Iclusig
16d804b6-4957-43ee-b18c-3b36ec37c5ac
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
ICLUSIG ® is indicated for the treatment of adult patients with: Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia (Ph+ ALL) Newly diagnosed Ph+ ALL in combination with chemotherapy. This indication is approved under accelerated approval based on minimal residual disease (MRD)-negative complete remission (CR) at the end of induction [see Clinical Studies (14) ] . Continued approval for this indication may be contingent upon verification of clinical benefit in a confirmatory trial(s). As monotherapy in Ph+ ALL for whom no other kinase inhibitors are indicated or T315I-positive Ph+ ALL. Chronic Myeloid Leukemia (CML) Chronic phase (CP) CML with resistance or intolerance to at least two prior kinase inhibitors. Accelerated phase (AP) or blast phase (BP) CML for whom no other kinase inhibitors are indicated. T315I-positive CML (chronic phase, accelerated phase, or blast phase).
Dosage and Administration
Recommended Dosage in Newly Diagnosed Ph+ ALL : Starting dose is 30 mg orally once daily in combination with chemotherapy, with a reduction to 15 mg once daily upon achievement of MRD-negative (≤0.01% BCR::ABL1/ABL1) CR at the end of induction. ( 2.1 ) Recommended Dosage in Monotherapy for Ph+ ALL for Whom No Other Kinase Inhibitors are Indicated or T315I-positive Ph+ ALL : Starting dose is 45 mg orally once daily. ( 2.1 ) Recommended Dosage in CP-CML : Starting dose is 45 mg orally once daily with a reduction to 15 mg once daily upon achievement of ≤1% BCR::ABL1 IS . ( 2.1 ) Recommended Dosage in AP-CML and BP-CML : Starting dose is 45 mg orally once daily. ( 2.1 ) Hepatic Impairment : See the Full Prescribing Information for dosage modifications for hepatic impairment. ( 2.4 ) ICLUSIG may be taken with or without food. ( 2.1 )
Contraindications
None.
Warnings and Precautions
Hypertension : Monitor blood pressure and manage hypertension as clinically indicated. Interrupt, dose reduce or stop ICLUSIG if hypertension is not medically controlled. ( 2.2 , 5.5 ) Pancreatitis : Monitor serum lipase. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on severity. Evaluate for pancreatitis when lipase elevation is accompanied by abdominal symptoms. ( 2.2 , 5.6 ) Neuropathy : Monitor for symptoms of peripheral and cranial neuropathy. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 2.2 , 5.8 ) Ocular Toxicity : Conduct comprehensive eye exams at baseline and periodically during treatment. ( 5.9 ) Hemorrhage : Monitor for hemorrhage and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 2.2 , 5.10 ) Fluid Retention : Monitor for fluid retention and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 2.2 , 5.11 ) Cardiac Arrhythmias : Monitor for signs or symptoms of arrhythmias and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 5.12 ) Myelosuppression : Obtain complete blood counts every 2 weeks for the first 3 months and then monthly or as clinically indicated. If ANC less than 1 × 10 9 /L or platelets less than 50 × 10 9 /L, interrupt ICLUSIG until ANC at least 1.5 × 10 9 /L and platelets at least 75 × 10 9 /L, then resume at same or reduced dose. ( 2.2 , 5.13 ) Tumor Lysis Syndrome : Ensure adequate hydration and correct elevated uric acid levels prior to initiating ICLUSIG. ( 5.14 ) Reversible Posterior Leukoencephalopathy Syndrome (RPLS) : Interrupt ICLUSIG until resolution. The safety of resumption of ICLUSIG in patients upon resolution of RPLS is unknown. ( 5.15 ) Impaired Wound Healing and Gastrointestinal Perforation : Withhold ICLUSIG for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of ICLUSIG after resolution of wound healing complications has not been established. ( 5.16 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.17 , 8.1 , 8.3 )
Adverse Reactions
Recommended dosage modifications of ICLUSIG for adverse reactions are provided in Table 1 and recommended dose reductions of ICLUSIG for adverse reactions are presented in Table 2. Table 1: Recommended Dosage Modifications for ICLUSIG for Adverse Reactions Adverse Reaction Severity ICLUSIG Dosage Modifications Based on CTCAE v5.0: Grade 1 mild, Grade 2 moderate, Grade 3 severe, Grade 4 life-threatening ULN = Upper Limit of Normal for the lab; AOE = Arterial Occlusive Event; VTE = Venous Thromboembolic Event; ANC = absolute neutrophil count AOE: cardiovascular or cerebrovascular [see Warnings and Precautions (5.1) ] Grade 1 Interrupt ICLUSIG until resolved, then resume at same dose. Grade 2 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Grade 3 or 4 Discontinue ICLUSIG. AOE: peripheral vascular and other or VTE [see Warnings and Precautions (5.1 , 5.2) ] Grade 1 Interrupt ICLUSIG until resolved, then resume at same dose. Grade 2 Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Grade 3 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Grade 4 Discontinue ICLUSIG. Heart Failure [see Warnings and Precautions (5.3) ] Grade 2 or 3 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Grade 4 Discontinue ICLUSIG. Hepatotoxicity [see Warnings and Precautions (5.4) ] AST or ALT greater than 3 times ULN Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. AST or ALT at least 3 times ULN concurrent with bilirubin greater than 2 times ULN and alkaline phosphatase less than 2 times ULN Discontinue ICLUSIG. Pancreatitis and Elevated Lipase [see Warnings and Precautions (5.6) ] Serum lipase greater than 1 to 1.5 times ULN Consider interrupting ICLUSIG until resolution, then resume at same dose. Serum lipase greater than 1.5 to 2 times ULN, 2 to 5 times ULN and asymptomatic, or asymptomatic radiologic pancreatitis Interrupt ICLUSIG until Grade 0 or 1 (less than 1.5 times ULN), then resume at next lower dose. Serum lipase greater than 2 to 5 times ULN and symptomatic, symptomatic Grade 3 pancreatitis, or serum lipase greater than 5 times ULN and asymptomatic Interrupt ICLUSIG until complete resolution of symptoms and after recovery of lipase elevation Grade 0 or 1, then resume at next lower dose. Symptomatic pancreatitis and serum lipase greater than 5 times ULN Discontinue ICLUSIG. Myelosuppression [see Warnings and Precautions (5.13) ] ANC less than 1 × 10 9 /L or Platelets less than 50 × 10 9 /L Interrupt ICLUSIG until ANC at least 1.5 × 10 9 /L and platelet at least 75 × 10 9 /L, then resume at same dose. If recurrence, interrupt ICLUSIG until resolution, then resume at next lower dose. Other Non-hematologic Adverse Reactions [see Warnings and Precautions (5.5 , 5.8 , 5.10 , 5.11 , 5.12) ] Grade 1 Interrupt ICLUSIG until resolved, then resume at same dose. Grade 2 Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Grade 3 or 4 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Table 2: Recommended Dose Reductions for ICLUSIG for Adverse Reactions Dose Reduction Dosage for Patients with CP-CML Dosage for Patients with AP-CML, BP-CML, and Ph+ ALL Monotherapy Dosage for Patients with Newly Diagnosed Ph+ ALL First 30 mg orally once daily 30 mg orally once daily 15 mg orally once daily Second 15 mg orally once daily 15 mg orally once daily 10 mg orally once daily Third 10 mg orally once daily Permanently discontinue ICLUSIG in patients unable to tolerate 15 mg orally once daily. Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily. Subsequent Reduction Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily.
Drug Interactions
Strong CYP3A Inhibitors : Avoid coadministration or reduce ICLUSIG dose if coadministration cannot be avoided. ( 2.3 , 7.1 ) Strong CYP3A Inducers : Avoid coadministration. ( 7.1 )
How Supplied
ICLUSIG tablets are available in the following configurations. Strength NDC Number Description Presentation 10 mg 63020-536-30 oval, white to off-white, biconvex film-coated tablets with debossed "NZ" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 15 mg 63020-535-30 round, white, biconvex film-coated tablets with debossed "A5" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 63020-535-60 60 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 30 mg 63020-533-30 round, white, biconvex film-coated tablets with debossed "C7" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 45 mg 63020-534-30 round, white, biconvex film-coated tablets with debossed "AP4" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure.
Storage and Handling
ICLUSIG tablets are available in the following configurations. Strength NDC Number Description Presentation 10 mg 63020-536-30 oval, white to off-white, biconvex film-coated tablets with debossed "NZ" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 15 mg 63020-535-30 round, white, biconvex film-coated tablets with debossed "A5" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 63020-535-60 60 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 30 mg 63020-533-30 round, white, biconvex film-coated tablets with debossed "C7" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 45 mg 63020-534-30 round, white, biconvex film-coated tablets with debossed "AP4" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure.
Description
Warnings and Precautions ( 5 ) 10/2025
Medication Information
Warnings and Precautions
Hypertension : Monitor blood pressure and manage hypertension as clinically indicated. Interrupt, dose reduce or stop ICLUSIG if hypertension is not medically controlled. ( 2.2 , 5.5 ) Pancreatitis : Monitor serum lipase. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on severity. Evaluate for pancreatitis when lipase elevation is accompanied by abdominal symptoms. ( 2.2 , 5.6 ) Neuropathy : Monitor for symptoms of peripheral and cranial neuropathy. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 2.2 , 5.8 ) Ocular Toxicity : Conduct comprehensive eye exams at baseline and periodically during treatment. ( 5.9 ) Hemorrhage : Monitor for hemorrhage and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 2.2 , 5.10 ) Fluid Retention : Monitor for fluid retention and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 2.2 , 5.11 ) Cardiac Arrhythmias : Monitor for signs or symptoms of arrhythmias and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. ( 5.12 ) Myelosuppression : Obtain complete blood counts every 2 weeks for the first 3 months and then monthly or as clinically indicated. If ANC less than 1 × 10 9 /L or platelets less than 50 × 10 9 /L, interrupt ICLUSIG until ANC at least 1.5 × 10 9 /L and platelets at least 75 × 10 9 /L, then resume at same or reduced dose. ( 2.2 , 5.13 ) Tumor Lysis Syndrome : Ensure adequate hydration and correct elevated uric acid levels prior to initiating ICLUSIG. ( 5.14 ) Reversible Posterior Leukoencephalopathy Syndrome (RPLS) : Interrupt ICLUSIG until resolution. The safety of resumption of ICLUSIG in patients upon resolution of RPLS is unknown. ( 5.15 ) Impaired Wound Healing and Gastrointestinal Perforation : Withhold ICLUSIG for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of ICLUSIG after resolution of wound healing complications has not been established. ( 5.16 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.17 , 8.1 , 8.3 )
Indications and Usage
ICLUSIG ® is indicated for the treatment of adult patients with: Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia (Ph+ ALL) Newly diagnosed Ph+ ALL in combination with chemotherapy. This indication is approved under accelerated approval based on minimal residual disease (MRD)-negative complete remission (CR) at the end of induction [see Clinical Studies (14) ] . Continued approval for this indication may be contingent upon verification of clinical benefit in a confirmatory trial(s). As monotherapy in Ph+ ALL for whom no other kinase inhibitors are indicated or T315I-positive Ph+ ALL. Chronic Myeloid Leukemia (CML) Chronic phase (CP) CML with resistance or intolerance to at least two prior kinase inhibitors. Accelerated phase (AP) or blast phase (BP) CML for whom no other kinase inhibitors are indicated. T315I-positive CML (chronic phase, accelerated phase, or blast phase).
Dosage and Administration
Recommended Dosage in Newly Diagnosed Ph+ ALL : Starting dose is 30 mg orally once daily in combination with chemotherapy, with a reduction to 15 mg once daily upon achievement of MRD-negative (≤0.01% BCR::ABL1/ABL1) CR at the end of induction. ( 2.1 ) Recommended Dosage in Monotherapy for Ph+ ALL for Whom No Other Kinase Inhibitors are Indicated or T315I-positive Ph+ ALL : Starting dose is 45 mg orally once daily. ( 2.1 ) Recommended Dosage in CP-CML : Starting dose is 45 mg orally once daily with a reduction to 15 mg once daily upon achievement of ≤1% BCR::ABL1 IS . ( 2.1 ) Recommended Dosage in AP-CML and BP-CML : Starting dose is 45 mg orally once daily. ( 2.1 ) Hepatic Impairment : See the Full Prescribing Information for dosage modifications for hepatic impairment. ( 2.4 ) ICLUSIG may be taken with or without food. ( 2.1 )
Contraindications
None.
Adverse Reactions
Recommended dosage modifications of ICLUSIG for adverse reactions are provided in Table 1 and recommended dose reductions of ICLUSIG for adverse reactions are presented in Table 2. Table 1: Recommended Dosage Modifications for ICLUSIG for Adverse Reactions Adverse Reaction Severity ICLUSIG Dosage Modifications Based on CTCAE v5.0: Grade 1 mild, Grade 2 moderate, Grade 3 severe, Grade 4 life-threatening ULN = Upper Limit of Normal for the lab; AOE = Arterial Occlusive Event; VTE = Venous Thromboembolic Event; ANC = absolute neutrophil count AOE: cardiovascular or cerebrovascular [see Warnings and Precautions (5.1) ] Grade 1 Interrupt ICLUSIG until resolved, then resume at same dose. Grade 2 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Grade 3 or 4 Discontinue ICLUSIG. AOE: peripheral vascular and other or VTE [see Warnings and Precautions (5.1 , 5.2) ] Grade 1 Interrupt ICLUSIG until resolved, then resume at same dose. Grade 2 Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Grade 3 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Grade 4 Discontinue ICLUSIG. Heart Failure [see Warnings and Precautions (5.3) ] Grade 2 or 3 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Grade 4 Discontinue ICLUSIG. Hepatotoxicity [see Warnings and Precautions (5.4) ] AST or ALT greater than 3 times ULN Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. AST or ALT at least 3 times ULN concurrent with bilirubin greater than 2 times ULN and alkaline phosphatase less than 2 times ULN Discontinue ICLUSIG. Pancreatitis and Elevated Lipase [see Warnings and Precautions (5.6) ] Serum lipase greater than 1 to 1.5 times ULN Consider interrupting ICLUSIG until resolution, then resume at same dose. Serum lipase greater than 1.5 to 2 times ULN, 2 to 5 times ULN and asymptomatic, or asymptomatic radiologic pancreatitis Interrupt ICLUSIG until Grade 0 or 1 (less than 1.5 times ULN), then resume at next lower dose. Serum lipase greater than 2 to 5 times ULN and symptomatic, symptomatic Grade 3 pancreatitis, or serum lipase greater than 5 times ULN and asymptomatic Interrupt ICLUSIG until complete resolution of symptoms and after recovery of lipase elevation Grade 0 or 1, then resume at next lower dose. Symptomatic pancreatitis and serum lipase greater than 5 times ULN Discontinue ICLUSIG. Myelosuppression [see Warnings and Precautions (5.13) ] ANC less than 1 × 10 9 /L or Platelets less than 50 × 10 9 /L Interrupt ICLUSIG until ANC at least 1.5 × 10 9 /L and platelet at least 75 × 10 9 /L, then resume at same dose. If recurrence, interrupt ICLUSIG until resolution, then resume at next lower dose. Other Non-hematologic Adverse Reactions [see Warnings and Precautions (5.5 , 5.8 , 5.10 , 5.11 , 5.12) ] Grade 1 Interrupt ICLUSIG until resolved, then resume at same dose. Grade 2 Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Grade 3 or 4 Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. Table 2: Recommended Dose Reductions for ICLUSIG for Adverse Reactions Dose Reduction Dosage for Patients with CP-CML Dosage for Patients with AP-CML, BP-CML, and Ph+ ALL Monotherapy Dosage for Patients with Newly Diagnosed Ph+ ALL First 30 mg orally once daily 30 mg orally once daily 15 mg orally once daily Second 15 mg orally once daily 15 mg orally once daily 10 mg orally once daily Third 10 mg orally once daily Permanently discontinue ICLUSIG in patients unable to tolerate 15 mg orally once daily. Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily. Subsequent Reduction Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily.
Drug Interactions
Strong CYP3A Inhibitors : Avoid coadministration or reduce ICLUSIG dose if coadministration cannot be avoided. ( 2.3 , 7.1 ) Strong CYP3A Inducers : Avoid coadministration. ( 7.1 )
Storage and Handling
ICLUSIG tablets are available in the following configurations. Strength NDC Number Description Presentation 10 mg 63020-536-30 oval, white to off-white, biconvex film-coated tablets with debossed "NZ" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 15 mg 63020-535-30 round, white, biconvex film-coated tablets with debossed "A5" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 63020-535-60 60 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 30 mg 63020-533-30 round, white, biconvex film-coated tablets with debossed "C7" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 45 mg 63020-534-30 round, white, biconvex film-coated tablets with debossed "AP4" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure.
How Supplied
ICLUSIG tablets are available in the following configurations. Strength NDC Number Description Presentation 10 mg 63020-536-30 oval, white to off-white, biconvex film-coated tablets with debossed "NZ" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 15 mg 63020-535-30 round, white, biconvex film-coated tablets with debossed "A5" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 63020-535-60 60 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 30 mg 63020-533-30 round, white, biconvex film-coated tablets with debossed "C7" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. 45 mg 63020-534-30 round, white, biconvex film-coated tablets with debossed "AP4" on one side and plain on the other side 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure.
