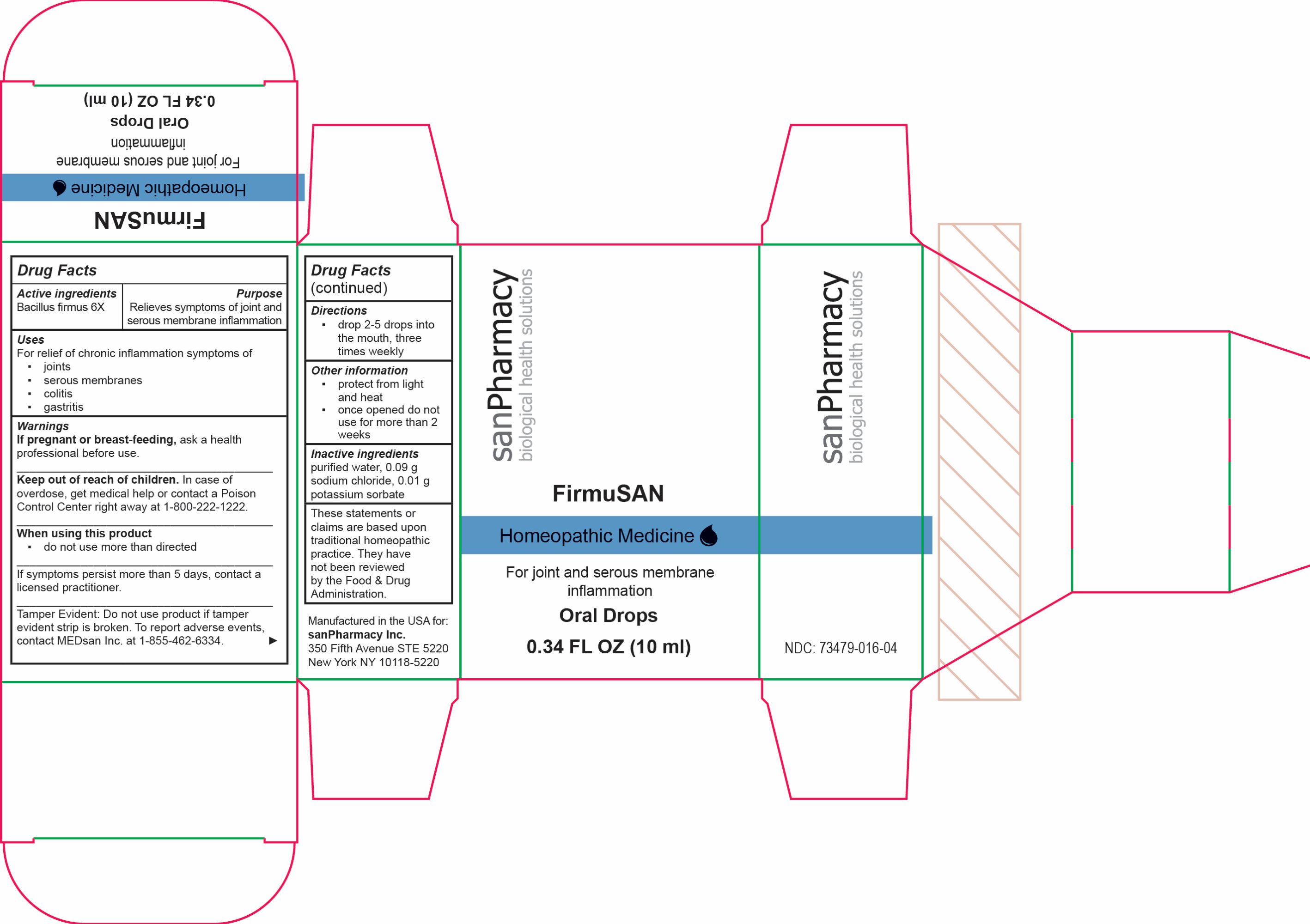
Firmusan Drops
173af2af-79bd-65cc-e063-6394a90acfce
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
For joint and serous membrane inflammation
Purpose
Relieves symptoms of joint and serous membrane inflammation
Medication Information
Purpose
Relieves symptoms of joint and serous membrane inflammation
Description
For joint and serous membrane inflammation
Dosage
drop 2-5 drops into the mouth, three times weekly
Section 42229-5
Manufactured in the USA for:
sanPharmacy Inc.
350 Fifth Avenue STE 5220
New York NY 10118-5220
Section 44425-7
protect from light and heat▪once opened do not use for more than 2 weeks
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222
Section 51945-4
FirmuSAN
Homeopathic Medicine
For joint and serous membrane inflammation
Oral Drops
0.34 FL OZ (10 ml)
sanPharmacy
biological health solutions
Warnings
If pregnant or breast-feeding, ask a health professional before use.
______________________________
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
______________________________
When using this product
▪ do not use more than directed
______________________________
If symptoms persist more than 5 days, contact a licensed practitioner.
______________________________
Tamper Evident: Do not use product if tamper evident strip is broken. To report adverse events, contact MEDsan Inc. at 1-855-462-6334.
Indications
For joint and serous membrane inflammation
Active Ingredients
Bacillus firmus 6X
Liability Statement
These statements or claims are based upon traditional homeopathic practice. They have not been reviewed by the Food & Drug Administration
Inactive Ingeredients
purified water, 0.09 g sodium chloride, 0.01 g potassium sorbate
Structured Label Content
Dosage
drop 2-5 drops into the mouth, three times weekly
Section 42229-5 (42229-5)
Manufactured in the USA for:
sanPharmacy Inc.
350 Fifth Avenue STE 5220
New York NY 10118-5220
Section 44425-7 (44425-7)
protect from light and heat▪once opened do not use for more than 2 weeks
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222
Section 51945-4 (51945-4)
FirmuSAN
Homeopathic Medicine
For joint and serous membrane inflammation
Oral Drops
0.34 FL OZ (10 ml)
sanPharmacy
biological health solutions
Purpose
Relieves symptoms of joint and serous membrane inflammation
Warnings
If pregnant or breast-feeding, ask a health professional before use.
______________________________
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
______________________________
When using this product
▪ do not use more than directed
______________________________
If symptoms persist more than 5 days, contact a licensed practitioner.
______________________________
Tamper Evident: Do not use product if tamper evident strip is broken. To report adverse events, contact MEDsan Inc. at 1-855-462-6334.
Indications
For joint and serous membrane inflammation
Active Ingredients (Active ingredients)
Bacillus firmus 6X
Liability Statement (Liability statement)
These statements or claims are based upon traditional homeopathic practice. They have not been reviewed by the Food & Drug Administration
Inactive Ingeredients (Inactive ingeredients)
purified water, 0.09 g sodium chloride, 0.01 g potassium sorbate
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:29.460476 · Updated: 2026-03-14T23:16:53.792884