These Highlights Do Not Include All The Information Needed To Use Zepatier Safely And Effectively. See Full Prescribing Information For Zepatier.
164dc02a-9180-426a-b8b5-04ab39d2bbd4
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Test all patients for evidence of current or prior hepatitis B virus (HBV) infection before initiating treatment with ZEPATIER. HBV reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals and were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure, and death. Monitor HCV/HBV coinfected patients for hepatitis flare or HBV reactivation during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated [see Warnings and Precautions (5.1) ].
Indications and Usage
ZEPATIER ® is indicated for the treatment of chronic hepatitis C virus (HCV) genotype 1 or 4 infection in adult and pediatric patients 12 years of age and older or weighing at least 30 kg. ZEPATIER is indicated for use with ribavirin in certain patient populations [see Dosage and Administration (2.2) ] .
Dosage and Administration
Testing Prior to Initiation of Therapy: Test all patients for HBV infection by measuring HBsAg and anti-HBc. ( 2.1 ) Genotype 1a: Testing for the presence of virus with NS5A resistance-associated polymorphisms is recommended. ( 2.1 ) Obtain hepatic laboratory testing. ( 2.1 ) Recommended dosage in adult and pediatric patients 12 years of age and older or weighing at least 30 kg: One tablet taken orally once daily with or without food. ( 2.2 ) Dosage Regimens and Durations for ZEPATIER in Patients with Genotype 1 or 4 HCV with or without Cirrhosis Patient Population Treatment Duration Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced Peginterferon alfa + ribavirin. without baseline NS5A polymorphisms Polymorphisms at amino acid positions 28, 30, 31, or 93. ZEPATIER 12 weeks Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced with baseline NS5A polymorphisms ZEPATIER + ribavirin 16 weeks Genotype 1b: Treatment-naïve or PegIFN/RBV-experienced ZEPATIER 12 weeks Genotype 1a or 1b: PegIFN/RBV/PI-experienced Peginterferon alfa + ribavirin + HCV NS3/4A protease inhibitor. ZEPATIER + ribavirin 12 weeks Genotype 4: Treatment-naïve ZEPATIER 12 weeks Genotype 4: PegIFN/RBV-experienced ZEPATIER + ribavirin 16 weeks HCV/HIV-1 co-infection: Follow the dosage recommendations in the table above. ( 2.2 ) Renal Impairment, including hemodialysis: No dosage adjustment of ZEPATIER is recommended. Refer to ribavirin prescribing information for ribavirin dosing and dosage modifications. ( 2.3 )
Warnings and Precautions
Risk of Hepatitis B Virus Reactivation: Test all patients for evidence of current or prior HBV infection before initiation of HCV treatment. Monitor HCV/HBV coinfected patients for HBV reactivation and hepatitis flare during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated. ( 5.1 ) ALT Elevations: Perform hepatic laboratory testing prior to therapy, at treatment week 8, and as clinically indicated. For patients receiving 16 weeks of therapy, perform additional hepatic laboratory testing at treatment week 12. For ALT elevations on ZEPATIER, follow recommendations in full prescribing information. ( 5.2 ) Risk of Hepatic Decompensation/Failure in Patients with Evidence of Advanced Liver Disease: Hepatic decompensation/failure, including fatal outcomes, have been reported mostly in patients with cirrhosis and baseline moderate or severe liver impairment (Child-Pugh B or C) treated with HCV NS3/4A protease inhibitor-containing regimens. Monitor for clinical and laboratory evidence of hepatic decompensation. Discontinue ZEPATIER in patients who develop evidence of hepatic decompensation/failure. ( 5.3 ) Risk Associated with Ribavirin Combination Treatment: If ZEPATIER is administered with ribavirin, the warnings and precautions for ribavirin also apply. ( 5.4 )
Contraindications
ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) due to the expected significantly increased grazoprevir plasma concentration and the increased risk of alanine aminotransferase (ALT) elevations [see Warnings and Precautions (5.2) , Use in Specific Populations (8.9) , and Clinical Pharmacology (12.3) ] . ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) or those with any history of hepatic decompensation due to the risk of hepatic decompensation [see Warnings and Precautions (5.3) , Use in Specific Populations (8.9) ] . ZEPATIER is contraindicated with inhibitors of organic anion transporting polypeptides 1B1/3 (OATP1B1/3) that are known or expected to significantly increase grazoprevir plasma concentrations, strong inducers of cytochrome P450 3A (CYP3A), and efavirenz [see Warnings and Precautions (5.5) , Drug Interactions (7) , and Clinical Pharmacology (12.3) ] . If ZEPATIER is administered with ribavirin, the contraindications to ribavirin also apply to this combination regimen. Refer to the ribavirin prescribing information for a list of contraindications for ribavirin. Table 2 lists drugs that are contraindicated with ZEPATIER. Table 2: Drugs that are Contraindicated with ZEPATIER Drug Class Drug(s) within Class that are Contraindicated Clinical Comment This table is not a comprehensive list of all drugs that strongly induce CYP3A. This table may not include all OATP1B1/3 inhibitors that significantly increase grazoprevir plasma concentrations. Anticonvulsants Phenytoin Carbamazepine May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. Antimycobacterials Rifampin May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. Herbal Products St. John's Wort (Hypericum perforatum) May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. HIV Medications Efavirenz Efavirenz is included as a strong CYP3A inducer in this table, since co-administration reduced grazoprevir exposure by ≥80% [see Table 9 ] . May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by CYP3A induction. HIV Medications Atazanavir Darunavir Lopinavir Saquinavir Tipranavir May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition. Immunosuppressants Cyclosporine May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition.
