These Highlights Do Not Include All The Information Needed To Use Winlevi Cream Safely And Effectively. See Full Prescribing Information For Winlevi Cream.
1673a84b-7f5c-47ab-a99c-1e3db21a6a09
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WINLEVI (clascoterone) cream is an androgen receptor inhibitor indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
Indications and Usage
WINLEVI (clascoterone) cream is an androgen receptor inhibitor indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
Dosage and Administration
Cleanse the affected area gently. After the skin is dry, apply a thin uniform layer of WINLEVI cream twice per day, in the morning and the evening, to the affected area. Avoid accidental transfer of WINLEVI cream into eyes, mouth or other mucous membranes. If contact with mucous membranes occurs, rinse thoroughly with water. WINLEVI cream is for topical use only. WINLEVI cream is not for ophthalmic, oral or vaginal use.
Warnings and Precautions
Local Irritation: Pruritus, burning, skin redness or peeling may be experienced with WINLEVI cream. If these effects occur, discontinue or reduce the frequency of application of WINLEVI cream. ( 5.1 ) Hypothalamic-pituitary-adrenal (HPA) axis suppression may occur during or after treatment with clascoterone. ( 5.2 ) Attempt to withdraw use if HPA axis suppression develops. ( 5.2 ) Pediatric patients may be more susceptible to systemic toxicity. ( 5.2 , 8.4 ) Hyperkalemia: Elevated potassium levels were observed in some subjects during the clinical trials. ( 12.2 )
Contraindications
None.
Adverse Reactions
Most common adverse reactions occurring in 7 to 12% of patients are erythema/reddening, pruritus and scaling/dryness. Additionally, edema, stinging, and burning occurred in >3% of patients and were reported in a similar percentage of subjects treated with vehicle. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Sun Pharmaceutical Industries, Inc. 1-800-818-4555 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch.
Storage and Handling
WINLEVI cream 1% is supplied in an epoxy-lined aluminum blind-end tube with a polypropylene cap closure: NDC 47335-994-36 60-gram tube
How Supplied
WINLEVI cream 1% is supplied in an epoxy-lined aluminum blind-end tube with a polypropylene cap closure: NDC 47335-994-36 60-gram tube
Medication Information
Warnings and Precautions
Local Irritation: Pruritus, burning, skin redness or peeling may be experienced with WINLEVI cream. If these effects occur, discontinue or reduce the frequency of application of WINLEVI cream. ( 5.1 ) Hypothalamic-pituitary-adrenal (HPA) axis suppression may occur during or after treatment with clascoterone. ( 5.2 ) Attempt to withdraw use if HPA axis suppression develops. ( 5.2 ) Pediatric patients may be more susceptible to systemic toxicity. ( 5.2 , 8.4 ) Hyperkalemia: Elevated potassium levels were observed in some subjects during the clinical trials. ( 12.2 )
Indications and Usage
WINLEVI (clascoterone) cream is an androgen receptor inhibitor indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
Dosage and Administration
Cleanse the affected area gently. After the skin is dry, apply a thin uniform layer of WINLEVI cream twice per day, in the morning and the evening, to the affected area. Avoid accidental transfer of WINLEVI cream into eyes, mouth or other mucous membranes. If contact with mucous membranes occurs, rinse thoroughly with water. WINLEVI cream is for topical use only. WINLEVI cream is not for ophthalmic, oral or vaginal use.
Contraindications
None.
Adverse Reactions
Most common adverse reactions occurring in 7 to 12% of patients are erythema/reddening, pruritus and scaling/dryness. Additionally, edema, stinging, and burning occurred in >3% of patients and were reported in a similar percentage of subjects treated with vehicle. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Sun Pharmaceutical Industries, Inc. 1-800-818-4555 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch.
