, and 71% were White. All patients had an ECOG performance status of 0 or 1. Seventy-eight percent received an anthracycline containing regimen.
PERJETA-treated patients and placebo-treated patients both received a median number of 18 cycles of anti-HER2 therapy. After a median follow-up of 45.4 months, a statistically significant improvement in IDFS was demonstrated in patients randomized to receive PERJETA compared with patients randomized to receive placebo. The efficacy results from APHINITY are summarized in Tables 10 and 11 and in Figure 3.
| PERJETA + trastuzumab + chemotherapy N=2400 |
Placebo + trastuzumab + chemotherapy N=2404 |
|
|---|---|---|
| HR=Hazard Ratio, CI=Confidence Interval | ||
| Invasive Disease Free Survival (IDFS) | ||
| Number (%) of patients with event | 171 (7.1%) | 210 (8.7%) |
| HR [95% CI] All analyses stratified by nodal status, protocol version, central hormone receptor status, and adjuvant chemotherapy regimen. Stratification factors are defined according to the randomization data for IDFS.
|
0.82 [0.67, 1.00] | |
| p-value (Log-Rank test, stratified) | 0.047 | |
| 3 year event-free rate 3-year event-free rate derived from Kaplan-Meier estimates , % [95% CI] |
94.1 [93.1, 95.0] | 93.2 [92.2, 94.3] |
| IDFS including second primary non-breast cancer | ||
| Number (%) of patients with event | 189 (7.9%) | 230 (9.6%) |
| HR [95% CI] | 0.83 [0.68, 1.00] | |
| 3 year event-free rate, % [95% CI] | 93.5 [92.5, 94.5] | 92.5 [91.4, 93.6] |
| Disease Free Survival (DFS) | ||
| Number (%) of patients with event | 192 (8.0%) | 236 (9.8%) |
| HR [95% CI] | 0.82 [0.68, 0.99] | |
| 3 year event-free rate, % [95% CI] | 93.4 [92.4, 94.4] | 92.3 [91.2, 93.4] |
|
Overall Survival (OS) Data from first interim analysis
|
||
| Number (%) of patients with event | 80 (3.3%) | 89 (3.7%) |
| HR [95% CI] | 0.89 [0.66, 1.21] | |
| 3 year event-free rate, % [95% CI] | 97.7 [97.0, 98.3] | 97.7 [97.1, 98.3] |
Figure 3 Kaplan-Meier Curve of Invasive Disease Free Survival from APHINITY (ITT Population)
| Population | Number of events/Total N (%) | IDFS at 3 year (%, 95% CI) |
Unstratified HR (95% CI) | ||
|---|---|---|---|---|---|
| PERJETA + trastuzumab + chemotherapy | Placebo + trastuzumab + chemotherapy | PERJETA + trastuzumab + chemotherapy | Placebo + trastuzumab + chemotherapy | ||
| Hormone Receptor Status | |||||
| Negative | 71/864 (8.2%) |
91/858 (10.6%) |
92.8 (90.8, 94.3) |
91.2 (89.0, 92.9) |
0.76 (0.56, 1.04) |
| Positive | 100/1536 (6.5%) |
119/1546 (7.7%) |
94.8 (93.5, 95.8) |
94.4 (93.1, 95.4) |
0.86 (0.66, 1.13) |
| Nodal Status | |||||
| Negative | 32/897 (3.6%) |
29/902 (3.2%) |
97.5 (96.3, 98.4) |
98.4 (97.3, 99.0) |
1.13 (0.68, 1.86) |
| Positive | 139/1503 (9.2%) |
181/1502 (12.1%) |
92.0 (90.5, 93.3) |
90.2 (88.5, 91.6) |
0.77 (0.62, 0.96) |
| Adjuvant Chemotherapy Regimen | |||||
| Anthracycline | 139/1865 (7.4%) |
171/1877 (9.1%) |
93.8 (92.6, 94.8) |
93.0 (91.8, 94.1) |
0.82 (0.66, 1.03) |
| Non-Anthracycline | 32/535 (6.0%) |
39/527 (7.4%) |
94.9 (92.6, 96.6) |
94.0 (91.5, 95.8) |
0.82 (0.51, 1.31) |
2.3 Recommended Dosage and Administration
The initial dose of PERJETA is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks by a dose of 420 mg administered as an intravenous infusion over 30 to 60 minutes.
When administered with PERJETA, the recommended initial dose of trastuzumab is 8 mg/kg administered as a 90-minute intravenous infusion, followed every 3 weeks by a dose of 6 mg/kg administered as an intravenous infusion over 30 to 90 minutes.
When administered with PERJETA, the recommended initial dose of trastuzumab hyaluronidase-oysk is 600 mg/10,000 units (600 mg trastuzumab and 10,000 units hyaluronidase) administered subcutaneously over approximately 2 to 5 minutes once every three weeks irrespective of the patient's body weight.
Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and taxane sequentially. PERJETA and trastuzumab or trastuzumab hyaluronidase-oysk can be given in any order. Administer taxane after PERJETA and trastuzumab or trastuzumab hyaluronidase-oysk. An observation period of 30 to 60 minutes is recommended after each PERJETA infusion and before commencement of any subsequent administration of trastuzumab or trastuzumab hyaluronidase-oysk, or taxane [see Warnings and Precautions (5.3)].