Description
Warnings and Precautions ( 5 ) 10/2025
Section 42229-5
Arterial Occlusive Events:
- Arterial occlusive events (AOEs), including fatalities, have occurred in ICLUSIG-treated patients. AOEs included fatal myocardial infarction, stroke, stenosis of large arterial vessels of the brain, severe peripheral vascular disease, and the need for urgent revascularization procedures. Patients with and without cardiovascular risk factors, including patients age 50 years or younger, experienced these events. Monitor for evidence of AOEs. Interrupt or discontinue ICLUSIG based on severity. Consider benefit-risk to guide a decision to restart ICLUSIG [see Dosage and Administration (2.2) , Warnings and Precautions (5.1)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 10/2025 | ||
|
MEDICATION GUIDE ICLUSIG® (eye-CLUE-sig) (ponatinib) tablets |
|||
|
What is the most important information I should know about ICLUSIG? ICLUSIG can cause serious side effects, including: Blood clots or blockage in your blood vessels (arteries and veins). Blood clots or blockage in your blood vessels may lead to heart attack, stroke, or death. A blood clot or blockage in your blood vessels can prevent proper blood flow to your heart, brain, bowels (intestines), legs, eyes, and other parts of your body. You may need emergency surgery or treatment in a hospital. Get medical help right away if you get any of the following symptoms: |
|||
|
|
||
| Blood clots or blockage in your blood vessels can happen in people with or without risk factors for heart and blood vessel disease, including people 50 years of age or younger. The most common risk factors for these problems are a history of high blood pressure (hypertension), high cholesterol, and heart disease. Blood clots or blockages in your blood vessels happen more often in people as they get older, and in people with a history of decreased blood flow, high blood pressure, diabetes, or high cholesterol. Heart problems. ICLUSIG can cause heart problems, including heart failure which can be serious and may lead to death. Heart failure means your heart does not pump blood well enough. ICLUSIG can also cause irregular, slow, or fast heartbeats and heart attack. Your healthcare provider will check you for heart problems during your treatment with ICLUSIG. Get medical help right away if you get any of the following symptoms: shortness of breath, chest pain, fast or irregular heartbeats, dizziness, or feel faint. Liver problems. ICLUSIG can cause liver problems, including liver failure, which can be severe and may lead to death. Your healthcare provider will do blood tests before and during your treatment with ICLUSIG to check for liver problems. Get medical help right away if you get any of these symptoms of liver problems during treatment:
|
|||
|
What is ICLUSIG?
ICLUSIG is a prescription medicine used to treat adults who have:
It is not known if ICLUSIG is safe and effective in children. |
|||
Before you take ICLUSIG, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
|
|
||
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I take ICLUSIG?
|
|||
|
What are the possible side effects of ICLUSIG? ICLUSIG may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
|
|
| The most common side effects of ICLUSIG when given with chemotherapy include: | |||
|
|
|
|
| Your healthcare provider may change your dose, temporarily stop, or permanently stop treatment with ICLUSIG if you have certain side effects. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of ICLUSIG. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store ICLUSIG?
Store ICLUSIG at room temperature between 68°F to 77°F (20°C to 25°C). Keep ICLUSIG and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of ICLUSIG
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ICLUSIG for a condition for which it was not prescribed. Do not give ICLUSIG to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about ICLUSIG that is written for health professionals. |
|||
|
What are the ingredients in ICLUSIG? TAKEDA and are registered trademarks of Takeda Pharmaceutical Company Limited. ©2025 Takeda Pharmaceuticals U.S.A., Inc. All rights reserved. For more information, go to www.iclusig.com or call 1-844-817-6468. ICL348 R12 |
Section 43683-2
| Warnings and Precautions (5) | 10/2025 |
Section 44425-7
Store ICLUSIG tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage
Overdoses with ICLUSIG were reported in clinical trials. One patient was estimated to have been administered 540 mg via nasogastric tube. Two hours after the overdosage, the patient had an uncorrected QT interval of 520 ms. Subsequent ECGs showed normal sinus rhythm with uncorrected QT intervals of 480 ms and 400 ms. The patient died 9 days after the overdosage from pneumonia and sepsis. Another patient self-administered 165 mg on Cycle 1 Day 2. The patient experienced fatigue and non-cardiac chest pain on Day 3. Multiple doses of 90 mg per day for 12 days in a patient resulted in pneumonia, systemic inflammatory response, atrial fibrillation, and a moderate pericardial effusion.
In the event of an overdosage, stop ICLUSIG, observe the patient and provide supportive treatment as appropriate.
11 Description
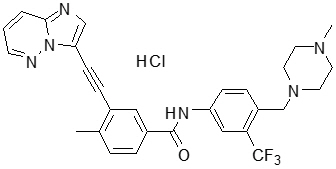
Ponatinib is a kinase inhibitor. The chemical name for ponatinib hydrochloride is 3-(imidazo[1,2-b]pyridazin-3-ylethynyl)-4-methyl-N-{4-[(4-methylpiperazin-1-yl)methyl]-3-(trifluoromethyl)phenyl}benzamide hydrochloride. The molecular formula is C29H28ClF3N6O which corresponds to a formula weight of 569.02 g/mol. Its structure is shown below:
Ponatinib HCl is an off-white to yellow powder with pKa of 2.77 and 7.8. The solubility of ponatinib in pH 1.7, 2.7, and 7.5 buffers is 7790 mcg/mL, 3.44 mcg/mL, and 0.16 mcg/mL, respectively, indicating a decrease in solubility with increasing pH. Each tablet for oral administration contains 10 mg, 15 mg, 30 mg or 45 mg of ponatinib equivalent to 10.68 mg, 16.03 mg, 32.05 mg, and 48.08 mg of ponatinib hydrochloride with the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (type B), colloidal silicon dioxide, magnesium stearate and a tablet coating. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol, and titanium dioxide.
5.8 Neuropathy
In PhALLCON, peripheral neuropathy occurred in 68% of 163 patients; 3.1% experienced Grade 3 or 4 peripheral neuropathy. The most frequent peripheral neuropathies were neuropathy peripheral (33%), paresthesia (22%), and peripheral sensory neuropathy (12%). The median time to onset of peripheral neuropathy was 1.1 month (range: 1 day to 17.2 months). Cranial neuropathy was reported in 0.6% of 163 patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, neuropathy occurred in 13% of patients. Peripheral neuropathy occurred in 11% of patients. The most frequently reported peripheral neuropathies were muscular weakness (3.2%), paresthesia (3.2%), hypoesthesia (2.1%) and neuropathy peripheral (2.1%). Cranial neuropathy developed in 2 patients. The median time to onset of peripheral neuropathy and cranial neuropathy was 1.1 years (range: 1 month to 4.1 years) and 3 years (range: 10.3 months to 5.2 years), respectively.
In PACE, neuropathy occurred in 22% of patients; 2.4% experienced Grade 3 or 4 neuropathy. Peripheral neuropathy occurred in 20% of 449 patients; 1.8% experienced Grade 3 or 4 peripheral neuropathy. The most frequent peripheral neuropathies were paresthesia (5%), neuropathy peripheral (4.5%), and hypoesthesia (3.6%). Cranial neuropathy developed in 3% of patients; 0.7% were Grade 3 or 4. The median time to onset of peripheral neuropathy and cranial neuropathy was 5.3 months (range: 1 day to 4.6 years) and 1.2 years (range: 18 days to 4 years), respectively.
Monitor patients for symptoms of neuropathy, such as hypoesthesia, hyperesthesia, paresthesia, discomfort, a burning sensation, neuropathic pain or weakness. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
5.10 Hemorrhage
Fatal and serious hemorrhage events have occurred in patients who received ICLUSIG.
In PhALLCON, hemorrhage occurred in 31% of 163 patients; 2.5% experienced a serious hemorrhage. Intracranial hemorrhage was the most frequently reported serious hemorrhage, occurring in 1.2% of patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, hemorrhage occurred in 13% of patients; 1 patient experienced a serious subdural hematoma.
In PACE, hemorrhage occurred in 28% of 449 patients; 6% experienced a serious hemorrhage and 1.3% experienced a fatal hemorrhage. The incidence of serious bleeding events was higher in patients with AP-CML, BP-CML, and Ph+ ALL. Gastrointestinal hemorrhage and subdural hematoma were the most frequently reported serious hemorrhages, each occurring in 0.9% of patients. Most hemorrhages occurred in patients with Grade 4 thrombocytopenia [see Warnings and Precautions (5.13)].
Monitor for hemorrhage and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
5.5 Hypertension
Serious or severe hypertension, including hypertensive crisis, has occurred in patients who received ICLUSIG.
In PhALLCON, hypertension occurred in 34% of 163 patients; 14% experienced serious or severe hypertension. Based on vital signs data, Grade 1 blood pressure elevation occurred in 15 out of 60 (25%) patients with normal initial blood pressure, Grade 2 occurred in 67 out of 134 (50%) patients with initial blood pressure of less than Grade 2, and Grade 3 occurred in 63 out of 160 (39%) patients with an initial blood pressure of less than Grade 3.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, hypertension events were reported in 37% of patients; 14% experienced serious or severe hypertension. Based on vital signs data, Grade 1 blood pressure elevation occurred in 8 out of 18 (44%) patients with normal initial blood pressure, Grade 2 occurred in 30 out of 81 (37%) patients with initial blood pressure of less than Grade 2, and Grade 3 occurred in 20 out of 92 (22%) patients with initial blood pressure of less than Grade 3. Three patients (3.2%) experienced hypertensive crisis.
In PACE, hypertension events were reported in 32% of 449 patients; 13% experienced serious or severe hypertension. Any post-baseline elevation of systolic or diastolic BP of Grade 2 or higher in patients with normal baseline blood pressure occurred in 44% of 449 patients. Grade 1 BP elevation occurred in 26%, Grade 2 in 45%, and Grade 3 in 26%. Two patients (<1%) experienced Grade 4 hypertension (hypertensive crisis).
Patients may require urgent clinical intervention for hypertension associated with confusion, headache, chest pain, or shortness of breath [see Adverse Reactions (6.1)]. Monitor blood pressure at baseline and as clinically indicated and manage hypertension as clinically indicated. Interrupt, dose reduce, or stop ICLUSIG if hypertension is not medically controlled [see Dosage and Administration (2.2)]. For significant worsening, labile or treatment-resistant hypertension, interrupt ICLUSIG and consider evaluating for renal artery stenosis.
5.6 Pancreatitis
Serious or severe pancreatitis has occurred in patients who received ICLUSIG.
In PhALLCON, pancreatitis occurred in 34% of 163 patients; 15% experienced serious or severe (Grade 3 or 4) pancreatitis. The median time to onset of pancreatitis was 8 days (range: 1 day to 2 years). In 7 patients with clinical pancreatitis that led to dose modification, pancreatitis resolved within 3 weeks. Laboratory abnormalities of amylase elevations occurred in 25% of patients, while lipase elevations occurred in 60% of patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, pancreatitis occurred in 29% of patients; 16% experienced serious or severe (Grade 3 or 4) pancreatitis. Pancreatitis resulted in discontinuation in 1.1% of patients and interruption and/or dose reduction in 23% of patients. The median time to onset of pancreatitis was 1 month (range: 3 days to 4.1 years). In two patients with clinical pancreatitis that led to dose modification or treatment discontinuation, pancreatitis resolved within 2 weeks. Laboratory abnormalities of amylase elevation occurred in 15% of patients, while lipase elevation occurred in 40% of patients.
In PACE, pancreatitis occurred in 26% of 449 patients; 17% experienced serious or severe (Grade 3 or 4) pancreatitis. Pancreatitis resulted in discontinuation in 0.4% of patients and interruption and/or dose reduction in 17% of patients. The median time to onset of pancreatitis was 29 days (range: 1 day to 4 years). Nineteen of the 28 cases of clinical pancreatitis that led to dose modification or treatment discontinuation resolved within 2 weeks. Laboratory abnormalities of amylase elevations occurred in 18% of patients, while lipase elevations occurred in 39% of patients.
Monitor serum lipase every 2 weeks for the first 2 months and then monthly thereafter or as clinically indicated. Consider additional serum lipase monitoring in patients with a history of pancreatitis or alcohol abuse. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on severity [see Dosage and Administration (2.2)]. Evaluate for pancreatitis when lipase elevation is accompanied by abdominal symptoms.
5.3 Heart Failure
Fatal, serious or severe heart failure events have occurred in patients who received ICLUSIG.
In PhALLCON, heart failure occurred in 6% of 163 patients; 1.2% experienced serious or severe (Grade 3 or 4) heart failure. The most frequently reported heart failure event (>1 patient) was increased brain natriuretic peptide (BNP) (2.5%).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, heart failure occurred in 20% of patients; 2.1% experienced serious or severe (Grade 3 or 4) heart failure. The most frequently reported heart failure events (>1 patient each) were left ventricular hypertrophy (5%), left ventricular dysfunction (5%), BNP increased (5%), cardiac failure (3.2%), left atrial dilatation (2.1%) and ejection fraction decreased (2.1%).
Fatal or serious heart failure occurred in PACE. Heart failure occurred in 9% of 449 patients; 7% experienced serious or severe (Grade 3 or higher) heart failure. The most frequently reported heart failure events (≥2%) were congestive cardiac failure (3.1%), decreased ejection fraction (2.9%), and cardiac failure (2%).
Monitor patients for signs or symptoms consistent with heart failure and manage heart failure as clinically indicated. Interrupt, then resume at reduced dose or discontinue ICLUSIG for new or worsening heart failure [see Dosage and Administration (2.2)].
8.4 Pediatric Use
Safety and effectiveness of ICLUSIG have not been established in pediatric patients.
8.5 Geriatric Use
Of the 163 patients with Ph+ALL who received ICLUSIG in PhALLCON, 21% were 65 years and older and 7% were 75 years and older. Overall, no differences in efficacy of ICLUSIG were observed between patients 65 years of age or older compared to younger patients. AOEs occurred in 21% (7/34) of patients 65 years and older and 2.3% (3/129) of patients less than 65 years of age.
Of the 94 patients with CP-CML who received ICLUSIG at a starting dose of 45 mg in OPTIC, 17% were 65 years and older and 2.1% were 75 years and older. Patients aged 65 years and older had a lower ≤1% BCR::ABL1IS rate at 12 months (27%) than patients less than 65 years of age (47%). By 60 months, patients aged 65 years and older had a ≤1% BCR::ABL1IS rate of 40% and patients less than 65 years of age had a rate of 64%. AOEs occurred in 38% (6/16) of patients 65 years and older and 14% (11/78) of patients less than 65 years of age [see Warnings and Precautions (5.1)].
Of the 449 patients who received ICLUSIG in PACE, 35% were 65 years and older and 8% were 75 years and older. In patients with CP-CML, patients aged 65 years and older had a lower major cytogenetic response rate (40%) as compared with patients less than 65 years of age (65%). In patients with AP-CML, BP-CML, and Ph+ ALL, patients aged 65 years and older had a similar hematologic response rate (45%) as compared with patients less than 65 years of age (44%). AOEs occurred in 35% (54/155) of patients 65 years and older and in 21% (61/294) of patients less than 65 years of age [see Warnings and Precautions (5.1)].
Patients aged 65 years or older are more likely to experience adverse reactions including vascular occlusion, decreased platelet count, peripheral edema, increased lipase, dyspnea, asthenia, muscle spasms, and decreased appetite. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
5.4 Hepatotoxicity
ICLUSIG can cause hepatotoxicity, including liver failure and death. Fulminant hepatic failure leading to death occurred in 3 patients, with hepatic failure occurring within 1 week of starting ICLUSIG in one of these patients. These fatal cases occurred in patients with BP-CML or Ph+ ALL treated with monotherapy.
In PhALLCON, hepatotoxicity occurred in 66% of 163 patients; 30% experienced Grade 3 or 4 hepatotoxicity. The median time to onset of hepatotoxicity was 15 days (range: 1 day to 10 months). The most frequent hepatotoxic events were elevated alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), bilirubin and alkaline phosphatase, decreased albumin and decreased blood fibrinogen. In 6% of the 73 patients who reported ALT or AST elevation, the elevations were not resolved by the date of the last follow-up.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, hepatotoxicity occurred in 34% of patients; 7% experienced Grade 3 or 4 hepatotoxicity. The median time to onset of hepatotoxicity was 4.1 months, with a range of 1 day to 4.8 years. The most frequent hepatotoxic events were elevations of ALT, AST, alkaline phosphatase, and GGT. In one of the 26 patients who reported ALT or AST elevation, the event was not resolved by the date of last follow-up.
In PACE, hepatotoxicity occurred in 32% of 449 patients; 13% experienced Grade 3 or 4 hepatotoxicity. The median time to onset of hepatotoxicity was 3.1 months, with a range of 1 day to 4.9 years. The most frequent hepatotoxic events were elevations of ALT, AST, GGT, bilirubin, and alkaline phosphatase. In 9% of the 88 patients who reported ALT or AST elevation, the event was not resolved by the date of last follow-up.
Monitor liver function tests at baseline, then at least monthly or as clinically indicated. Interrupt, then resume at reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
4 Contraindications
None.
5.9 Ocular Toxicity
Serious ocular toxicities leading to blindness or blurred vision have occurred in ICLUSIG-treated patients.
In PhALLCON, ocular toxicities occurred in 33% of 163 patients; 1.8% experienced a serious or severe ocular toxicity. The most frequent ocular toxicities were blurred vision and dry eye. Retinal toxicities occurred in 4.3% of patients; 0.6% experienced a Grade 3 retinal vein occlusion. The most frequent retinal toxicity event (>1 patient) was retinal hemorrhage (1.8%).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, ocular toxicities occurred in 15% of patients; 1.1% experienced a serious or severe ocular toxicity. The most frequent ocular toxicities were dry eye, blurred vision and eye pain. Retinal toxicities occurred in 4.3% of patients, including age-related macular degeneration, arteriosclerotic retinopathy, retinal vascular disorder and retinal vein occlusion (1.1% each).