Storage and Handling
WINLEVI cream 1% is supplied in an epoxy-lined aluminum blind-end tube with a polypropylene cap closure: NDC 47335-994-36 60-gram tube
How Supplied
WINLEVI cream 1% is supplied in an epoxy-lined aluminum blind-end tube with a polypropylene cap closure: NDC 47335-994-36 60-gram tube
Description
WINLEVI (clascoterone) cream is an androgen receptor inhibitor indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
Section 42229-5
Risk Summary
There are no available data on WINLEVI cream use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, subcutaneous administration of clascoterone to pregnant rats and rabbits during organogenesis at doses 8 or 39 times the maximum recommended human dose (MRHD), respectively, increased malformations in rats and post-implantation loss and resorptions in rabbits (see Data ).
The background risk of major birth defects and miscarriage for the indicated population is unknown. Adverse outcomes in pregnancy occur regardless of the health of the mother or the use of medications. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Section 42230-3
|
Patient Information
|
|
Important Information:WINLEVI cream is for use on the skin only (topical). Do not use WINLEVI cream in or on your eyes, mouth, or vagina. |
|
What is WINLEVI cream?
|
|
|
How should I use WINLEVI cream?
|
|
What should I avoid while using WINLEVI cream?
|
|
What are the possible side effects of WINLEVI cream?
The most common side effects of WINLEVI creaminclude reddening, scaling or dryness, and itching of the skin being treated.
|
|
How should I store WINLEVI cream?
Keep WINLEVI cream and all medicines out of the reach of children. |
|
General Information about the safe and effective use of WINLEVI cream.
|
|
What are the ingredients in WINLEVI cream?
Manufactured in Italy for: Sun Pharmaceutical Industries Limited Distributed by: Sun Pharmaceutical Industries, Inc., Cranbury, NJ 08512 WINLEVI is a licensed trademark of Cassiopea, S.p.A. exclusively licensed by Sun Pharmaceutical Industries, Ltd. US Patents: https://www.cassiopea.com/patents ©2022 Sun Pharmaceutical Industries Limited, All right reserved. This Patient Information has been approved by the U.S. Food and Drug Administration |
|
Issued: 07/2022 2000405-3 |
Section 44425-7
Prior to Dispensing:Store the product in a refrigerator between 36°F and 46°F (2°C and 8°C). Do not freeze.
Section 51945-4
NDC 47335-994-36
Rx Only
WINLEVI ®
Clascoterone cream 1%
For Topical Use Only
11 Description
WINLEVI (clascoterone) cream contains clascoterone, an androgen receptor inhibitor, in a cream base for topical dermatologic use. WINLEVI cream is a white to almost white cream.
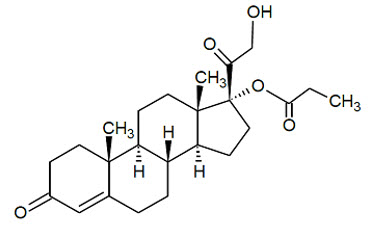
Chemically, clascoterone is cortexolone-17α propionate. Clascoterone is a white to almost white powder, practically insoluble in water. The compound has the empirical formula C 24H 34O 5and molecular weight of 402.5 g/mol. The structural formula is shown below.
Each gram of WINLEVI cream 1% contains 10 mg of clascoterone in a cream base of cetyl alcohol, citric acid monohydrate, edetate disodium, mineral oil, mono- and di-glycerides, polysorbate 80, propylene glycol, purified water, and vitamin E.
8.4 Pediatric Use
Safety and effectiveness of WINLEVI cream for the topical treatment of acne vulgaris have been established in 641 pediatric patients, aged 12 to 18 years in two identical multicenter, randomized, double-blind, vehicle-controlled, 12-week trials and 2 open-label pharmacokinetic studies [see Clinical Studies (14) ].
Safety and effectiveness of WINLEVI cream for the topical treatment of acne vulgaris has not been established in pediatric patients under 12 years of age.