In patients receiving an anthracycline-based regimen, administer PERJETA and trastuzumab or trastuzumab hyaluronidase-oysk after completion of the anthracycline.
5.4 Hypersensitivity Reactions/anaphylaxis
PERJETA can cause hypersensitivity reactions, including anaphylaxis.
In CLEOPATRA, the overall frequency of hypersensitivity/anaphylaxis reactions was 11% in PERJETA-treated patients, with Grade 3 – 4 hypersensitivity reactions and anaphylaxis occurring in 2% of patients.
In NeoSphere, TRYPHAENA, BERENICE, and APHINITY, hypersensitivity/anaphylaxis events were consistent with those observed in CLEOPATRA. In APHINITY, the overall frequency of hypersensitivity/anaphylaxis was 5% in the PERJETA treated group. The incidence was highest in the PERJETA plus TCH treated group (8%) with 1% Grade 3 – 4 events.
Observe patients closely for hypersensitivity reactions. Severe hypersensitivity, including anaphylaxis and fatal events, have been observed in patients treated with PERJETA [see Clinical Trials Experience (6.1)]. Angioedema has been described in post-marketing reports. Medications to treat such reactions, as well as emergency equipment, should be available for immediate use prior to administration of PERJETA. PERJETA is contraindicated in patients with known hypersensitivity to pertuzumab or to any of its excipients [see Contraindications (4)].
Principal Display Panel 14 Ml Vial Carton
NDC 50242-145-01
Perjeta®
(Pertuzumab)
Injection
420 mg/ 14 mL
(30 mg/mL)
Dilute Prior To Use
For Intravenous Infusion Only
Single-Dose Vial
Discard Unused Portion
No preservative.
Rx only
1 vial
Genentech
11029352
2.1 Evaluation and Testing Before Initiating Perjeta
Assess left ventricular ejection fraction (LVEF) prior to initiation of PERJETA and at regular intervals during treatment [see Boxed Warning, Dosage and Administration (2.4), Warnings and Precautions (5.1)].
Verify the pregnancy status of females of reproductive potential prior to the initiation of PERJETA [see Warnings and Precautions (5.2), Use in Specific Populations (8.1, 8.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of pertuzumab.
Studies have not been performed to evaluate the mutagenic potential of pertuzumab.
No specific fertility studies in animals have been performed to evaluate the effect of pertuzumab. No adverse effects on male and female reproductive organs were observed in repeat-dose toxicity studies of up to six months duration in cynomolgus monkeys.
Warning: Left Ventricular Dysfunction and Embryo Fetal Toxicity
- Left Ventricular Dysfunction: PERJETA can cause subclinical and clinical cardiac failure manifesting as decreased LVEF and CHF. Evaluate cardiac function prior to and during treatment. Discontinue PERJETA treatment for a confirmed clinically significant decrease in left ventricular function [see Dosage and Administration (2.3), Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
- Embryo-fetal Toxicity: Exposure to PERJETA can cause embryo-fetal death and birth defects. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1) (8.3)].
Structured Label Content
Section 42229-5 (42229-5)
Metastatic Breast Cancer (MBC)
When administered with PERJETA, the recommended initial dose of docetaxel is 75 mg/m2 administered as an intravenous infusion. The dose may be escalated to 100 mg/m2 administered every 3 weeks if the initial dose is well tolerated.
Section 44425-7 (44425-7)
Storage and Handling
Store vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton until time of use to protect from light.
Do not freeze. Do not shake.
DO NOT FREEZE. DO NOT SHAKE.
11 Description (11 DESCRIPTION)
Pertuzumab is a recombinant humanized monoclonal antibody and HER2/neu receptor antagonist that targets the extracellular dimerization domain (Subdomain II) of the human epidermal growth factor receptor 2 protein (HER2). Pertuzumab is produced by recombinant DNA technology in a mammalian cell (Chinese Hamster Ovary) culture. Pertuzumab has an approximate molecular weight of 148 kDa.
PERJETA (pertuzumab) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brown solution for intravenous infusion. Each single-dose vial contains 420 mg of pertuzumab,glacial acetic acid (9.2 mg), histidine (43.5 mg), polysorbate 20 (2.8 mg), and sucrose (575.1 mg) with a pH of 6.
8.4 Pediatric Use
The safety and effectiveness of PERJETA have not been established in pediatric patients.
8.5 Geriatric Use
In CLEOPATRA, NeoSphere, TRYPHAENA, BERENICE, and APHINITY, 464 patients who received PERJETA were ≥ 65 years of age and 47 were ≥ 75 years of age.
The incidence of adverse reactions was increased in patients aged ≥ 65 years of age compared to patients aged < 65 years of age for decreased appetite, anemia, decreased weight, asthenia, dysgeusia, peripheral neuropathy, and hypomagnesemia.
No overall differences in efficacy of PERJETA were observed in patients aged ≥ 65 and <65 years of age. Clinical studies did not include sufficient numbers of patients aged ≥ 75 years to determine if these patients respond differently than younger patients.
