16aa3bcc-81f7-6532-e063-6394a90ace29
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
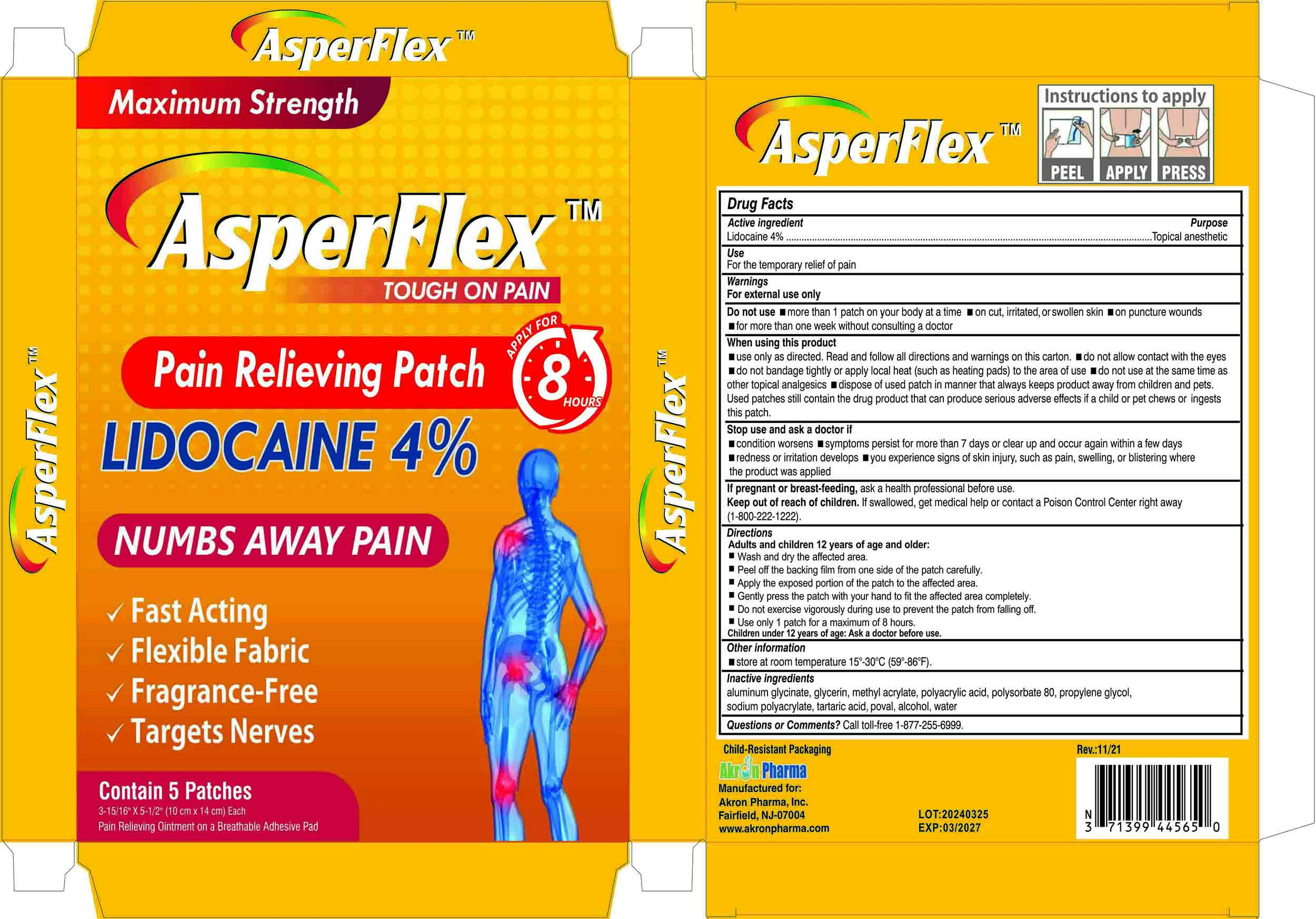
LIDOCAINE PATCH
Medication Information
Warnings and Precautions
For external use only
Indications and Usage
For the temporary relief of pain
Dosage and Administration
Adults and children 12 years of age and older:
Wash and dry the affected area.
Peel off the backing film from one side of the patch carefully
Apply the exposed portion of the patch to the affected area.
Gently press the patch with your hand to fit the affected area completely.
Do not exercise vigorously during use to prevent the patch from falling off.
Use only 1 patch for a maximum of 8 hours.
Children under 12 years of age: Ask a doctor before use
Description
LIDOCAINE PATCH
Section 42229-5
LIDOCAINE PATCH
Section 44425-7
store at room temperature 15°-30C (59-86F).
Section 50565-1
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9
condition worsens
symptoms persist for more than 7 days or clear up and occur again within a few days
redness or irritation develops
you experience signs of skin injury, such as pain, sweling, or blistering where
the product was applied
Section 50567-7
use only as directed. Read and follow all directions and warnings on this carton.
do not allow contact with the eyes
heat (such as heating pads) to the area of use
do not use at the same time as other topical analgesics
dispose of used patch in manner that always keeps product away from children and pets.
Used patches stll contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
Section 50570-1
more than 1 patch on your body at a time
on cut,irritated,or swollen skin
on puncture wounds
for more than one week without consulting a doctor
Section 51727-6
aluminum glycinate
glycerin
methyl acrylate
polyacrylic acid
polysorbate 80
propylene glycol
sodium polyacrylate
tartaric acid
poval
alcohol
Section 51945-4
Section 55105-1
Topical anesthetic
Section 55106-9
Lidocaine 4%
Structured Label Content
Indications and Usage (34067-9)
For the temporary relief of pain
Dosage and Administration (34068-7)
Adults and children 12 years of age and older:
Wash and dry the affected area.
Peel off the backing film from one side of the patch carefully
Apply the exposed portion of the patch to the affected area.
Gently press the patch with your hand to fit the affected area completely.
Do not exercise vigorously during use to prevent the patch from falling off.
Use only 1 patch for a maximum of 8 hours.
Children under 12 years of age: Ask a doctor before use
Warnings and Precautions (34071-1)
For external use only
Section 42229-5 (42229-5)
LIDOCAINE PATCH
Section 44425-7 (44425-7)
store at room temperature 15°-30C (59-86F).
Section 50565-1 (50565-1)
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9 (50566-9)
condition worsens
symptoms persist for more than 7 days or clear up and occur again within a few days
redness or irritation develops
you experience signs of skin injury, such as pain, sweling, or blistering where
the product was applied
Section 50567-7 (50567-7)
use only as directed. Read and follow all directions and warnings on this carton.
do not allow contact with the eyes
heat (such as heating pads) to the area of use
do not use at the same time as other topical analgesics
dispose of used patch in manner that always keeps product away from children and pets.
Used patches stll contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
Section 50570-1 (50570-1)
more than 1 patch on your body at a time
on cut,irritated,or swollen skin
on puncture wounds
for more than one week without consulting a doctor
Section 51727-6 (51727-6)
aluminum glycinate
glycerin
methyl acrylate
polyacrylic acid
polysorbate 80
propylene glycol
sodium polyacrylate
tartaric acid
poval
alcohol
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Topical anesthetic
Section 55106-9 (55106-9)
Lidocaine 4%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:00.810183 · Updated: 2026-03-14T23:02:51.977564