Hypothalamic-pituitary-adrenal (HPA) axis suppression was observed in 2/22 (9%) adolescent subjects. All subjects returned to normal HPA axis function at follow-up 4 weeks after stopping the treatment [see Clinical Pharmacology (12.2) ].Children may be more susceptible to systemic toxicity when treated with clascoterone [see Pharmacodynamics (12.2) ].
8.5 Geriatric Use
Clinical studies of WINLEVI cream did not include sufficient numbers of subjects aged 65 years of age and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies
The safety and efficacy of WINLEVI cream 1% applied twice daily for 12 weeks for the treatment of acne vulgaris were assessed in two identically-designed, multicenter, randomized, double-blind, vehicle-controlled clinical trials (Trial 1 [NCT02608450] and Trial 2 [NCT02608476]) enrolling 1440 subjects with facial acne vulgaris. The trials enrolled subjects 9 years or older with Investigator's Global Assessment (IGA) of moderate or severe facial acne vulgaris (score of 3 or 4), 30 to 75 inflammatory lesions (papules, pustules and nodules), and 30 to 100 non-inflammatory lesions (open and closed comedones).
A total of 1421 subjects 12 years and older with facial acne vulgaris were enrolled. Of these subjects, 641 (45%) were 12 to 17 years of age, and 780 (55%) were 18 years of age or older. In addition, 62% of the subjects were female, and 91% were Caucasian. At baseline, subjects had a mean inflammatory lesion count of 42.4 and a mean non-inflammatory lesion count of 61.4. Additionally, approximately 83% of subjects had an IGA score of 3 ("moderate").
Efficacy was assessed at Week 12 by the proportion of subjects in each treatment group with at least a 2-point reduction in IGA compared to baseline and an IGA score of 0 (clear) or 1 (almost clear), absolute change and percent change from baseline in non-inflammatory and inflammatory lesions. The IGA success rate and mean absolute and percent reduction from baseline in acne lesion counts after 12 weeks of treatment for subjects 12 years of age and older are presented in the following table.
Table 2. Clinical Efficacy of WINLEVI Cream 1% in Subjects with Acne Vulgaris at Week 12
|
Trial 1 |
Trial 2 |
|||
|
WINLEVI Cream 1%
|
Vehicle Cream
|
WINLEVI Cream 1%
|
Vehicle Cream
|
|
|
IGA Success a |
18.8% |
8.7% |
20.9% |
6.6% |
|
Difference from Vehicle
|
10.1%
|
14.3%
|
||
|
Non-inflammatory Lesions |
||||
|
Mean Absolute Reduction |
20.4 |
13.0 |
19.5 |
10.8 |
|
Difference from Vehicle
|
7.3
|
8.7
|
||
|
Mean Percent Reduction |
32.6% |
21.8% |
29.6% |
15.7% |
|
Difference from Vehicle
|
10.8%
|
13.8%
|
||
|
Inflammatory Lesions |
||||
|
Mean Absolute Reduction |
19.3 |
15.4 |
20.1 |
12.6 |
|
Difference from Vehicle
|
3.9
|
7.5
|
||
|
Mean Percent Reduction |
44.6% |
36.3% |
47.1% |
29.7% |
|
Difference from Vehicle
|
8.3%
|
17.5%
|
||
|
aInvestigator Global Assessment (IGA) success was defined as at least a 2-point reduction in IGA compared to baseline and an IGA score of 0 (clear) or 1 (almost clear). |
4 Contraindications
None.
6 Adverse Reactions
Most common adverse reactions occurring in 7 to 12% of patients are erythema/reddening, pruritus and scaling/dryness. Additionally, edema, stinging, and burning occurred in >3% of patients and were reported in a similar percentage of subjects treated with vehicle. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Sun Pharmaceutical Industries, Inc. 1-800-818-4555 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch.
1 Indications and Usage
WINLEVI (clascoterone) cream is an androgen receptor inhibitor indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
12.1 Mechanism of Action
Clascoterone is an androgen receptor inhibitor. The mechanism of action of WINLEVI cream for the topical treatment of acne vulgaris is unknown.