Adverse Reactions
The following adverse reaction is described below and elsewhere in the labeling: Increased Risk of ALT Elevations [see Warnings and Precautions (5.2) ].
Drug Interactions
If dose adjustments of concomitant medications are made due to treatment with ZEPATIER, doses should be readjusted after administration of ZEPATIER is completed. Clearance of HCV infection with direct-acting antivirals may lead to changes in hepatic function, which may impact the safe and effective use of concomitant medications. For example, altered blood glucose control resulting in serious symptomatic hypoglycemia has been reported in diabetic patients in postmarketing case reports and published epidemiological studies. Management of hypoglycemia in these cases required either discontinuation or dose modification of concomitant medications used for diabetes treatment. Frequent monitoring of relevant laboratory parameters (e.g., International Normalized Ratio [INR] in patients taking warfarin, blood glucose levels in diabetic patients) or drug concentrations of concomitant medications such as CYP450 substrates with a narrow therapeutic index (e.g., certain immunosuppressants) is recommended to ensure safe and effective use. Dose adjustments of concomitant medications may be necessary. Table 6 provides a listing of established or potentially clinically significant drug interactions. The drug interactions described are based on studies conducted with either ZEPATIER, the components of ZEPATIER (elbasvir [EBR] and grazoprevir [GZR]) as individual agents, or are predicted drug interactions that may occur with ZEPATIER [see Contraindications (4) , Warnings and Precautions (5.5) , and Clinical Pharmacology (12.3) ] . Table 6: Potentially Significant Drug Interactions: Alteration in Dose May Be Recommended Based on Results from Drug Interaction Studies or Predicted Interactions This table is not all inclusive. Concomitant Drug Class: Drug Name Effect on Concentration ↓ = decrease, ↑ = increase Clinical Comment Antibiotics : Nafcillin ↓ EBR ↓ GZR Co-administration of ZEPATIER with nafcillin may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. Antifungals: oral Ketoconazole These interactions have been studied in healthy adults. ↑ EBR ↑ GZR Co-administration of oral ketoconazole is not recommended. Endothelin Antagonists: Bosentan ↓ EBR ↓ GZR Co-administration of ZEPATIER with bosentan may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. Immunosuppressants: Tacrolimus ↑ tacrolimus Frequent monitoring of tacrolimus whole blood concentrations, changes in renal function, and tacrolimus-associated adverse events upon the initiation of co-administration is recommended. HIV Medications: Etravirine ↓ EBR ↓ GZR Co-administration of ZEPATIER with etravirine may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. Elvitegravir/ cobicistat/ emtricitabine/ tenofovir (disoproxil fumarate or alafenamide) ↑ EBR ↑ GZR Co-administration of cobicistat-containing regimens is not recommended. HMG-CoA Reductase Inhibitors See Drug Interactions (7.3) for a list of HMG Co-A reductase inhibitors without clinically relevant interactions with ZEPATIER. : Atorvastatin ↑ atorvastatin The dose of atorvastatin should not exceed a daily dose of 20 mg when co-administered with ZEPATIER. Rosuvastatin ↑ rosuvastatin The dose of rosuvastatin should not exceed a daily dose of 10 mg when co-administered with ZEPATIER. Fluvastatin Lovastatin Simvastatin ↑ fluvastatin ↑ lovastatin ↑ simvastatin Statin-associated adverse events such as myopathy should be closely monitored. The lowest necessary dose should be used when co-administered with ZEPATIER. Wakefulness-Promoting Agents: Modafinil ↓ EBR ↓ GZR Co-administration of ZEPATIER with modafinil may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended.
