These Highlights Do Not Include All The Information Needed To Use Aristada®
17a8d11b-73b0-4833-a0b4-cf1ef85edefb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions ( 5.1 )] .
Indications and Usage
ARISTADA is indicated for the treatment of schizophrenia in adults [see Clinical Studies ( 14 )].
Dosage and Administration
Administer ARISTADA by intramuscular injection in the deltoid (441 mg dose only) or gluteal (441 mg, 662 mg, 882 mg or 1064 mg) muscle by a healthcare professional ( 2.1 ). For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ARISTADA ( 2.1 ). There are two options for initiating treatment with ARISTADA: Option #1: Administer one injection of 675 mg of ARISTADA INITIO ® and one 30 mg dose of oral aripiprazole in conjunction with the first ARISTADA injection. ( 2.1 ). Option #2: Administer 21 consecutive days of oral aripiprazole in conjunction with the first ARISTADA injection ( 2.1 ). ARISTADA can be initiated at a dose of 441 mg, 662 mg or 882 mg administered monthly, 882 mg dose every 6 weeks, or 1064 mg dose every 2 months ( 2.1 ). Dosing regimen adjustments may be required for missed doses ( 2.2 ). Dose adjustments are required for 1) known CYP2D6 poor metabolizers and 2) for patients taking CYP3A4 inhibitors, CYP2D6 inhibitors, or CYP3A4 inducers for more than 2 weeks ( 2.4 ).
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemia attack, including fatalities) ( 5.2 ). Potential for Dosing and Medication Errors : Substitution and dispensing errors between ARISTADA and ARISTADA INITIO could occur. Do not substitute ARISTADA INITIO for ARISTADA ( 5.3 ). Neuroleptic Malignant Syndrome : Manage with immediate discontinuation and close monitoring ( 5.4 ). Tardive Dyskinesia : Discontinue if clinically appropriate ( 5.5 ). Metabolic Changes : Monitor for hyperglycemia, dyslipidemia, and weight gain ( 5.6 ). Pathological Gambling and Other Compulsive Behaviors : Consider dose reduction or discontinuation ( 5.7 ). Orthostatic Hypotension : Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope ( 5.8 ). Leukopenia, Neutropenia, and Agranulocytosis : Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors ( 5.10 ). Seizures : Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold ( 5.11 ). Potential for Cognitive and Motor Impairment : Use caution when operating machinery ( 5.12 ).
Contraindications
ARISTADA is contraindicated in patients with a known hypersensitivity reaction to aripiprazole. Hypersensitivity reactions have ranged from pruritus/urticaria to anaphylaxis [see Adverse Reactions ( 6 )].
Adverse Reactions
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly patients with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated patients. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning , Warnings and Precautions ( 5.1 )].
How Supplied
ARISTADA extended-release injectable suspension is available in strengths of 441 mg in 1.6 mL, 662 mg in 2.4 mL, 882 mg in 3.2 mL and 1064 mg in 3.9 mL. The kit contains a 5-mL pre-filled syringe containing ARISTADA as a sterile white to off-white aqueous extended-release injectable suspension with safety needles. The 441 mg strength kit (NDC 65757-401-03 ; light blue label ) contains three safety needles; a 1-inch (25 mm) 21 gauge, a 1½-inch (38 mm) 20 gauge, and a 2-inch (50 mm) 20 gauge needle. The 662 mg strength kit (NDC 65757-402-03 ; green label ) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle. The 882 mg strength kit (NDC 65757-403-03 ; burgundy label ) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle. The 1064 mg strength kit (NDC 65757-404-03 ; dark blue label ) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
Medication Information
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemia attack, including fatalities) ( 5.2 ). Potential for Dosing and Medication Errors : Substitution and dispensing errors between ARISTADA and ARISTADA INITIO could occur. Do not substitute ARISTADA INITIO for ARISTADA ( 5.3 ). Neuroleptic Malignant Syndrome : Manage with immediate discontinuation and close monitoring ( 5.4 ). Tardive Dyskinesia : Discontinue if clinically appropriate ( 5.5 ). Metabolic Changes : Monitor for hyperglycemia, dyslipidemia, and weight gain ( 5.6 ). Pathological Gambling and Other Compulsive Behaviors : Consider dose reduction or discontinuation ( 5.7 ). Orthostatic Hypotension : Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope ( 5.8 ). Leukopenia, Neutropenia, and Agranulocytosis : Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors ( 5.10 ). Seizures : Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold ( 5.11 ). Potential for Cognitive and Motor Impairment : Use caution when operating machinery ( 5.12 ).
Indications and Usage
ARISTADA is indicated for the treatment of schizophrenia in adults [see Clinical Studies ( 14 )].
Dosage and Administration
Administer ARISTADA by intramuscular injection in the deltoid (441 mg dose only) or gluteal (441 mg, 662 mg, 882 mg or 1064 mg) muscle by a healthcare professional ( 2.1 ). For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ARISTADA ( 2.1 ). There are two options for initiating treatment with ARISTADA: Option #1: Administer one injection of 675 mg of ARISTADA INITIO ® and one 30 mg dose of oral aripiprazole in conjunction with the first ARISTADA injection. ( 2.1 ). Option #2: Administer 21 consecutive days of oral aripiprazole in conjunction with the first ARISTADA injection ( 2.1 ). ARISTADA can be initiated at a dose of 441 mg, 662 mg or 882 mg administered monthly, 882 mg dose every 6 weeks, or 1064 mg dose every 2 months ( 2.1 ). Dosing regimen adjustments may be required for missed doses ( 2.2 ). Dose adjustments are required for 1) known CYP2D6 poor metabolizers and 2) for patients taking CYP3A4 inhibitors, CYP2D6 inhibitors, or CYP3A4 inducers for more than 2 weeks ( 2.4 ).
Contraindications
ARISTADA is contraindicated in patients with a known hypersensitivity reaction to aripiprazole. Hypersensitivity reactions have ranged from pruritus/urticaria to anaphylaxis [see Adverse Reactions ( 6 )].
Adverse Reactions
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly patients with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated patients. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning , Warnings and Precautions ( 5.1 )].
How Supplied
ARISTADA extended-release injectable suspension is available in strengths of 441 mg in 1.6 mL, 662 mg in 2.4 mL, 882 mg in 3.2 mL and 1064 mg in 3.9 mL. The kit contains a 5-mL pre-filled syringe containing ARISTADA as a sterile white to off-white aqueous extended-release injectable suspension with safety needles. The 441 mg strength kit (NDC 65757-401-03 ; light blue label ) contains three safety needles; a 1-inch (25 mm) 21 gauge, a 1½-inch (38 mm) 20 gauge, and a 2-inch (50 mm) 20 gauge needle. The 662 mg strength kit (NDC 65757-402-03 ; green label ) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle. The 882 mg strength kit (NDC 65757-403-03 ; burgundy label ) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle. The 1064 mg strength kit (NDC 65757-404-03 ; dark blue label ) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions ( 5.1 )] .
Section 42229-5
Hyperglycemia/ Diabetes Mellitus
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. There have been reports of hyperglycemia in patients treated with oral aripiprazole. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Given these confounders, the relationship between atypical antipsychotic use and hyperglycemia-related adverse events is not completely understood. However, epidemiological studies suggest an increased risk of hyperglycemia-related adverse reactions in patients treated with the atypical antipsychotics.
Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should be monitored regularly for worsening of glucose control. Patients with risk factors for diabetes mellitus (e.g., obesity, family history of diabetes) who are starting treatment with atypical antipsychotics should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients require continuation of anti-diabetic treatment despite discontinuation of the suspect drug.
In the long-term, open-label schizophrenia study with ARISTADA, 14% of patients with normal hemoglobin A1c (<5.7%) at baseline developed elevated levels (≥5.7%) post-baseline.
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised 12/2023 |
|
|
MEDICATION GUIDE
ARISTADA ® (air-is-TAH-dah) (aripiprazole lauroxil) extended-release injectable suspension, for intramuscular use |
||
|
What is the most important information I should know about ARISTADA?
|
||
|
What is ARISTADA?