5.1 Local Skin Reactions
WINLEVI cream may induce local irritation (erythema/redness, pruritus, scaling/ dryness). Concomitant use with other potentially irritating topical products (medicated or abrasive soaps and cleansers, soaps and cosmetics that have a strong drying effect and products with high concentrations of alcohol, astringents, spices or lime) should be limited.
The product should not be applied to cuts, abrasions, eczematous or sunburned skin.
5 Warnings and Precautions
- Local Irritation: Pruritus, burning, skin redness or peeling may be experienced with WINLEVI cream. If these effects occur, discontinue or reduce the frequency of application of WINLEVI cream. ( 5.1)
- Hypothalamic-pituitary-adrenal (HPA) axis suppression may occur during or after treatment with clascoterone. ( 5.2)
- Attempt to withdraw use if HPA axis suppression develops. ( 5.2)
- Pediatric patients may be more susceptible to systemic toxicity. ( 5.2, 8.4)
- Hyperkalemia: Elevated potassium levels were observed in some subjects during the clinical trials. ( 12.2)
2 Dosage and Administration
Cleanse the affected area gently. After the skin is dry, apply a thin uniform layer of WINLEVI cream twice per day, in the morning and the evening, to the affected area. Avoid accidental transfer of WINLEVI cream into eyes, mouth or other mucous membranes. If contact with mucous membranes occurs, rinse thoroughly with water.
WINLEVI cream is for topical use only. WINLEVI cream is not for ophthalmic, oral or vaginal use.
3 Dosage Form and Strengths
Cream 1%. Each gram of WINLEVI cream contains 10 mg of clascoterone in a white to almost white cream.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In two identical multicenter, randomized, double-blind, vehicle-controlled trials, 1421 subjects 12 years and older with facial acne vulgaris applied WINLEVI cream or vehicle twice daily for 12 weeks. Overall, 62% of the subjects were female, and 38% were male, 91% of the patients were Caucasian, and the mean age was 19.7 years.
Local skin reactions (edema, erythema/redness, pruritus, scaling/dryness, skin atrophy, stinging/burning, striae rubrea, telangiectasia) were observed during the 12-week treatment and occurred in a similar percentage of subjects treated with vehicle. Local skin reactions reported by ≥ 1% of subjects treated with WINLEVI cream are shown in the following table.
Table 1. Incidence of New or Worsening Local Skin Reactions Reported by ≥ 1% of Subjects Treated with WINLEVI Cream After Day 1 in 12-Week Controlled Clinical Trials
|
WINLEVI Cream 1%
|
Vehicle Cream
|
|
|
aThe denominators for calculating the percentages were the 674 of 709 subjects treated with WINLEVI cream and 656 of 712 subjects treated with vehicle in these trials who had local skin reaction results reported after Day 1. |
||
|
Edema |
24 (3.6%) |
23 (3.5%) |
|
Erythema/redness |
82 (12.2%) |
101 (15.4%) |
|
Pruritus |
52 (7.7%) |
54 (8.2%) |
|
Scaling/dryness |
71 (10.5%) |
68 (10.4%) |
|
Skin atrophy |
11 (1.6%) |
17 (2.6%) |
|
Stinging/burning |
28 (4.2%) |
28 (4.3%) |
|
Striae rubrae |
17 (2.5%) |
10 (1.5%) |
|
Telangiectasia |
8 (1.2%) |
12 (1.8%) |
The following adverse reactions associated with the use of WINLEVI cream were identified in clinical trials and long-term safety studies.
Metabolism: hyperkalemia [see Clinical Pharmacology (12.2) ]
Reproductive: polycystic ovaries, amenorrhea.
17 Patient Counseling Information
- Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Avoid applying WINLEVI Cream to damaged skin (such as cuts, abrasions), eczematous areas, and sunburned skin.
