Actinel®
16660d82-524b-42ca-8581-48237caa13b2
34390-5
HUMAN OTC DRUG LABEL
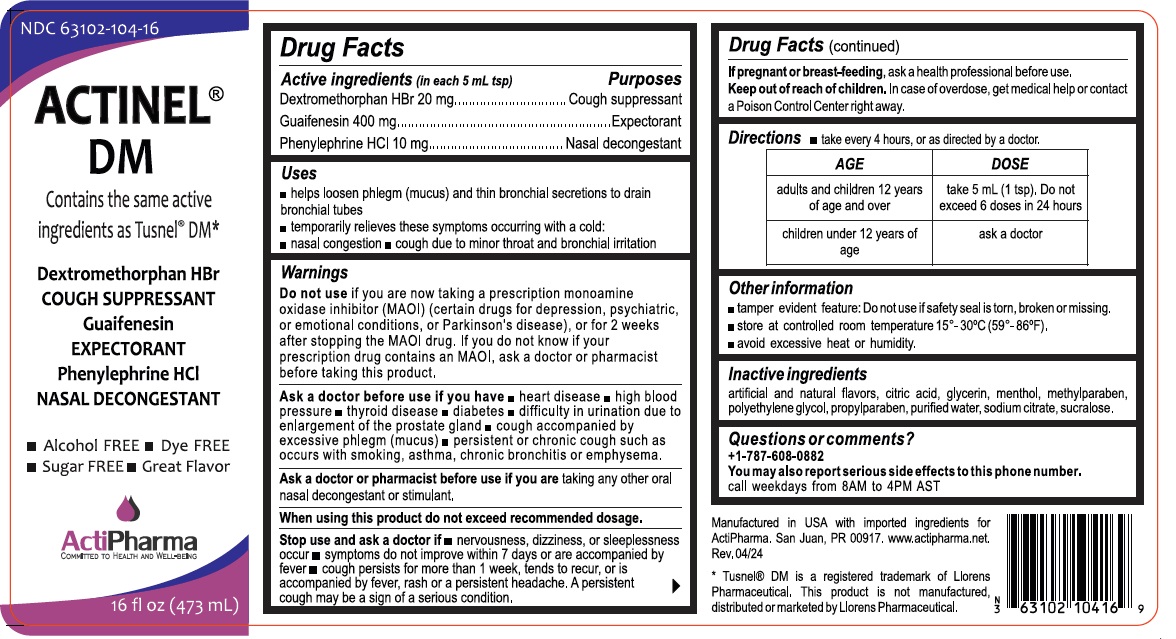
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dextromethorphan HBr 20 mg Guaifenesin 400 mg Phenylephrine HCl 10 mg
Purpose
Cough suppressant Expectorant Nasal decongestant
Medication Information
Purpose
Cough suppressant Expectorant Nasal decongestant
Description
Dextromethorphan HBr 20 mg Guaifenesin 400 mg Phenylephrine HCl 10 mg
Uses
• helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes
• temporarily relieves these symptoms occurring with a cold:
• nasal congestion • cough due to minor throat and bronchial irritation
Section 42229-5
Contains the same active ingredients as Tusnel® DM*
• Alcohol FREE • Dye FREE
• Sugar FREE • Great Flavor
Manufactured in USA with imported ingredients for ActiPharma. San Juan, PR 00917. www.actipharma.net.
* Tusnel® DM is a registered trademark of Llorens Pharmaceutical. This product is not manufactured, distributed or marketed by Llorens Pharmaceutical.
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Purposes
Cough suppressant
Expectorant
Nasal decongestant
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have • heart disease • high blood pressure • thyroid disease • diabetes • difficulty in urination due to enlargement of the prostate gland • cough accompanied by excessive phlegm (mucus) • persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema.
Ask a doctor or pharmacist before use if you are taking any other oral nasal decongestant or stimulant.
When using this produ ct do not exceed recommended dosage.
Stop use and ask a doctor if • nervousness, dizziness, or sleeplessness occur • symptoms do not improve within 7 days or are accompanied by fever • cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash or a persistent headache. A persistent cough may be a sign of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Packaging
Directions
- take every 4 hours, or as directed by a doctor.
| AGE | DOSE |
| adults and children 12 years of age and over |
take 5mL (1tsp). Do not exceed 6 doses in 24 hours |
| Children under 12 years of age | ask a doctor |
Drug Facts
Other Information
• tamper evident feature: Do not use if safety seal is torn, broken or missing.
• store at controlled room temperature 15°- 30°C (59°- 86°F).
• avoid excessive heat or humidity.
Inactive Ingredients
artificial and natural flavors, citric acid, glycerin, menthol, methylparaben, polyethylene glycol, propylparaben, purified water, sodium citrate, sucralose.
Questions Or Comments?
+1-787-608-0882
You may also report serious side effects to this phone number.
call weekdays from 8AM to 4PM AST
Active Ingredients (in Each 5 Ml Tsp)
Dextromethorphan HBr 20 mg
Guaifenesin 400 mg
Phenylephrine HCl 10 mg
Structured Label Content
Uses
• helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes
• temporarily relieves these symptoms occurring with a cold:
• nasal congestion • cough due to minor throat and bronchial irritation
Section 42229-5 (42229-5)
Contains the same active ingredients as Tusnel® DM*
• Alcohol FREE • Dye FREE
• Sugar FREE • Great Flavor
Manufactured in USA with imported ingredients for ActiPharma. San Juan, PR 00917. www.actipharma.net.
* Tusnel® DM is a registered trademark of Llorens Pharmaceutical. This product is not manufactured, distributed or marketed by Llorens Pharmaceutical.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Purposes
Cough suppressant
Expectorant
Nasal decongestant
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have • heart disease • high blood pressure • thyroid disease • diabetes • difficulty in urination due to enlargement of the prostate gland • cough accompanied by excessive phlegm (mucus) • persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema.
Ask a doctor or pharmacist before use if you are taking any other oral nasal decongestant or stimulant.
When using this produ ct do not exceed recommended dosage.
Stop use and ask a doctor if • nervousness, dizziness, or sleeplessness occur • symptoms do not improve within 7 days or are accompanied by fever • cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash or a persistent headache. A persistent cough may be a sign of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Packaging
Directions
- take every 4 hours, or as directed by a doctor.
| AGE | DOSE |
| adults and children 12 years of age and over |
take 5mL (1tsp). Do not exceed 6 doses in 24 hours |
| Children under 12 years of age | ask a doctor |
Drug Facts
Other Information (Other information)
• tamper evident feature: Do not use if safety seal is torn, broken or missing.
• store at controlled room temperature 15°- 30°C (59°- 86°F).
• avoid excessive heat or humidity.
Inactive Ingredients (Inactive ingredients)
artificial and natural flavors, citric acid, glycerin, menthol, methylparaben, polyethylene glycol, propylparaben, purified water, sodium citrate, sucralose.
Questions Or Comments? (Questions or comments?)
+1-787-608-0882
You may also report serious side effects to this phone number.
call weekdays from 8AM to 4PM AST
Active Ingredients (in Each 5 Ml Tsp) (Active ingredients (in each 5 mL tsp))
Dextromethorphan HBr 20 mg
Guaifenesin 400 mg
Phenylephrine HCl 10 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:24.049337 · Updated: 2026-03-14T23:03:27.182760