In PACE, ocular toxicities occurred in 30% of 449 patients; 3.6% experienced a serious or severe ocular toxicity. The most frequent ocular toxicities were dry eye, blurred vision, and eye pain. Retinal toxicities occurred in 3.6% of patients. The most frequent retinal toxicities were macular edema, retinal vein occlusion, retinal hemorrhage, and vitreous floaters (0.7% each).
Conduct comprehensive eye exams at baseline and periodically during treatment.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Arterial Occlusive Events [see Warnings and Precautions (5.1)]
- Venous Thromboembolic Events [see Warnings and Precautions (5.2)]
- Heart Failure [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warnings and Precautions (5.4)]
- Hypertension [see Warnings and Precautions (5.5)]
- Pancreatitis [see Warnings and Precautions (5.6)]
- Neuropathy [see Warnings and Precautions (5.8)]
- Ocular Toxicity [see Warnings and Precautions (5.9)]
- Hemorrhage [see Warnings and Precautions (5.10)]
- Fluid Retention [see Warnings and Precautions (5.11)]
- Cardiac Arrhythmias [see Warnings and Precautions (5.12)]
- Myelosuppression [see Warnings and Precautions (5.13)]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.14)]
- Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.15)]
- Impaired Wound Healing and Gastrointestinal Perforation [see Warnings and Precautions (5.16)]
7 Drug Interactions
5.11 Fluid Retention
Fatal and serious fluid retention events have occurred in patients who received ICLUSIG.
In PhALLCON, fluid retention occurred in 24% of 163 patients; 1.2% experienced serious fluid retention, including pericardial effusion (1.2%). The most frequent occurrences of fluid retention were peripheral edema (11%) and pleural effusion (6%).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, fluid retention occurred in 6% of patients. The most frequent fluid retention events were peripheral edema (3.2%), hydrothorax (2.1%) and pleural effusion (2.1%).
In PACE, fluid retention events occurred in 33% of 449 patients; 4.5% experienced serious fluid retention. One instance of brain edema was fatal. Serious fluid retention included pleural effusion (1.6%), pericardial effusion (1.6%), and angioedema (0.4%). The most frequent fluid retention events were peripheral edema (17%), pleural effusion (9%), pericardial effusion (4.2%) and peripheral swelling (3.8%).
Monitor for fluid retention and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
12.2 Pharmacodynamics
In PACE, the dose intensity-safety relationship indicated that there are significant increases in Grade ≥3 adverse reactions (hypertension, thrombocytopenia, pancreatitis, neutropenia, rash, ALT increase, AST increase, lipase increase, myelosuppression) over the dose range of 15 mg to 45 mg. In addition to dose, increased age and history of ischemia, hypertension, diabetes, or hypercholesterolemia were also contributory factors to a higher incidence of AOEs.
In OPTIC, an exposure-response relationship between ponatinib exposure and molecular response rate at 12 months was observed. A relationship between higher ponatinib exposures and higher incidence of adverse reactions, including thrombocytopenia (Grade ≥3) and AOEs, was observed.
In vitro, there was no significant inhibition of platelet aggregation with ponatinib at concentrations seen clinically and up to 0.7 mcg/mL (1.23 μM).
12.3 Pharmacokinetics
Ponatinib administered to patients with cancer exhibited approximately dose proportional increases in both steady-state Cmax and AUC over the dose range of 2 mg to 60 mg (0.04 to 1.33 times the approved maximum recommended starting dose). The mean (CV%) Cmax and AUC(0-24) of ICLUSIG 45 mg orally once daily at presumed steady-state in patients with advanced hematologic malignancies were 73 ng/mL (74%) and 1253 ng∙hr/mL (73%), respectively. The mean (CV%) Cmax and AUC(0-24) of ICLUSIG 30 mg orally once daily at presumed steady-state in patients with advanced hematologic malignancies were 65 ng/mL (28%) and 1080 ng∙hr/mL (29%), respectively. Exposure increased by approximately 90% (median) [range: 20% to 440%] between the first dose and presumed steady-state.
5.13 Myelosuppression
In PhALLCON, neutropenia occurred in 66% (Grade 3 or 4 occurred in 63%), thrombocytopenia occurred in 65% (Grade 3 or 4 occurred in 62%) and anemia occurred in 53% (Grade 3 or 4 occurred in 38%) of 163 patients. The median time to onset of Grade 3 or 4 myelosuppression was 27 days (range: 1 day to 9.2 months).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, thrombocytopenia occurred in 66% (Grade 3 or 4 occurred in 31%), neutropenia occurred in 56% (Grade 3 or 4 occurred in 22%), and anemia occurred in 38% of patients (Grade 3 or 4 occurred in 14%). The median time to onset of Grade 3 or 4 myelosuppression was 1.3 months (range: 1 day to 1.2 years).
In PACE, neutropenia occurred in 56% (Grade 3 or 4 occurred in 34%), thrombocytopenia occurred in 63% (Grade 3 or 4 occurred in 40%), and anemia occurred in 52% of patients (Grade 3 or 4 occurred in 20%). The incidence of myelosuppression was greater in patients with AP-CML, BP-CML, and Ph+ ALL than in patients with CP-CML. Severe myelosuppression (Grade 3 or 4) was observed early in treatment, with a median onset time of 29 days (range: 1 day to 4.1 years).
Obtain complete blood counts every 2 weeks for the first 3 months and then monthly or as clinically indicated. If ANC less than 1 × 109/L or platelets less than 50 × 109/L, interrupt ICLUSIG until ANC at least 1.5 × 109/L and platelets at least 75 × 109/L, then resume at same or reduced dose [see Dosage and Administration (2.2)].
8.6 Hepatic Impairment
Patients with hepatic impairment are more likely to experience adverse reactions compared to patients with normal hepatic function. For patients with CP-CML, AP-CML, BP-CML, and Ph+ ALL receiving monotherapy, reduce the starting dose of ICLUSIG for patients with pre-existing hepatic impairment (Child-Pugh A, B, or C). For patients with newly diagnosed Ph+ ALL, dosage adjustment is not recommended when administering ICLUSIG to patients with mild hepatic impairment (Child-Pugh A). Clinical data in patients with newly diagnosed Ph+ ALL with pre-existing moderate or severe hepatic impairment (Child-Pugh B or C) is not available and patients should be closely monitored for potential increased incidence of adverse reactions. Modify the ICLUSIG dosage in the event of adverse reactions [see Dosage and Administration (2.2, 2.4), Clinical Pharmacology (12.3)]. The safety of multiple doses, or doses higher than 30 mg, has not been studied in patients with hepatic impairment.
1 Indications and Usage
ICLUSIG® is indicated for the treatment of adult patients with:
Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia (Ph+ ALL)
- Newly diagnosed Ph+ ALL in combination with chemotherapy.
This indication is approved under accelerated approval based on minimal residual disease (MRD)-negative complete remission (CR) at the end of induction [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification of clinical benefit in a confirmatory trial(s). - As monotherapy in Ph+ ALL for whom no other kinase inhibitors are indicated or T315I-positive Ph+ ALL.
Chronic Myeloid Leukemia (CML)
- Chronic phase (CP) CML with resistance or intolerance to at least two prior kinase inhibitors.
- Accelerated phase (AP) or blast phase (BP) CML for whom no other kinase inhibitors are indicated.
- T315I-positive CML (chronic phase, accelerated phase, or blast phase).
12.1 Mechanism of Action
Ponatinib is a kinase inhibitor. Ponatinib inhibited the in vitro tyrosine kinase activity of ABL and T315I mutant ABL with IC50 concentrations of 0.4 nM and 2.0 nM, respectively. Ponatinib inhibited the in vitro activity of additional kinases with IC50 concentrations between 0.1 nM and 20 nM, including members of the VEGFR, PDGFR, FGFR, EPH receptors and SRC families of kinases, and KIT, RET, TIE2, and FLT3. Ponatinib inhibited the in vitro viability of cells expressing native or mutant BCR::ABL, including T315I. In mice, treatment with ponatinib reduced the size of tumors expressing native or T315I mutant BCR::ABL when compared to controls.
5.12 Cardiac Arrhythmias
In PhALLCON, cardiac arrhythmia events occurred in 22% of 163 patients; 2.5% experienced Grade 3 or 4 cardiac arrhythmias, including tachycardia, syncope, atrial fibrillation and supraventricular tachycardia (0.6%, each).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, cardiac arrhythmias occurred in 27% of patients; 5% experienced Grade 3 or 4 cardiac arrhythmias including atrial fibrillation, cardio-respiratory arrest, supraventricular extrasystoles, supraventricular tachycardia and syncope (1.1%, each).
In PACE, cardiac arrhythmias occurred in 20% of 449 patients; 7% experienced Grade 3 or 4 cardiac arrhythmias. Ventricular arrhythmias occurred in 3.4% of the 89 patients who reported an arrhythmia, with one event being Grade 3 or 4. Symptomatic bradyarrhythmias that led to pacemaker implantation occurred in 1% of patients. Atrial fibrillation was the most frequent cardiac arrhythmia (8%), with 3.3% being Grade 3 or 4. Other Grade 3 or 4 arrhythmia events included syncope (2%), tachycardia and bradycardia (0.4% each), and QT interval prolongation, atrial flutter, sinus bradycardia, supraventricular tachycardia, ventricular tachycardia, atrial tachycardia, atrioventricular block complete, cardio-respiratory arrest, loss of consciousness, and sinus node dysfunction (0.2% each). For 31 patients, the arrythmia led to hospitalization.
Monitor for signs and symptoms suggestive of slow heart rate (fainting, dizziness) or rapid heart rate (chest pain, palpitations or dizziness) and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity.
5.14 Tumor Lysis Syndrome
In PhALLCON, serious tumor lysis syndrome (TLS) developed in 0.6% of 163 patients. Hyperuricemia occurred in 10% of patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, serious TLS developed in 1.1% of patients. Hyperuricemia occurred in 2.1% of patients.
In PACE, serious TLS developed in 0.4% of 449 patients. One case occurred in a patient with advanced AP-CML and 1 case occurred in a patient with BP-CML. Hyperuricemia occurred in 7% of patients.
Ensure adequate hydration and treat high uric acid levels prior to initiating ICLUSIG.
5 Warnings and Precautions
- Hypertension: Monitor blood pressure and manage hypertension as clinically indicated. Interrupt, dose reduce or stop ICLUSIG if hypertension is not medically controlled. (2.2, 5.5)
- Pancreatitis: Monitor serum lipase. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on severity. Evaluate for pancreatitis when lipase elevation is accompanied by abdominal symptoms. (2.2, 5.6)
- Neuropathy: Monitor for symptoms of peripheral and cranial neuropathy. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (2.2, 5.8)
- Ocular Toxicity: Conduct comprehensive eye exams at baseline and periodically during treatment. (5.9)
- Hemorrhage: Monitor for hemorrhage and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (2.2, 5.10)
- Fluid Retention: Monitor for fluid retention and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (2.2, 5.11)
- Cardiac Arrhythmias: Monitor for signs or symptoms of arrhythmias and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (5.12)
- Myelosuppression: Obtain complete blood counts every 2 weeks for the first 3 months and then monthly or as clinically indicated. If ANC less than 1 × 109/L or platelets less than 50 × 109/L, interrupt ICLUSIG until ANC at least 1.5 × 109/L and platelets at least 75 × 109/L, then resume at same or reduced dose. (2.2, 5.13)
- Tumor Lysis Syndrome: Ensure adequate hydration and correct elevated uric acid levels prior to initiating ICLUSIG. (5.14)
- Reversible Posterior Leukoencephalopathy Syndrome (RPLS): Interrupt ICLUSIG until resolution. The safety of resumption of ICLUSIG in patients upon resolution of RPLS is unknown. (5.15)
- Impaired Wound Healing and Gastrointestinal Perforation: Withhold ICLUSIG for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of ICLUSIG after resolution of wound healing complications has not been established. (5.16)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.17, 8.1, 8.3)
5.17 Embryo Fetal Toxicity
Based on its mechanism of action and findings from animal studies, ICLUSIG can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, oral administration of ponatinib to pregnant rats during organogenesis caused adverse developmental effects at exposures lower than human exposures at the maximum recommended human dose of 45 mg/day. Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with ICLUSIG and for 3 weeks after the last dose [see Use in Specific Populations (8.1, 8.3)].
2 Dosage and Administration
- Recommended Dosage in Newly Diagnosed Ph+ ALL: Starting dose is 30 mg orally once daily in combination with chemotherapy, with a reduction to 15 mg once daily upon achievement of MRD-negative (≤0.01% BCR::ABL1/ABL1) CR at the end of induction. (2.1)
- Recommended Dosage in Monotherapy for Ph+ ALL for Whom No Other Kinase Inhibitors are Indicated or T315I-positive Ph+ ALL: Starting dose is 45 mg orally once daily. (2.1)
- Recommended Dosage in CP-CML: Starting dose is 45 mg orally once daily with a reduction to 15 mg once daily upon achievement of ≤1% BCR::ABL1IS. (2.1)
- Recommended Dosage in AP-CML and BP-CML: Starting dose is 45 mg orally once daily. (2.1)
- Hepatic Impairment: See the Full Prescribing Information for dosage modifications for hepatic impairment. (2.4)
- ICLUSIG may be taken with or without food. (2.1)
3 Dosage Forms and Strengths
Tablets, film-coated:
- 10 mg of ponatinib: Oval, white to off-white, biconvex, debossed "NZ" on one side and plain on the other side
- 15 mg of ponatinib: Round, white, biconvex, debossed "A5" on one side and plain on the other side
- 30 mg of ponatinib: Round, white, biconvex, debossed "C7" on one side and plain on the other side
- 45 mg of ponatinib: Round, white, biconvex, debossed "AP4" on one side and plain on the other side
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ICLUSIG. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Blood and Lymphatic System Disorders: Thrombotic microangiopathy
Endocrine Disorders: Hyperthyroidism
Gastrointestinal Disorders: Gastrointestinal perforation, fistula
Metabolism and Nutrition Disorders: Dehydration
Nervous System Disorders: Reversible posterior leukoencephalopathy syndrome (RPLS)
Skin and Subcutaneous Tissue Disorders: Severe cutaneous reaction (e.g., Erythema multiforme, Stevens-Johnson syndrome), impaired wound healing, panniculitis (including erythema nodosum)
Vascular Disorders: Arterial (including aortic) aneurysms, dissections, and rupture
5.1 Arterial Occlusive Events
Arterial occlusive events (AOEs), including fatalities, occurred in patients who received ICLUSIG [see Adverse Reactions (6.1)].
In PhALLCON, 6% of 163 patients experienced AOEs, of which 3.1%, 1.8%, and 1.2% experienced cardiovascular, cerebrovascular, and peripheral vascular AOEs, respectively. The median time to onset of the first AOE was 11.3 months (range: 8 days to 2.8 years). Grade 3 or 4 AOEs occurred in 3.7% of patients; the most frequent Grade 3 or 4 AOEs were myocardial infarction (1.2%), peripheral arterial occlusive disease (1.2%), angina pectoris and cerebrovascular accident (0.6% each). Fatal AOE of sudden death occurred in 1 patient (0.6%). AOEs were more frequent with increasing age [see Use in Specific Populations (8.5)].
In PhALLCON, patients with uncontrolled hypertension, hypertriglyceridemia, or diabetes were excluded. Patients with clinically significant, uncontrolled, or active cardiovascular disease, including any history of myocardial infarction, peripheral vascular infarction, revascularization procedure, venous thromboembolism, clinically significant atrial/ventricular tachyarrhythmias, unstable angina, or congestive heart failure within the 6 months prior to the first dose of ICLUSIG, were also excluded.
In OPTIC, of the 94 patients who received a starting dose of 45 mg (45 mg → 15 mg), 18% experienced AOEs, of which 11%, 4.3%, and 3.2% experienced cardiovascular, cerebrovascular or peripheral vascular AOEs, respectively. The median time to onset of the first cardiovascular, cerebrovascular, or peripheral vascular event was 9.4 months (range: 12 days to 5.7 years), 11.7 months (range: 15 days to 1.6 years), and 6.3 months (range: 23 days to 3.6 years), respectively. Grade 3 or 4 AOEs occurred in 7% of patients; the most frequent Grade 3 or 4 AOEs were myocardial infarction, acute coronary syndrome, arterial thrombosis, ischemic stroke, ischemic cerebral infarction, subclavian artery stenosis and unstable angina (1.1% each). Fatal AOEs occurred in 4 patients (4.3%); including sudden death (2.1%), myocardial ischemia (1.1%) and myocardial infarction (1.1%). AOEs were more frequent with increasing age [see Use in Specific Populations (8.5)].
In OPTIC, patients with uncontrolled hypertension or diabetes and patients with clinically significant, uncontrolled, or active cardiovascular disease, including any history of myocardial infarction, peripheral vascular infarction, revascularization procedure, congestive heart failure, venous thromboembolism, or clinically significant atrial/ventricular arrhythmias, were excluded.