Storage and Handling
Each ZEPATIER tablet contains 50 mg elbasvir and 100 mg grazoprevir, is beige, oval-shaped, film-coated, debossed with "770" on one side and plain on the other. The tablets are packaged into a carton (NDC 0006-3074-02) containing two (2) 14-count child-resistant dose packs for a total of 28 tablets.
How Supplied
Each ZEPATIER tablet contains 50 mg elbasvir and 100 mg grazoprevir, is beige, oval-shaped, film-coated, debossed with "770" on one side and plain on the other. The tablets are packaged into a carton (NDC 0006-3074-02) containing two (2) 14-count child-resistant dose packs for a total of 28 tablets.
Medication Information
Warnings and Precautions
Risk of Hepatitis B Virus Reactivation: Test all patients for evidence of current or prior HBV infection before initiation of HCV treatment. Monitor HCV/HBV coinfected patients for HBV reactivation and hepatitis flare during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated. ( 5.1 ) ALT Elevations: Perform hepatic laboratory testing prior to therapy, at treatment week 8, and as clinically indicated. For patients receiving 16 weeks of therapy, perform additional hepatic laboratory testing at treatment week 12. For ALT elevations on ZEPATIER, follow recommendations in full prescribing information. ( 5.2 ) Risk of Hepatic Decompensation/Failure in Patients with Evidence of Advanced Liver Disease: Hepatic decompensation/failure, including fatal outcomes, have been reported mostly in patients with cirrhosis and baseline moderate or severe liver impairment (Child-Pugh B or C) treated with HCV NS3/4A protease inhibitor-containing regimens. Monitor for clinical and laboratory evidence of hepatic decompensation. Discontinue ZEPATIER in patients who develop evidence of hepatic decompensation/failure. ( 5.3 ) Risk Associated with Ribavirin Combination Treatment: If ZEPATIER is administered with ribavirin, the warnings and precautions for ribavirin also apply. ( 5.4 )
Indications and Usage
ZEPATIER ® is indicated for the treatment of chronic hepatitis C virus (HCV) genotype 1 or 4 infection in adult and pediatric patients 12 years of age and older or weighing at least 30 kg. ZEPATIER is indicated for use with ribavirin in certain patient populations [see Dosage and Administration (2.2) ] .
Dosage and Administration
Testing Prior to Initiation of Therapy: Test all patients for HBV infection by measuring HBsAg and anti-HBc. ( 2.1 ) Genotype 1a: Testing for the presence of virus with NS5A resistance-associated polymorphisms is recommended. ( 2.1 ) Obtain hepatic laboratory testing. ( 2.1 ) Recommended dosage in adult and pediatric patients 12 years of age and older or weighing at least 30 kg: One tablet taken orally once daily with or without food. ( 2.2 ) Dosage Regimens and Durations for ZEPATIER in Patients with Genotype 1 or 4 HCV with or without Cirrhosis Patient Population Treatment Duration Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced Peginterferon alfa + ribavirin. without baseline NS5A polymorphisms Polymorphisms at amino acid positions 28, 30, 31, or 93. ZEPATIER 12 weeks Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced with baseline NS5A polymorphisms ZEPATIER + ribavirin 16 weeks Genotype 1b: Treatment-naïve or PegIFN/RBV-experienced ZEPATIER 12 weeks Genotype 1a or 1b: PegIFN/RBV/PI-experienced Peginterferon alfa + ribavirin + HCV NS3/4A protease inhibitor. ZEPATIER + ribavirin 12 weeks Genotype 4: Treatment-naïve ZEPATIER 12 weeks Genotype 4: PegIFN/RBV-experienced ZEPATIER + ribavirin 16 weeks HCV/HIV-1 co-infection: Follow the dosage recommendations in the table above. ( 2.2 ) Renal Impairment, including hemodialysis: No dosage adjustment of ZEPATIER is recommended. Refer to ribavirin prescribing information for ribavirin dosing and dosage modifications. ( 2.3 )
Contraindications
ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) due to the expected significantly increased grazoprevir plasma concentration and the increased risk of alanine aminotransferase (ALT) elevations [see Warnings and Precautions (5.2) , Use in Specific Populations (8.9) , and Clinical Pharmacology (12.3) ] . ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) or those with any history of hepatic decompensation due to the risk of hepatic decompensation [see Warnings and Precautions (5.3) , Use in Specific Populations (8.9) ] . ZEPATIER is contraindicated with inhibitors of organic anion transporting polypeptides 1B1/3 (OATP1B1/3) that are known or expected to significantly increase grazoprevir plasma concentrations, strong inducers of cytochrome P450 3A (CYP3A), and efavirenz [see Warnings and Precautions (5.5) , Drug Interactions (7) , and Clinical Pharmacology (12.3) ] . If ZEPATIER is administered with ribavirin, the contraindications to ribavirin also apply to this combination regimen. Refer to the ribavirin prescribing information for a list of contraindications for ribavirin. Table 2 lists