Based on a population pharmacokinetic analysis, no significant difference was observed in the pharmacokinetics of pertuzumab between patients < 65 years (n=306) and patients ≥ 65 years (n=175).
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of PERJETA or of other pertuzumab products.
Patients in CLEOPATRA were tested at multiple time-points for anti-pertuzumab antibodies. 3% (13/389) of patients in the PERJETA-treated group and 7% (25/372) of patients in the placebo-treated group tested positive for anti-pertuzumab antibodies. Of these 38 patients, none experienced anaphylactic/hypersensitivity reactions that were clearly related to anti-pertuzumab antibodies. The presence of pertuzumab in patient serum at the levels expected at the time of ADA sampling can interfere with the ability of this assay to detect anti-pertuzumab antibodies. In addition, the assay may be detecting antibodies to trastuzumab. As a result, data may not accurately reflect the true incidence of anti-pertuzumab antibody development.
In the neoadjuvant period of BERENICE, 0.3% (1/383) of patients treated with PERJETA tested positive for anti-pertuzumab antibodies. This patient did not experience any anaphylactic/hypersensitivity reactions.
Because of limited immunogenicity data, the clinical impact of anti-pertuzumab antibodies is unknown. There was no identified clinically significant effect of anti-pertuzumab antibodies on the safety of PERJETA.
4 Contraindications (4 CONTRAINDICATIONS)
PERJETA is contraindicated in patients with known hypersensitivity to pertuzumab or to any of its excipients [see Warnings and Precautions (5.4)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the label:
- Left Ventricular Dysfunction [see Warnings and Precautions (5.1)]
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.2)]
- Infusion-Related Reactions [see Warnings and Precautions (5.3)]
- Hypersensitivity Reactions/Anaphylaxis [see Warnings and Precautions (5.4)]
8.6 Renal Impairment
Dose adjustments of PERJETA are not needed in patients with mild (creatinine clearance [CLcr] 60 to 90 mL/min) or moderate (CLcr 30 to 60 mL/min) renal impairment. No dose adjustment can be recommended for patients with severe renal impairment (CLcr less than 30 mL/min) because of the limited pharmacokinetic data available [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of PERJETA have not been fully characterized.
12.3 Pharmacokinetics
Based on a population PK analysis that included 481 patients, pertuzumab demonstrated linear pharmacokinetics at a dose range of 2 – 25 mg/kg.
With an initial dose of 840 mg followed by a maintenance dose of 420 mg every three weeks thereafter, the steady-state concentration of pertuzumab was reached after the first maintenance dose.
2.2 Patient Selection
Select patients based on HER2 protein overexpression or HER2 gene amplification in tumor specimens [see Indications and Usage (1) and Clinical Studies (14)]. Assessment of HER2 protein overexpression and HER2 gene amplification should be performed using FDA-approved tests specific for breast cancer by laboratories with demonstrated proficiency.
Information on the FDA-approved tests for the detection of HER2 protein overexpression and HER2 gene amplification is available at: http://www.fda.gov/CompanionDiagnostics.
Improper assay performance, including use of suboptimally fixed tissue, failure to utilize specified reagents, deviation from specific assay instructions, and failure to include appropriate controls for assay validation, can lead to unreliable results.
1 Indications and Usage (1 INDICATIONS AND USAGE)
PERJETA is a HER2/neu receptor antagonist indicated for:
- Use in combination with trastuzumab and docetaxel for treatment of adults with HER2-positive metastatic breast cancer (MBC) who have not received prior anti-HER2 therapy or chemotherapy for metastatic disease. (1.1)
- Use in combination with trastuzumab and chemotherapy as
- neoadjuvant treatment of adults with HER2-positive, locally advanced, inflammatory, or early stage breast cancer (either greater than 2 cm in diameter or node positive) as part of a complete treatment regimen for early breast cancer. (1.2, 2.2, 14.2)
- adjuvant treatment of adults with HER2-positive early breast cancer at high risk of recurrence (1.2, 2.2, 14.3)
12.1 Mechanism of Action
Pertuzumab targets the extracellular dimerization domain (Subdomain II) of the human epidermal growth factor receptor 2 protein (HER2) and, thereby, blocks ligand-dependent heterodimerization of HER2 with other HER family members, including EGFR, HER3, and HER4. As a result, pertuzumab inhibits ligand-initiated intracellular signaling through two major signal pathways, mitogen-activated protein (MAP) kinase, and phosphoinositide 3-kinase (PI3K). Inhibition of these signaling pathways can result in cell growth arrest and apoptosis, respectively. In addition, pertuzumab mediates antibody-dependent cell-mediated cytotoxicity (ADCC).
While pertuzumab alone inhibited the proliferation of human tumor cells, the combination of pertuzumab and trastuzumab augmented anti-tumor activity in HER2-overexpressing xenograft models.