In PACE, 26% of 449 patients experienced AOEs, of which 15%, 7%, and 11% experienced cardiovascular, cerebrovascular, and peripheral vascular AOEs, respectively. Some patients experienced recurrent or multisite vascular occlusion. The median time to onset of the first cardiovascular, cerebrovascular, and peripheral vascular AOEs was 1 year (range: 1 day to 4.1 years), 1.4 years (range: 2 days to 4.5 years), and 2 years (range: 10 days to 4.9 years), respectively. Grade 3 or 4 AOEs occurred in 14% of patients; the most frequent Grade 3 or 4 AOEs were peripheral arterial occlusive disease (3.1%), myocardial infarction (2%), coronary artery disease (1.6%), and cerebral infarction (1.6%). Fatal AOEs occurred in 9 patients (2%); the most frequent fatal AOE was cardiac arrest (0.9%).
In PACE, fatal and life-threatening AOEs occurred within 2 weeks of starting treatment at 45 mg, and at dose levels as low as 15 mg per day. Patients with and without cardiovascular risk factors, including patients age 50 years or younger, experienced AOEs. AOEs were more frequent with increasing age [see Use in Specific Populations (8.5)] and in patients with history of ischemia, hypertension, diabetes, or hypercholesterolemia. The most common risk factors in patients with AOEs were history of hypertension (67%; 77/115), hypercholesterolemia (59%; 68/115), and non-ischemic cardiac disease (43%; 49/115).
In PACE, patients developed heart failure concurrent or subsequent to a myocardial ischemic event [see Warnings and Precautions (5.3)]. Patients required revascularization procedures (coronary, cerebrovascular, and peripheral arterial). ICLUSIG caused stenosis over multiple segments in major arterial vessels that supply the brain (e.g., carotid, vertebral, middle cerebral artery). Patients developed digital or distal extremity necrosis and required amputations. Renal artery stenosis associated with worsening, labile or treatment-resistant hypertension occurred in some ICLUSIG-treated patients [see Warnings and Precautions (5.5)].
In PACE, patients with uncontrolled hypertriglyceridemia and patients with clinically significant or active cardiovascular disease, including any history of clinically significant atrial/ventricular arrhythmias or history of myocardial infarction, unstable angina, or congestive heart failure within the 3 months prior to the first dose of ICLUSIG, were excluded [see Adverse Reactions (6.1)]. Consider whether the benefits of ICLUSIG are expected to exceed the risks.
Monitor for evidence of AOEs. Interrupt, then resume at the same or decreased dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)]. Consider benefit-risk to guide a decision to restart ICLUSIG.
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most common adverse reactions identified in the Highlights of the Prescribing Information are based on two safety populations. The first is from a pooled safety population of 543 patients with CML or resistant or intolerant Ph+ ALL (OPTIC and PACE studies) who received ICLUSIG as a single agent at a starting dose of 45 mg orally once daily. In this pooled safety population, the most common (>20%) adverse reactions were rash and related conditions, arthralgia, abdominal pain, fatigue, headache, constipation, hypertension, dry skin, hepatotoxicity, fluid retention and edema, pyrexia, pancreatitis/lipase elevation, nausea, hemorrhage, anemia, AOEs and cardiac arrhythmias. The most common Grade 3 or 4 laboratory abnormalities (>20%) were platelet count decreased, neutrophil cell count decreased, and white blood cell decreased.
The second safety population is from 163 patients with newly diagnosed Ph+ ALL (PhALLCON study) who received ICLUSIG in combination with chemotherapy at a starting dose of 30 mg orally once daily. The most common adverse reactions (>20%) included hepatotoxicity, arthralgia, rash and related conditions, headache, pyrexia, abdominal pain, constipation, fatigue, nausea, oral mucositis, hypertension, pancreatitis/lipase elevation, neuropathy peripheral, hemorrhage, febrile neutropenia, fluid retention and edema, vomiting, paresthesia, and cardiac arrhythmias. The most common Grade 3 or 4 laboratory abnormalities (>20%) included decreased white blood cell count, decreased neutrophil cell count, decreased platelet count, decreased lymphocyte cell count, decreased hemoglobin, increased lipase, and increased ALT.
Newly Diagnosed Ph+ ALL
The safety of ICLUSIG was evaluated in PhALLCON, a randomized, active-controlled, multicenter trial conducted in patients with newly diagnosed Ph+ ALL [see Clinical Studies (14)]. Patients received ICLUSIG (n=163) or imatinib 600 mg (n=81) in combination with reduced-intensity chemotherapy followed by continued treatment with ICLUSIG or imatinib as a single agent (imatinib in combination with chemotherapy is not an approved regimen in adult patients). In the ICLUSIG arm, patients received a starting dosage of ICLUSIG 30 mg orally once daily in combination with chemotherapy, with a reduction to 15 mg orally once daily upon achievement of MRD-negative CR at the end of induction. The median duration of exposure was 9.0 months (range: <1 month to 4.2 years) in the ICLUSIG arm and 5.2 months (range: <1 month to 4.4 years) in the imatinib arm.
Patients with uncontrolled hypertension, hypertriglyceridemia, or diabetes and patients with clinically significant, uncontrolled, or active cardiovascular disease, including any history of myocardial infarction, peripheral vascular infarction, revascularization procedure, venous thromboembolism, clinically significant atrial/ventricular tachyarrhythmias, history of myocardial infarction, unstable angina, or congestive heart failure within the 6 months prior to the first dose of ICLUSIG, were excluded.
Serious adverse reactions occurred in 63% of patients receiving ICLUSIG in combination with chemotherapy. Serious adverse reactions in >2% of patients included febrile neutropenia (18%), pyrexia (6%), thrombocytopenia (4.3%), sepsis (3.7%), septic shock (3.7%), anemia (2.5%), hemorrhage (2.5%), neutropenia (2.5%), pancreatitis (2.5%), peripheral neuropathy (2.5%), pneumonia (2.5%) and acute kidney injury (2.5%). Fatal adverse reactions occurred in 6% of patients who received ICLUSIG in combination with chemotherapy, including sepsis (3.7%), sudden death, pneumonitis and respiratory failure (0.6%, each).
Permanent discontinuation of ICLUSIG due to adverse reactions occurred in 13% of patients. Adverse reactions resulting in permanent discontinuation of ICLUSIG in >2% of patients included arterial occlusive events and sepsis.
Dosage modifications (dose interruption or reduction) of ICLUSIG due to adverse reactions occurred in 71% of patients. Adverse reactions leading to dose interruption or reduction of ICLUSIG in >5% of patients included increased ALT, neutropenia, increased lipase, thrombocytopenia, increased AST, febrile neutropenia, and abdominal pain.
Table 4 summarizes the adverse reactions in patients receiving ICLUSIG or imatinib in combination with chemotherapy in PhALLCON.
| Adverse Reaction | ICLUSIG 30 mg → 15 mg with Chemotherapy (n = 163) |
Imatinib 600 mg with Chemotherapy (n = 81) |
||
|---|---|---|---|---|
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| Graded using CTCAE v5.0 | ||||
| Hepatobiliary Disorders | ||||
| Hepatotoxicity | 66 | 30 | 57 | 14 |
| Musculoskeletal and Connective Tissue Disorders | ||||
| Arthralgia Includes arthralgia, arthritis, back pain, flank pain, intervertebral disc degeneration, joint swelling, osteoarthritis, neck pain, pain, pain in extremity, pain of skin, sciatica, spinal pain, tendonitis, and tenosynovitis.
|
47 | 4.3 | 35 | 1.2 |
| Myalgia | 13 | 1.2 | 10 | 1.2 |
| Nervous System Disorders | ||||
| Headache | 45 | 1.8 | 43 | 1.2 |
| Neuropathy peripheral | 33 | 1.2 | 24 | 1.2 |
| Paresthesia | 22 | 0 | 10 | 0 |
| Peripheral sensory neuropathy | 12 | 0 | 12 | 0 |
| Skin and Subcutaneous Tissue Disorders | ||||
| Rash and related conditions | 47 | 1.2 | 33 | 1.2 |
| Gastrointestinal Disorders | ||||
| Abdominal pain Includes abdominal discomfort, abdominal distension, abdominal pain, abdominal pain lower, abdominal pain upper, chronic gastritis, colitis, enteritis, enterocolitis, gastric ulcer, gastritis, gastroenteritis, gastrointestinal pain, gastroesophageal reflux disease, and helicobacter gastritis.
|
43 | 4.9 | 28 | 0 |
| Constipation | 41 | 0.6 | 21 | 1.2 |
| Nausea | 37 | 3.1 | 52 | 7 |
| Oral mucositis | 35 | 4.9 | 30 | 10 |
| Pancreatitis/lipase elevation | 34 | 15 | 37 | 20 |
| Vomiting | 24 | 1.2 | 40 | 2.5 |
| Diarrhea | 20 | 0 | 35 | 2.5 |
| General Disorders | ||||
| Pyrexia | 44 | 4.3 | 26 | 2.5 |
| Fatigue or asthenia | 40 | 2.5 | 38 | 3.7 |
| Fluid retention and edema | 24 | 0.6 | 48 | 3.7 |
| Vascular Disorders | ||||
| Hypertension | 34 | 14 | 15 | 7 |
| Hemorrhage | 31 | 1.8 | 30 | 7 |
| Venous thromboembolic events | 12 | 3.1 | 10 | 2.5 |
| Blood and Lymphatic System Disorders | ||||
| Febrile neutropenia | 28 | 25 | 22 | 20 |
| Metabolism and Nutrition Disorders | ||||
| Impaired glucose tolerance | 20 | 4.9 | 20 | 9 |
| Hyperlipidemia | 16 | 1.2 | 15 | 1.2 |
| Decreased appetite | 10 | 0 | 19 | 3.7 |
| Cardiac Disorders | ||||
| Cardiac arrhythmias | 22 | 2.5 | 17 | 6 |
| Infections | ||||
| Sepsis Includes abdominal sepsis, bacteremia, bacterial sepsis, device-related sepsis, escherichia bacteremia, fungemia, klebsiella bacteremia, klebsiella sepsis, neutropenic sepsis, pseudomonal sepsis, sepsis, septic shock, staphylococcal bacteremia, staphylococcal sepsis, streptococcal bacteremia, and urosepsis.
|
17 | 12 | 15 | 11 |
| Pneumonia | 11 | 7 | 11 | 6 |
| Respiratory, Thoracic, and Mediastinal Disorders | ||||
| Cough | 17 | 0 | 6 | 0 |
| Dyspnea | 13 | 1.2 | 4.9 | 2.5 |
Clinically relevant adverse reactions in ≤10% of patients receiving ICLUSIG with chemotherapy: urinary tract infection (10%), arterial occlusive events (6%), cardiac failure (6%), and acute kidney injury (4.3%).
Table 5 summarizes the laboratory abnormalities in PhALLCON for patients who received ICLUSIG or imatinib in combination with chemotherapy.
| Laboratory Abnormality | ICLUSIG 30 mg → 15 mg with Chemotherapy (n = 163) |
Imatinib 600 mg with Chemotherapy (n = 81) |
||
|---|---|---|---|---|
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase Graded using CTCAE v5.0 |
||||
| Hematologic Laboratory Tests | ||||
| White blood cell decreased | 79 | 71 | 78 | 70 |
| Lymphocyte cell count decreased | 77 | 61 | 94 | 89 |
| Neutrophil cell count decreased | 66 | 63 | 57 | 53 |
| Platelet count decreased | 65 | 62 | 64 | 53 |
| Hemoglobin decreased | 53 | 38 | 59 | 49 |
| Liver Function Tests | ||||
| ALT increased | 69 | 21 | 62 | 7 |
| AST increased | 53 | 7 | 48 | 6 |
| Alkaline phosphatase increased | 44 | 1.2 | 24 | 0 |
| Total bilirubin increased | 25 | 0.6 | 24 | 0 |
| Direct bilirubin increased | 24 | 4.3 | 24 | 1.2 |
| Pancreatic Enzymes | ||||
| Lipase increased | 60 | 24 | 78 | 38 |
| Amylase increased | 25 | 6 | 35 | 7 |
| Chemistry | ||||
| Calcium decreased | 67 | 3.1 | 69 | 4.9 |
| Phosphate decreased | 58 | 16 | 85 | 36 |
| Potassium decreased | 44 | 10 | 74 | 25 |
| Albumin decreased | 42 | 1.8 | 56 | 0 |
| Glucose increased | 34 | 2.5 | 38 | 2.5 |
| Creatinine increased | 34 | 3.7 | 48 | 4.9 |
| Sodium decreased | 32 | 3.1 | 35 | 3.7 |
| Potassium increased | 31 | 3.7 | 12 | 0 |
| Magnesium decreased | 15 | 0.6 | 31 | 2.5 |
5.2 Venous Thromboembolic Events
Serious or severe VTEs have occurred in patients who received ICLUSIG.
In PhALLCON, VTEs occurred in 12% of 163 patients, including serious or severe (Grade 3 or 4) in 3.1%. VTEs included deep vein thrombosis (6%), superficial vein thrombosis (2.5%), embolism (1.8%), pulmonary embolism and thrombosis (1.2% each), and jugular vein thrombosis and retinal vein occlusion (0.6% each). The median time to onset of the first VTE event was 2.5 months (range: 6 days to 1.8 years).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, 2 patients experienced a VTE (Grade 1 retinal vein occlusion and grade 2 phlebitis).
In PACE, VTEs occurred in 6% of 449 patients, including serious or severe (Grade 3 or 4) in 5.8%. VTEs included deep venous thrombosis (2.2%), pulmonary embolism (1.8%), superficial thrombophlebitis (0.7%), retinal vein occlusion (0.7%), and retinal vein thrombosis (0.4%) with vision loss. VTEs occurred in 10% of the 62 patients with BP-CML, 9% of the 32 patients with Ph+ ALL, 6% of the 270 patients with CP-CML, and 3.5% of the 85 patients with AP-CML.
Monitor for evidence of VTEs. Interrupt, then resume at the same or decreased dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
ICLUSIG tablets are available in the following configurations.
| Strength | NDC Number | Description | Presentation |
|---|---|---|---|
| 10 mg | 63020-536-30 | oval, white to off-white, biconvex film-coated tablets with debossed "NZ" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
| 15 mg | 63020-535-30 | round, white, biconvex film-coated tablets with debossed "A5" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
| 63020-535-60 | 60 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. | ||
| 30 mg | 63020-533-30 | round, white, biconvex film-coated tablets with debossed "C7" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
| 45 mg | 63020-534-30 | round, white, biconvex film-coated tablets with debossed "AP4" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
2.2 Dosage Modifications for Adverse Reactions
Recommended dosage modifications of ICLUSIG for adverse reactions are provided in Table 1 and recommended dose reductions of ICLUSIG for adverse reactions are presented in Table 2.
| Adverse Reaction | Severity | ICLUSIG Dosage Modifications |
|---|---|---|
| Based on CTCAE v5.0: Grade 1 mild, Grade 2 moderate, Grade 3 severe, Grade 4 life-threatening ULN = Upper Limit of Normal for the lab; AOE = Arterial Occlusive Event; VTE = Venous Thromboembolic Event; ANC = absolute neutrophil count |
||
| AOE: cardiovascular or cerebrovascular [see Warnings and Precautions (5.1)] |
Grade 1 | Interrupt ICLUSIG until resolved, then resume at same dose. |
| Grade 2 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
|
| Grade 3 or 4 | Discontinue ICLUSIG. | |
| AOE: peripheral vascular and other or VTE [see Warnings and Precautions (5.1, 5.2)] |
Grade 1 | Interrupt ICLUSIG until resolved, then resume at same dose. |
| Grade 2 | Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. |
|
| Grade 3 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
|
| Grade 4 | Discontinue ICLUSIG. | |
| Heart Failure [see Warnings and Precautions (5.3)] |
Grade 2 or 3 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
| Grade 4 | Discontinue ICLUSIG. | |
| Hepatotoxicity [see Warnings and Precautions (5.4)] |
AST or ALT greater than 3 times ULN | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. |
| AST or ALT at least 3 times ULN concurrent with bilirubin greater than 2 times ULN and alkaline phosphatase less than 2 times ULN | Discontinue ICLUSIG. | |
| Pancreatitis and Elevated Lipase [see Warnings and Precautions (5.6)] |
Serum lipase greater than 1 to 1.5 times ULN | Consider interrupting ICLUSIG until resolution, then resume at same dose. |
| Serum lipase greater than 1.5 to 2 times ULN, 2 to 5 times ULN and asymptomatic, or asymptomatic radiologic pancreatitis | Interrupt ICLUSIG until Grade 0 or 1 (less than 1.5 times ULN), then resume at next lower dose. | |
| Serum lipase greater than 2 to 5 times ULN and symptomatic, symptomatic Grade 3 pancreatitis, or serum lipase greater than 5 times ULN and asymptomatic | Interrupt ICLUSIG until complete resolution of symptoms and after recovery of lipase elevation Grade 0 or 1, then resume at next lower dose. | |
| Symptomatic pancreatitis and serum lipase greater than 5 times ULN | Discontinue ICLUSIG. | |
| Myelosuppression [see Warnings and Precautions (5.13)] |
ANC less than 1 × 109/L or Platelets less than 50 × 109/L |
Interrupt ICLUSIG until ANC at least 1.5 × 109/L and platelet at least 75 × 109/L, then resume at same dose. If recurrence, interrupt ICLUSIG until resolution, then resume at next lower dose. |
| Other Non-hematologic Adverse Reactions [see Warnings and Precautions (5.5, 5.8, 5.10, 5.11, 5.12)] |
Grade 1 | Interrupt ICLUSIG until resolved, then resume at same dose. |
| Grade 2 | Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. |
|
| Grade 3 or 4 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
| Dose Reduction | Dosage for Patients with CP-CML | Dosage for Patients with AP-CML, BP-CML, and Ph+ ALL Monotherapy | Dosage for Patients with Newly Diagnosed Ph+ ALL |
|---|---|---|---|
| First | 30 mg orally once daily | 30 mg orally once daily | 15 mg orally once daily |
| Second | 15 mg orally once daily | 15 mg orally once daily | 10 mg orally once daily |
| Third | 10 mg orally once daily | Permanently discontinue ICLUSIG in patients unable to tolerate 15 mg orally once daily. | Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily. |
| Subsequent Reduction | Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily. |
2.4 Dosage for Patients With Hepatic Impairment
For patients with CP-CML, AP-CML, BP-CML, and Ph+ ALL receiving monotherapy, reduce the starting dose of ICLUSIG from 45 mg orally once daily to 30 mg orally once daily in patients with pre-existing hepatic impairment (Child-Pugh A, B, or C).