drugs that are contraindicated with ZEPATIER. Table 2: Drugs that are Contraindicated with ZEPATIER Drug Class Drug(s) within Class that are Contraindicated Clinical Comment This table is not a comprehensive list of all drugs that strongly induce CYP3A. This table may not include all OATP1B1/3 inhibitors that significantly increase grazoprevir plasma concentrations. Anticonvulsants Phenytoin Carbamazepine May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. Antimycobacterials Rifampin May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. Herbal Products St. John's Wort (Hypericum perforatum) May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. HIV Medications Efavirenz Efavirenz is included as a strong CYP3A inducer in this table, since co-administration reduced grazoprevir exposure by ≥80% [see Table 9 ] . May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by CYP3A induction. HIV Medications Atazanavir Darunavir Lopinavir Saquinavir Tipranavir May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition. Immunosuppressants Cyclosporine May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition.
Adverse Reactions
The following adverse reaction is described below and elsewhere in the labeling: Increased Risk of ALT Elevations [see Warnings and Precautions (5.2) ].
Drug Interactions
If dose adjustments of concomitant medications are made due to treatment with ZEPATIER, doses should be readjusted after administration of ZEPATIER is completed. Clearance of HCV infection with direct-acting antivirals may lead to changes in hepatic function, which may impact the safe and effective use of concomitant medications. For example, altered blood glucose control resulting in serious symptomatic hypoglycemia has been reported in diabetic patients in postmarketing case reports and published epidemiological studies. Management of hypoglycemia in these cases required either discontinuation or dose modification of concomitant medications used for diabetes treatment. Frequent monitoring of relevant laboratory parameters (e.g., International Normalized Ratio [INR] in patients taking warfarin, blood glucose levels in diabetic patients) or drug concentrations of concomitant medications such as CYP450 substrates with a narrow therapeutic index (e.g., certain immunosuppressants) is recommended to ensure safe and effective use. Dose adjustments of concomitant medications may be necessary. Table 6 provides a listing of established or potentially clinically significant drug interactions. The drug interactions described are based on studies conducted with either ZEPATIER, the components of ZEPATIER (elbasvir [EBR] and grazoprevir [GZR]) as individual agents, or are predicted drug interactions that may occur with ZEPATIER [see Contraindications (4) , Warnings and Precautions (5.5) , and Clinical Pharmacology (12.3) ] . Table 6: Potentially Significant Drug Interactions: Alteration in Dose May Be Recommended Based on Results from Drug Interaction Studies or Predicted Interactions This table is not all inclusive. Concomitant Drug Class: Drug Name Effect on Concentration ↓ = decrease, ↑ = increase Clinical Comment Antibiotics : Nafcillin ↓ EBR ↓ GZR Co-administration of ZEPATIER with nafcillin may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. Antifungals: oral Ketoconazole These interactions have been studied in healthy adults. ↑ EBR ↑ GZR Co-administration of oral ketoconazole is not recommended. Endothelin Antagonists: Bosentan ↓ EBR ↓ GZR Co-administration of ZEPATIER with bosentan may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. Immunosuppressants: Tacrolimus ↑ tacrolimus Frequent monitoring of tacrolimus whole blood concentrations, changes in renal function, and tacrolimus-associated adverse events upon the initiation of co-administration is recommended. HIV Medications: Etravirine ↓ EBR ↓ GZR Co-administration of ZEPATIER with etravirine may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. Elvitegravir/ cobicistat/ emtricitabine/ tenofovir (disoproxil fumarate or alafenamide) ↑ EBR ↑ GZR Co-administration of cobicistat-containing regimens is not recommended. HMG-CoA Reductase Inhibitors See Drug Interactions (7.3) for a list of HMG Co-A reductase inhibitors without clinically relevant interactions with ZEPATIER. : Atorvastatin ↑ atorvastatin The dose of atorvastatin should not exceed a daily dose of 20 mg when co-administered with ZEPATIER. Rosuvastatin ↑ rosuvastatin The dose of rosuvastatin should not exceed a daily dose of 10 mg when co-administered with ZEPATIER. Fluvastatin Lovastatin Simvastatin ↑ fluvastatin ↑ lovastatin ↑ simvastatin Statin-associated adverse events such as myopathy should be closely monitored. The lowest necessary dose should be used when co-administered with ZEPATIER. Wakefulness-Promoting Agents: Modafinil ↓ EBR ↓ GZR Co-administration of ZEPATIER with modafinil may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended.