- Avoid concomitant use of other potentially irritating topical products (medicated or not).
16 How Supplied/storage and Handling
WINLEVI cream 1% is supplied in an epoxy-lined aluminum blind-end tube with a polypropylene cap closure:
|
NDC 47335-994-36 |
60-gram tube |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Clascoterone cream (0.1%, 1%, or 5%) was not carcinogenic after daily topical administration in a 2-year carcinogenicity study in rats. An increased incidence of the non-neoplastic finding of atrophy of the skin and subcutis at the application site was reported in males and females treated with 1% and 5% clascoterone cream.
Clascoterone was not mutagenic in the Ames reverse mutation assay and was not clastogenic in the in vitro human lymphocyte chromosomal aberration assay. In rats, clascoterone administered via subcutaneous injection did not induce micronuclei in the bone marrow at 500 or 1000 mg/kg but a slight increase in micronuclei occurred in 2 of 5 rats at 2000 mg/kg. The response was considered equivocal. Overall, the weight of evidence indicates that clascoterone does not represent a genotoxic risk.
In a fertility and early embryonic development study in rats, clascoterone was administered subcutaneously at doses of 0.5, 2.5, or 12.5 mg/kg/day from 2 – 4 weeks before mating through mating. Clascoterone increased pre-implantation loss at 12.5 mg/kg/day (163 times the MRHD based on AUC comparison). Clascoterone had no effects on mating or fertility in rats at doses up to 12.5 mg/kg/day (163 times the MRHD based on AUC comparison). No effects were noted on development at doses up to 2.5 mg/kg/day (33 times the MRHD based on AUC comparison).
5.2 Hypothalamic Pituitary Adrenal (hpa) Axis Suppression
Hypothalamic-pituitary-adrenal (HPA) axis suppression was observed and may occur during or after treatment with clascoterone. In the PK trial, all subjects returned to normal HPA axis function at follow-up 4 weeks after stopping treatment [see Clinical Pharmacology (12.2) ]. Conditions which augment systemic absorption include use over large surface areas, prolonged use, and the use of occlusive dressings.
If HPA axis suppression develops, an attempt should be made to withdraw the drug.
Pediatric patients may be more susceptible to systemic toxicity.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There are no available data on WINLEVI cream use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, subcutaneous administration of clascoterone to pregnant rats and rabbits during organogenesis at doses 8 or 39 times the maximum recommended human dose (MRHD), respectively, increased malformations in rats and post-implantation loss and resorptions in rabbits (see Data ).
The background risk of major birth defects and miscarriage for the indicated population is unknown. Adverse outcomes in pregnancy occur regardless of the health of the mother or the use of medications. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Section 42230-3 (42230-3)
|
Patient Information
|
|
Important Information:WINLEVI cream is for use on the skin only (topical). Do not use WINLEVI cream in or on your eyes, mouth, or vagina. |
|
What is WINLEVI cream?
|
|
|
How should I use WINLEVI cream?
|
|
What should I avoid while using WINLEVI cream?
|
|
What are the possible side effects of WINLEVI cream?
The most common side effects of WINLEVI creaminclude reddening, scaling or dryness, and itching of the skin being treated.
|
|
How should I store WINLEVI cream?
Keep WINLEVI cream and all medicines out of the reach of children. |
|
General Information about the safe and effective use of WINLEVI cream.
|
|
What are the ingredients in WINLEVI cream?
Manufactured in Italy for: Sun Pharmaceutical Industries Limited Distributed by: Sun Pharmaceutical Industries, Inc., Cranbury, NJ 08512 WINLEVI is a licensed trademark of Cassiopea, S.p.A. exclusively licensed by Sun Pharmaceutical Industries, Ltd. US Patents: https://www.cassiopea.com/patents ©2022 Sun Pharmaceutical Industries Limited, All right reserved. This Patient Information has been approved by the U.S. Food and Drug Administration |
|
Issued: 07/2022 2000405-3 |
Section 44425-7 (44425-7)
Prior to Dispensing:Store the product in a refrigerator between 36°F and 46°F (2°C and 8°C). Do not freeze.