For patients with newly diagnosed Ph+ ALL, no dosage adjustment is recommended when administering ICLUSIG to patients with mild hepatic impairment (Child-Pugh A). Closely monitor patients with moderate or severe hepatic impairment (Child-Pugh B or C) and modify the ICLUSIG dosage in the event of adverse reactions [see Dosage and Administration (2.2), Use in Specific Populations (8.6)].
8.3 Females and Males of Reproductive Potential
ICLUSIG can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)].
Principal Display Panel 10 Mg Tablet Bottle Label
NDC 63020-536-30
ICLUSIG®
(ponatinib) tablets
10 mg
Each tablet contains 10 mg ponatinib
equivalent to 10.68 mg ponatinib HCl
Dispense Attached Medication Guide
30 tablets
Rx only
Takeda
Principal Display Panel 15 Mg Tablet Bottle Label
NDC 63020-535-30
ICLUSIG®
(ponatinib) tablets
15 mg
Each tablet contains 15 mg ponatinib
equivalent to 16.03 mg ponatinib HCl
Dispense Attached Medication Guide
30 tablets
Rx only
Takeda
Principal Display Panel 30 Mg Tablet Bottle Label
NDC 63020-533-30
ICLUSIG®
(ponatinib) tablets
30 mg
Each tablet contains 30 mg ponatinib
equivalent to 32.05 mg ponatinib HCl
Dispense Attached Medication Guide
Takeda
Principal Display Panel 45 Mg Tablet Bottle Label
NDC 63020-534-30
ICLUSIG®
(ponatinib) tablets
45 mg
Each tablet contains 45 mg ponatinib
equivalent to 48.08 mg ponatinib HCl
Dispense Attached Medication Guide
Takeda
5.15 Reversible Posterior Leukoencephalopathy Syndrome
Reversible posterior leukoencephalopathy syndrome (RPLS; also known as Posterior Reversible Encephalopathy Syndrome) has been reported in patients who received ICLUSIG. Patients can present with hypertension, seizure, headache, decreased alertness, altered mental functioning, vision loss, and other visual and neurological disturbances. Magnetic resonance imaging (MRI) is necessary to confirm the diagnosis. Interrupt ICLUSIG until resolution. The safety of resumption of ICLUSIG in patients upon resolution of RPLS is unknown.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2 year carcinogenicity study, male and female rats were administered daily oral doses of ponatinib of 0.05 mg/kg/day, 0.1 mg/kg/day, 0.2 mg/kg/day and 0.2 mg/kg/day, 0.4 mg/kg/day, and 0.8 mg/kg/day, respectively. Exposures in animals at the highest dose tested were 0.3- to 0.8-fold the human exposure (based on AUC) at doses of 15 mg and 45 mg daily. Ponatinib induced a statistically significant increase in malignant squamous neoplasms of the clitoral gland in females at 0.8 mg/kg/day.
Ponatinib was not mutagenic in a bacterial mutagenesis (Ames) assay, was not clastogenic in a chromosome aberration assay in human lymphocytes, nor was it clastogenic in an in vivo mouse micronucleus assay at oral doses up to 2000 mg/kg.
Ponatinib may impair female fertility. In a fertility study in male and female rats, female fertility parameters were reduced at 1.5 mg/kg/day with exposure equivalent to 0.43 times and 1.23 times of human daily steady-state AUC at the maximum recommended human dose of 45 mg/day (AUC = 1296 h∙ng/mL) and 15 mg/day (451.8 h∙ng/mL), respectively. Evidence of pre- and post-implantation loss of embryos was observed in female rats. Although there were no effects on male fertility parameters in the rat fertility study, repeat dose toxicology studies in monkeys showed degeneration of epithelium of the testes in monkeys at exposures approximately 3.3 times the plasma drug exposure (AUC) in patients receiving the maximum recommended human dose of 45 mg/day.
5.7 Increased Toxicity in Newly Diagnosed Chronic Phase Cml
In a prospective randomized clinical trial in the first line treatment of newly diagnosed patients with CP-CML, single agent ICLUSIG 45 mg once daily increased the risk of serious adverse reactions 2-fold compared to single agent imatinib 400 mg once daily. The median exposure to treatment was less than 6 months. The trial was halted for safety.
Arterial and venous thrombosis and occlusions occurred at least twice as frequently in the ICLUSIG arm compared to the imatinib arm. Compared to imatinib-treated patients, ICLUSIG-treated patients exhibited a greater incidence of myelosuppression, pancreatitis, hepatotoxicity, cardiac failure, hypertension, and skin and subcutaneous tissue disorders. ICLUSIG is not indicated and is not recommended for the treatment of patients with newly diagnosed CP-CML.
5.16 Impaired Wound Healing and Gastrointestinal Perforation
Impaired wound healing occurred in patients receiving ICLUSIG [see Adverse Reactions (6.2)]. Withhold ICLUSIG for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of ICLUSIG after resolution of wound healing complications has not been established.
Gastrointestinal perforation or fistula occurred in patients receiving ICLUSIG [see Adverse Reactions (6.2)]. Permanently discontinue in patients with gastrointestinal perforation.
2.3 Dosage Modification for Coadministration of Strong Cyp3a Inhibitors
Avoid coadministration of ICLUSIG with strong CYP3A inhibitors. If coadministration of a strong CYP3A inhibitor cannot be avoided, reduce the dosage of ICLUSIG as recommended in Table 3.
After the strong CYP3A inhibitor has been discontinued for 3 to 5 elimination half-lives, resume the ICLUSIG dosage that was tolerated prior to initiating the strong CYP3A inhibitor [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
| Current ICLUSIG Dosage | Recommended ICLUSIG Dosage with a Strong CYP3A Inhibitor |
|---|---|
| 45 mg orally once daily | 30 mg orally once daily |
| 30 mg orally once daily | 15 mg orally once daily |
| 15 mg orally once daily | 10 mg orally once daily |
| 10 mg orally once daily | Avoid coadministration of ICLUSIG with a strong CYP3A inhibitor |
Warning: Arterial Occlusive Events, Venous Thromboembolic Events, Heart Failure, and Hepatotoxicity
WARNING: ARTERIAL OCCLUSIVE EVENTS, VENOUS THROMBOEMBOLIC EVENTS, HEART FAILURE, and HEPATOTOXICITY
See full prescribing information for complete boxed warning.
- Arterial occlusive events (AOEs), including fatalities, have occurred in ICLUSIG-treated patients. AOEs included fatal myocardial infarction, stroke, stenosis of large arterial vessels of the brain, severe peripheral vascular disease, and the need for urgent revascularization procedures. Patients with and without cardiovascular risk factors, including patients age 50 years or younger, experienced these events. Monitor for evidence of AOEs. Interrupt or discontinue ICLUSIG based on severity. Consider benefit-risk to guide a decision to restart ICLUSIG. (2.2, 5.1)
- Venous thromboembolic events (VTEs) have occurred in ICLUSIG-treated patients. Monitor for evidence of VTEs. Interrupt or discontinue ICLUSIG based on severity. (2.2, 5.2)
- Heart failure, including fatalities, occurred in ICLUSIG-treated patients. Monitor for heart failure and manage patients as clinically indicated. Interrupt or discontinue ICLUSIG for new or worsening heart failure. (2.2, 5.3)
- Hepatotoxicity, liver failure and death have occurred in ICLUSIG-treated patients. Monitor liver function tests. Interrupt or discontinue ICLUSIG based on severity. (2.2, 5.4)
Structured Label Content
Section 42229-5 (42229-5)
Arterial Occlusive Events:
- Arterial occlusive events (AOEs), including fatalities, have occurred in ICLUSIG-treated patients. AOEs included fatal myocardial infarction, stroke, stenosis of large arterial vessels of the brain, severe peripheral vascular disease, and the need for urgent revascularization procedures. Patients with and without cardiovascular risk factors, including patients age 50 years or younger, experienced these events. Monitor for evidence of AOEs. Interrupt or discontinue ICLUSIG based on severity. Consider benefit-risk to guide a decision to restart ICLUSIG [see Dosage and Administration (2.2) , Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 10/2025 | ||
|
MEDICATION GUIDE ICLUSIG® (eye-CLUE-sig) (ponatinib) tablets |
|||
|
What is the most important information I should know about ICLUSIG? ICLUSIG can cause serious side effects, including: Blood clots or blockage in your blood vessels (arteries and veins). Blood clots or blockage in your blood vessels may lead to heart attack, stroke, or death. A blood clot or blockage in your blood vessels can prevent proper blood flow to your heart, brain, bowels (intestines), legs, eyes, and other parts of your body. You may need emergency surgery or treatment in a hospital. Get medical help right away if you get any of the following symptoms: |
|||
|
|
||
| Blood clots or blockage in your blood vessels can happen in people with or without risk factors for heart and blood vessel disease, including people 50 years of age or younger. The most common risk factors for these problems are a history of high blood pressure (hypertension), high cholesterol, and heart disease. Blood clots or blockages in your blood vessels happen more often in people as they get older, and in people with a history of decreased blood flow, high blood pressure, diabetes, or high cholesterol. Heart problems. ICLUSIG can cause heart problems, including heart failure which can be serious and may lead to death. Heart failure means your heart does not pump blood well enough. ICLUSIG can also cause irregular, slow, or fast heartbeats and heart attack. Your healthcare provider will check you for heart problems during your treatment with ICLUSIG. Get medical help right away if you get any of the following symptoms: shortness of breath, chest pain, fast or irregular heartbeats, dizziness, or feel faint. Liver problems. ICLUSIG can cause liver problems, including liver failure, which can be severe and may lead to death. Your healthcare provider will do blood tests before and during your treatment with ICLUSIG to check for liver problems. Get medical help right away if you get any of these symptoms of liver problems during treatment:
|
|||
|
What is ICLUSIG?
ICLUSIG is a prescription medicine used to treat adults who have:
It is not known if ICLUSIG is safe and effective in children. |
|||
Before you take ICLUSIG, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
|
|
||
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I take ICLUSIG?
|
|||
|
What are the possible side effects of ICLUSIG? ICLUSIG may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
|
|
| The most common side effects of ICLUSIG when given with chemotherapy include: | |||
|
|
|
|
| Your healthcare provider may change your dose, temporarily stop, or permanently stop treatment with ICLUSIG if you have certain side effects. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of ICLUSIG. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store ICLUSIG?
Store ICLUSIG at room temperature between 68°F to 77°F (20°C to 25°C). Keep ICLUSIG and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of ICLUSIG
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ICLUSIG for a condition for which it was not prescribed. Do not give ICLUSIG to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about ICLUSIG that is written for health professionals. |
|||
|
What are the ingredients in ICLUSIG? TAKEDA and are registered trademarks of Takeda Pharmaceutical Company Limited. ©2025 Takeda Pharmaceuticals U.S.A., Inc. All rights reserved. For more information, go to www.iclusig.com or call 1-844-817-6468. ICL348 R12 |
Section 43683-2 (43683-2)
| Warnings and Precautions (5) | 10/2025 |
Section 44425-7 (44425-7)
Store ICLUSIG tablets at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Overdoses with ICLUSIG were reported in clinical trials. One patient was estimated to have been administered 540 mg via nasogastric tube. Two hours after the overdosage, the patient had an uncorrected QT interval of 520 ms. Subsequent ECGs showed normal sinus rhythm with uncorrected QT intervals of 480 ms and 400 ms. The patient died 9 days after the overdosage from pneumonia and sepsis. Another patient self-administered 165 mg on Cycle 1 Day 2. The patient experienced fatigue and non-cardiac chest pain on Day 3. Multiple doses of 90 mg per day for 12 days in a patient resulted in pneumonia, systemic inflammatory response, atrial fibrillation, and a moderate pericardial effusion.
In the event of an overdosage, stop ICLUSIG, observe the patient and provide supportive treatment as appropriate.
11 Description (11 DESCRIPTION)
Ponatinib is a kinase inhibitor. The chemical name for ponatinib hydrochloride is 3-(imidazo[1,2-b]pyridazin-3-ylethynyl)-4-methyl-N-{4-[(4-methylpiperazin-1-yl)methyl]-3-(trifluoromethyl)phenyl}benzamide hydrochloride. The molecular formula is C29H28ClF3N6O which corresponds to a formula weight of 569.02 g/mol. Its structure is shown below:
Ponatinib HCl is an off-white to yellow powder with pKa of 2.77 and 7.8. The solubility of ponatinib in pH 1.7, 2.7, and 7.5 buffers is 7790 mcg/mL, 3.44 mcg/mL, and 0.16 mcg/mL, respectively, indicating a decrease in solubility with increasing pH. Each tablet for oral administration contains 10 mg, 15 mg, 30 mg or 45 mg of ponatinib equivalent to 10.68 mg, 16.03 mg, 32.05 mg, and 48.08 mg of ponatinib hydrochloride with the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (type B), colloidal silicon dioxide, magnesium stearate and a tablet coating. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol, and titanium dioxide.
5.8 Neuropathy
In PhALLCON, peripheral neuropathy occurred in 68% of 163 patients; 3.1% experienced Grade 3 or 4 peripheral neuropathy. The most frequent peripheral neuropathies were neuropathy peripheral (33%), paresthesia (22%), and peripheral sensory neuropathy (12%). The median time to onset of peripheral neuropathy was 1.1 month (range: 1 day to 17.2 months). Cranial neuropathy was reported in 0.6% of 163 patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, neuropathy occurred in 13% of patients. Peripheral neuropathy occurred in 11% of patients. The most frequently reported peripheral neuropathies were muscular weakness (3.2%), paresthesia (3.2%), hypoesthesia (2.1%) and neuropathy peripheral (2.1%). Cranial neuropathy developed in 2 patients. The median time to onset of peripheral neuropathy and cranial neuropathy was 1.1 years (range: 1 month to 4.1 years) and 3 years (range: 10.3 months to 5.2 years), respectively.
In PACE, neuropathy occurred in 22% of patients; 2.4% experienced Grade 3 or 4 neuropathy. Peripheral neuropathy occurred in 20% of 449 patients; 1.8% experienced Grade 3 or 4 peripheral neuropathy. The most frequent peripheral neuropathies were paresthesia (5%), neuropathy peripheral (4.5%), and hypoesthesia (3.6%). Cranial neuropathy developed in 3% of patients; 0.7% were Grade 3 or 4. The median time to onset of peripheral neuropathy and cranial neuropathy was 5.3 months (range: 1 day to 4.6 years) and 1.2 years (range: 18 days to 4 years), respectively.
Monitor patients for symptoms of neuropathy, such as hypoesthesia, hyperesthesia, paresthesia, discomfort, a burning sensation, neuropathic pain or weakness. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
5.10 Hemorrhage
Fatal and serious hemorrhage events have occurred in patients who received ICLUSIG.
In PhALLCON, hemorrhage occurred in 31% of 163 patients; 2.5% experienced a serious hemorrhage. Intracranial hemorrhage was the most frequently reported serious hemorrhage, occurring in 1.2% of patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, hemorrhage occurred in 13% of patients; 1 patient experienced a serious subdural hematoma.
In PACE, hemorrhage occurred in 28% of 449 patients; 6% experienced a serious hemorrhage and 1.3% experienced a fatal hemorrhage. The incidence of serious bleeding events was higher in patients with AP-CML, BP-CML, and Ph+ ALL. Gastrointestinal hemorrhage and subdural hematoma were the most frequently reported serious hemorrhages, each occurring in 0.9% of patients. Most hemorrhages occurred in patients with Grade 4 thrombocytopenia [see Warnings and Precautions (5.13)].
Monitor for hemorrhage and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
5.5 Hypertension
Serious or severe hypertension, including hypertensive crisis, has occurred in patients who received ICLUSIG.
In PhALLCON, hypertension occurred in 34% of 163 patients; 14% experienced serious or severe hypertension. Based on vital signs data, Grade 1 blood pressure elevation occurred in 15 out of 60 (25%) patients with normal initial blood pressure, Grade 2 occurred in 67 out of 134 (50%) patients with initial blood pressure of less than Grade 2, and Grade 3 occurred in 63 out of 160 (39%) patients with an initial blood pressure of less than Grade 3.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, hypertension events were reported in 37% of patients; 14% experienced serious or severe hypertension. Based on vital signs data, Grade 1 blood pressure elevation occurred in 8 out of 18 (44%) patients with normal initial blood pressure, Grade 2 occurred in 30 out of 81 (37%) patients with initial blood pressure of less than Grade 2, and Grade 3 occurred in 20 out of 92 (22%) patients with initial blood pressure of less than Grade 3. Three patients (3.2%) experienced hypertensive crisis.
