17f49334-1ad3-1d81-e063-6294a90afa14
17f49334-1ad3-1d81-e063-6294a90afa14
34390-5
HUMAN OTC DRUG LABEL
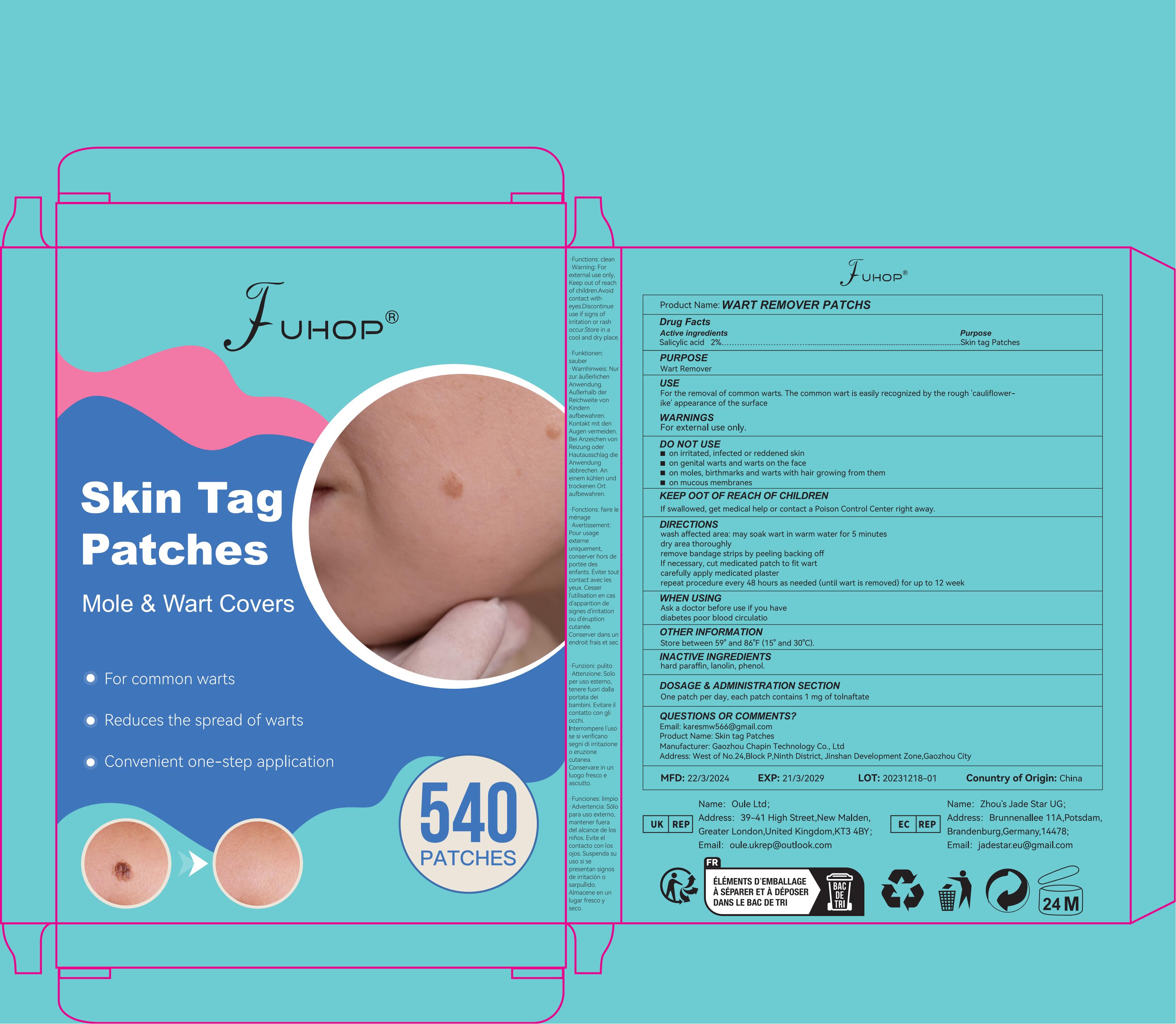
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Wart Remover
Description
Salicylic acid 20%
Medication Information
Warnings
For external use only.
Purpose
Wart Remover
Directions
wash affected area: may soak wart in warm water for 5 minutes
dry area thoroughly
remove bandage strips by peeling backing off
If necessary, cut medicated patch to fit wart
carefully apply medicated plaster
repeat procedure every 48 hours as needed (until wart is removed) for up to 12 week
Other Information
Store between 59° and 86°F (15° and 30°C).
Inactive Ingredients
Ethylhexyl Acrylate ·Polyethylene Terephthalate
Description
Salicylic acid 20%
Use
For the removal of common warts. The common wart is easily recognized by the rough ‘cauliflower-like’ appearance of the surface.
Section 51945-4
Do Not Use
on irritated, infected or reddened skin
on genital warts and warts on the face
on moles, birthmarks and warts with hair growing from them
on mucous membranes
When Using
Ask a doctor before use if you have
diabetes poor blood circulatio
Active Ingredient
Salicylic acid 20%
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away
Dosage & Administration Section
One patch per day, each patch contains1 mg of tolnaftate
Structured Label Content
Warnings (WARNINGS)
For external use only.
Purpose (PURPOSE)
Wart Remover
Directions (DIRECTIONS)
wash affected area: may soak wart in warm water for 5 minutes
dry area thoroughly
remove bandage strips by peeling backing off
If necessary, cut medicated patch to fit wart
carefully apply medicated plaster
repeat procedure every 48 hours as needed (until wart is removed) for up to 12 week
Other Information (OTHER INFORMATION)
Store between 59° and 86°F (15° and 30°C).
Inactive Ingredients (INACTIVE INGREDIENTS)
Ethylhexyl Acrylate ·Polyethylene Terephthalate
Use (USE)
For the removal of common warts. The common wart is easily recognized by the rough ‘cauliflower-like’ appearance of the surface.
Section 51945-4 (51945-4)
Do Not Use (DO NOT USE)
on irritated, infected or reddened skin
on genital warts and warts on the face
on moles, birthmarks and warts with hair growing from them
on mucous membranes
When Using (WHEN USING)
Ask a doctor before use if you have
diabetes poor blood circulatio
Active Ingredient (ACTIVE INGREDIENT)
Salicylic acid 20%
Keep Out of Reach of Children (KEEP OUT OF REACH OF CHILDREN)
If swallowed, get medical help or contact a Poison Control Center right away
Dosage & Administration Section (DOSAGE & ADMINISTRATION SECTION)
One patch per day, each patch contains1 mg of tolnaftate
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:03.886518 · Updated: 2026-03-14T23:02:55.861635