diphenhydramine hcl 2 % + zinc acetate 0.1%, a+ health
178ed513-4f22-8f11-e063-6294a90a109b
34390-5
HUMAN OTC DRUG LABEL
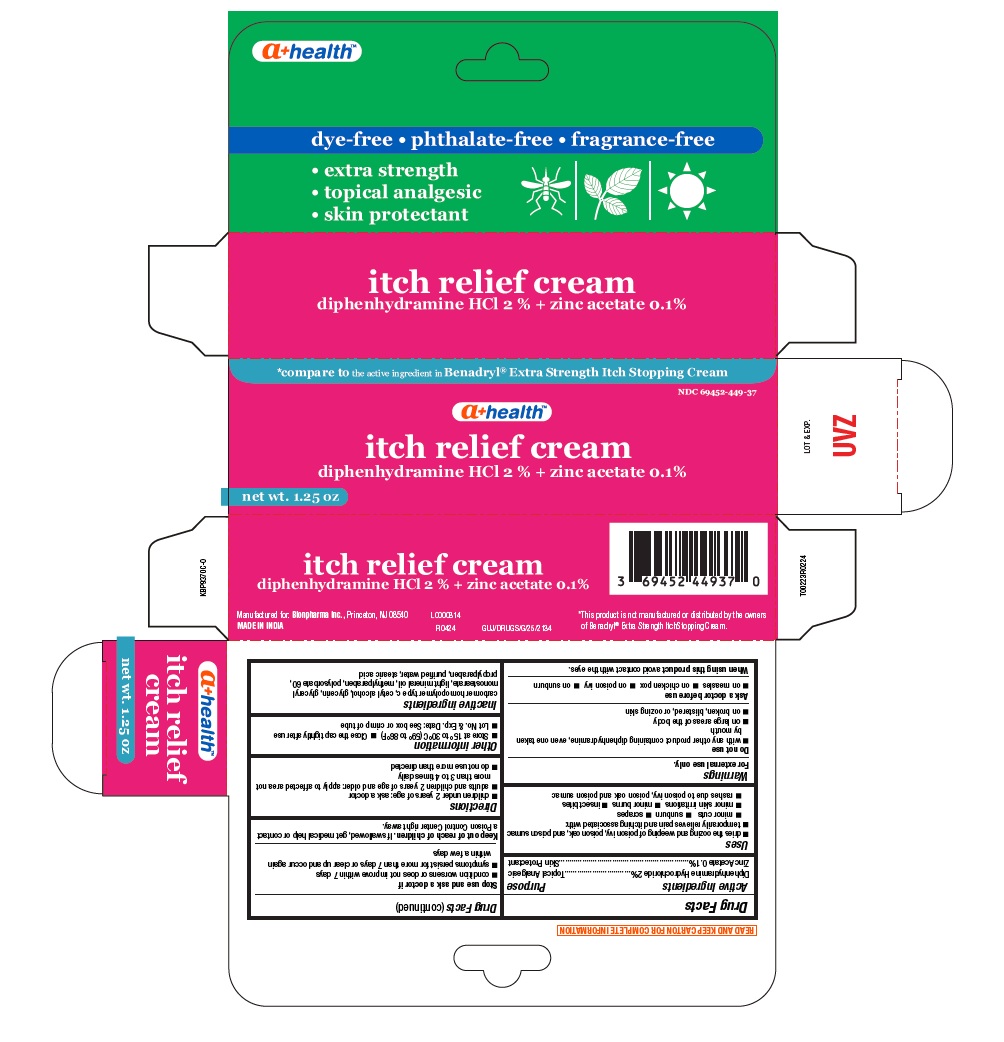
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Topical Analgesic Skin Protectant
Description
Diphenhydramine Hydrochloride 2% Zinc Acetate 0.1%
Medication Information
Warnings
For external use only.