ARISTADA is a prescription medicine used to treat schizophrenia in adults. It is not known if ARISTADA is safe and effective in children under 18 years of age. |
||
| Do not receive ARISTADA if you are allergic to aripiprazole or any of the ingredients in ARISTADA. See the end of this Medication Guide for a complete list of ingredients in ARISTADA. | ||
Before receiving ARISTADA, tell your healthcare provider about all of your medical conditions, including if you:
ARISTADA and other medicines may affect each other causing possible serious side effects. ARISTADA may affect the way other medicines work, and other medicines may affect how ARISTADA works. Your healthcare provider can tell you if it is safe to receive ARISTADA with your other medicines. Do not start or stop any medicines while receiving ARISTADA without talking to your healthcare provider first. |
||
How should I receive ARISTADA?
|
||
|
What should I avoid while receiving ARISTADA?
|
||
|
What are the possible side effects of ARISTADA?
ARISTADA may cause serious side effects, including:
|
||
These are not all the possible side effects of ARISTADA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about ARISTADA
If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about ARISTADA that is written for health professionals. |
||
|
What are the ingredients in ARISTADA?
Active ingredient: aripiprazole lauroxil Inactive ingredients: sorbitan monolaurate, polysorbate 20, sodium chloride, sodium phosphate dibasic anhydrous, sodium phosphate monobasic dihydrate and water for injection Manufactured and marketed by: Alkermes, Inc., Waltham, MA 02451 For more information, go to www.ARISTADA.com or call 1-866-274-7823. |
Section 51945-4
Principal Display Panel - 441 mg/1.6 mL Carton Label
NDC: 65757-401-03
Rx only
Aristada
®
aripiprazole lauroxil
extended-release injectable suspension
441 mg
For deltoid or gluteal intramuscular injection only
Single-dose injection - Entire Content of Syringe Must Be
Administered by Healthcare Professional Only
441 mg/1.6 mL administered monthly.
For dosing and administration instructions, please
see accompanying full prescribing information.
Dispense enclosed Medication Guide to Patient.
Store at room temperature 20°C to 25°C (68°F to 77°F)
with excursions permitted between 15°C to 30°C (59°F to 86°F)
Keep out of reach of children
5.9 Falls
Antipsychotics including ARISTADA may cause somnolence, postural hypotension, or motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for those patients on long-term antipsychotic therapy.
16.2 Storage
Store at room temperature 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (between 59°F and 86°F).
5.11 Seizures
As with other antipsychotic drugs, use ARISTADA cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in a population of 65 years or older.
11 Description
ARISTADA contains aripiprazole lauroxil, an atypical antipsychotic.
The chemical name of aripiprazole lauroxil is 7-{4-[4-(2,3-dichlorophenyl)-piperazin-1-yl]butoxy}-2-oxo-3,4-dihydro-2H-quinolin-1-yl)methyl dodecanoate. The empirical formula is C36H51Cl2N3O4 and its molecular weight is 660.7 g/mol. The chemical structure is:
ARISTADA is available as a white to off-white sterile aqueous extended-release injectable suspension for intramuscular injection in the following strengths of aripiprazole lauroxil (and deliverable volumes from a single-dose pre-filled syringe): 441 mg (1.6 mL), 662 mg (2.4 mL), 882 mg (3.2 mL) and 1064 mg (3.9 mL). The inactive ingredients include sorbitan monolaurate (3.8 mg/mL), polysorbate 20 (1.5 mg/mL), sodium chloride (6.1 mg/mL), sodium phosphate dibasic anhydrous (0.62 mg/mL), sodium phosphate monobasic dihydrate (0.52 mg/mL) and water for injection.
5.14 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. ARISTADA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia.
2.2 Missed Doses
When a dose of ARISTADA is missed, administer the next injection of ARISTADA as soon as possible. Depending on the time elapsed since the last ARISTADA injection, supplement the next ARISTADA injection as recommended in Table 3 below.
| Dose of Patient's Last ARISTADA Injection | Length of Time Since Last Injection | ||
|---|---|---|---|
|
a The patient should supplement with the same dose of oral aripiprazole as when the patient began ARISTADA (see Table 2). |
|||
| 441 mg | ≤ 6 weeks | > 6 and ≤ 7 weeks | > 7 weeks |
| 662 mg | ≤ 8 weeks | > 8 and ≤ 12 weeks | > 12 weeks |
| 882 mg | ≤ 8 weeks | > 8 and ≤ 12 weeks | > 12 weeks |
| 1064 mg | ≤ 10 weeks | > 10 and ≤ 12 weeks | > 12 weeks |
| Dosage and Administration for Re-initiation of ARISTADA | No Supplementation Required | Supplement with a Single Dose of ARISTADA INITIO OR 7 Days of Oral Aripiprazolea | Re-initiate with a Single Dose of ARISTADA INITIO and a Single Dose of Oral Aripiprazole 30 mg OR supplement with 21 Days of Oral Aripiprazolea |
2.3 Early Dosing
The recommended ARISTADA dosing interval is monthly for the 441 mg, 662 mg and 882 mg doses, every 6 weeks for the 882 mg dose, or every 2 months for the 1064 mg dose and should be maintained. In the event of early dosing, an ARISTADA injection should not be given earlier than 14 days after the previous injection.
16.1 How Supplied
ARISTADA extended-release injectable suspension is available in strengths of 441 mg in 1.6 mL, 662 mg in 2.4 mL, 882 mg in 3.2 mL and 1064 mg in 3.9 mL. The kit contains a 5-mL pre-filled syringe containing ARISTADA as a sterile white to off-white aqueous extended-release injectable suspension with safety needles.
- The 441 mg strength kit (NDC 65757-401-03; light blue label) contains three safety needles; a 1-inch (25 mm) 21 gauge, a 1½-inch (38 mm) 20 gauge, and a 2-inch (50 mm) 20 gauge needle.
- The 662 mg strength kit (NDC 65757-402-03; green label) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
- The 882 mg strength kit (NDC 65757-403-03; burgundy label) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
- The 1064 mg strength kit (NDC 65757-404-03; dark blue label) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
8.4 Pediatric Use
Safety and effectiveness of ARISTADA in patients <18 years of age have not been evaluated.
8.5 Geriatric Use
Safety and effectiveness of ARISTADA in patients >65 years of age have not been evaluated.
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1, 5.2)].
4 Contraindications
ARISTADA is contraindicated in patients with a known hypersensitivity reaction to aripiprazole. Hypersensitivity reactions have ranged from pruritus/urticaria to anaphylaxis [see Adverse Reactions (6)].
6 Adverse Reactions
The following are discussed in more details in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-related Psychosis [see Boxed Warning, Warnings and Precautions ( 5.1 )]
- Cerebrovascular Adverse Reactions, Including Stroke [see Boxed Warning, Warnings and Precautions ( 5.2 )]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions ( 5.4 )]
- Tardive Dyskinesia [see Warnings and Precautions ( 5.5 )]
- Metabolic Changes [see Warnings and Precautions ( 5.6 )]
- Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions ( 5.7 )]
- Orthostatic Hypotension [see Warnings and Precautions ( 5.8 )]
- Falls [see Warnings and Precautions ( 5.9 )]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions ( 5.10 )]
- Seizures [see Warnings and Precautions ( 5.11 )]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions ( 5.12 )]
- Body Temperature Regulation [see Warnings and Precautions ( 5.13 )]
- Dysphagia [see Warnings and Precautions ( 5.14 )]
10.1 Human Experience
Common adverse reactions (reported in at least 5% of all overdose cases) reported with oral aripiprazole overdosage (alone or in combination with other substances) include vomiting, somnolence, and tremor. Other clinically important signs and symptoms observed in one or more patients with aripiprazole overdoses (alone or with other substances) include acidosis, aggression, aspartate aminotransferase increased, atrial fibrillation, bradycardia, coma, confusional state, convulsion, blood creatine phosphokinase increased, depressed level of consciousness, hypertension, hypokalemia, hypotension, lethargy, loss of consciousness, QRS complex prolonged, QT prolonged, pneumonia aspiration, respiratory arrest, status epilepticus, and tachycardia.
12.2 Pharmacodynamics
Aripiprazole exhibits high affinity for dopamine D2 and D3 (Kis 0.34 and 0.8 nM respectively), serotonin 5-HT1A and 5-HT2A receptors (Kis 1.7 and 3.4 nM respectively), moderate affinity for dopamine D4, serotonin 5-HT2C and 5-HT7, alpha1-adrenergic and histamine H1 receptors (Kis 44 nM, 15 nM, 39 nM, 57 nM, and 61 nM, respectively), and moderate affinity for the serotonin reuptake site (Ki 98 nM). Aripiprazole has no appreciable affinity for cholinergic muscarinic receptors (IC50 > 1000 nM). Actions at receptors other than D2, 5-HT1A, and 5-HT2A could explain some of the adverse reactions of aripiprazole (e.g., the orthostatic hypotension observed with aripiprazole may be explained by its antagonist activity at adrenergic alpha1 receptors).