5.2 Embryo Fetal Toxicity (5.2 Embryo-Fetal Toxicity)
Based on its mechanism of action and findings in animal studies, PERJETA can cause fetal harm when administered to a pregnant woman. PERJETA is a HER2/neu receptor antagonist. Cases of oligohydramnios and oligohydramnios sequence manifesting as pulmonary hypoplasia, skeletal abnormalities, and neonatal death have been reported with use of another HER2/neu receptor antagonist (trastuzumab) during pregnancy. In an animal reproduction study, administration of pertuzumab to pregnant cynomolgus monkeys during the period of organogenesis resulted in oligohydramnios, delayed fetal kidney development, and embryo-fetal death at exposures 2.5 to 20 times the exposure in humans at the recommended dose, based on Cmax.
Verify the pregnancy status of females of reproductive potential prior to the initiation of PERJETA. Advise pregnant women and females of reproductive potential that exposure to PERJETA in combination with trastuzumab during pregnancy or within 7 months prior to conception can result in fetal harm, including embryo-fetal death or birth defects. Advise females of reproductive potential to use effective contraception during treatment and for 7 months following the last dose of PERJETA in combination with trastuzumab [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Infusion-Related Reactions: PERJETA can cause serious infusion reactions, including fatal events: Monitor for signs and symptoms. If a significant infusion-associated reaction occurs, slow or interrupt the infusion and administer appropriate medical therapies. (5.3)
- Hypersensitivity Reactions/Anaphylaxis: PERJETA can cause hypersensitivity reactions, including anaphylaxis: Monitor for signs and symptoms, including angioedema. If a severe hypersensitivity reaction/anaphylaxis occurs, discontinue the infusion immediately and administer appropriate medical therapies. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For intravenous infusion only. Do not administer as an intravenous push or bolus. (2.5)
- HER2 testing: Perform using FDA-approved tests by laboratories with demonstrated proficiency. (2.1)
- The initial PERJETA dose is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks thereafter by 420 mg administered as a 30 to 60 minute intravenous infusion. (2.3)
- MBC: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and docetaxel every 3 weeks. (2.3)
- Neoadjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy preoperatively every 3 weeks for 3 to 6 cycles. (2.3)
- Adjuvant: Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and chemotherapy postoperatively every 3 weeks for a total of 1 year (up to 18 cycles). (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 420 mg/14 mL (30 mg/mL) clear to slightly opalescent and colorless to pale brown solution in a single-dose vial
1.2 Early Breast Cancer (ebc) (1.2 Early Breast Cancer (EBC))
PERJETA is indicated for use in combination with trastuzumab and chemotherapy for
- the neoadjuvant treatment of adults with HER2-positive, locally advanced, inflammatory, or early stage breast cancer (either greater than 2 cm in diameter or node positive) as part of a complete treatment regimen for early breast cancer [see Dosage and Administration (2.2) and Clinical Studies (14.2)].
- the adjuvant treatment of adults with HER2-positive early breast cancer at high risk of recurrence [see Dosage and Administration (2.2) and Clinical Studies (14.3)].
14.1 Metastatic Breast Cancer
CLEOPATRA (NCT00567190) was a multicenter, double-blind, placebo-controlled trial of 808 patients with HER2-positive metastatic breast cancer. HER2 overexpression was defined as a score of 3+ IHC or FISH amplification ratio of 2.0 or greater as determined by a central laboratory. Patients were randomly allocated 1:1 to receive placebo plus trastuzumab and docetaxel or PERJETA plus trastuzumab and docetaxel. Randomization was stratified by prior treatment (prior or no prior adjuvant/neoadjuvant anti-HER2 therapy or chemotherapy) and geographic region (Europe, North America, South America, and Asia). Patients with prior adjuvant or neoadjuvant therapy were required to have a disease-free interval of greater than 12 months before trial enrollment.
PERJETA was given intravenously at an initial dose of 840 mg, followed by 420 mg every 3 weeks thereafter. Trastuzumab was given intravenously at an initial dose of 8 mg/kg, followed by 6 mg/kg every 3 weeks thereafter. Patients were treated with PERJETA and trastuzumab until progression of disease, withdrawal of consent, or unacceptable toxicity. Docetaxel was given as an initial dose of 75 mg/m2 by intravenous infusion every 3 weeks for at least 6 cycles. The docetaxel dose could be escalated to 100 mg/m2 at the investigator's discretion if the initial dose was well tolerated. At the time of the primary analysis, the mean number of cycles of study treatment administered was 16.2 in the placebo-treated group and 19.9 in the PERJETA-treated group.
The major efficacy outcome measure of CLEOPATRA was progression-free survival (PFS) as assessed by an independent review facility (IRF). PFS was defined as the time from the date of randomization to the date of disease progression or death (from any cause) if the death occurred within 18 weeks of the last tumor assessment. Additional endpoints included overall survival (OS), PFS (investigator-assessed), objective response rate (ORR), and duration of response.