Storage and Handling
Each ZEPATIER tablet contains 50 mg elbasvir and 100 mg grazoprevir, is beige, oval-shaped, film-coated, debossed with "770" on one side and plain on the other. The tablets are packaged into a carton (NDC 0006-3074-02) containing two (2) 14-count child-resistant dose packs for a total of 28 tablets.
How Supplied
Each ZEPATIER tablet contains 50 mg elbasvir and 100 mg grazoprevir, is beige, oval-shaped, film-coated, debossed with "770" on one side and plain on the other. The tablets are packaged into a carton (NDC 0006-3074-02) containing two (2) 14-count child-resistant dose packs for a total of 28 tablets.
Description
Test all patients for evidence of current or prior hepatitis B virus (HBV) infection before initiating treatment with ZEPATIER. HBV reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals and were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure, and death. Monitor HCV/HBV coinfected patients for hepatitis flare or HBV reactivation during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated [see Warnings and Precautions (5.1) ].
Section 34083-6
Carcinogenesis and Mutagenesis
Elbasvir and grazoprevir were not genotoxic in a battery of in vitro or in vivo assays, including microbial mutagenesis, chromosomal aberration in Chinese Hamster Ovary cells, and in in vivo rat micronucleus assays.
Carcinogenicity studies with elbasvir or grazoprevir have not been conducted.
If ZEPATIER is administered in a regimen containing ribavirin, the information for ribavirin on carcinogenesis and mutagenesis also applies to this combination regimen. Refer to the ribavirin prescribing information for information on carcinogenesis and mutagenesis.
Section 42229-5
Testing for HBV Infection
Test all patients for evidence of current or prior HBV infection by measuring hepatitis B surface antigen (HBsAg) and hepatitis B core antibody (anti-HBc) before initiating HCV treatment with ZEPATIER [see Warnings and Precautions (5.1)].
Section 42230-3
|
Patient Information ZEPATIER® (ZEP-ah-teer) (elbasvir and grazoprevir) tablets |
What you need to know about ZEPATIER
|
|
What is the most important information I should know about ZEPATIER? |
ZEPATIER can cause serious side effects, including,
Hepatitis B virus reactivation: Before starting treatment with ZEPATIER, your healthcare provider will do blood tests to check for hepatitis B virus infection. If you have ever had hepatitis B virus infection, the hepatitis B virus could become active again during or after treatment of hepatitis C virus infection with ZEPATIER. Hepatitis B virus becoming active again (called reactivation) may cause serious liver problems including liver failure and death. Your healthcare provider will monitor you if you are at risk for hepatitis B virus reactivation during treatment and after you stop taking ZEPATIER.
For more information about side effects, see the section "What are the possible side effects of ZEPATIER?"
| What is ZEPATIER®? |
ZEPATIER is a prescription medicine used with or without ribavirin to treat chronic (long-lasting) hepatitis C virus (HCV) genotypes 1 or 4 infection in adults and children 12 years of age and older or weighing at least 66 pounds (30 kilograms).
ZEPATIER should not be used in children younger than 12 years of age who weigh less than 66 pounds (30 kilograms).
It is not known if ZEPATIER is safe or effective in people awaiting a liver transplant or people who have had a liver transplant.
| Who should not take ZEPATIER? |
Do not take ZEPATIER if you have certain liver problems.
| What should I tell my healthcare provider before taking ZEPATIER? |
Before taking ZEPATIER, tell your healthcare provider about all of your medical conditions, including if you:
- have ever had hepatitis B virus infection
- have liver problems other than hepatitis C
- have ever taken any medicine for hepatitis C
- have HIV
- have had or are waiting for a liver transplant
- are pregnant or trying to get pregnant. ZEPATIER has not been studied in pregnant women. We do not know if ZEPATIER will harm your baby while you are pregnant.
- Males and females who take ZEPATIER and ribavirin should also read the ribavirin Medication Guide for important pregnancy, contraception, and infertility information.
- are breastfeeding or plan to breastfeed. We do not know if ZEPATIER gets in your breast milk and gets passed to your baby.