12.3 Pharmacokinetics
ARISTADA is a prodrug of aripiprazole and its activity is primarily due to aripiprazole, and to a lesser extent dehydro-aripiprazole (major metabolite of aripiprazole), which has been shown to have affinities for D2 receptors similar to aripiprazole and represents 30-40% of the aripiprazole exposure in plasma.
5.6 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that include hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. While all drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
2.1 Recommended Dosage
ARISTADA is only to be administered as an intramuscular injection by a healthcare professional. For patients who have never taken aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ARISTADA. Due to the half-life of oral aripiprazole, it may take up to 2 weeks to fully assess tolerability. Refer to the prescribing information of oral aripiprazole for the recommended dosage and administration of the oral formulation.
There are two ways to initiate treatment with ARISTADA:
- Option #1: Administer one intramuscular injection of ARISTADA INITIO 675 mg (in either the deltoid or gluteal muscle) and one dose of oral aripiprazole 30 mg in conjunction with the first ARISTADA injection.
- The first ARISTADA injection may be administered on the same day as ARISTADA INITIO or up to 10 days thereafter. See the ARISTADA INITIO prescribing information for additional information regarding administration of ARISTADA INITIO.
- Avoid injecting both ARISTADA INITIO and ARISTADA concomitantly into the same deltoid or gluteal muscle.
- Option #2: Administer 21 consecutive days of oral aripiprazole in conjunction with the first ARISTADA injection.
Depending on individual patient's needs, treatment with ARISTADA can be initiated at a dose of 441 mg, 662 mg or 882 mg administered monthly, 882 mg administered every 6 weeks or 1064 mg administered every 2 months. The 441 mg, 662 mg, 882 mg and 1064 mg doses correspond to 300 mg, 450 mg, 600 mg and 724 mg of aripiprazole, respectively [see Clinical Pharmacology (12.3)].
| Dose | Dosing Frequency | Site of Intramuscular Injection |
|---|---|---|
| 441 mg | Monthly | Deltoid or Gluteal |
| 662 mg | Monthly | Gluteal |
| 882 mg | Monthly or every 6 weeks | Gluteal |
| 1064 mg | Every 2 months | Gluteal |
Use the following ARISTADA doses for patients who are stabilized on oral aripiprazole, as shown in Table 2.
| Oral Aripiprazole Dose | Intramuscular ARISTADA Dose |
|---|---|
| 10 mg per day | 441 mg every month |
| 15 mg per day | 662 mg every month 882 mg every 6 weeks 1064 mg every 2 months |
| 20 mg or higher per day | 882 mg every month |
In conjunction with the first ARISTADA injection, administer a single injection of ARISTADA INITIO and one dose of oral aripiprazole 30 mg, or continue treatment with oral aripiprazole for 21 consecutive days [see Recommended Dosage ( 2.1)].
Adjust the ARISTADA dose as needed. When making dose and dosing interval adjustments, consider the pharmacokinetics and prolonged-release characteristics of ARISTADA [see Clinical Pharmacology ( 12.3 )].
5.5 Tardive Dyskinesia
A syndrome of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase, but the syndrome can develop after relatively brief treatment periods at low doses, although this is uncommon.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome and may thus mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, ARISTADA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that is known to respond to antipsychotic drugs. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient treated with ARISTADA drug discontinuation should be considered. However, some patients may require treatment with ARISTADA despite the presence of the syndrome.
1 Indications and Usage
ARISTADA is indicated for the treatment of schizophrenia in adults [see Clinical Studies (14)].
12.1 Mechanism of Action
Aripiprazole lauroxil is a prodrug of aripiprazole. Following intramuscular injection, aripiprazole lauroxil is likely converted by enzyme-mediated hydrolysis to N-hydroxymethyl aripiprazole, which is then hydrolyzed to aripiprazole. The mechanism of action of aripiprazole in schizophrenia is unknown. However, efficacy could be mediated through a combination of partial agonist activity at dopamine D2 and serotonin 5-HT1A receptors and antagonist activity at 5-HT2A receptors.
5 Warnings and Precautions
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemia attack, including fatalities) (5.2).
- Potential for Dosing and Medication Errors: Substitution and dispensing errors between ARISTADA and ARISTADA INITIO could occur. Do not substitute ARISTADA INITIO for ARISTADA (5.3).
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring (5.4).
- Tardive Dyskinesia: Discontinue if clinically appropriate (5.5).
- Metabolic Changes: Monitor for hyperglycemia, dyslipidemia, and weight gain (5.6).
- Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation (5.7).
- Orthostatic Hypotension: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope (5.8).
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors (5.10).
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold (5.11).
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery (5.12).
2 Dosage and Administration
- Administer ARISTADA by intramuscular injection in the deltoid (441 mg dose only) or gluteal (441 mg, 662 mg, 882 mg or 1064 mg) muscle by a healthcare professional (2.1).
- For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ARISTADA (2.1).
- There are two options for initiating treatment with ARISTADA:
- ARISTADA can be initiated at a dose of 441 mg, 662 mg or 882 mg administered monthly, 882 mg dose every 6 weeks, or 1064 mg dose every 2 months (2.1).
- Dosing regimen adjustments may be required for missed doses (2.2).
- Dose adjustments are required for 1) known CYP2D6 poor metabolizers and 2) for patients taking CYP3A4 inhibitors, CYP2D6 inhibitors, or CYP3A4 inducers for more than 2 weeks (2.4).
5.8 Orthostatic Hypotension
Aripiprazole may cause orthostatic hypotension, perhaps due to its α1-adrenergic receptor antagonism. Associated adverse reactions related to orthostatic hypotension can include dizziness, lightheadedness and tachycardia. Generally, these risks are greatest at the beginning of treatment and during dose escalation. Patients at increased risk of these adverse reactions or at increased risk of developing complications from hypotension include those with dehydration, hypovolemia, treatment with antihypertensive medication, history of cardiovascular disease (e.g., heart failure, myocardial infarction, ischemia, or conduction abnormalities), history of cerebrovascular disease, as well as patients who are antipsychotic-naïve. In such patients, consider using a lower starting dose, and monitor orthostatic vital signs.
Orthostatic hypotension was reported for one patient in the ARISTADA 882 mg group (0.5%) and no patients in the ARISTADA 441 mg and placebo groups in the 12-week schizophrenia efficacy study [see Clinical Studies (14)]. In the long-term open-label schizophrenia study, orthostatic hypotension was reported for 1 (0.2%) patient treated with ARISTADA. Orthostatic hypotension was defined as a decrease in systolic blood pressure ≥20 mmHg accompanied by an increase in heart rate ≥25 bpm when comparing standing to supine values.
3 Dosage Forms and Strengths
ARISTADA is a white to off-white aqueous extended-release injectable suspension provided in a single-dose pre-filled syringe.
ARISTADA is available as described in Table 6.
| Dose Strength | Volume | Inject Intramuscularly | Color Label |
|---|---|---|---|
| 441 mg | 1.6 mL | Deltoid or Gluteal Muscle | Light Blue |
| 662 mg | 2.4 mL | Gluteal Muscle Only | Green |
| 882 mg | 3.2 mL | Gluteal Muscle Only | Burgundy |
| 1064 mg | 3.9 mL | Gluteal Muscle Only | Dark Blue |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of oral aripiprazole. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure: occurrences of allergic reaction (anaphylactic reaction, angioedema, laryngospasm, pruritus/urticaria, or oropharyngeal spasm), pathological gambling, hiccups, blood glucose fluctuation, oculogyric crisis, drug reaction with eosinophilia and systemic symptoms (DRESS), and fecal incontinence.
8.6 Cyp2d6 Poor Metabolizers
Dosage adjustment is recommended in known CYP2D6 poor metabolizers due to high aripiprazole concentrations. Approximately 8% of Caucasians and 3-8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers (PM) [see Dosage and Administration ( 2.4 ), Clinical Pharmacology ( 12.3 )].
10.2 Management of Overdosage
In case of overdosage, call the Poison control center immediately at 1-800-222-1222.
8 Use in Specific Populations
8.8 Other Specific Populations
No dosage adjustment for ARISTADA is required on the basis of a patient's sex, race, or smoking status [see Clinical Pharmacology ( 12.3 )].