Patient demographic and baseline characteristics were balanced between the treatment arms. The median age was 54 (range 22 to 89 years), 59% were White, 32% were Asian, and 4% were Black. All were female with the exception of 2 patients (0.2%). Seventeen percent (17%) of patients were enrolled in North America, 14% in South America, 38% in Europe, and 31% in Asia. Tumor prognostic characteristics, including hormone receptor status (positive 48%, negative 50%), presence of visceral disease (78%) and non-visceral disease only (22%) were similar in the study arms. Approximately half of the patients received prior adjuvant or neoadjuvant anti-HER2 therapy or chemotherapy (PERJETA 46%, placebo 47%). Among patients with hormone receptor positive tumors, 45% received prior adjuvant hormonal therapy and 11% received hormonal therapy for metastatic disease. Eleven percent of patients received prior adjuvant or neoadjuvant trastuzumab.
CLEOPATRA demonstrated a statistically significant improvement in IRF-assessed PFS in the PERJETA-treated group compared with the placebo-treated group. The results for investigator-assessed PFS were comparable to those observed for IRF-assessed PFS. A statistically significant OS improvement was demonstrated for the PERJETA-treated group compared with the placebo-treated group) with the final OS analysis. OS results in patient subgroups were consistent with those observed for IRF-assessed PFS with the exception of the subgroup of patients with disease limited to non-visceral metastasis [HR=1.11 (95% CI: 0.66, 1.85)].
| Endpoint | PERJETA + trastuzumab + docetaxel n=402 |
Placebo + trastuzumab + docetaxel n=406 |
|---|---|---|
| CI=Confidence Interval | ||
|
Progression-Free Survival
(independent review) |
||
| Number of events (%) | 191 (47.5%) | 242 (59.6%) |
| Median (months) | 18.5 | 12.4 |
| Hazard Ratio (95% CI) |
0.62 (0.51, 0.75) |
|
| p-value | < 0.0001 | |
|
Overall Survival
Final analysis of overall survival performed when 389 patients had died.
(final) |
||
| Deaths (%) | 168 (41.8%) | 221 (54.4%) |
| Median (months) | 56.5 | 40.8 |
| Hazard Ratio (95% CI) |
0.68 (0.56, 0.84) |
|
| p-value | 0.0002 | |
|
Objective Response Rate
(independent review) |
n = 343 | n = 336 |
| Objective response (CR + PR) | 275 (80.2%) | 233 (69.3%) |
| Complete response (CR) (%) | 19 (5.5%) | 14 (4.2%) |
| Partial Response (PR) (%) | 256 (74.6%) | 219 (65.2%) |
| Difference in ORR (95% CI) |
10.8% (4.2%, 17.5%) |
|
| p-value | 0.0011 | |
|
Duration of Response
Median (months) |
20.2 | 12.5 |
Consistent results were observed across several patient subgroups including age (< 65 or ≥ 65 years), race, geographic region, prior adjuvant/neoadjuvant anti-HER2 therapy or chemotherapy (yes or no), and prior adjuvant/neoadjuvant trastuzumab (yes or no). In the subgroup of patients with hormone receptor-negative disease (n=408), the hazard ratio was 0.55 (95% CI: 0.42, 0.72). In the subgroup of patients with hormone receptor-positive disease (n=388), the hazard ratio was 0.72 (95% CI: 0.55, 0.95). In the subgroup of patients with disease limited to non-visceral metastasis (n=178), the hazard ratio was 0.96 (95% CI: 0.61, 1.52).
Figure 1 Kaplan-Meier Curve of Progression-Free Survival for CLEOPATRA (IRF-Assessed)
Figure 2 Kaplan-Meier Curve of Overall Survival for CLEOPATRA (Final Analysis)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Females and Males of Reproductive Potential: Verify the pregnancy status of females prior to initiation of PERJETA. (8.3)
5.3 Infusion Related Reactions (5.3 Infusion-Related Reactions)
PERJETA can cause serious infusion reactions, including fatal events [see Adverse Reactions (6.1)].
In CLEOPATRA, on the first day, when only PERJETA was administered, infusion-related reactions occurred in 13% of patients and < 1% were Grade 3 or 4. The most common infusion reactions (≥ 1%) were pyrexia, chills, fatigue, headache, asthenia, hypersensitivity, and vomiting. During the second cycle when all drugs were administered on the same day, the most common infusion reactions in the PERJETA-treated group (≥ 1%) were fatigue, dysgeusia, hypersensitivity, myalgia, and vomiting.
In APHINITY, when PERJETA was administered in combination with trastuzumab and chemotherapy on the same day, infusion-related reactions occurred in 21% of patients with <1% of patients experiencing Grade 3-4 events.
Observe patients closely for 60 minutes after the first infusion and for 30 minutes after subsequent infusions of PERJETA. If a significant infusion-related reaction occurs, slow or interrupt the infusion, and administer appropriate medical therapies. Monitor patients carefully until complete resolution of signs and symptoms. Consider permanent discontinuation in patients with severe infusion reactions [see Dosage and Administration (2.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.1 Left Ventricular Dysfunction
PERJETA can cause left ventricular dysfunction, including symptomatic heart failure. Decreases in LVEF have been reported with drugs that block HER2 activity, including PERJETA.
Assess LVEF prior to initiation of PERJETA and at regular intervals during treatment to ensure that LVEF is within normal limits. If the LVEF declines and has not improved, or has declined further at the subsequent assessment, consider permanent discontinuation of PERJETA and trastuzumab [see Dosage Modification for Adverse Reactions (2.5)].