In PACE, hypertension events were reported in 32% of 449 patients; 13% experienced serious or severe hypertension. Any post-baseline elevation of systolic or diastolic BP of Grade 2 or higher in patients with normal baseline blood pressure occurred in 44% of 449 patients. Grade 1 BP elevation occurred in 26%, Grade 2 in 45%, and Grade 3 in 26%. Two patients (<1%) experienced Grade 4 hypertension (hypertensive crisis).
Patients may require urgent clinical intervention for hypertension associated with confusion, headache, chest pain, or shortness of breath [see Adverse Reactions (6.1)]. Monitor blood pressure at baseline and as clinically indicated and manage hypertension as clinically indicated. Interrupt, dose reduce, or stop ICLUSIG if hypertension is not medically controlled [see Dosage and Administration (2.2)]. For significant worsening, labile or treatment-resistant hypertension, interrupt ICLUSIG and consider evaluating for renal artery stenosis.
5.6 Pancreatitis
Serious or severe pancreatitis has occurred in patients who received ICLUSIG.
In PhALLCON, pancreatitis occurred in 34% of 163 patients; 15% experienced serious or severe (Grade 3 or 4) pancreatitis. The median time to onset of pancreatitis was 8 days (range: 1 day to 2 years). In 7 patients with clinical pancreatitis that led to dose modification, pancreatitis resolved within 3 weeks. Laboratory abnormalities of amylase elevations occurred in 25% of patients, while lipase elevations occurred in 60% of patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, pancreatitis occurred in 29% of patients; 16% experienced serious or severe (Grade 3 or 4) pancreatitis. Pancreatitis resulted in discontinuation in 1.1% of patients and interruption and/or dose reduction in 23% of patients. The median time to onset of pancreatitis was 1 month (range: 3 days to 4.1 years). In two patients with clinical pancreatitis that led to dose modification or treatment discontinuation, pancreatitis resolved within 2 weeks. Laboratory abnormalities of amylase elevation occurred in 15% of patients, while lipase elevation occurred in 40% of patients.
In PACE, pancreatitis occurred in 26% of 449 patients; 17% experienced serious or severe (Grade 3 or 4) pancreatitis. Pancreatitis resulted in discontinuation in 0.4% of patients and interruption and/or dose reduction in 17% of patients. The median time to onset of pancreatitis was 29 days (range: 1 day to 4 years). Nineteen of the 28 cases of clinical pancreatitis that led to dose modification or treatment discontinuation resolved within 2 weeks. Laboratory abnormalities of amylase elevations occurred in 18% of patients, while lipase elevations occurred in 39% of patients.
Monitor serum lipase every 2 weeks for the first 2 months and then monthly thereafter or as clinically indicated. Consider additional serum lipase monitoring in patients with a history of pancreatitis or alcohol abuse. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on severity [see Dosage and Administration (2.2)]. Evaluate for pancreatitis when lipase elevation is accompanied by abdominal symptoms.
5.3 Heart Failure
Fatal, serious or severe heart failure events have occurred in patients who received ICLUSIG.
In PhALLCON, heart failure occurred in 6% of 163 patients; 1.2% experienced serious or severe (Grade 3 or 4) heart failure. The most frequently reported heart failure event (>1 patient) was increased brain natriuretic peptide (BNP) (2.5%).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, heart failure occurred in 20% of patients; 2.1% experienced serious or severe (Grade 3 or 4) heart failure. The most frequently reported heart failure events (>1 patient each) were left ventricular hypertrophy (5%), left ventricular dysfunction (5%), BNP increased (5%), cardiac failure (3.2%), left atrial dilatation (2.1%) and ejection fraction decreased (2.1%).
Fatal or serious heart failure occurred in PACE. Heart failure occurred in 9% of 449 patients; 7% experienced serious or severe (Grade 3 or higher) heart failure. The most frequently reported heart failure events (≥2%) were congestive cardiac failure (3.1%), decreased ejection fraction (2.9%), and cardiac failure (2%).
Monitor patients for signs or symptoms consistent with heart failure and manage heart failure as clinically indicated. Interrupt, then resume at reduced dose or discontinue ICLUSIG for new or worsening heart failure [see Dosage and Administration (2.2)].
8.4 Pediatric Use
Safety and effectiveness of ICLUSIG have not been established in pediatric patients.
8.5 Geriatric Use
Of the 163 patients with Ph+ALL who received ICLUSIG in PhALLCON, 21% were 65 years and older and 7% were 75 years and older. Overall, no differences in efficacy of ICLUSIG were observed between patients 65 years of age or older compared to younger patients. AOEs occurred in 21% (7/34) of patients 65 years and older and 2.3% (3/129) of patients less than 65 years of age.
Of the 94 patients with CP-CML who received ICLUSIG at a starting dose of 45 mg in OPTIC, 17% were 65 years and older and 2.1% were 75 years and older. Patients aged 65 years and older had a lower ≤1% BCR::ABL1IS rate at 12 months (27%) than patients less than 65 years of age (47%). By 60 months, patients aged 65 years and older had a ≤1% BCR::ABL1IS rate of 40% and patients less than 65 years of age had a rate of 64%. AOEs occurred in 38% (6/16) of patients 65 years and older and 14% (11/78) of patients less than 65 years of age [see Warnings and Precautions (5.1)].
Of the 449 patients who received ICLUSIG in PACE, 35% were 65 years and older and 8% were 75 years and older. In patients with CP-CML, patients aged 65 years and older had a lower major cytogenetic response rate (40%) as compared with patients less than 65 years of age (65%). In patients with AP-CML, BP-CML, and Ph+ ALL, patients aged 65 years and older had a similar hematologic response rate (45%) as compared with patients less than 65 years of age (44%). AOEs occurred in 35% (54/155) of patients 65 years and older and in 21% (61/294) of patients less than 65 years of age [see Warnings and Precautions (5.1)].
Patients aged 65 years or older are more likely to experience adverse reactions including vascular occlusion, decreased platelet count, peripheral edema, increased lipase, dyspnea, asthenia, muscle spasms, and decreased appetite. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
5.4 Hepatotoxicity
ICLUSIG can cause hepatotoxicity, including liver failure and death. Fulminant hepatic failure leading to death occurred in 3 patients, with hepatic failure occurring within 1 week of starting ICLUSIG in one of these patients. These fatal cases occurred in patients with BP-CML or Ph+ ALL treated with monotherapy.
In PhALLCON, hepatotoxicity occurred in 66% of 163 patients; 30% experienced Grade 3 or 4 hepatotoxicity. The median time to onset of hepatotoxicity was 15 days (range: 1 day to 10 months). The most frequent hepatotoxic events were elevated alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), bilirubin and alkaline phosphatase, decreased albumin and decreased blood fibrinogen. In 6% of the 73 patients who reported ALT or AST elevation, the elevations were not resolved by the date of the last follow-up.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, hepatotoxicity occurred in 34% of patients; 7% experienced Grade 3 or 4 hepatotoxicity. The median time to onset of hepatotoxicity was 4.1 months, with a range of 1 day to 4.8 years. The most frequent hepatotoxic events were elevations of ALT, AST, alkaline phosphatase, and GGT. In one of the 26 patients who reported ALT or AST elevation, the event was not resolved by the date of last follow-up.
In PACE, hepatotoxicity occurred in 32% of 449 patients; 13% experienced Grade 3 or 4 hepatotoxicity. The median time to onset of hepatotoxicity was 3.1 months, with a range of 1 day to 4.9 years. The most frequent hepatotoxic events were elevations of ALT, AST, GGT, bilirubin, and alkaline phosphatase. In 9% of the 88 patients who reported ALT or AST elevation, the event was not resolved by the date of last follow-up.
Monitor liver function tests at baseline, then at least monthly or as clinically indicated. Interrupt, then resume at reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
5.9 Ocular Toxicity
Serious ocular toxicities leading to blindness or blurred vision have occurred in ICLUSIG-treated patients.
In PhALLCON, ocular toxicities occurred in 33% of 163 patients; 1.8% experienced a serious or severe ocular toxicity. The most frequent ocular toxicities were blurred vision and dry eye. Retinal toxicities occurred in 4.3% of patients; 0.6% experienced a Grade 3 retinal vein occlusion. The most frequent retinal toxicity event (>1 patient) was retinal hemorrhage (1.8%).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, ocular toxicities occurred in 15% of patients; 1.1% experienced a serious or severe ocular toxicity. The most frequent ocular toxicities were dry eye, blurred vision and eye pain. Retinal toxicities occurred in 4.3% of patients, including age-related macular degeneration, arteriosclerotic retinopathy, retinal vascular disorder and retinal vein occlusion (1.1% each).
In PACE, ocular toxicities occurred in 30% of 449 patients; 3.6% experienced a serious or severe ocular toxicity. The most frequent ocular toxicities were dry eye, blurred vision, and eye pain. Retinal toxicities occurred in 3.6% of patients. The most frequent retinal toxicities were macular edema, retinal vein occlusion, retinal hemorrhage, and vitreous floaters (0.7% each).
Conduct comprehensive eye exams at baseline and periodically during treatment.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Arterial Occlusive Events [see Warnings and Precautions (5.1)]
- Venous Thromboembolic Events [see Warnings and Precautions (5.2)]
- Heart Failure [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warnings and Precautions (5.4)]
- Hypertension [see Warnings and Precautions (5.5)]
- Pancreatitis [see Warnings and Precautions (5.6)]
- Neuropathy [see Warnings and Precautions (5.8)]
- Ocular Toxicity [see Warnings and Precautions (5.9)]
- Hemorrhage [see Warnings and Precautions (5.10)]
- Fluid Retention [see Warnings and Precautions (5.11)]
- Cardiac Arrhythmias [see Warnings and Precautions (5.12)]
- Myelosuppression [see Warnings and Precautions (5.13)]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.14)]
- Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.15)]
- Impaired Wound Healing and Gastrointestinal Perforation [see Warnings and Precautions (5.16)]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.11 Fluid Retention
Fatal and serious fluid retention events have occurred in patients who received ICLUSIG.
In PhALLCON, fluid retention occurred in 24% of 163 patients; 1.2% experienced serious fluid retention, including pericardial effusion (1.2%). The most frequent occurrences of fluid retention were peripheral edema (11%) and pleural effusion (6%).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, fluid retention occurred in 6% of patients. The most frequent fluid retention events were peripheral edema (3.2%), hydrothorax (2.1%) and pleural effusion (2.1%).
In PACE, fluid retention events occurred in 33% of 449 patients; 4.5% experienced serious fluid retention. One instance of brain edema was fatal. Serious fluid retention included pleural effusion (1.6%), pericardial effusion (1.6%), and angioedema (0.4%). The most frequent fluid retention events were peripheral edema (17%), pleural effusion (9%), pericardial effusion (4.2%) and peripheral swelling (3.8%).
Monitor for fluid retention and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
12.2 Pharmacodynamics
In PACE, the dose intensity-safety relationship indicated that there are significant increases in Grade ≥3 adverse reactions (hypertension, thrombocytopenia, pancreatitis, neutropenia, rash, ALT increase, AST increase, lipase increase, myelosuppression) over the dose range of 15 mg to 45 mg. In addition to dose, increased age and history of ischemia, hypertension, diabetes, or hypercholesterolemia were also contributory factors to a higher incidence of AOEs.
In OPTIC, an exposure-response relationship between ponatinib exposure and molecular response rate at 12 months was observed. A relationship between higher ponatinib exposures and higher incidence of adverse reactions, including thrombocytopenia (Grade ≥3) and AOEs, was observed.
In vitro, there was no significant inhibition of platelet aggregation with ponatinib at concentrations seen clinically and up to 0.7 mcg/mL (1.23 μM).
12.3 Pharmacokinetics
Ponatinib administered to patients with cancer exhibited approximately dose proportional increases in both steady-state Cmax and AUC over the dose range of 2 mg to 60 mg (0.04 to 1.33 times the approved maximum recommended starting dose). The mean (CV%) Cmax and AUC(0-24) of ICLUSIG 45 mg orally once daily at presumed steady-state in patients with advanced hematologic malignancies were 73 ng/mL (74%) and 1253 ng∙hr/mL (73%), respectively. The mean (CV%) Cmax and AUC(0-24) of ICLUSIG 30 mg orally once daily at presumed steady-state in patients with advanced hematologic malignancies were 65 ng/mL (28%) and 1080 ng∙hr/mL (29%), respectively. Exposure increased by approximately 90% (median) [range: 20% to 440%] between the first dose and presumed steady-state.
5.13 Myelosuppression
In PhALLCON, neutropenia occurred in 66% (Grade 3 or 4 occurred in 63%), thrombocytopenia occurred in 65% (Grade 3 or 4 occurred in 62%) and anemia occurred in 53% (Grade 3 or 4 occurred in 38%) of 163 patients. The median time to onset of Grade 3 or 4 myelosuppression was 27 days (range: 1 day to 9.2 months).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, thrombocytopenia occurred in 66% (Grade 3 or 4 occurred in 31%), neutropenia occurred in 56% (Grade 3 or 4 occurred in 22%), and anemia occurred in 38% of patients (Grade 3 or 4 occurred in 14%). The median time to onset of Grade 3 or 4 myelosuppression was 1.3 months (range: 1 day to 1.2 years).
In PACE, neutropenia occurred in 56% (Grade 3 or 4 occurred in 34%), thrombocytopenia occurred in 63% (Grade 3 or 4 occurred in 40%), and anemia occurred in 52% of patients (Grade 3 or 4 occurred in 20%). The incidence of myelosuppression was greater in patients with AP-CML, BP-CML, and Ph+ ALL than in patients with CP-CML. Severe myelosuppression (Grade 3 or 4) was observed early in treatment, with a median onset time of 29 days (range: 1 day to 4.1 years).
Obtain complete blood counts every 2 weeks for the first 3 months and then monthly or as clinically indicated. If ANC less than 1 × 109/L or platelets less than 50 × 109/L, interrupt ICLUSIG until ANC at least 1.5 × 109/L and platelets at least 75 × 109/L, then resume at same or reduced dose [see Dosage and Administration (2.2)].
8.6 Hepatic Impairment
Patients with hepatic impairment are more likely to experience adverse reactions compared to patients with normal hepatic function. For patients with CP-CML, AP-CML, BP-CML, and Ph+ ALL receiving monotherapy, reduce the starting dose of ICLUSIG for patients with pre-existing hepatic impairment (Child-Pugh A, B, or C). For patients with newly diagnosed Ph+ ALL, dosage adjustment is not recommended when administering ICLUSIG to patients with mild hepatic impairment (Child-Pugh A). Clinical data in patients with newly diagnosed Ph+ ALL with pre-existing moderate or severe hepatic impairment (Child-Pugh B or C) is not available and patients should be closely monitored for potential increased incidence of adverse reactions. Modify the ICLUSIG dosage in the event of adverse reactions [see Dosage and Administration (2.2, 2.4), Clinical Pharmacology (12.3)]. The safety of multiple doses, or doses higher than 30 mg, has not been studied in patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ICLUSIG® is indicated for the treatment of adult patients with:
Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia (Ph+ ALL)
- Newly diagnosed Ph+ ALL in combination with chemotherapy.
This indication is approved under accelerated approval based on minimal residual disease (MRD)-negative complete remission (CR) at the end of induction [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification of clinical benefit in a confirmatory trial(s). - As monotherapy in Ph+ ALL for whom no other kinase inhibitors are indicated or T315I-positive Ph+ ALL.
Chronic Myeloid Leukemia (CML)
- Chronic phase (CP) CML with resistance or intolerance to at least two prior kinase inhibitors.
- Accelerated phase (AP) or blast phase (BP) CML for whom no other kinase inhibitors are indicated.
- T315I-positive CML (chronic phase, accelerated phase, or blast phase).
12.1 Mechanism of Action
Ponatinib is a kinase inhibitor. Ponatinib inhibited the in vitro tyrosine kinase activity of ABL and T315I mutant ABL with IC50 concentrations of 0.4 nM and 2.0 nM, respectively. Ponatinib inhibited the in vitro activity of additional kinases with IC50 concentrations between 0.1 nM and 20 nM, including members of the VEGFR, PDGFR, FGFR, EPH receptors and SRC families of kinases, and KIT, RET, TIE2, and FLT3. Ponatinib inhibited the in vitro viability of cells expressing native or mutant BCR::ABL, including T315I. In mice, treatment with ponatinib reduced the size of tumors expressing native or T315I mutant BCR::ABL when compared to controls.
5.12 Cardiac Arrhythmias
In PhALLCON, cardiac arrhythmia events occurred in 22% of 163 patients; 2.5% experienced Grade 3 or 4 cardiac arrhythmias, including tachycardia, syncope, atrial fibrillation and supraventricular tachycardia (0.6%, each).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, cardiac arrhythmias occurred in 27% of patients; 5% experienced Grade 3 or 4 cardiac arrhythmias including atrial fibrillation, cardio-respiratory arrest, supraventricular extrasystoles, supraventricular tachycardia and syncope (1.1%, each).
In PACE, cardiac arrhythmias occurred in 20% of 449 patients; 7% experienced Grade 3 or 4 cardiac arrhythmias. Ventricular arrhythmias occurred in 3.4% of the 89 patients who reported an arrhythmia, with one event being Grade 3 or 4. Symptomatic bradyarrhythmias that led to pacemaker implantation occurred in 1% of patients. Atrial fibrillation was the most frequent cardiac arrhythmia (8%), with 3.3% being Grade 3 or 4. Other Grade 3 or 4 arrhythmia events included syncope (2%), tachycardia and bradycardia (0.4% each), and QT interval prolongation, atrial flutter, sinus bradycardia, supraventricular tachycardia, ventricular tachycardia, atrial tachycardia, atrioventricular block complete, cardio-respiratory arrest, loss of consciousness, and sinus node dysfunction (0.2% each). For 31 patients, the arrythmia led to hospitalization.