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.13 Body Temperature Regulation
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing ARISTADA for patients who will be experiencing conditions which may contribute to an elevation in core body temperature, (e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration).
8.7 Hepatic and Renal Impairment
No dosage adjustment for ARISTADA is required based on a patient's hepatic function (mild to severe hepatic impairment, Child-Pugh score between 5 and 15), or renal function (mild to severe renal impairment, glomerular filtration rate between 15 and 90 mL/minute) [see Clinical Pharmacology ( 12.3 )].
17 Patient Counseling Information
Advise patients to read FDA-approved patient labeling (Medication Guide).
5.4 Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) may occur in association with antipsychotic drugs, including ARISTADA. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases in which the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system pathology.
The management of NMS should include: (1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy; (2) intensive symptomatic treatment and medical monitoring; and (3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient appears to require antipsychotic drug treatment after recovery from NMS, reintroduction of drug therapy should be closely monitored, since recurrences of NMS have been reported.
2.5 Important Administration Instructions
The kit contains a syringe containing ARISTADA sterile aqueous extended-release injectable suspension and 2 or 3 safety needles depending on dose (a 2-inch 20 gauge needle with yellow needle hub, a 1 ½-inch 20 gauge needle with yellow needle hub, and a 1-inch 21 gauge needle with green needle hub (441 mg kit only)) for intramuscular injection. All materials should be stored at room temperature.
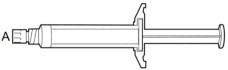
A | 5 mL syringe containing ARISTADA sterile aqueous extended-release injectable suspension
B | 20 gauge needle, 2-inch with yellow needle hub
C | 20 gauge needle, 1½-inch with yellow needle hub
D | 21 gauge needle, 1-inch with green needle hub
1. TAP and vigorously SHAKE the syringe.
1a. Tap the syringe at least 10 times to dislodge any material which may have settled.
1b. Shake the syringe vigorously for a minimum of 30 seconds to ensure a uniform suspension. If the syringe is not used within 15 minutes, shake again for 30 seconds.
2. SELECT the injection needle.
2a. Select injection site.
2b. Select needle length based on injection site. For patients with a larger amount of subcutaneous tissue overlaying the injection site muscle, use the longer of the needles provided.
| Injection Site | Needle Length |
| 441 mg dose | |
| Deltoid | 21 gauge, 1-inch or 20 gauge, 1½-inch |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
| 662 mg dose | |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
| 882 mg dose | |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
| 1064 mg dose | |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
[see Dosage and Administration ( 2.1 )]
3. ATTACH the injection needle.
Attach the appropriate needle securely with a clockwise twisting motion. Do NOT overtighten. Overtightening could lead to needle hub cracking.
4. PRIME the syringe to remove air.
4a. Bring the syringe into upright position and tap the syringe to bring air to the top.
4b. Depress the plunger rod to remove air until a few drops are released. It is normal to see small air bubbles remaining in the syringe.
5. Inject in a RAPID and CONTINUOUS manner. Product requires a RAPID injection. Do not hesitate. Administer the entire content intramuscularly. Do not inject by any other route.
6. DISPOSE of the needle. Cover the needle by pressing the safety device. Dispose of used and unused items in a proper waste container.
13.2 Animal Toxicology And/or Pharmacology
Intramuscular administration of aripiprazole lauroxil to rats and dogs was associated with injection site tissue reactions at all doses in rats treated up to 6 months at doses of 15, 29, and 103 mg/animal (which are approximately 0.3 to 2 times and 0.5 to 3 times the MRHD of 1064 mg on mg/m2 basis in males and females, respectively) and in dogs treated up to 9 months at doses of 147, 662, and 2058 mg/animal (which are approximately 0.5 to 6 times and 0.6 to 8 times the MRHD in males and females, respectively on mg/m2 basis). These injection site tissue reactions consisted of localized granulomatous inflammation and granuloma formation. Transiently impaired limb function and swelling occurred in dogs. The granulomas did not completely resolve 2 months following the last injection in the 6 month rat study and 4 months following the last injection in the 9 month dog study (the low dose groups were not examined for reversibility in these studies).
Orally administered aripiprazole produced retinal degeneration in albino rats in a 26-week chronic toxicity study at a dose of 60 mg/kg, which is 19 times the oral MRHD of 30 mg/day on mg/m2 basis, and in a 2-year carcinogenicity study at doses of 40 mg/kg and 60 mg/kg, which are 13 and 19 times the oral MRHD on mg/m2 basis and 7 to 14 times human exposure at the oral MRHD based on AUC. Evaluation of the retinas of albino mice and of monkeys did not reveal evidence of retinal degeneration. Additional studies to further evaluate the mechanism have not been performed. The relevance of this finding to human risk is unknown.
2.4 Dose Adjustments for Cyp450 Considerations
Refer to the prescribing information for oral aripiprazole for recommendations regarding dosage adjustments due to drug interactions, for the first 21 days when the patient is taking 21 days of oral aripiprazole concomitantly with the first dose of ARISTADA. Avoid initiating ARISTADA treatment with ARISTADA INITIO in patients requiring dose adjustments.
Once stabilized on ARISTADA, refer to the dosing recommendations below for patients taking strong CYP2D6 inhibitors, strong CYP3A4 inhibitors, or strong CYP3A4 inducers:
- No dosage changes recommended for ARISTADA, if CYP450 modulators are added for less than 2 weeks.
- Make dose changes to ARISTADA if CYP450 modulators are added for greater than 2 weeks (see Table 4).
| Concomitant Medicine | Dose Change for ARISTADA a |
|---|---|
|
a For the 882 mg dose administered every 6 weeks and the 1064 mg administered every 2 months, the next lower strength should be 441 mg administered monthly. |
|
| Strong CYP3A4 Inhibitor | Reduce the dose of ARISTADA to the next lower strength. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. For patients known to be poor metabolizers of CYP2D6: Reduce dose to 441 mg from 662 mg, 882 mg, or 1064 mg. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. |
| Strong CYP2D6 Inhibitor | Reduce the dose of ARISTADA to the next lower strength. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. For patients known to be poor metabolizers of CYP2D6: No dose adjustment required. |
| Both Strong CYP3A4 Inhibitor and Strong CYP2D6 Inhibitor | Avoid use for patients at 662 mg, 882 mg, or 1064 mg dose. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. |
| CYP3A4 Inducers | No dose adjustment for 662 mg, 882 mg, or 1064 mg dose; increase the 441 mg dose to 662 mg. |
5.3 Potential for Dosing and Medication Errors
Medication errors, including substitution and dispensing errors, between ARISTADA and ARISTADA INITIO could occur. ARISTADA INITIO is for single administration in contrast to ARISTADA which is administered monthly, every 6 weeks, or every 8 weeks [see Dosage and Administration (2.1)]. Do not substitute ARISTADA INITIO for ARISTADA because of differing pharmacokinetic profiles [see Clinical Pharmacology ( 12.3 )].
5.10 Leukopenia, Neutropenia, and Agranulocytosis
In clinical trials and/or postmarketing experience, events of leukopenia and neutropenia have been reported temporally related to antipsychotic agents. Agranulocytosis has also been reported.
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC)/absolute neutrophil count (ANC) and history of drug-induced leukopenia/neutropenia. In patients with a history of a clinically significant low WBC/ANC or drug-induced leukopenia/neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of ARISTADA at the first sign of a clinical significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue ARISTADA in patients with severe neutropenia (absolute neutrophil count <1000/mm3) and follow their WBC until recovery.
5.12 Potential for Cognitive and Motor Impairment
ARISTADA, like other antipsychotics, has the potential to impair judgment, thinking or motor skills. Patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that therapy with ARISTADA does not affect them adversely.
5.2 Cerebrovascular Adverse Reactions, Including Stroke
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly patients with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated patients. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions ( 5.1 )].
5.7 Pathological Gambling and Other Compulsive Behaviors
Post-marketing case reports suggest that patients can experience intense urges, particularly for gambling, and the inability to control these urges while taking aripiprazole. Other compulsive urges, reported less frequently include: sexual urges, shopping, eating or binge eating, and other impulsive or compulsive behaviors. Because patients may not recognize these behaviors as abnormal, it is important for prescribers to ask patients or their caregivers specifically about the development of new or intense gambling urges, compulsive sexual urges, compulsive shopping, binge or compulsive eating, or other urges while being treated with aripiprazole. It should be noted that impulse-control symptoms can be associated with the underlying disorder. In some cases, although not all, urges were reported to have stopped when the dose was reduced or the medication was discontinued. Compulsive behaviors may result in harm for the patient and others if not recognized. Consider dose reduction or stopping the medication if a patient develops such urges.