In the PERJETA-treated patients with MBC in CLEOPATRA, left ventricular dysfunction occurred in 4% of patients and symptomatic left ventricular systolic dysfunction (LVSD) (congestive heart failure) occurred in 1% of patients. Patients who received prior anthracyclines or prior radiotherapy to the chest area may be at higher risk of decreased LVEF or left ventricular dysfunction.
In patients receiving PERJETA as a neoadjuvant treatment in combination with trastuzumab and docetaxel in NeoSphere, LVEF decline > 10% and a drop to < 50% occurred in 8% of patients and left ventricular dysfunction occurred in 3% of patients. LVEF recovered to ≥ 50% in all these patients.
In patients receiving neoadjuvant PERJETA in TRYPHAENA, LVEF decline > 10% and a drop to < 50% occurred in 7% of patients treated with PERJETA plus trastuzumab and FEC followed by PERJETA plus trastuzumab and docetaxel, 16% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC, and 11% of patients treated with PERJETA in combination with TCH. Left ventricular dysfunction occurred in 6% of patients treated with PERJETA plus trastuzumab and FEC followed by PERJETA plus trastuzumab and docetaxel, 4% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC, and 3% of patients treated with PERJETA in combination with TCH. Symptomatic LVSD occurred in 4% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC, 1% of patients treated with PERJETA in combination with TCH, and none of the patients treated with PERJETA plus trastuzumab and FEC followed by PERJETA plus trastuzumab and docetaxel. LVEF recovered to ≥ 50% in all but one patient.
In patients receiving neoadjuvant PERJETA in BERENICE, in the neoadjuvant period, LVEF decline ≥ 10% and a drop to < 50% as measured by ECHO/MUGA assessment occurred in 7% of patients treated with PERJETA plus trastuzumab and paclitaxel following ddAC, and 2% of patients treated with PERJETA plus trastuzumab and docetaxel following FEC. Ejection fraction decreased (asymptomatic LVD) occurred in 7% of patients treated with PERJETA plus trastuzumab and paclitaxel following ddAC and 4% of the patients treated with PERJETA plus trastuzumab and docetaxel following FEC in the neoadjuvant period. Symptomatic LVSD (NYHA Class III/IV Congestive Heart Failure) occurred in 2% of patients treated with PERJETA plus trastuzumab and paclitaxel following ddAC and none of the patients treated with PERJETA plus trastuzumab and docetaxel following FEC in the neoadjuvant period.
In patients receiving adjuvant PERJETA in APHINITY, the incidence of symptomatic heart failure (NYHA Class III/IV) with a LVEF decline ≥ 10% and a drop to < 50% was 0.6%. Of the patients who experienced symptomatic heart failure, 47% of PERJETA-treated patients had recovered (defined as 2 consecutive LVEF measurements above 50%) at the data cutoff. The majority of the events (86%) were reported in anthracycline-treated patients. Asymptomatic or mildly symptomatic (NYHA Class II) declines in LVEF ≥ 10% and a drop to < 50% were reported in 3% of PERJETA-treated patients, of whom 80% recovered at the data cutoff.
PERJETA has not been studied in patients with a pretreatment LVEF value of < 50%, a prior history of CHF, decreases in LVEF to < 50% during prior trastuzumab therapy, or conditions that could impair left ventricular function such as uncontrolled hypertension, recent myocardial infarction, serious cardiac arrhythmia requiring treatment or a cumulative prior anthracycline exposure to > 360 mg/m2 of doxorubicin or its equivalent.
1.1 Metastatic Breast Cancer (mbc) (1.1 Metastatic Breast Cancer (MBC))
PERJETA is indicated for use in combination with trastuzumab and docetaxel for the treatment of adults with HER2-positive metastatic breast cancer who have not received prior anti-HER2 therapy or chemotherapy for metastatic disease [see Dosage and Administration (2.2) and Clinical Studies (14.1)].
2.6 Preparation for Administration
Administer as an intravenous infusion only. Do not administer as an intravenous push or bolus. Do not mix PERJETA with other drugs.
14.3 Adjuvant Treatment of Breast Cancer
APHINITY (NCT01358877) was a multicenter, randomized, double-blind, placebo-controlled study conducted in 4804 patients with HER2-positive early breast cancer who had their primary tumor excised prior to randomization. Patients were then randomized to receive PERJETA or placebo, in combination with adjuvant trastuzumab and chemotherapy. Randomization was stratified by the following factors: region, nodal status, protocol version, central hormone receptor status, and adjuvant chemotherapy regimen.
Investigators selected one of the following anthracycline-based or non-anthracycline-based chemotherapy regimens for individual patients:
- 3 or 4 cycles of FEC (5-FU 500-600 mg/m2, epirubicin 90-120 mg/m2, cyclophosphamide 500-600 mg/m2) or FAC (5-FU 500-600 mg/m2, doxorubicin 50 mg/m2, cyclophosphamide 500-600 mg/m2), followed by 3 or 4 cycles of docetaxel (75 mg/m2 which could be escalated to 100 mg/m2 every 3 weeks) or 12 cycles of weekly paclitaxel (80 mg/m2).