Section 51945-4 (51945-4)
NDC 47335-994-36
Rx Only
WINLEVI ®
Clascoterone cream 1%
For Topical Use Only
11 Description (11 DESCRIPTION)
WINLEVI (clascoterone) cream contains clascoterone, an androgen receptor inhibitor, in a cream base for topical dermatologic use. WINLEVI cream is a white to almost white cream.
Chemically, clascoterone is cortexolone-17α propionate. Clascoterone is a white to almost white powder, practically insoluble in water. The compound has the empirical formula C 24H 34O 5and molecular weight of 402.5 g/mol. The structural formula is shown below.
Each gram of WINLEVI cream 1% contains 10 mg of clascoterone in a cream base of cetyl alcohol, citric acid monohydrate, edetate disodium, mineral oil, mono- and di-glycerides, polysorbate 80, propylene glycol, purified water, and vitamin E.
8.4 Pediatric Use
Safety and effectiveness of WINLEVI cream for the topical treatment of acne vulgaris have been established in 641 pediatric patients, aged 12 to 18 years in two identical multicenter, randomized, double-blind, vehicle-controlled, 12-week trials and 2 open-label pharmacokinetic studies [see Clinical Studies (14) ].
Safety and effectiveness of WINLEVI cream for the topical treatment of acne vulgaris has not been established in pediatric patients under 12 years of age.
Hypothalamic-pituitary-adrenal (HPA) axis suppression was observed in 2/22 (9%) adolescent subjects. All subjects returned to normal HPA axis function at follow-up 4 weeks after stopping the treatment [see Clinical Pharmacology (12.2) ].Children may be more susceptible to systemic toxicity when treated with clascoterone [see Pharmacodynamics (12.2) ].
8.5 Geriatric Use
Clinical studies of WINLEVI cream did not include sufficient numbers of subjects aged 65 years of age and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of WINLEVI cream 1% applied twice daily for 12 weeks for the treatment of acne vulgaris were assessed in two identically-designed, multicenter, randomized, double-blind, vehicle-controlled clinical trials (Trial 1 [NCT02608450] and Trial 2 [NCT02608476]) enrolling 1440 subjects with facial acne vulgaris. The trials enrolled subjects 9 years or older with Investigator's Global Assessment (IGA) of moderate or severe facial acne vulgaris (score of 3 or 4), 30 to 75 inflammatory lesions (papules, pustules and nodules), and 30 to 100 non-inflammatory lesions (open and closed comedones).
A total of 1421 subjects 12 years and older with facial acne vulgaris were enrolled. Of these subjects, 641 (45%) were 12 to 17 years of age, and 780 (55%) were 18 years of age or older. In addition, 62% of the subjects were female, and 91% were Caucasian. At baseline, subjects had a mean inflammatory lesion count of 42.4 and a mean non-inflammatory lesion count of 61.4. Additionally, approximately 83% of subjects had an IGA score of 3 ("moderate").
Efficacy was assessed at Week 12 by the proportion of subjects in each treatment group with at least a 2-point reduction in IGA compared to baseline and an IGA score of 0 (clear) or 1 (almost clear), absolute change and percent change from baseline in non-inflammatory and inflammatory lesions. The IGA success rate and mean absolute and percent reduction from baseline in acne lesion counts after 12 weeks of treatment for subjects 12 years of age and older are presented in the following table.