- Talk to your healthcare provider about the best way to feed your baby during treatment with ZEPATIER.
| Are you taking other medicines? |
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. ZEPATIER may affect the way other medicines work, and other medicines may affect how ZEPATIER works. Some medicines cannot be taken with ZEPATIER. Your healthcare provider can tell you if it is safe to take ZEPATIER with other medicines.
- Know the medicines you take. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with this medicine.
- Do not start taking a new medicine without telling your healthcare provider.
| How do I take ZEPATIER? |
- Take 1 ZEPATIER tablet at the same time every day.
- ZEPATIER comes in a blister package of individually-packaged tablets. Keep the tablets in this package until you are ready to take your dose.
- Take ZEPATIER exactly as your healthcare provider tells you to take it.
- Take ZEPATIER with or without food.
- Do not stop taking ZEPATIER without first talking with your healthcare provider.
- If you take more than your prescribed dose, call your healthcare provider right away.
| What if I forget to take ZEPATIER? |
- Do not take two doses of ZEPATIER at the same time to make up for a missed dose.
- If you are not sure what to do, call your healthcare provider or pharmacist. It is important that you do not miss or skip doses of ZEPATIER during treatment.
|
What are the possible side effects of ZEPATIER? |
ZEPATIER can cause serious side effects, including:
Hepatitis B virus reactivation. See "What is the most important information I should know about ZEPATIER?"
Signs of liver problems. ZEPATIER may cause increases in your liver-related blood tests. This could be a sign of serious liver problems. Your healthcare provider will do blood tests to check your liver before and during treatment with ZEPATIER. Tell your healthcare provider right away if you get any of the following symptoms or if they get worse during treatment with ZEPATIER:
|
|
Common side effects of ZEPATIER when used without ribavirin include:
|
|
Common side effects of ZEPATIER when used with ribavirin include:
|
|
If you have any side effect that bothers you or that does not go away, tell your healthcare provider.
There may be other side effects to ZEPATIER that are not listed.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. For more information or medical advice call your doctor.
| Where should I keep ZEPATIER? |
- Keep ZEPATIER in its original packaging (blister package) until you are ready to take it. Do not take the tablets out of the original blister package to store in another container such as a pill box. This is important because the tablets are sensitive to moisture. The package is designed to protect them.
- Keep ZEPATIER at room temperature.
- Keep ZEPATIER and all medicines out of the reach of children.
| General information about ZEPATIER |
- Medicines are sometimes prescribed for purposes other than those listed in the patient information. Do not use ZEPATIER for a condition for which it was not prescribed. Do not give ZEPATIER to other people, even if they have the same condition. It may harm them.
- If you would like more information, talk with your healthcare provider or pharmacist. You can ask them for information about ZEPATIER that was written for health professionals.
- For more information, call the company that makes ZEPATIER at 1-877-888-4231 or go to www.ZEPATIER.com.
| What are the ingredients in ZEPATIER? |
The active ingredients are: elbasvir and grazoprevir.
The inactive ingredients are: colloidal silicon dioxide, copovidone, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, mannitol, microcrystalline cellulose, sodium chloride, sodium lauryl sulfate, and vitamin E polyethylene glycol succinate.
The tablets are film-coated with a coating material containing the following inactive ingredients: carnauba wax, ferrosoferric oxide, hypromellose, iron oxide red, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Section 43683-2
Section 44425-7
Store ZEPATIER in the original blister package until use to protect from moisture.
Store ZEPATIER at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (between 59°F to 86°F) [see USP Controlled Room Temperature].
8.7 Race
Higher elbasvir and grazoprevir plasma concentrations were observed in Asians compared to Caucasians. Asians experienced a higher rate of late ALT elevations in clinical trials [see Warnings and Precautions (5.2)]. However, no dose adjustment of ZEPATIER is recommended based on race/ethnicity [see Clinical Pharmacology (12.3)].
8.6 Gender
Higher elbasvir and grazoprevir plasma concentrations were observed in females compared to males. Females experienced a higher rate of late ALT elevations in clinical trials [see Warnings and Precautions (5.2)]. However, no dose adjustment of ZEPATIER is recommended based on gender [see Clinical Pharmacology (12.3)].
10 Overdosage
Human experience of overdose with ZEPATIER is limited. No specific antidote is available for overdose with ZEPATIER. In case of overdose, it is recommended that the patient be monitored for any signs or symptoms of adverse reactions and appropriate symptomatic treatment instituted.
Hemodialysis does not remove elbasvir or grazoprevir since elbasvir and grazoprevir are highly bound to plasma protein [see Clinical Pharmacology (12.3)].