7.1 Drugs Having Clinically Important Interactions With Aristada
| Strong CYP3A4 Inhibitors and CYP2D6 Inhibitors | |
| Clinical Impact: | The concomitant use of oral aripiprazole with strong CYP3A4 or CYP2D6 inhibitors increased the exposure of aripiprazole compared to the use of oral aripiprazole alone [see Clinical Pharmacology ( 12.3 )]. |
| Intervention: | With concomitant use of ARISTADA with a strong CYP3A4 inhibitor or CYP2D6 inhibitor for more than 2 weeks, reduce the ARISTADA dose [see Dosage and Administration ( 2.4 )]. |
| Examples: | itraconazole, clarithromycin, quinidine, fluoxetine, paroxetine |
| Strong CYP3A4 Inducers | |
| Clinical Impact: | The concomitant use of oral aripiprazole and carbamazepine decreased the exposure of aripiprazole compared to the use of oral aripiprazole alone [see Clinical Pharmacology ( 12.3 )]. |
| Intervention: | With concomitant use of ARISTADA with a strong CYP3A4 inducer for more than 2 weeks consider increasing the ARISTADA dose [see Dosage and Administration ( 2.4 )]. |
| Examples: | carbamazepine, rifampin |
| Antihypertensive Drugs | |
| Clinical Impact: | Due to its alpha adrenergic antagonism, aripiprazole has the potential to enhance the effect of certain antihypertensive agents. |
| Intervention: | Monitor blood pressure and adjust dose accordingly [see Warnings and Precautions ( 5.8 )]. |
| Examples: | carvedilol, lisinopril, prazosin |
| Benzodiazepines | |
| Clinical Impact: | The intensity of sedation was greater with the combination of oral aripiprazole and lorazepam as compared to that observed with aripiprazole alone. The orthostatic hypotension observed was greater with the combination as compared to that observed with lorazepam alone [see Warnings and Precautions ( 5.8 )]. |
| Intervention: | Monitor sedation and blood pressure. Adjust dose accordingly. |
| Example: | lorazepam |
7.2 Drugs Having No Clinically Important Interactions With Aristada
Based on pharmacokinetic studies with oral aripiprazole, no dosage adjustment of ARISTADA is required when administered concomitantly with famotidine, valproate, or lithium [see Clinical Pharmacology ( 12.3 )].
In addition, no dosage adjustment is necessary for substrates of CYP2D6 (e.g., dextromethorphan, fluoxetine, paroxetine, or venlafaxine), CYP2C9 (e.g., warfarin), CYP2C19 (e.g., omeprazole, warfarin, escitalopram), or CYP3A4 (e.g., dextromethorphan) when co-administered with ARISTADA. Additionally, no dosage adjustment is necessary for valproate, lithium, lamotrigine, or sertraline when co-administered with ARISTADA [see Clinical Pharmacology ( 12.3 )].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group.
Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions ( 5.2 )].
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
Structured Label Content
Section 42229-5 (42229-5)
Hyperglycemia/ Diabetes Mellitus
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. There have been reports of hyperglycemia in patients treated with oral aripiprazole. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Given these confounders, the relationship between atypical antipsychotic use and hyperglycemia-related adverse events is not completely understood. However, epidemiological studies suggest an increased risk of hyperglycemia-related adverse reactions in patients treated with the atypical antipsychotics.
Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should be monitored regularly for worsening of glucose control. Patients with risk factors for diabetes mellitus (e.g., obesity, family history of diabetes) who are starting treatment with atypical antipsychotics should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients require continuation of anti-diabetic treatment despite discontinuation of the suspect drug.
In the long-term, open-label schizophrenia study with ARISTADA, 14% of patients with normal hemoglobin A1c (<5.7%) at baseline developed elevated levels (≥5.7%) post-baseline.
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised 12/2023 |
|
|
MEDICATION GUIDE
ARISTADA ® (air-is-TAH-dah) (aripiprazole lauroxil) extended-release injectable suspension, for intramuscular use |
||
|
What is the most important information I should know about ARISTADA?
|
||
|
What is ARISTADA?
ARISTADA is a prescription medicine used to treat schizophrenia in adults. It is not known if ARISTADA is safe and effective in children under 18 years of age. |
||
| Do not receive ARISTADA if you are allergic to aripiprazole or any of the ingredients in ARISTADA. See the end of this Medication Guide for a complete list of ingredients in ARISTADA. | ||
Before receiving ARISTADA, tell your healthcare provider about all of your medical conditions, including if you:
ARISTADA and other medicines may affect each other causing possible serious side effects. ARISTADA may affect the way other medicines work, and other medicines may affect how ARISTADA works. Your healthcare provider can tell you if it is safe to receive ARISTADA with your other medicines. Do not start or stop any medicines while receiving ARISTADA without talking to your healthcare provider first. |
||
How should I receive ARISTADA?
|
||
|
What should I avoid while receiving ARISTADA?
|
||
|
What are the possible side effects of ARISTADA?
ARISTADA may cause serious side effects, including:
|
||
These are not all the possible side effects of ARISTADA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about ARISTADA
If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about ARISTADA that is written for health professionals. |
||
|
What are the ingredients in ARISTADA?
Active ingredient: aripiprazole lauroxil Inactive ingredients: sorbitan monolaurate, polysorbate 20, sodium chloride, sodium phosphate dibasic anhydrous, sodium phosphate monobasic dihydrate and water for injection Manufactured and marketed by: Alkermes, Inc., Waltham, MA 02451 For more information, go to www.ARISTADA.com or call 1-866-274-7823. |
Section 51945-4 (51945-4)
Principal Display Panel - 441 mg/1.6 mL Carton Label
NDC: 65757-401-03
Rx only
Aristada
®
aripiprazole lauroxil
extended-release injectable suspension
441 mg
For deltoid or gluteal intramuscular injection only
Single-dose injection - Entire Content of Syringe Must Be
Administered by Healthcare Professional Only
441 mg/1.6 mL administered monthly.
For dosing and administration instructions, please
see accompanying full prescribing information.
Dispense enclosed Medication Guide to Patient.
Store at room temperature 20°C to 25°C (68°F to 77°F)
with excursions permitted between 15°C to 30°C (59°F to 86°F)
Keep out of reach of children
5.9 Falls
Antipsychotics including ARISTADA may cause somnolence, postural hypotension, or motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for those patients on long-term antipsychotic therapy.
16.2 Storage
Store at room temperature 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (between 59°F and 86°F).
5.11 Seizures
As with other antipsychotic drugs, use ARISTADA cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in a population of 65 years or older.
11 Description (11 DESCRIPTION)
ARISTADA contains aripiprazole lauroxil, an atypical antipsychotic.
The chemical name of aripiprazole lauroxil is 7-{4-[4-(2,3-dichlorophenyl)-piperazin-1-yl]butoxy}-2-oxo-3,4-dihydro-2H-quinolin-1-yl)methyl dodecanoate. The empirical formula is C36H51Cl2N3O4 and its molecular weight is 660.7 g/mol. The chemical structure is:
ARISTADA is available as a white to off-white sterile aqueous extended-release injectable suspension for intramuscular injection in the following strengths of aripiprazole lauroxil (and deliverable volumes from a single-dose pre-filled syringe): 441 mg (1.6 mL), 662 mg (2.4 mL), 882 mg (3.2 mL) and 1064 mg (3.9 mL). The inactive ingredients include sorbitan monolaurate (3.8 mg/mL), polysorbate 20 (1.5 mg/mL), sodium chloride (6.1 mg/mL), sodium phosphate dibasic anhydrous (0.62 mg/mL), sodium phosphate monobasic dihydrate (0.52 mg/mL) and water for injection.
5.14 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. ARISTADA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia.