Monitor for signs and symptoms suggestive of slow heart rate (fainting, dizziness) or rapid heart rate (chest pain, palpitations or dizziness) and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity.
5.14 Tumor Lysis Syndrome
In PhALLCON, serious tumor lysis syndrome (TLS) developed in 0.6% of 163 patients. Hyperuricemia occurred in 10% of patients.
In OPTIC, of the 94 patients who received a starting dose of 45 mg, serious TLS developed in 1.1% of patients. Hyperuricemia occurred in 2.1% of patients.
In PACE, serious TLS developed in 0.4% of 449 patients. One case occurred in a patient with advanced AP-CML and 1 case occurred in a patient with BP-CML. Hyperuricemia occurred in 7% of patients.
Ensure adequate hydration and treat high uric acid levels prior to initiating ICLUSIG.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypertension: Monitor blood pressure and manage hypertension as clinically indicated. Interrupt, dose reduce or stop ICLUSIG if hypertension is not medically controlled. (2.2, 5.5)
- Pancreatitis: Monitor serum lipase. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on severity. Evaluate for pancreatitis when lipase elevation is accompanied by abdominal symptoms. (2.2, 5.6)
- Neuropathy: Monitor for symptoms of peripheral and cranial neuropathy. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (2.2, 5.8)
- Ocular Toxicity: Conduct comprehensive eye exams at baseline and periodically during treatment. (5.9)
- Hemorrhage: Monitor for hemorrhage and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (2.2, 5.10)
- Fluid Retention: Monitor for fluid retention and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (2.2, 5.11)
- Cardiac Arrhythmias: Monitor for signs or symptoms of arrhythmias and manage patients as clinically indicated. Interrupt, then resume at the same or reduced dose or discontinue ICLUSIG based on recurrence/severity. (5.12)
- Myelosuppression: Obtain complete blood counts every 2 weeks for the first 3 months and then monthly or as clinically indicated. If ANC less than 1 × 109/L or platelets less than 50 × 109/L, interrupt ICLUSIG until ANC at least 1.5 × 109/L and platelets at least 75 × 109/L, then resume at same or reduced dose. (2.2, 5.13)
- Tumor Lysis Syndrome: Ensure adequate hydration and correct elevated uric acid levels prior to initiating ICLUSIG. (5.14)
- Reversible Posterior Leukoencephalopathy Syndrome (RPLS): Interrupt ICLUSIG until resolution. The safety of resumption of ICLUSIG in patients upon resolution of RPLS is unknown. (5.15)
- Impaired Wound Healing and Gastrointestinal Perforation: Withhold ICLUSIG for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of ICLUSIG after resolution of wound healing complications has not been established. (5.16)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.17, 8.1, 8.3)
5.17 Embryo Fetal Toxicity (5.17 Embryo-Fetal Toxicity)
Based on its mechanism of action and findings from animal studies, ICLUSIG can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, oral administration of ponatinib to pregnant rats during organogenesis caused adverse developmental effects at exposures lower than human exposures at the maximum recommended human dose of 45 mg/day. Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with ICLUSIG and for 3 weeks after the last dose [see Use in Specific Populations (8.1, 8.3)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended Dosage in Newly Diagnosed Ph+ ALL: Starting dose is 30 mg orally once daily in combination with chemotherapy, with a reduction to 15 mg once daily upon achievement of MRD-negative (≤0.01% BCR::ABL1/ABL1) CR at the end of induction. (2.1)
- Recommended Dosage in Monotherapy for Ph+ ALL for Whom No Other Kinase Inhibitors are Indicated or T315I-positive Ph+ ALL: Starting dose is 45 mg orally once daily. (2.1)
- Recommended Dosage in CP-CML: Starting dose is 45 mg orally once daily with a reduction to 15 mg once daily upon achievement of ≤1% BCR::ABL1IS. (2.1)
- Recommended Dosage in AP-CML and BP-CML: Starting dose is 45 mg orally once daily. (2.1)
- Hepatic Impairment: See the Full Prescribing Information for dosage modifications for hepatic impairment. (2.4)
- ICLUSIG may be taken with or without food. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets, film-coated:
- 10 mg of ponatinib: Oval, white to off-white, biconvex, debossed "NZ" on one side and plain on the other side
- 15 mg of ponatinib: Round, white, biconvex, debossed "A5" on one side and plain on the other side
- 30 mg of ponatinib: Round, white, biconvex, debossed "C7" on one side and plain on the other side
- 45 mg of ponatinib: Round, white, biconvex, debossed "AP4" on one side and plain on the other side
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ICLUSIG. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Blood and Lymphatic System Disorders: Thrombotic microangiopathy
Endocrine Disorders: Hyperthyroidism
Gastrointestinal Disorders: Gastrointestinal perforation, fistula
Metabolism and Nutrition Disorders: Dehydration
Nervous System Disorders: Reversible posterior leukoencephalopathy syndrome (RPLS)
Skin and Subcutaneous Tissue Disorders: Severe cutaneous reaction (e.g., Erythema multiforme, Stevens-Johnson syndrome), impaired wound healing, panniculitis (including erythema nodosum)
Vascular Disorders: Arterial (including aortic) aneurysms, dissections, and rupture
5.1 Arterial Occlusive Events
Arterial occlusive events (AOEs), including fatalities, occurred in patients who received ICLUSIG [see Adverse Reactions (6.1)].
In PhALLCON, 6% of 163 patients experienced AOEs, of which 3.1%, 1.8%, and 1.2% experienced cardiovascular, cerebrovascular, and peripheral vascular AOEs, respectively. The median time to onset of the first AOE was 11.3 months (range: 8 days to 2.8 years). Grade 3 or 4 AOEs occurred in 3.7% of patients; the most frequent Grade 3 or 4 AOEs were myocardial infarction (1.2%), peripheral arterial occlusive disease (1.2%), angina pectoris and cerebrovascular accident (0.6% each). Fatal AOE of sudden death occurred in 1 patient (0.6%). AOEs were more frequent with increasing age [see Use in Specific Populations (8.5)].
In PhALLCON, patients with uncontrolled hypertension, hypertriglyceridemia, or diabetes were excluded. Patients with clinically significant, uncontrolled, or active cardiovascular disease, including any history of myocardial infarction, peripheral vascular infarction, revascularization procedure, venous thromboembolism, clinically significant atrial/ventricular tachyarrhythmias, unstable angina, or congestive heart failure within the 6 months prior to the first dose of ICLUSIG, were also excluded.
In OPTIC, of the 94 patients who received a starting dose of 45 mg (45 mg → 15 mg), 18% experienced AOEs, of which 11%, 4.3%, and 3.2% experienced cardiovascular, cerebrovascular or peripheral vascular AOEs, respectively. The median time to onset of the first cardiovascular, cerebrovascular, or peripheral vascular event was 9.4 months (range: 12 days to 5.7 years), 11.7 months (range: 15 days to 1.6 years), and 6.3 months (range: 23 days to 3.6 years), respectively. Grade 3 or 4 AOEs occurred in 7% of patients; the most frequent Grade 3 or 4 AOEs were myocardial infarction, acute coronary syndrome, arterial thrombosis, ischemic stroke, ischemic cerebral infarction, subclavian artery stenosis and unstable angina (1.1% each). Fatal AOEs occurred in 4 patients (4.3%); including sudden death (2.1%), myocardial ischemia (1.1%) and myocardial infarction (1.1%). AOEs were more frequent with increasing age [see Use in Specific Populations (8.5)].
In OPTIC, patients with uncontrolled hypertension or diabetes and patients with clinically significant, uncontrolled, or active cardiovascular disease, including any history of myocardial infarction, peripheral vascular infarction, revascularization procedure, congestive heart failure, venous thromboembolism, or clinically significant atrial/ventricular arrhythmias, were excluded.
In PACE, 26% of 449 patients experienced AOEs, of which 15%, 7%, and 11% experienced cardiovascular, cerebrovascular, and peripheral vascular AOEs, respectively. Some patients experienced recurrent or multisite vascular occlusion. The median time to onset of the first cardiovascular, cerebrovascular, and peripheral vascular AOEs was 1 year (range: 1 day to 4.1 years), 1.4 years (range: 2 days to 4.5 years), and 2 years (range: 10 days to 4.9 years), respectively. Grade 3 or 4 AOEs occurred in 14% of patients; the most frequent Grade 3 or 4 AOEs were peripheral arterial occlusive disease (3.1%), myocardial infarction (2%), coronary artery disease (1.6%), and cerebral infarction (1.6%). Fatal AOEs occurred in 9 patients (2%); the most frequent fatal AOE was cardiac arrest (0.9%).
In PACE, fatal and life-threatening AOEs occurred within 2 weeks of starting treatment at 45 mg, and at dose levels as low as 15 mg per day. Patients with and without cardiovascular risk factors, including patients age 50 years or younger, experienced AOEs. AOEs were more frequent with increasing age [see Use in Specific Populations (8.5)] and in patients with history of ischemia, hypertension, diabetes, or hypercholesterolemia. The most common risk factors in patients with AOEs were history of hypertension (67%; 77/115), hypercholesterolemia (59%; 68/115), and non-ischemic cardiac disease (43%; 49/115).
In PACE, patients developed heart failure concurrent or subsequent to a myocardial ischemic event [see Warnings and Precautions (5.3)]. Patients required revascularization procedures (coronary, cerebrovascular, and peripheral arterial). ICLUSIG caused stenosis over multiple segments in major arterial vessels that supply the brain (e.g., carotid, vertebral, middle cerebral artery). Patients developed digital or distal extremity necrosis and required amputations. Renal artery stenosis associated with worsening, labile or treatment-resistant hypertension occurred in some ICLUSIG-treated patients [see Warnings and Precautions (5.5)].
In PACE, patients with uncontrolled hypertriglyceridemia and patients with clinically significant or active cardiovascular disease, including any history of clinically significant atrial/ventricular arrhythmias or history of myocardial infarction, unstable angina, or congestive heart failure within the 3 months prior to the first dose of ICLUSIG, were excluded [see Adverse Reactions (6.1)]. Consider whether the benefits of ICLUSIG are expected to exceed the risks.
Monitor for evidence of AOEs. Interrupt, then resume at the same or decreased dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)]. Consider benefit-risk to guide a decision to restart ICLUSIG.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most common adverse reactions identified in the Highlights of the Prescribing Information are based on two safety populations. The first is from a pooled safety population of 543 patients with CML or resistant or intolerant Ph+ ALL (OPTIC and PACE studies) who received ICLUSIG as a single agent at a starting dose of 45 mg orally once daily. In this pooled safety population, the most common (>20%) adverse reactions were rash and related conditions, arthralgia, abdominal pain, fatigue, headache, constipation, hypertension, dry skin, hepatotoxicity, fluid retention and edema, pyrexia, pancreatitis/lipase elevation, nausea, hemorrhage, anemia, AOEs and cardiac arrhythmias. The most common Grade 3 or 4 laboratory abnormalities (>20%) were platelet count decreased, neutrophil cell count decreased, and white blood cell decreased.
The second safety population is from 163 patients with newly diagnosed Ph+ ALL (PhALLCON study) who received ICLUSIG in combination with chemotherapy at a starting dose of 30 mg orally once daily. The most common adverse reactions (>20%) included hepatotoxicity, arthralgia, rash and related conditions, headache, pyrexia, abdominal pain, constipation, fatigue, nausea, oral mucositis, hypertension, pancreatitis/lipase elevation, neuropathy peripheral, hemorrhage, febrile neutropenia, fluid retention and edema, vomiting, paresthesia, and cardiac arrhythmias. The most common Grade 3 or 4 laboratory abnormalities (>20%) included decreased white blood cell count, decreased neutrophil cell count, decreased platelet count, decreased lymphocyte cell count, decreased hemoglobin, increased lipase, and increased ALT.
Newly Diagnosed Ph+ ALL
The safety of ICLUSIG was evaluated in PhALLCON, a randomized, active-controlled, multicenter trial conducted in patients with newly diagnosed Ph+ ALL [see Clinical Studies (14)]. Patients received ICLUSIG (n=163) or imatinib 600 mg (n=81) in combination with reduced-intensity chemotherapy followed by continued treatment with ICLUSIG or imatinib as a single agent (imatinib in combination with chemotherapy is not an approved regimen in adult patients). In the ICLUSIG arm, patients received a starting dosage of ICLUSIG 30 mg orally once daily in combination with chemotherapy, with a reduction to 15 mg orally once daily upon achievement of MRD-negative CR at the end of induction. The median duration of exposure was 9.0 months (range: <1 month to 4.2 years) in the ICLUSIG arm and 5.2 months (range: <1 month to 4.4 years) in the imatinib arm.
Patients with uncontrolled hypertension, hypertriglyceridemia, or diabetes and patients with clinically significant, uncontrolled, or active cardiovascular disease, including any history of myocardial infarction, peripheral vascular infarction, revascularization procedure, venous thromboembolism, clinically significant atrial/ventricular tachyarrhythmias, history of myocardial infarction, unstable angina, or congestive heart failure within the 6 months prior to the first dose of ICLUSIG, were excluded.
Serious adverse reactions occurred in 63% of patients receiving ICLUSIG in combination with chemotherapy. Serious adverse reactions in >2% of patients included febrile neutropenia (18%), pyrexia (6%), thrombocytopenia (4.3%), sepsis (3.7%), septic shock (3.7%), anemia (2.5%), hemorrhage (2.5%), neutropenia (2.5%), pancreatitis (2.5%), peripheral neuropathy (2.5%), pneumonia (2.5%) and acute kidney injury (2.5%). Fatal adverse reactions occurred in 6% of patients who received ICLUSIG in combination with chemotherapy, including sepsis (3.7%), sudden death, pneumonitis and respiratory failure (0.6%, each).
Permanent discontinuation of ICLUSIG due to adverse reactions occurred in 13% of patients. Adverse reactions resulting in permanent discontinuation of ICLUSIG in >2% of patients included arterial occlusive events and sepsis.
Dosage modifications (dose interruption or reduction) of ICLUSIG due to adverse reactions occurred in 71% of patients. Adverse reactions leading to dose interruption or reduction of ICLUSIG in >5% of patients included increased ALT, neutropenia, increased lipase, thrombocytopenia, increased AST, febrile neutropenia, and abdominal pain.
Table 4 summarizes the adverse reactions in patients receiving ICLUSIG or imatinib in combination with chemotherapy in PhALLCON.
| Adverse Reaction | ICLUSIG 30 mg → 15 mg with Chemotherapy (n = 163) |
Imatinib 600 mg with Chemotherapy (n = 81) |
||
|---|---|---|---|---|
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| Graded using CTCAE v5.0 | ||||
| Hepatobiliary Disorders | ||||
| Hepatotoxicity | 66 | 30 | 57 | 14 |
| Musculoskeletal and Connective Tissue Disorders | ||||
| Arthralgia Includes arthralgia, arthritis, back pain, flank pain, intervertebral disc degeneration, joint swelling, osteoarthritis, neck pain, pain, pain in extremity, pain of skin, sciatica, spinal pain, tendonitis, and tenosynovitis.
|
47 | 4.3 | 35 | 1.2 |
| Myalgia | 13 | 1.2 | 10 | 1.2 |
| Nervous System Disorders | ||||
| Headache | 45 | 1.8 | 43 | 1.2 |
| Neuropathy peripheral | 33 | 1.2 | 24 | 1.2 |
| Paresthesia | 22 | 0 | 10 | 0 |
| Peripheral sensory neuropathy | 12 | 0 | 12 | 0 |
| Skin and Subcutaneous Tissue Disorders | ||||
| Rash and related conditions | 47 | 1.2 | 33 | 1.2 |
| Gastrointestinal Disorders | ||||
| Abdominal pain Includes abdominal discomfort, abdominal distension, abdominal pain, abdominal pain lower, abdominal pain upper, chronic gastritis, colitis, enteritis, enterocolitis, gastric ulcer, gastritis, gastroenteritis, gastrointestinal pain, gastroesophageal reflux disease, and helicobacter gastritis.
|
43 | 4.9 | 28 | 0 |
| Constipation | 41 | 0.6 | 21 | 1.2 |
| Nausea | 37 | 3.1 | 52 | 7 |
| Oral mucositis | 35 | 4.9 | 30 | 10 |
| Pancreatitis/lipase elevation | 34 | 15 | 37 | 20 |
| Vomiting | 24 | 1.2 | 40 | 2.5 |
| Diarrhea | 20 | 0 | 35 | 2.5 |
| General Disorders | ||||
| Pyrexia | 44 | 4.3 | 26 | 2.5 |
| Fatigue or asthenia | 40 | 2.5 | 38 | 3.7 |
| Fluid retention and edema | 24 | 0.6 | 48 | 3.7 |
| Vascular Disorders | ||||
| Hypertension | 34 | 14 | 15 | 7 |
| Hemorrhage | 31 | 1.8 | 30 | 7 |
| Venous thromboembolic events | 12 | 3.1 | 10 | 2.5 |
| Blood and Lymphatic System Disorders | ||||
| Febrile neutropenia | 28 | 25 | 22 | 20 |
| Metabolism and Nutrition Disorders | ||||
| Impaired glucose tolerance | 20 | 4.9 | 20 | 9 |
| Hyperlipidemia | 16 | 1.2 | 15 | 1.2 |
| Decreased appetite | 10 | 0 | 19 | 3.7 |
| Cardiac Disorders | ||||
| Cardiac arrhythmias | 22 | 2.5 | 17 | 6 |
| Infections | ||||
| Sepsis Includes abdominal sepsis, bacteremia, bacterial sepsis, device-related sepsis, escherichia bacteremia, fungemia, klebsiella bacteremia, klebsiella sepsis, neutropenic sepsis, pseudomonal sepsis, sepsis, septic shock, staphylococcal bacteremia, staphylococcal sepsis, streptococcal bacteremia, and urosepsis.
|
17 | 12 | 15 | 11 |
| Pneumonia | 11 | 7 | 11 | 6 |
| Respiratory, Thoracic, and Mediastinal Disorders | ||||
| Cough | 17 | 0 | 6 | 0 |
| Dyspnea | 13 | 1.2 | 4.9 | 2.5 |
Clinically relevant adverse reactions in ≤10% of patients receiving ICLUSIG with chemotherapy: urinary tract infection (10%), arterial occlusive events (6%), cardiac failure (6%), and acute kidney injury (4.3%).