11 Description
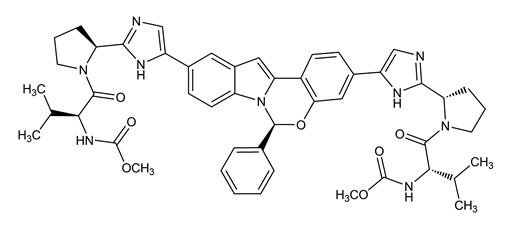
ZEPATIER is a fixed-dose combination tablet containing elbasvir and grazoprevir for oral administration.
Elbasvir is an HCV NS5A inhibitor, and grazoprevir is an HCV NS3/4A protease inhibitor.
Each tablet contains 50 mg elbasvir and 100 mg grazoprevir. The tablets include the following inactive ingredients: colloidal silicon dioxide, copovidone, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, mannitol, microcrystalline cellulose, sodium chloride, sodium lauryl sulfate, and vitamin E polyethylene glycol succinate. The tablets are film-coated with a coating material containing the following inactive ingredients: carnauba wax, ferrosoferric oxide, hypromellose, iron oxide red, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin.
8.4 Pediatric Use
The safety, efficacy, and pharmacokinetics of ZEPATIER was evaluated in an open-label clinical trial (MK-5172-079), which included 22 subjects (n=21, genotype 1; n=1, genotype 4) 12 years of age and older who received ZEPATIER for 12 weeks. The safety, pharmacokinetics, and efficacy observed in this trial were comparable to those observed in adults [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.6)].
Safety and effectiveness of ZEPATIER have not been established in pediatric patients younger than 12 years of age who weigh less than 30 kg.
8.5 Geriatric Use
Clinical trials of ZEPATIER with or without ribavirin included 187 subjects aged 65 years and over. Higher elbasvir and grazoprevir plasma concentrations were observed in subjects aged 65 years and over. A higher rate of late ALT elevations was observed in subjects aged 65 years and over in clinical trials [see Warnings and Precautions (5.2)]. However, no dosage adjustment of ZEPATIER is recommended in geriatric patients [see Clinical Pharmacology (12.3)].
4 Contraindications
- ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) due to the expected significantly increased grazoprevir plasma concentration and the increased risk of alanine aminotransferase (ALT) elevations [see Warnings and Precautions (5.2), Use in Specific Populations (8.9), and Clinical Pharmacology (12.3)].
- ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) or those with any history of hepatic decompensation due to the risk of hepatic decompensation [see Warnings and Precautions (5.3), Use in Specific Populations (8.9)].
- ZEPATIER is contraindicated with inhibitors of organic anion transporting polypeptides 1B1/3 (OATP1B1/3) that are known or expected to significantly increase grazoprevir plasma concentrations, strong inducers of cytochrome P450 3A (CYP3A), and efavirenz [see Warnings and Precautions (5.5), Drug Interactions (7), and Clinical Pharmacology (12.3)].
- If ZEPATIER is administered with ribavirin, the contraindications to ribavirin also apply to this combination regimen. Refer to the ribavirin prescribing information for a list of contraindications for ribavirin.
Table 2 lists drugs that are contraindicated with ZEPATIER.
| Drug Class | Drug(s) within Class that are Contraindicated | Clinical Comment This table is not a comprehensive list of all drugs that strongly induce CYP3A. This table may not include all OATP1B1/3 inhibitors that significantly increase grazoprevir plasma concentrations.
|
|---|---|---|
| Anticonvulsants | Phenytoin Carbamazepine |
May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. |
| Antimycobacterials | Rifampin | May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. |
| Herbal Products | St. John's Wort (Hypericum perforatum) | May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. |
| HIV Medications | Efavirenz Efavirenz is included as a strong CYP3A inducer in this table, since co-administration reduced grazoprevir exposure by ≥80% [see Table 9].
|
May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by CYP3A induction. |
| HIV Medications | Atazanavir Darunavir Lopinavir Saquinavir Tipranavir |
May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition. |
| Immunosuppressants | Cyclosporine | May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition. |
6 Adverse Reactions
The following adverse reaction is described below and elsewhere in the labeling:
- Increased Risk of ALT Elevations [see Warnings and Precautions (5.2)].