2.2 Missed Doses
When a dose of ARISTADA is missed, administer the next injection of ARISTADA as soon as possible. Depending on the time elapsed since the last ARISTADA injection, supplement the next ARISTADA injection as recommended in Table 3 below.
| Dose of Patient's Last ARISTADA Injection | Length of Time Since Last Injection | ||
|---|---|---|---|
|
a The patient should supplement with the same dose of oral aripiprazole as when the patient began ARISTADA (see Table 2). |
|||
| 441 mg | ≤ 6 weeks | > 6 and ≤ 7 weeks | > 7 weeks |
| 662 mg | ≤ 8 weeks | > 8 and ≤ 12 weeks | > 12 weeks |
| 882 mg | ≤ 8 weeks | > 8 and ≤ 12 weeks | > 12 weeks |
| 1064 mg | ≤ 10 weeks | > 10 and ≤ 12 weeks | > 12 weeks |
| Dosage and Administration for Re-initiation of ARISTADA | No Supplementation Required | Supplement with a Single Dose of ARISTADA INITIO OR 7 Days of Oral Aripiprazolea | Re-initiate with a Single Dose of ARISTADA INITIO and a Single Dose of Oral Aripiprazole 30 mg OR supplement with 21 Days of Oral Aripiprazolea |
2.3 Early Dosing
The recommended ARISTADA dosing interval is monthly for the 441 mg, 662 mg and 882 mg doses, every 6 weeks for the 882 mg dose, or every 2 months for the 1064 mg dose and should be maintained. In the event of early dosing, an ARISTADA injection should not be given earlier than 14 days after the previous injection.
16.1 How Supplied
ARISTADA extended-release injectable suspension is available in strengths of 441 mg in 1.6 mL, 662 mg in 2.4 mL, 882 mg in 3.2 mL and 1064 mg in 3.9 mL. The kit contains a 5-mL pre-filled syringe containing ARISTADA as a sterile white to off-white aqueous extended-release injectable suspension with safety needles.
- The 441 mg strength kit (NDC 65757-401-03; light blue label) contains three safety needles; a 1-inch (25 mm) 21 gauge, a 1½-inch (38 mm) 20 gauge, and a 2-inch (50 mm) 20 gauge needle.
- The 662 mg strength kit (NDC 65757-402-03; green label) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
- The 882 mg strength kit (NDC 65757-403-03; burgundy label) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
- The 1064 mg strength kit (NDC 65757-404-03; dark blue label) contains two safety needles; a 1½-inch (38 mm) 20 gauge and a 2-inch (50 mm) 20 gauge needle.
8.4 Pediatric Use
Safety and effectiveness of ARISTADA in patients <18 years of age have not been evaluated.
8.5 Geriatric Use
Safety and effectiveness of ARISTADA in patients >65 years of age have not been evaluated.
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1, 5.2)].
4 Contraindications (4 CONTRAINDICATIONS)
ARISTADA is contraindicated in patients with a known hypersensitivity reaction to aripiprazole. Hypersensitivity reactions have ranged from pruritus/urticaria to anaphylaxis [see Adverse Reactions (6)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are discussed in more details in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-related Psychosis [see Boxed Warning, Warnings and Precautions ( 5.1 )]
- Cerebrovascular Adverse Reactions, Including Stroke [see Boxed Warning, Warnings and Precautions ( 5.2 )]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions ( 5.4 )]
- Tardive Dyskinesia [see Warnings and Precautions ( 5.5 )]
- Metabolic Changes [see Warnings and Precautions ( 5.6 )]
- Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions ( 5.7 )]
- Orthostatic Hypotension [see Warnings and Precautions ( 5.8 )]
- Falls [see Warnings and Precautions ( 5.9 )]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions ( 5.10 )]
- Seizures [see Warnings and Precautions ( 5.11 )]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions ( 5.12 )]
- Body Temperature Regulation [see Warnings and Precautions ( 5.13 )]
- Dysphagia [see Warnings and Precautions ( 5.14 )]
10.1 Human Experience
Common adverse reactions (reported in at least 5% of all overdose cases) reported with oral aripiprazole overdosage (alone or in combination with other substances) include vomiting, somnolence, and tremor. Other clinically important signs and symptoms observed in one or more patients with aripiprazole overdoses (alone or with other substances) include acidosis, aggression, aspartate aminotransferase increased, atrial fibrillation, bradycardia, coma, confusional state, convulsion, blood creatine phosphokinase increased, depressed level of consciousness, hypertension, hypokalemia, hypotension, lethargy, loss of consciousness, QRS complex prolonged, QT prolonged, pneumonia aspiration, respiratory arrest, status epilepticus, and tachycardia.
12.2 Pharmacodynamics
Aripiprazole exhibits high affinity for dopamine D2 and D3 (Kis 0.34 and 0.8 nM respectively), serotonin 5-HT1A and 5-HT2A receptors (Kis 1.7 and 3.4 nM respectively), moderate affinity for dopamine D4, serotonin 5-HT2C and 5-HT7, alpha1-adrenergic and histamine H1 receptors (Kis 44 nM, 15 nM, 39 nM, 57 nM, and 61 nM, respectively), and moderate affinity for the serotonin reuptake site (Ki 98 nM). Aripiprazole has no appreciable affinity for cholinergic muscarinic receptors (IC50 > 1000 nM). Actions at receptors other than D2, 5-HT1A, and 5-HT2A could explain some of the adverse reactions of aripiprazole (e.g., the orthostatic hypotension observed with aripiprazole may be explained by its antagonist activity at adrenergic alpha1 receptors).
12.3 Pharmacokinetics
ARISTADA is a prodrug of aripiprazole and its activity is primarily due to aripiprazole, and to a lesser extent dehydro-aripiprazole (major metabolite of aripiprazole), which has been shown to have affinities for D2 receptors similar to aripiprazole and represents 30-40% of the aripiprazole exposure in plasma.
5.6 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that include hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. While all drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
2.1 Recommended Dosage
ARISTADA is only to be administered as an intramuscular injection by a healthcare professional. For patients who have never taken aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ARISTADA. Due to the half-life of oral aripiprazole, it may take up to 2 weeks to fully assess tolerability. Refer to the prescribing information of oral aripiprazole for the recommended dosage and administration of the oral formulation.
There are two ways to initiate treatment with ARISTADA:
- Option #1: Administer one intramuscular injection of ARISTADA INITIO 675 mg (in either the deltoid or gluteal muscle) and one dose of oral aripiprazole 30 mg in conjunction with the first ARISTADA injection.
- The first ARISTADA injection may be administered on the same day as ARISTADA INITIO or up to 10 days thereafter. See the ARISTADA INITIO prescribing information for additional information regarding administration of ARISTADA INITIO.
- Avoid injecting both ARISTADA INITIO and ARISTADA concomitantly into the same deltoid or gluteal muscle.
- Option #2: Administer 21 consecutive days of oral aripiprazole in conjunction with the first ARISTADA injection.
Depending on individual patient's needs, treatment with ARISTADA can be initiated at a dose of 441 mg, 662 mg or 882 mg administered monthly, 882 mg administered every 6 weeks or 1064 mg administered every 2 months. The 441 mg, 662 mg, 882 mg and 1064 mg doses correspond to 300 mg, 450 mg, 600 mg and 724 mg of aripiprazole, respectively [see Clinical Pharmacology (12.3)].
| Dose | Dosing Frequency | Site of Intramuscular Injection |
|---|---|---|
| 441 mg | Monthly | Deltoid or Gluteal |
| 662 mg | Monthly | Gluteal |
| 882 mg | Monthly or every 6 weeks | Gluteal |
| 1064 mg | Every 2 months | Gluteal |
Use the following ARISTADA doses for patients who are stabilized on oral aripiprazole, as shown in Table 2.
| Oral Aripiprazole Dose | Intramuscular ARISTADA Dose |
|---|---|
| 10 mg per day | 441 mg every month |
| 15 mg per day | 662 mg every month 882 mg every 6 weeks 1064 mg every 2 months |
| 20 mg or higher per day | 882 mg every month |
In conjunction with the first ARISTADA injection, administer a single injection of ARISTADA INITIO and one dose of oral aripiprazole 30 mg, or continue treatment with oral aripiprazole for 21 consecutive days [see Recommended Dosage ( 2.1)].
Adjust the ARISTADA dose as needed. When making dose and dosing interval adjustments, consider the pharmacokinetics and prolonged-release characteristics of ARISTADA [see Clinical Pharmacology ( 12.3 )].
5.5 Tardive Dyskinesia
A syndrome of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase, but the syndrome can develop after relatively brief treatment periods at low doses, although this is uncommon.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome and may thus mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, ARISTADA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that is known to respond to antipsychotic drugs. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient treated with ARISTADA drug discontinuation should be considered. However, some patients may require treatment with ARISTADA despite the presence of the syndrome.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ARISTADA is indicated for the treatment of schizophrenia in adults [see Clinical Studies (14)].