Do not use
■ with any other product containing diphenhydramine, even one taken by mouth
■ on large areas of the body
■ on broken, blistered, or oozing skin
Uses
■ dries the oozing and weeping of poison ivy, poison oak, and poison sumac
■ temporarily relieves pain and itching associated with:
■ minor cuts ■ sunburn ■ scrapes
■ minor skin irritations ■ minor burns ■ insect bites
■ rashes due to poison ivy, poison oak and poison sumac
Purpose
Topical Analgesic
Skin Protectant
Directions
■ children under 2 years of age: ask a doctor
■ adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
■ do not use more than directed
Other Information
■ Store at 15° to 30°C (59° to 86°F) ■ Close the cap tightly after use
■ Lot No. & Exp. Date: See box or crimp of tube
Inactive Ingredients
carbomer homopolymer type c, cetyl alcohol, glycerin, glyceryl monostearate, light mineral oil, methylparaben, polysorbate 60,
propylparaben, purified water, stearic acid
Description
Diphenhydramine Hydrochloride 2% Zinc Acetate 0.1%
Section 42229-5
READ AND KEEP CARTON FOR COMPLETE INFORMATION
*This product is not manufactured or distributed by the owners of Benadryl® Extra Strength ItchStopping Cream.
Manufactured for: Bionpharma Inc., Princeton, NJ 08540
MADE IN INDIA
GUJ/DRUGS/G/25/2134
L0000814
R0424
LOT & EXP.
Tube Carton
* compare to the active ingredient in Benadryl® Extra Strength Itch Stopping Cream
NDC 69452-449-37
a+ health TM
itch relief cream
diphenhydramine HCl 2 % + zinc acetate 0.1%
net wt. 1.25 oz
dye-free • phthalate-free • fragrance-free
• extra strength
• topical analgesic
• skin protectant
Active Ingredient
Diphenhydramine Hydrochloride 2%
Zinc Acetate 0.1%
Ask A Doctor Before Use
■ on measles ■ on chicken pox ■ on poison ivy ■ on sunburn
When Using This Product
avoid contact with the eyes.
Stop Use and Ask A Doctor If
■ condition worsens or does not improve within 7 days
■ symptoms persist for more than 7 days or clear up and occur again within a few days
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Warnings
For external use only.
Do not use
■ with any other product containing diphenhydramine, even one taken by mouth
■ on large areas of the body
■ on broken, blistered, or oozing skin
Uses
■ dries the oozing and weeping of poison ivy, poison oak, and poison sumac
■ temporarily relieves pain and itching associated with:
■ minor cuts ■ sunburn ■ scrapes
■ minor skin irritations ■ minor burns ■ insect bites
■ rashes due to poison ivy, poison oak and poison sumac
Purpose
Topical Analgesic
Skin Protectant
Directions
■ children under 2 years of age: ask a doctor
■ adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
■ do not use more than directed
Other Information (Other information)
■ Store at 15° to 30°C (59° to 86°F) ■ Close the cap tightly after use
■ Lot No. & Exp. Date: See box or crimp of tube
Inactive Ingredients (Inactive ingredients)
carbomer homopolymer type c, cetyl alcohol, glycerin, glyceryl monostearate, light mineral oil, methylparaben, polysorbate 60,
propylparaben, purified water, stearic acid
Section 42229-5 (42229-5)
READ AND KEEP CARTON FOR COMPLETE INFORMATION
*This product is not manufactured or distributed by the owners of Benadryl® Extra Strength ItchStopping Cream.
Manufactured for: Bionpharma Inc., Princeton, NJ 08540
MADE IN INDIA
GUJ/DRUGS/G/25/2134
L0000814
R0424
LOT & EXP.
Tube Carton (tube carton)
* compare to the active ingredient in Benadryl® Extra Strength Itch Stopping Cream
NDC 69452-449-37
a+ health TM
itch relief cream
diphenhydramine HCl 2 % + zinc acetate 0.1%
net wt. 1.25 oz
dye-free • phthalate-free • fragrance-free
• extra strength
• topical analgesic
• skin protectant
Active Ingredient (Active ingredient)
Diphenhydramine Hydrochloride 2%
Zinc Acetate 0.1%
Ask A Doctor Before Use (Ask a doctor before use)
■ on measles ■ on chicken pox ■ on poison ivy ■ on sunburn
When Using This Product (When using this product)
avoid contact with the eyes.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
■ condition worsens or does not improve within 7 days
■ symptoms persist for more than 7 days or clear up and occur again within a few days
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:18.008932 · Updated: 2026-03-14T23:03:17.077951