- 4 cycles of AC (doxorubicin 60 mg/m2 and cyclophosphamide 500-600 mg/m2) or EC (epirubicin 90-120 mg/m2 and cyclophosphamide 500-600 mg/m2) either every 3 weeks or every 2 weeks with GCSF support, followed by docetaxel (100 mg/m2 for 3 cycles or 75 mg/m2 for first cycle and 100 mg/m2 for subsequent three cycles, or 75 mg/m2 for four cycles) or 12 cycles of weekly paclitaxel (80 mg/m2).
- 6 cycles of docetaxel (75 mg/m2) in combination with carboplatin (AUC 6)
PERJETA and trastuzumab were administered intravenously every 3 weeks starting on Day 1 of the first taxane-containing cycle, for a total of 52 weeks (up to 18 cycles) or until recurrence, withdrawal of consent, or unmanageable toxicity.
After completion of chemotherapy, patients received radiotherapy and/or hormone therapy as per investigator's discretion.
The major efficacy outcome of the study was invasive disease-free survival (IDFS), defined as the time from randomization to first occurrence of ipsilateral local or regional invasive breast cancer recurrence, distant recurrence, contralateral invasive breast cancer, or death from any cause. Additional efficacy endpoints were IDFS including second primary non-breast cancer, disease-free survival (DFS), and overall survival (OS).
Demographics were balanced between the two treatment arms. The median age was 51 years (range 18-86), 13% of patients were 65 or older, and over 99% of patients were female. Sixty-three percent of patients had node-positive disease, 64% had hormone receptor-positive disease, and 71% were White. All patients had an ECOG performance status of 0 or 1. Seventy-eight percent received an anthracycline containing regimen.
PERJETA-treated patients and placebo-treated patients both received a median number of 18 cycles of anti-HER2 therapy. After a median follow-up of 45.4 months, a statistically significant improvement in IDFS was demonstrated in patients randomized to receive PERJETA compared with patients randomized to receive placebo. The efficacy results from APHINITY are summarized in Tables 10 and 11 and in Figure 3.
| PERJETA + trastuzumab + chemotherapy N=2400 |
Placebo + trastuzumab + chemotherapy N=2404 |
|
|---|---|---|
| HR=Hazard Ratio, CI=Confidence Interval | ||
| Invasive Disease Free Survival (IDFS) | ||
| Number (%) of patients with event | 171 (7.1%) | 210 (8.7%) |
| HR [95% CI] All analyses stratified by nodal status, protocol version, central hormone receptor status, and adjuvant chemotherapy regimen. Stratification factors are defined according to the randomization data for IDFS.
|
0.82 [0.67, 1.00] | |
| p-value (Log-Rank test, stratified) | 0.047 | |
| 3 year event-free rate 3-year event-free rate derived from Kaplan-Meier estimates , % [95% CI] |
94.1 [93.1, 95.0] | 93.2 [92.2, 94.3] |
| IDFS including second primary non-breast cancer | ||
| Number (%) of patients with event | 189 (7.9%) | 230 (9.6%) |
| HR [95% CI] | 0.83 [0.68, 1.00] | |
| 3 year event-free rate, % [95% CI] | 93.5 [92.5, 94.5] | 92.5 [91.4, 93.6] |
| Disease Free Survival (DFS) | ||
| Number (%) of patients with event | 192 (8.0%) | 236 (9.8%) |
| HR [95% CI] | 0.82 [0.68, 0.99] | |
| 3 year event-free rate, % [95% CI] | 93.4 [92.4, 94.4] | 92.3 [91.2, 93.4] |
|
Overall Survival (OS) Data from first interim analysis
|
||
| Number (%) of patients with event | 80 (3.3%) | 89 (3.7%) |
| HR [95% CI] | 0.89 [0.66, 1.21] | |
| 3 year event-free rate, % [95% CI] | 97.7 [97.0, 98.3] | 97.7 [97.1, 98.3] |
Figure 3 Kaplan-Meier Curve of Invasive Disease Free Survival from APHINITY (ITT Population)
| Population | Number of events/Total N (%) | IDFS at 3 year (%, 95% CI) |
Unstratified HR (95% CI) | ||
|---|---|---|---|---|---|
| PERJETA + trastuzumab + chemotherapy | Placebo + trastuzumab + chemotherapy | PERJETA + trastuzumab + chemotherapy | Placebo + trastuzumab + chemotherapy | ||
| Hormone Receptor Status | |||||
| Negative | 71/864 (8.2%) |
91/858 (10.6%) |
92.8 (90.8, 94.3) |
91.2 (89.0, 92.9) |
0.76 (0.56, 1.04) |
| Positive | 100/1536 (6.5%) |
119/1546 (7.7%) |
94.8 (93.5, 95.8) |
94.4 (93.1, 95.4) |
0.86 (0.66, 1.13) |
| Nodal Status | |||||
| Negative | 32/897 (3.6%) |
29/902 (3.2%) |
97.5 (96.3, 98.4) |
98.4 (97.3, 99.0) |
1.13 (0.68, 1.86) |
| Positive | 139/1503 (9.2%) |
181/1502 (12.1%) |
92.0 (90.5, 93.3) |
90.2 (88.5, 91.6) |
0.77 (0.62, 0.96) |
| Adjuvant Chemotherapy Regimen | |||||
| Anthracycline | 139/1865 (7.4%) |
171/1877 (9.1%) |
93.8 (92.6, 94.8) |
93.0 (91.8, 94.1) |
0.82 (0.66, 1.03) |
| Non-Anthracycline | 32/535 (6.0%) |
39/527 (7.4%) |
94.9 (92.6, 96.6) |
94.0 (91.5, 95.8) |
0.82 (0.51, 1.31) |
2.3 Recommended Dosage and Administration
The initial dose of PERJETA is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks by a dose of 420 mg administered as an intravenous infusion over 30 to 60 minutes.