12.1 Mechanism of Action
Aripiprazole lauroxil is a prodrug of aripiprazole. Following intramuscular injection, aripiprazole lauroxil is likely converted by enzyme-mediated hydrolysis to N-hydroxymethyl aripiprazole, which is then hydrolyzed to aripiprazole. The mechanism of action of aripiprazole in schizophrenia is unknown. However, efficacy could be mediated through a combination of partial agonist activity at dopamine D2 and serotonin 5-HT1A receptors and antagonist activity at 5-HT2A receptors.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemia attack, including fatalities) (5.2).
- Potential for Dosing and Medication Errors: Substitution and dispensing errors between ARISTADA and ARISTADA INITIO could occur. Do not substitute ARISTADA INITIO for ARISTADA (5.3).
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring (5.4).
- Tardive Dyskinesia: Discontinue if clinically appropriate (5.5).
- Metabolic Changes: Monitor for hyperglycemia, dyslipidemia, and weight gain (5.6).
- Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation (5.7).
- Orthostatic Hypotension: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope (5.8).
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell (WBC) count. Consider discontinuation if clinically significant decline in WBC in the absence of other causative factors (5.10).
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold (5.11).
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery (5.12).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer ARISTADA by intramuscular injection in the deltoid (441 mg dose only) or gluteal (441 mg, 662 mg, 882 mg or 1064 mg) muscle by a healthcare professional (2.1).
- For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ARISTADA (2.1).
- There are two options for initiating treatment with ARISTADA:
- ARISTADA can be initiated at a dose of 441 mg, 662 mg or 882 mg administered monthly, 882 mg dose every 6 weeks, or 1064 mg dose every 2 months (2.1).
- Dosing regimen adjustments may be required for missed doses (2.2).
- Dose adjustments are required for 1) known CYP2D6 poor metabolizers and 2) for patients taking CYP3A4 inhibitors, CYP2D6 inhibitors, or CYP3A4 inducers for more than 2 weeks (2.4).
5.8 Orthostatic Hypotension
Aripiprazole may cause orthostatic hypotension, perhaps due to its α1-adrenergic receptor antagonism. Associated adverse reactions related to orthostatic hypotension can include dizziness, lightheadedness and tachycardia. Generally, these risks are greatest at the beginning of treatment and during dose escalation. Patients at increased risk of these adverse reactions or at increased risk of developing complications from hypotension include those with dehydration, hypovolemia, treatment with antihypertensive medication, history of cardiovascular disease (e.g., heart failure, myocardial infarction, ischemia, or conduction abnormalities), history of cerebrovascular disease, as well as patients who are antipsychotic-naïve. In such patients, consider using a lower starting dose, and monitor orthostatic vital signs.
Orthostatic hypotension was reported for one patient in the ARISTADA 882 mg group (0.5%) and no patients in the ARISTADA 441 mg and placebo groups in the 12-week schizophrenia efficacy study [see Clinical Studies (14)]. In the long-term open-label schizophrenia study, orthostatic hypotension was reported for 1 (0.2%) patient treated with ARISTADA. Orthostatic hypotension was defined as a decrease in systolic blood pressure ≥20 mmHg accompanied by an increase in heart rate ≥25 bpm when comparing standing to supine values.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
ARISTADA is a white to off-white aqueous extended-release injectable suspension provided in a single-dose pre-filled syringe.
ARISTADA is available as described in Table 6.
| Dose Strength | Volume | Inject Intramuscularly | Color Label |
|---|---|---|---|
| 441 mg | 1.6 mL | Deltoid or Gluteal Muscle | Light Blue |
| 662 mg | 2.4 mL | Gluteal Muscle Only | Green |
| 882 mg | 3.2 mL | Gluteal Muscle Only | Burgundy |
| 1064 mg | 3.9 mL | Gluteal Muscle Only | Dark Blue |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of oral aripiprazole. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure: occurrences of allergic reaction (anaphylactic reaction, angioedema, laryngospasm, pruritus/urticaria, or oropharyngeal spasm), pathological gambling, hiccups, blood glucose fluctuation, oculogyric crisis, drug reaction with eosinophilia and systemic symptoms (DRESS), and fecal incontinence.
8.6 Cyp2d6 Poor Metabolizers (8.6 CYP2D6 Poor Metabolizers)
Dosage adjustment is recommended in known CYP2D6 poor metabolizers due to high aripiprazole concentrations. Approximately 8% of Caucasians and 3-8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers (PM) [see Dosage and Administration ( 2.4 ), Clinical Pharmacology ( 12.3 )].
10.2 Management of Overdosage
In case of overdosage, call the Poison control center immediately at 1-800-222-1222.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
8.8 Other Specific Populations
No dosage adjustment for ARISTADA is required on the basis of a patient's sex, race, or smoking status [see Clinical Pharmacology ( 12.3 )].
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.13 Body Temperature Regulation
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing ARISTADA for patients who will be experiencing conditions which may contribute to an elevation in core body temperature, (e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration).
8.7 Hepatic and Renal Impairment
No dosage adjustment for ARISTADA is required based on a patient's hepatic function (mild to severe hepatic impairment, Child-Pugh score between 5 and 15), or renal function (mild to severe renal impairment, glomerular filtration rate between 15 and 90 mL/minute) [see Clinical Pharmacology ( 12.3 )].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read FDA-approved patient labeling (Medication Guide).
5.4 Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) may occur in association with antipsychotic drugs, including ARISTADA. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases in which the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system pathology.
The management of NMS should include: (1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy; (2) intensive symptomatic treatment and medical monitoring; and (3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient appears to require antipsychotic drug treatment after recovery from NMS, reintroduction of drug therapy should be closely monitored, since recurrences of NMS have been reported.
2.5 Important Administration Instructions
The kit contains a syringe containing ARISTADA sterile aqueous extended-release injectable suspension and 2 or 3 safety needles depending on dose (a 2-inch 20 gauge needle with yellow needle hub, a 1 ½-inch 20 gauge needle with yellow needle hub, and a 1-inch 21 gauge needle with green needle hub (441 mg kit only)) for intramuscular injection. All materials should be stored at room temperature.
A | 5 mL syringe containing ARISTADA sterile aqueous extended-release injectable suspension
B | 20 gauge needle, 2-inch with yellow needle hub
C | 20 gauge needle, 1½-inch with yellow needle hub
D | 21 gauge needle, 1-inch with green needle hub
1. TAP and vigorously SHAKE the syringe.
1a. Tap the syringe at least 10 times to dislodge any material which may have settled.
1b. Shake the syringe vigorously for a minimum of 30 seconds to ensure a uniform suspension. If the syringe is not used within 15 minutes, shake again for 30 seconds.
2. SELECT the injection needle.
2a. Select injection site.
2b. Select needle length based on injection site. For patients with a larger amount of subcutaneous tissue overlaying the injection site muscle, use the longer of the needles provided.
| Injection Site | Needle Length |
| 441 mg dose | |
| Deltoid | 21 gauge, 1-inch or 20 gauge, 1½-inch |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
| 662 mg dose | |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
| 882 mg dose | |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
| 1064 mg dose | |
| Gluteal | 20 gauge, 1½-inch or 20 gauge, 2-inch |
[see Dosage and Administration ( 2.1 )]
3. ATTACH the injection needle.
Attach the appropriate needle securely with a clockwise twisting motion. Do NOT overtighten. Overtightening could lead to needle hub cracking.
4. PRIME the syringe to remove air.
4a. Bring the syringe into upright position and tap the syringe to bring air to the top.
4b. Depress the plunger rod to remove air until a few drops are released. It is normal to see small air bubbles remaining in the syringe.
5. Inject in a RAPID and CONTINUOUS manner. Product requires a RAPID injection. Do not hesitate. Administer the entire content intramuscularly. Do not inject by any other route.
6. DISPOSE of the needle. Cover the needle by pressing the safety device. Dispose of used and unused items in a proper waste container.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Intramuscular administration of aripiprazole lauroxil to rats and dogs was associated with injection site tissue reactions at all doses in rats treated up to 6 months at doses of 15, 29, and 103 mg/animal (which are approximately 0.3 to 2 times and 0.5 to 3 times the MRHD of 1064 mg on mg/m2 basis in males and females, respectively) and in dogs treated up to 9 months at doses of 147, 662, and 2058 mg/animal (which are approximately 0.5 to 6 times and 0.6 to 8 times the MRHD in males and females, respectively on mg/m2 basis). These injection site tissue reactions consisted of localized granulomatous inflammation and granuloma formation. Transiently impaired limb function and swelling occurred in dogs. The granulomas did not completely resolve 2 months following the last injection in the 6 month rat study and 4 months following the last injection in the 9 month dog study (the low dose groups were not examined for reversibility in these studies).