Table 2. Clinical Efficacy of WINLEVI Cream 1% in Subjects with Acne Vulgaris at Week 12
|
Trial 1 |
Trial 2 |
|||
|
WINLEVI Cream 1%
|
Vehicle Cream
|
WINLEVI Cream 1%
|
Vehicle Cream
|
|
|
IGA Success a |
18.8% |
8.7% |
20.9% |
6.6% |
|
Difference from Vehicle
|
10.1%
|
14.3%
|
||
|
Non-inflammatory Lesions |
||||
|
Mean Absolute Reduction |
20.4 |
13.0 |
19.5 |
10.8 |
|
Difference from Vehicle
|
7.3
|
8.7
|
||
|
Mean Percent Reduction |
32.6% |
21.8% |
29.6% |
15.7% |
|
Difference from Vehicle
|
10.8%
|
13.8%
|
||
|
Inflammatory Lesions |
||||
|
Mean Absolute Reduction |
19.3 |
15.4 |
20.1 |
12.6 |
|
Difference from Vehicle
|
3.9
|
7.5
|
||
|
Mean Percent Reduction |
44.6% |
36.3% |
47.1% |
29.7% |
|
Difference from Vehicle
|
8.3%
|
17.5%
|
||
|
aInvestigator Global Assessment (IGA) success was defined as at least a 2-point reduction in IGA compared to baseline and an IGA score of 0 (clear) or 1 (almost clear). |
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions occurring in 7 to 12% of patients are erythema/reddening, pruritus and scaling/dryness. Additionally, edema, stinging, and burning occurred in >3% of patients and were reported in a similar percentage of subjects treated with vehicle. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Sun Pharmaceutical Industries, Inc. 1-800-818-4555 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch.
1 Indications and Usage (1 INDICATIONS AND USAGE)
WINLEVI (clascoterone) cream is an androgen receptor inhibitor indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
12.1 Mechanism of Action
Clascoterone is an androgen receptor inhibitor. The mechanism of action of WINLEVI cream for the topical treatment of acne vulgaris is unknown.
5.1 Local Skin Reactions
WINLEVI cream may induce local irritation (erythema/redness, pruritus, scaling/ dryness). Concomitant use with other potentially irritating topical products (medicated or abrasive soaps and cleansers, soaps and cosmetics that have a strong drying effect and products with high concentrations of alcohol, astringents, spices or lime) should be limited.
The product should not be applied to cuts, abrasions, eczematous or sunburned skin.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Local Irritation: Pruritus, burning, skin redness or peeling may be experienced with WINLEVI cream. If these effects occur, discontinue or reduce the frequency of application of WINLEVI cream. ( 5.1)
- Hypothalamic-pituitary-adrenal (HPA) axis suppression may occur during or after treatment with clascoterone. ( 5.2)
- Attempt to withdraw use if HPA axis suppression develops. ( 5.2)
- Pediatric patients may be more susceptible to systemic toxicity. ( 5.2, 8.4)
- Hyperkalemia: Elevated potassium levels were observed in some subjects during the clinical trials. ( 12.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Cleanse the affected area gently. After the skin is dry, apply a thin uniform layer of WINLEVI cream twice per day, in the morning and the evening, to the affected area. Avoid accidental transfer of WINLEVI cream into eyes, mouth or other mucous membranes. If contact with mucous membranes occurs, rinse thoroughly with water.
WINLEVI cream is for topical use only. WINLEVI cream is not for ophthalmic, oral or vaginal use.
3 Dosage Form and Strengths (3 DOSAGE FORM AND STRENGTHS)
Cream 1%. Each gram of WINLEVI cream contains 10 mg of clascoterone in a white to almost white cream.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In two identical multicenter, randomized, double-blind, vehicle-controlled trials, 1421 subjects 12 years and older with facial acne vulgaris applied WINLEVI cream or vehicle twice daily for 12 weeks. Overall, 62% of the subjects were female, and 38% were male, 91% of the patients were Caucasian, and the mean age was 19.7 years.
Local skin reactions (edema, erythema/redness, pruritus, scaling/dryness, skin atrophy, stinging/burning, striae rubrea, telangiectasia) were observed during the 12-week treatment and occurred in a similar percentage of subjects treated with vehicle. Local skin reactions reported by ≥ 1% of subjects treated with WINLEVI cream are shown in the following table.