When administered with PERJETA, the recommended initial dose of trastuzumab is 8 mg/kg administered as a 90-minute intravenous infusion, followed every 3 weeks by a dose of 6 mg/kg administered as an intravenous infusion over 30 to 90 minutes.
When administered with PERJETA, the recommended initial dose of trastuzumab hyaluronidase-oysk is 600 mg/10,000 units (600 mg trastuzumab and 10,000 units hyaluronidase) administered subcutaneously over approximately 2 to 5 minutes once every three weeks irrespective of the patient's body weight.
Administer PERJETA, trastuzumab or trastuzumab hyaluronidase-oysk, and taxane sequentially. PERJETA and trastuzumab or trastuzumab hyaluronidase-oysk can be given in any order. Administer taxane after PERJETA and trastuzumab or trastuzumab hyaluronidase-oysk. An observation period of 30 to 60 minutes is recommended after each PERJETA infusion and before commencement of any subsequent administration of trastuzumab or trastuzumab hyaluronidase-oysk, or taxane [see Warnings and Precautions (5.3)].
In patients receiving an anthracycline-based regimen, administer PERJETA and trastuzumab or trastuzumab hyaluronidase-oysk after completion of the anthracycline.
5.4 Hypersensitivity Reactions/anaphylaxis (5.4 Hypersensitivity Reactions/Anaphylaxis)
PERJETA can cause hypersensitivity reactions, including anaphylaxis.
In CLEOPATRA, the overall frequency of hypersensitivity/anaphylaxis reactions was 11% in PERJETA-treated patients, with Grade 3 – 4 hypersensitivity reactions and anaphylaxis occurring in 2% of patients.
In NeoSphere, TRYPHAENA, BERENICE, and APHINITY, hypersensitivity/anaphylaxis events were consistent with those observed in CLEOPATRA. In APHINITY, the overall frequency of hypersensitivity/anaphylaxis was 5% in the PERJETA treated group. The incidence was highest in the PERJETA plus TCH treated group (8%) with 1% Grade 3 – 4 events.
Observe patients closely for hypersensitivity reactions. Severe hypersensitivity, including anaphylaxis and fatal events, have been observed in patients treated with PERJETA [see Clinical Trials Experience (6.1)]. Angioedema has been described in post-marketing reports. Medications to treat such reactions, as well as emergency equipment, should be available for immediate use prior to administration of PERJETA. PERJETA is contraindicated in patients with known hypersensitivity to pertuzumab or to any of its excipients [see Contraindications (4)].
Principal Display Panel 14 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 14 mL Vial Carton)
NDC 50242-145-01
Perjeta®
(Pertuzumab)
Injection
420 mg/ 14 mL
(30 mg/mL)
Dilute Prior To Use
For Intravenous Infusion Only
Single-Dose Vial
Discard Unused Portion
No preservative.
Rx only
1 vial
Genentech
11029352
2.1 Evaluation and Testing Before Initiating Perjeta
Assess left ventricular ejection fraction (LVEF) prior to initiation of PERJETA and at regular intervals during treatment [see Boxed Warning, Dosage and Administration (2.4), Warnings and Precautions (5.1)].
Verify the pregnancy status of females of reproductive potential prior to the initiation of PERJETA [see Warnings and Precautions (5.2), Use in Specific Populations (8.1, 8.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of pertuzumab.
Studies have not been performed to evaluate the mutagenic potential of pertuzumab.
No specific fertility studies in animals have been performed to evaluate the effect of pertuzumab. No adverse effects on male and female reproductive organs were observed in repeat-dose toxicity studies of up to six months duration in cynomolgus monkeys.
Warning: Left Ventricular Dysfunction and Embryo Fetal Toxicity (WARNING: LEFT VENTRICULAR DYSFUNCTION and EMBRYO-FETAL TOXICITY)
- Left Ventricular Dysfunction: PERJETA can cause subclinical and clinical cardiac failure manifesting as decreased LVEF and CHF. Evaluate cardiac function prior to and during treatment. Discontinue PERJETA treatment for a confirmed clinically significant decrease in left ventricular function [see Dosage and Administration (2.3), Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
- Embryo-fetal Toxicity: Exposure to PERJETA can cause embryo-fetal death and birth defects. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1) (8.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:56.009276 · Updated: 2026-03-14T22:38:58.661986