Orally administered aripiprazole produced retinal degeneration in albino rats in a 26-week chronic toxicity study at a dose of 60 mg/kg, which is 19 times the oral MRHD of 30 mg/day on mg/m2 basis, and in a 2-year carcinogenicity study at doses of 40 mg/kg and 60 mg/kg, which are 13 and 19 times the oral MRHD on mg/m2 basis and 7 to 14 times human exposure at the oral MRHD based on AUC. Evaluation of the retinas of albino mice and of monkeys did not reveal evidence of retinal degeneration. Additional studies to further evaluate the mechanism have not been performed. The relevance of this finding to human risk is unknown.
2.4 Dose Adjustments for Cyp450 Considerations (2.4 Dose Adjustments for CYP450 Considerations)
Refer to the prescribing information for oral aripiprazole for recommendations regarding dosage adjustments due to drug interactions, for the first 21 days when the patient is taking 21 days of oral aripiprazole concomitantly with the first dose of ARISTADA. Avoid initiating ARISTADA treatment with ARISTADA INITIO in patients requiring dose adjustments.
Once stabilized on ARISTADA, refer to the dosing recommendations below for patients taking strong CYP2D6 inhibitors, strong CYP3A4 inhibitors, or strong CYP3A4 inducers:
- No dosage changes recommended for ARISTADA, if CYP450 modulators are added for less than 2 weeks.
- Make dose changes to ARISTADA if CYP450 modulators are added for greater than 2 weeks (see Table 4).
| Concomitant Medicine | Dose Change for ARISTADA a |
|---|---|
|
a For the 882 mg dose administered every 6 weeks and the 1064 mg administered every 2 months, the next lower strength should be 441 mg administered monthly. |
|
| Strong CYP3A4 Inhibitor | Reduce the dose of ARISTADA to the next lower strength. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. For patients known to be poor metabolizers of CYP2D6: Reduce dose to 441 mg from 662 mg, 882 mg, or 1064 mg. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. |
| Strong CYP2D6 Inhibitor | Reduce the dose of ARISTADA to the next lower strength. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. For patients known to be poor metabolizers of CYP2D6: No dose adjustment required. |
| Both Strong CYP3A4 Inhibitor and Strong CYP2D6 Inhibitor | Avoid use for patients at 662 mg, 882 mg, or 1064 mg dose. No dosage adjustment is necessary in patients taking 441 mg ARISTADA, if tolerated. |
| CYP3A4 Inducers | No dose adjustment for 662 mg, 882 mg, or 1064 mg dose; increase the 441 mg dose to 662 mg. |
5.3 Potential for Dosing and Medication Errors
Medication errors, including substitution and dispensing errors, between ARISTADA and ARISTADA INITIO could occur. ARISTADA INITIO is for single administration in contrast to ARISTADA which is administered monthly, every 6 weeks, or every 8 weeks [see Dosage and Administration (2.1)]. Do not substitute ARISTADA INITIO for ARISTADA because of differing pharmacokinetic profiles [see Clinical Pharmacology ( 12.3 )].
5.10 Leukopenia, Neutropenia, and Agranulocytosis
In clinical trials and/or postmarketing experience, events of leukopenia and neutropenia have been reported temporally related to antipsychotic agents. Agranulocytosis has also been reported.
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC)/absolute neutrophil count (ANC) and history of drug-induced leukopenia/neutropenia. In patients with a history of a clinically significant low WBC/ANC or drug-induced leukopenia/neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of ARISTADA at the first sign of a clinical significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue ARISTADA in patients with severe neutropenia (absolute neutrophil count <1000/mm3) and follow their WBC until recovery.
5.12 Potential for Cognitive and Motor Impairment
ARISTADA, like other antipsychotics, has the potential to impair judgment, thinking or motor skills. Patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that therapy with ARISTADA does not affect them adversely.
5.2 Cerebrovascular Adverse Reactions, Including Stroke
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly patients with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated patients. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions ( 5.1 )].
5.7 Pathological Gambling and Other Compulsive Behaviors
Post-marketing case reports suggest that patients can experience intense urges, particularly for gambling, and the inability to control these urges while taking aripiprazole. Other compulsive urges, reported less frequently include: sexual urges, shopping, eating or binge eating, and other impulsive or compulsive behaviors. Because patients may not recognize these behaviors as abnormal, it is important for prescribers to ask patients or their caregivers specifically about the development of new or intense gambling urges, compulsive sexual urges, compulsive shopping, binge or compulsive eating, or other urges while being treated with aripiprazole. It should be noted that impulse-control symptoms can be associated with the underlying disorder. In some cases, although not all, urges were reported to have stopped when the dose was reduced or the medication was discontinued. Compulsive behaviors may result in harm for the patient and others if not recognized. Consider dose reduction or stopping the medication if a patient develops such urges.
7.1 Drugs Having Clinically Important Interactions With Aristada (7.1 Drugs Having Clinically Important Interactions with ARISTADA)
| Strong CYP3A4 Inhibitors and CYP2D6 Inhibitors | |
| Clinical Impact: | The concomitant use of oral aripiprazole with strong CYP3A4 or CYP2D6 inhibitors increased the exposure of aripiprazole compared to the use of oral aripiprazole alone [see Clinical Pharmacology ( 12.3 )]. |
| Intervention: | With concomitant use of ARISTADA with a strong CYP3A4 inhibitor or CYP2D6 inhibitor for more than 2 weeks, reduce the ARISTADA dose [see Dosage and Administration ( 2.4 )]. |
| Examples: | itraconazole, clarithromycin, quinidine, fluoxetine, paroxetine |
| Strong CYP3A4 Inducers | |
| Clinical Impact: | The concomitant use of oral aripiprazole and carbamazepine decreased the exposure of aripiprazole compared to the use of oral aripiprazole alone [see Clinical Pharmacology ( 12.3 )]. |
| Intervention: | With concomitant use of ARISTADA with a strong CYP3A4 inducer for more than 2 weeks consider increasing the ARISTADA dose [see Dosage and Administration ( 2.4 )]. |
| Examples: | carbamazepine, rifampin |
| Antihypertensive Drugs | |
| Clinical Impact: | Due to its alpha adrenergic antagonism, aripiprazole has the potential to enhance the effect of certain antihypertensive agents. |
| Intervention: | Monitor blood pressure and adjust dose accordingly [see Warnings and Precautions ( 5.8 )]. |
| Examples: | carvedilol, lisinopril, prazosin |
| Benzodiazepines | |
| Clinical Impact: | The intensity of sedation was greater with the combination of oral aripiprazole and lorazepam as compared to that observed with aripiprazole alone. The orthostatic hypotension observed was greater with the combination as compared to that observed with lorazepam alone [see Warnings and Precautions ( 5.8 )]. |
| Intervention: | Monitor sedation and blood pressure. Adjust dose accordingly. |
| Example: | lorazepam |
7.2 Drugs Having No Clinically Important Interactions With Aristada (7.2 Drugs Having No Clinically Important Interactions with ARISTADA)
Based on pharmacokinetic studies with oral aripiprazole, no dosage adjustment of ARISTADA is required when administered concomitantly with famotidine, valproate, or lithium [see Clinical Pharmacology ( 12.3 )].
In addition, no dosage adjustment is necessary for substrates of CYP2D6 (e.g., dextromethorphan, fluoxetine, paroxetine, or venlafaxine), CYP2C9 (e.g., warfarin), CYP2C19 (e.g., omeprazole, warfarin, escitalopram), or CYP3A4 (e.g., dextromethorphan) when co-administered with ARISTADA. Additionally, no dosage adjustment is necessary for valproate, lithium, lamotrigine, or sertraline when co-administered with ARISTADA [see Clinical Pharmacology ( 12.3 )].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis (5.1 Increased Mortality in Elderly Patients with Dementia-related Psychosis)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group.
Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions ( 5.2 )].
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis (WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ARISTADA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:36.932927 · Updated: 2026-03-14T22:22:49.782835