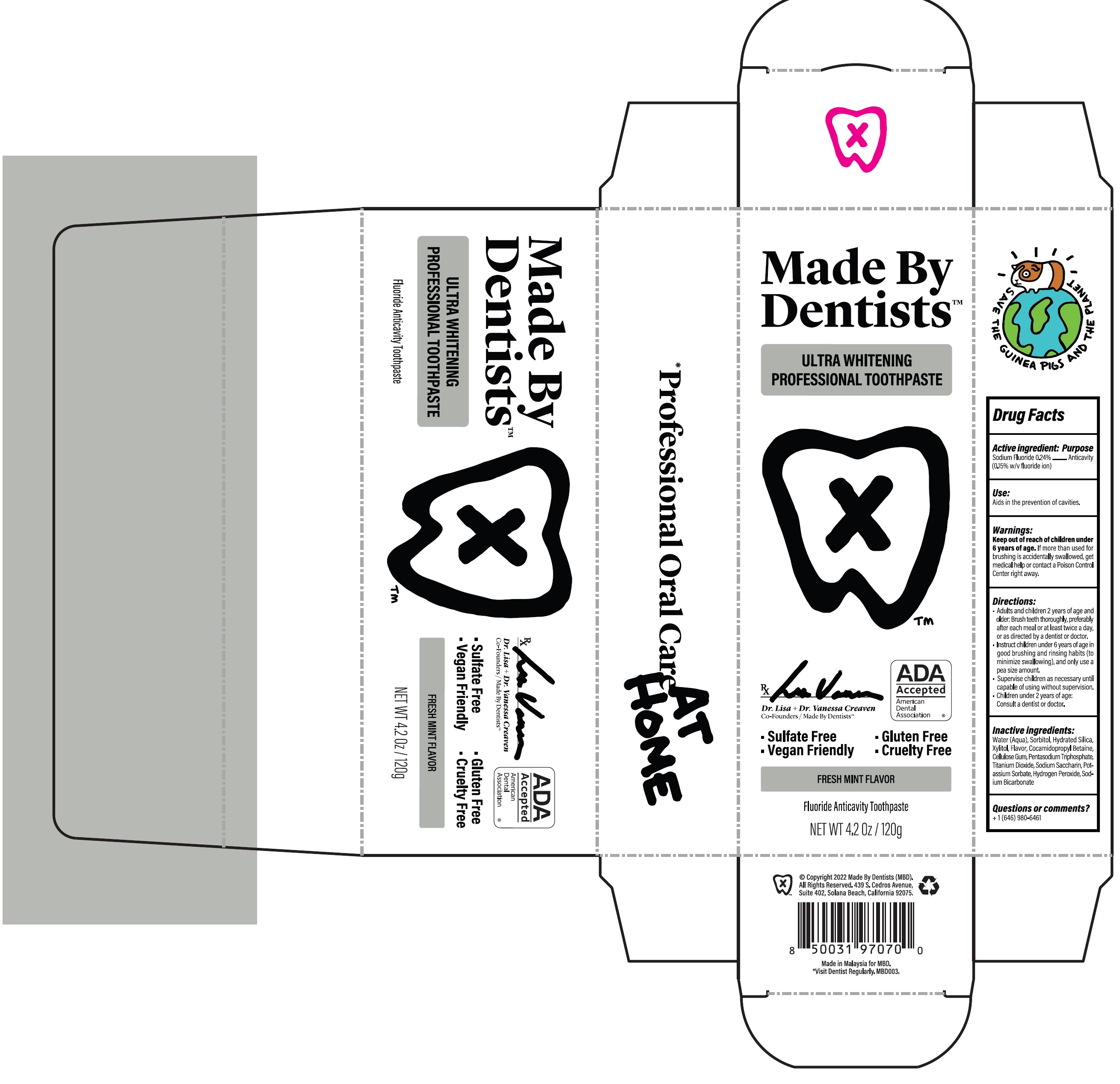
Made By Dentists Ultra Whitening Professional
177b26c8-6ad2-2e78-e063-6394a90afe6b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Purpose
Anticavity
Medication Information
Purpose
Anticavity
Description
Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Use:
Aids in the prevention of cavities.
Label
Drug Facts
Directions:
- Adults and children 2 years of age and older: Brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or doctor.
- Instruct children under 6 years of age in good brushing and rinsing habits (to minimize swallowing), and only use a pea size amount.
- Supervise children as necessary until capable of using without supervision.
- Children under 2 years of age: Consult a dentist or doctor.
Active Ingredient:
Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Label: 62776 005 02
Inactive Ingredients:
Water (Aqua), Sorbitol, Hydrated Silica, Xylitol, Flavor, Cocamidopropyl Betaine, Cellulose Gum, Pentasodium Triphosphate, Titanium Dioxide, Sordium Saccharin, Potassium Sorbate, Hydrogen Peroxide, Sodium Bicarbonate
Questions Or Comments?
+ 1 (646) 980-6461
Keep Out of Reach of Children Under 6 Years of Age.
If more than used for brushing is accidentlly swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use:
Aids in the prevention of cavities.
Label
Purpose
Anticavity
Drug Facts
Directions:
- Adults and children 2 years of age and older: Brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or doctor.
- Instruct children under 6 years of age in good brushing and rinsing habits (to minimize swallowing), and only use a pea size amount.
- Supervise children as necessary until capable of using without supervision.
- Children under 2 years of age: Consult a dentist or doctor.
Active Ingredient: (Active ingredient:)
Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Label: 62776 005 02 (Label: 62776-005-02)
Inactive Ingredients: (Inactive ingredients:)
Water (Aqua), Sorbitol, Hydrated Silica, Xylitol, Flavor, Cocamidopropyl Betaine, Cellulose Gum, Pentasodium Triphosphate, Titanium Dioxide, Sordium Saccharin, Potassium Sorbate, Hydrogen Peroxide, Sodium Bicarbonate
Questions Or Comments? (Questions or comments?)
+ 1 (646) 980-6461
Keep Out of Reach of Children Under 6 Years of Age. (Keep out of reach of children under 6 years of age.)
If more than used for brushing is accidentlly swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:22.776750 · Updated: 2026-03-14T23:09:06.809873