Table 1. Incidence of New or Worsening Local Skin Reactions Reported by ≥ 1% of Subjects Treated with WINLEVI Cream After Day 1 in 12-Week Controlled Clinical Trials
|
WINLEVI Cream 1%
|
Vehicle Cream
|
|
|
aThe denominators for calculating the percentages were the 674 of 709 subjects treated with WINLEVI cream and 656 of 712 subjects treated with vehicle in these trials who had local skin reaction results reported after Day 1. |
||
|
Edema |
24 (3.6%) |
23 (3.5%) |
|
Erythema/redness |
82 (12.2%) |
101 (15.4%) |
|
Pruritus |
52 (7.7%) |
54 (8.2%) |
|
Scaling/dryness |
71 (10.5%) |
68 (10.4%) |
|
Skin atrophy |
11 (1.6%) |
17 (2.6%) |
|
Stinging/burning |
28 (4.2%) |
28 (4.3%) |
|
Striae rubrae |
17 (2.5%) |
10 (1.5%) |
|
Telangiectasia |
8 (1.2%) |
12 (1.8%) |
The following adverse reactions associated with the use of WINLEVI cream were identified in clinical trials and long-term safety studies.
Metabolism: hyperkalemia [see Clinical Pharmacology (12.2) ]
Reproductive: polycystic ovaries, amenorrhea.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
- Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Avoid applying WINLEVI Cream to damaged skin (such as cuts, abrasions), eczematous areas, and sunburned skin.
- Avoid concomitant use of other potentially irritating topical products (medicated or not).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
WINLEVI cream 1% is supplied in an epoxy-lined aluminum blind-end tube with a polypropylene cap closure:
|
NDC 47335-994-36 |
60-gram tube |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Clascoterone cream (0.1%, 1%, or 5%) was not carcinogenic after daily topical administration in a 2-year carcinogenicity study in rats. An increased incidence of the non-neoplastic finding of atrophy of the skin and subcutis at the application site was reported in males and females treated with 1% and 5% clascoterone cream.
Clascoterone was not mutagenic in the Ames reverse mutation assay and was not clastogenic in the in vitro human lymphocyte chromosomal aberration assay. In rats, clascoterone administered via subcutaneous injection did not induce micronuclei in the bone marrow at 500 or 1000 mg/kg but a slight increase in micronuclei occurred in 2 of 5 rats at 2000 mg/kg. The response was considered equivocal. Overall, the weight of evidence indicates that clascoterone does not represent a genotoxic risk.
In a fertility and early embryonic development study in rats, clascoterone was administered subcutaneously at doses of 0.5, 2.5, or 12.5 mg/kg/day from 2 – 4 weeks before mating through mating. Clascoterone increased pre-implantation loss at 12.5 mg/kg/day (163 times the MRHD based on AUC comparison). Clascoterone had no effects on mating or fertility in rats at doses up to 12.5 mg/kg/day (163 times the MRHD based on AUC comparison). No effects were noted on development at doses up to 2.5 mg/kg/day (33 times the MRHD based on AUC comparison).
5.2 Hypothalamic Pituitary Adrenal (hpa) Axis Suppression (5.2 Hypothalamic-pituitary-adrenal (HPA) Axis Suppression)
Hypothalamic-pituitary-adrenal (HPA) axis suppression was observed and may occur during or after treatment with clascoterone. In the PK trial, all subjects returned to normal HPA axis function at follow-up 4 weeks after stopping treatment [see Clinical Pharmacology (12.2) ]. Conditions which augment systemic absorption include use over large surface areas, prolonged use, and the use of occlusive dressings.
If HPA axis suppression develops, an attempt should be made to withdraw the drug.
Pediatric patients may be more susceptible to systemic toxicity.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:40.738430 · Updated: 2026-03-14T22:20:59.493265