Table 5 summarizes the laboratory abnormalities in PhALLCON for patients who received ICLUSIG or imatinib in combination with chemotherapy.
| Laboratory Abnormality | ICLUSIG 30 mg → 15 mg with Chemotherapy (n = 163) |
Imatinib 600 mg with Chemotherapy (n = 81) |
||
|---|---|---|---|---|
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase Graded using CTCAE v5.0 |
||||
| Hematologic Laboratory Tests | ||||
| White blood cell decreased | 79 | 71 | 78 | 70 |
| Lymphocyte cell count decreased | 77 | 61 | 94 | 89 |
| Neutrophil cell count decreased | 66 | 63 | 57 | 53 |
| Platelet count decreased | 65 | 62 | 64 | 53 |
| Hemoglobin decreased | 53 | 38 | 59 | 49 |
| Liver Function Tests | ||||
| ALT increased | 69 | 21 | 62 | 7 |
| AST increased | 53 | 7 | 48 | 6 |
| Alkaline phosphatase increased | 44 | 1.2 | 24 | 0 |
| Total bilirubin increased | 25 | 0.6 | 24 | 0 |
| Direct bilirubin increased | 24 | 4.3 | 24 | 1.2 |
| Pancreatic Enzymes | ||||
| Lipase increased | 60 | 24 | 78 | 38 |
| Amylase increased | 25 | 6 | 35 | 7 |
| Chemistry | ||||
| Calcium decreased | 67 | 3.1 | 69 | 4.9 |
| Phosphate decreased | 58 | 16 | 85 | 36 |
| Potassium decreased | 44 | 10 | 74 | 25 |
| Albumin decreased | 42 | 1.8 | 56 | 0 |
| Glucose increased | 34 | 2.5 | 38 | 2.5 |
| Creatinine increased | 34 | 3.7 | 48 | 4.9 |
| Sodium decreased | 32 | 3.1 | 35 | 3.7 |
| Potassium increased | 31 | 3.7 | 12 | 0 |
| Magnesium decreased | 15 | 0.6 | 31 | 2.5 |
5.2 Venous Thromboembolic Events
Serious or severe VTEs have occurred in patients who received ICLUSIG.
In PhALLCON, VTEs occurred in 12% of 163 patients, including serious or severe (Grade 3 or 4) in 3.1%. VTEs included deep vein thrombosis (6%), superficial vein thrombosis (2.5%), embolism (1.8%), pulmonary embolism and thrombosis (1.2% each), and jugular vein thrombosis and retinal vein occlusion (0.6% each). The median time to onset of the first VTE event was 2.5 months (range: 6 days to 1.8 years).
In OPTIC, of the 94 patients who received a starting dose of 45 mg, 2 patients experienced a VTE (Grade 1 retinal vein occlusion and grade 2 phlebitis).
In PACE, VTEs occurred in 6% of 449 patients, including serious or severe (Grade 3 or 4) in 5.8%. VTEs included deep venous thrombosis (2.2%), pulmonary embolism (1.8%), superficial thrombophlebitis (0.7%), retinal vein occlusion (0.7%), and retinal vein thrombosis (0.4%) with vision loss. VTEs occurred in 10% of the 62 patients with BP-CML, 9% of the 32 patients with Ph+ ALL, 6% of the 270 patients with CP-CML, and 3.5% of the 85 patients with AP-CML.
Monitor for evidence of VTEs. Interrupt, then resume at the same or decreased dose or discontinue ICLUSIG based on recurrence/severity [see Dosage and Administration (2.2)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ICLUSIG tablets are available in the following configurations.
| Strength | NDC Number | Description | Presentation |
|---|---|---|---|
| 10 mg | 63020-536-30 | oval, white to off-white, biconvex film-coated tablets with debossed "NZ" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
| 15 mg | 63020-535-30 | round, white, biconvex film-coated tablets with debossed "A5" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
| 63020-535-60 | 60 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. | ||
| 30 mg | 63020-533-30 | round, white, biconvex film-coated tablets with debossed "C7" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
| 45 mg | 63020-534-30 | round, white, biconvex film-coated tablets with debossed "AP4" on one side and plain on the other side | 30 tablets in a wide-mouth white high density polyethylene (HDPE) bottle with a desiccant canister and induction sealed child resistant closure. |
2.2 Dosage Modifications for Adverse Reactions
Recommended dosage modifications of ICLUSIG for adverse reactions are provided in Table 1 and recommended dose reductions of ICLUSIG for adverse reactions are presented in Table 2.
| Adverse Reaction | Severity | ICLUSIG Dosage Modifications |
|---|---|---|
| Based on CTCAE v5.0: Grade 1 mild, Grade 2 moderate, Grade 3 severe, Grade 4 life-threatening ULN = Upper Limit of Normal for the lab; AOE = Arterial Occlusive Event; VTE = Venous Thromboembolic Event; ANC = absolute neutrophil count |
||
| AOE: cardiovascular or cerebrovascular [see Warnings and Precautions (5.1)] |
Grade 1 | Interrupt ICLUSIG until resolved, then resume at same dose. |
| Grade 2 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
|
| Grade 3 or 4 | Discontinue ICLUSIG. | |
| AOE: peripheral vascular and other or VTE [see Warnings and Precautions (5.1, 5.2)] |
Grade 1 | Interrupt ICLUSIG until resolved, then resume at same dose. |
| Grade 2 | Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. |
|
| Grade 3 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
|
| Grade 4 | Discontinue ICLUSIG. | |
| Heart Failure [see Warnings and Precautions (5.3)] |
Grade 2 or 3 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
| Grade 4 | Discontinue ICLUSIG. | |
| Hepatotoxicity [see Warnings and Precautions (5.4)] |
AST or ALT greater than 3 times ULN | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. |
| AST or ALT at least 3 times ULN concurrent with bilirubin greater than 2 times ULN and alkaline phosphatase less than 2 times ULN | Discontinue ICLUSIG. | |
| Pancreatitis and Elevated Lipase [see Warnings and Precautions (5.6)] |
Serum lipase greater than 1 to 1.5 times ULN | Consider interrupting ICLUSIG until resolution, then resume at same dose. |
| Serum lipase greater than 1.5 to 2 times ULN, 2 to 5 times ULN and asymptomatic, or asymptomatic radiologic pancreatitis | Interrupt ICLUSIG until Grade 0 or 1 (less than 1.5 times ULN), then resume at next lower dose. | |
| Serum lipase greater than 2 to 5 times ULN and symptomatic, symptomatic Grade 3 pancreatitis, or serum lipase greater than 5 times ULN and asymptomatic | Interrupt ICLUSIG until complete resolution of symptoms and after recovery of lipase elevation Grade 0 or 1, then resume at next lower dose. | |
| Symptomatic pancreatitis and serum lipase greater than 5 times ULN | Discontinue ICLUSIG. | |
| Myelosuppression [see Warnings and Precautions (5.13)] |
ANC less than 1 × 109/L or Platelets less than 50 × 109/L |
Interrupt ICLUSIG until ANC at least 1.5 × 109/L and platelet at least 75 × 109/L, then resume at same dose. If recurrence, interrupt ICLUSIG until resolution, then resume at next lower dose. |
| Other Non-hematologic Adverse Reactions [see Warnings and Precautions (5.5, 5.8, 5.10, 5.11, 5.12)] |
Grade 1 | Interrupt ICLUSIG until resolved, then resume at same dose. |
| Grade 2 | Interrupt ICLUSIG until Grade 0 or 1, then resume at same dose. If recurrence, interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. |
|
| Grade 3 or 4 | Interrupt ICLUSIG until Grade 0 or 1, then resume at next lower dose. Discontinue ICLUSIG if recurrence. |
| Dose Reduction | Dosage for Patients with CP-CML | Dosage for Patients with AP-CML, BP-CML, and Ph+ ALL Monotherapy | Dosage for Patients with Newly Diagnosed Ph+ ALL |
|---|---|---|---|
| First | 30 mg orally once daily | 30 mg orally once daily | 15 mg orally once daily |
| Second | 15 mg orally once daily | 15 mg orally once daily | 10 mg orally once daily |
| Third | 10 mg orally once daily | Permanently discontinue ICLUSIG in patients unable to tolerate 15 mg orally once daily. | Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily. |
| Subsequent Reduction | Permanently discontinue ICLUSIG in patients unable to tolerate 10 mg orally once daily. |
2.4 Dosage for Patients With Hepatic Impairment (2.4 Dosage for Patients with Hepatic Impairment)
For patients with CP-CML, AP-CML, BP-CML, and Ph+ ALL receiving monotherapy, reduce the starting dose of ICLUSIG from 45 mg orally once daily to 30 mg orally once daily in patients with pre-existing hepatic impairment (Child-Pugh A, B, or C).
For patients with newly diagnosed Ph+ ALL, no dosage adjustment is recommended when administering ICLUSIG to patients with mild hepatic impairment (Child-Pugh A). Closely monitor patients with moderate or severe hepatic impairment (Child-Pugh B or C) and modify the ICLUSIG dosage in the event of adverse reactions [see Dosage and Administration (2.2), Use in Specific Populations (8.6)].
8.3 Females and Males of Reproductive Potential
ICLUSIG can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)].
Principal Display Panel 10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label)
NDC 63020-536-30
ICLUSIG®
(ponatinib) tablets
10 mg
Each tablet contains 10 mg ponatinib
equivalent to 10.68 mg ponatinib HCl
Dispense Attached Medication Guide
30 tablets
Rx only
Takeda
Principal Display Panel 15 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 15 mg Tablet Bottle Label)
NDC 63020-535-30
ICLUSIG®
(ponatinib) tablets
15 mg
Each tablet contains 15 mg ponatinib
equivalent to 16.03 mg ponatinib HCl
Dispense Attached Medication Guide
30 tablets
Rx only
Takeda
Principal Display Panel 30 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 30 mg Tablet Bottle Label)
NDC 63020-533-30
ICLUSIG®
(ponatinib) tablets
30 mg
Each tablet contains 30 mg ponatinib
equivalent to 32.05 mg ponatinib HCl
Dispense Attached Medication Guide
Takeda
Principal Display Panel 45 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 45 mg Tablet Bottle Label)
NDC 63020-534-30
ICLUSIG®
(ponatinib) tablets
45 mg
Each tablet contains 45 mg ponatinib
equivalent to 48.08 mg ponatinib HCl
Dispense Attached Medication Guide
Takeda
5.15 Reversible Posterior Leukoencephalopathy Syndrome
Reversible posterior leukoencephalopathy syndrome (RPLS; also known as Posterior Reversible Encephalopathy Syndrome) has been reported in patients who received ICLUSIG. Patients can present with hypertension, seizure, headache, decreased alertness, altered mental functioning, vision loss, and other visual and neurological disturbances. Magnetic resonance imaging (MRI) is necessary to confirm the diagnosis. Interrupt ICLUSIG until resolution. The safety of resumption of ICLUSIG in patients upon resolution of RPLS is unknown.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2 year carcinogenicity study, male and female rats were administered daily oral doses of ponatinib of 0.05 mg/kg/day, 0.1 mg/kg/day, 0.2 mg/kg/day and 0.2 mg/kg/day, 0.4 mg/kg/day, and 0.8 mg/kg/day, respectively. Exposures in animals at the highest dose tested were 0.3- to 0.8-fold the human exposure (based on AUC) at doses of 15 mg and 45 mg daily. Ponatinib induced a statistically significant increase in malignant squamous neoplasms of the clitoral gland in females at 0.8 mg/kg/day.
Ponatinib was not mutagenic in a bacterial mutagenesis (Ames) assay, was not clastogenic in a chromosome aberration assay in human lymphocytes, nor was it clastogenic in an in vivo mouse micronucleus assay at oral doses up to 2000 mg/kg.
Ponatinib may impair female fertility. In a fertility study in male and female rats, female fertility parameters were reduced at 1.5 mg/kg/day with exposure equivalent to 0.43 times and 1.23 times of human daily steady-state AUC at the maximum recommended human dose of 45 mg/day (AUC = 1296 h∙ng/mL) and 15 mg/day (451.8 h∙ng/mL), respectively. Evidence of pre- and post-implantation loss of embryos was observed in female rats. Although there were no effects on male fertility parameters in the rat fertility study, repeat dose toxicology studies in monkeys showed degeneration of epithelium of the testes in monkeys at exposures approximately 3.3 times the plasma drug exposure (AUC) in patients receiving the maximum recommended human dose of 45 mg/day.
5.7 Increased Toxicity in Newly Diagnosed Chronic Phase Cml (5.7 Increased Toxicity in Newly Diagnosed Chronic Phase CML)
In a prospective randomized clinical trial in the first line treatment of newly diagnosed patients with CP-CML, single agent ICLUSIG 45 mg once daily increased the risk of serious adverse reactions 2-fold compared to single agent imatinib 400 mg once daily. The median exposure to treatment was less than 6 months. The trial was halted for safety.
Arterial and venous thrombosis and occlusions occurred at least twice as frequently in the ICLUSIG arm compared to the imatinib arm. Compared to imatinib-treated patients, ICLUSIG-treated patients exhibited a greater incidence of myelosuppression, pancreatitis, hepatotoxicity, cardiac failure, hypertension, and skin and subcutaneous tissue disorders. ICLUSIG is not indicated and is not recommended for the treatment of patients with newly diagnosed CP-CML.
5.16 Impaired Wound Healing and Gastrointestinal Perforation
Impaired wound healing occurred in patients receiving ICLUSIG [see Adverse Reactions (6.2)]. Withhold ICLUSIG for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of ICLUSIG after resolution of wound healing complications has not been established.
Gastrointestinal perforation or fistula occurred in patients receiving ICLUSIG [see Adverse Reactions (6.2)]. Permanently discontinue in patients with gastrointestinal perforation.
2.3 Dosage Modification for Coadministration of Strong Cyp3a Inhibitors (2.3 Dosage Modification for Coadministration of Strong CYP3A Inhibitors)
Avoid coadministration of ICLUSIG with strong CYP3A inhibitors. If coadministration of a strong CYP3A inhibitor cannot be avoided, reduce the dosage of ICLUSIG as recommended in Table 3.
After the strong CYP3A inhibitor has been discontinued for 3 to 5 elimination half-lives, resume the ICLUSIG dosage that was tolerated prior to initiating the strong CYP3A inhibitor [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
| Current ICLUSIG Dosage | Recommended ICLUSIG Dosage with a Strong CYP3A Inhibitor |
|---|---|
| 45 mg orally once daily | 30 mg orally once daily |
| 30 mg orally once daily | 15 mg orally once daily |
| 15 mg orally once daily | 10 mg orally once daily |
| 10 mg orally once daily | Avoid coadministration of ICLUSIG with a strong CYP3A inhibitor |
Warning: Arterial Occlusive Events, Venous Thromboembolic Events, Heart Failure, and Hepatotoxicity (WARNING: ARTERIAL OCCLUSIVE EVENTS, VENOUS THROMBOEMBOLIC EVENTS, HEART FAILURE, and HEPATOTOXICITY)
WARNING: ARTERIAL OCCLUSIVE EVENTS, VENOUS THROMBOEMBOLIC EVENTS, HEART FAILURE, and HEPATOTOXICITY
See full prescribing information for complete boxed warning.
- Arterial occlusive events (AOEs), including fatalities, have occurred in ICLUSIG-treated patients. AOEs included fatal myocardial infarction, stroke, stenosis of large arterial vessels of the brain, severe peripheral vascular disease, and the need for urgent revascularization procedures. Patients with and without cardiovascular risk factors, including patients age 50 years or younger, experienced these events. Monitor for evidence of AOEs. Interrupt or discontinue ICLUSIG based on severity. Consider benefit-risk to guide a decision to restart ICLUSIG. (2.2, 5.1)
- Venous thromboembolic events (VTEs) have occurred in ICLUSIG-treated patients. Monitor for evidence of VTEs. Interrupt or discontinue ICLUSIG based on severity. (2.2, 5.2)
- Heart failure, including fatalities, occurred in ICLUSIG-treated patients. Monitor for heart failure and manage patients as clinically indicated. Interrupt or discontinue ICLUSIG for new or worsening heart failure. (2.2, 5.3)
- Hepatotoxicity, liver failure and death have occurred in ICLUSIG-treated patients. Monitor liver function tests. Interrupt or discontinue ICLUSIG based on severity. (2.2, 5.4)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:17.715178 · Updated: 2026-03-14T22:48:00.481820