7 Drug Interactions
- Co-administration of ZEPATIER with moderate CYP3A inducers is not recommended as they may decrease the plasma concentration of ZEPATIER. (7)
- Co-administration of ZEPATIER with certain strong CYP3A inhibitors is not recommended as they may increase the plasma concentration of ZEPATIER. (7)
- Clearance of HCV infection with direct-acting antivirals may lead to changes in hepatic function, which may impact safe and effective use of concomitant medications. Frequent monitoring of relevant laboratory parameters (INR or blood glucose) and dose adjustments of certain concomitant medications may be necessary. (7.2)
- Consult the full prescribing information prior to and during treatment for potential drug interactions. (4, 5.5, 7, 12.3)
2.3 Renal Impairment
No dosage adjustment of ZEPATIER is recommended in patients with any degree of renal impairment including patients on hemodialysis. Administer ZEPATIER with or without ribavirin according to the recommendations in Table 1 [see Use in Specific Populations (8.8) and Clinical Studies (14.4)]. Refer to the ribavirin tablet prescribing information for the correct ribavirin dosage for patients with CrCl less than or equal to 50 mL per minute.
8.8 Renal Impairment
No dosage adjustment of ZEPATIER is recommended in patients with any degree of renal impairment including patients receiving hemodialysis [see Clinical Pharmacology (12.3)]. Administer ZEPATIER with or without ribavirin according to recommendations in Table 1 [see Dosage and Administration (2.2, 2.3)]. Refer to the prescribing information for ribavirin tablets for renal dosage adjustment of ribavirin in patients with CrCl less than or equal to 50 mL per minute.
12.3 Pharmacokinetics
The pharmacokinetic properties of elbasvir and grazoprevir have been evaluated in non-HCV-infected adult subjects and in HCV-infected adult subjects. Elbasvir pharmacokinetics were similar in healthy subjects and HCV-infected subjects and were approximately dose-proportional over the range of 5-100 mg once daily. Grazoprevir oral exposures are approximately 2-fold greater in HCV-infected subjects as compared to healthy subjects. Grazoprevir pharmacokinetics increased in a greater than dose-proportional manner over the range of 10-800 mg once daily in HCV-infected subjects. Ribavirin co-administration with ZEPATIER had no clinically relevant impact on plasma AUC and Cmax of elbasvir and grazoprevir compared to administration of ZEPATIER alone. The geometric mean steady-state pharmacokinetic parameter values for elbasvir and grazoprevir in non-cirrhotic HCV-infected subjects are provided in Table 7. Following once daily administration of ZEPATIER to HCV-infected subjects, elbasvir and grazoprevir reached steady state within approximately 6 days.
| Geometric Mean (90% Confidence Interval) | |||
|---|---|---|---|
| AUC0-24 (ng•hr/mL) | Cmax (ng/mL) | C24 (ng/mL) | |
| Elbasvir | 1920 (1880, 1960) | 121 (118, 123) | 48.4 (47.3, 49.6) |
| Grazoprevir | 1420 (1400, 1530) | 165 (161, 176) | 18.0 (17.8, 19.9) |
2.4 Hepatic Impairment
No dosage adjustment of ZEPATIER is recommended in patients with mild hepatic impairment (Child-Pugh A). ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) or those with any history of prior hepatic decompensation [see Contraindications (4), Warnings and Precautions (5.3), Use in Specific Populations (8.9), and Clinical Pharmacology (12.3)].
8.9 Hepatic Impairment
No dosage adjustment of ZEPATIER is recommended in patients with mild hepatic impairment (Child-Pugh A). ZEPATIER is contraindicated in patients with moderate hepatic impairment (Child-Pugh B) due to the lack of clinical safety and efficacy experience in HCV-infected Child-Pugh B patients, and in patients with severe hepatic impairment (Child-Pugh C) due to a 12-fold increase in grazoprevir exposure in non-HCV infected Child-Pugh C subjects. In addition, postmarketing cases of hepatic decompensation/failure have been reported in patients with advanced liver disease [see Dosage and Administration (2.4), Contraindications (4), Warnings and Precautions (5.3), and Clinical Pharmacology (12.3)].
The safety and efficacy of ZEPATIER have not been established in patients awaiting liver transplant or in liver transplant recipients.
1 Indications and Usage
ZEPATIER® is indicated for the treatment of chronic hepatitis C virus (HCV) genotype 1 or 4 infection in adult and pediatric patients 12 years of age and older or weighing at least 30 kg.
ZEPATIER is indicated for use with ribavirin in certain patient populations [see Dosage and Administration (2.2)].
12.1 Mechanism of Action
ZEPATIER is a fixed-dose combination of elbasvir and grazoprevir which are direct-acting antiviral agents against the hepatitis C virus [see Microbiology (12.4)].
5 Warnings and Precautions
- Risk of Hepatitis B Virus Reactivation: Test all patients for evidence of current or prior HBV infection before initiation of HCV treatment. Monitor HCV/HBV coinfected patients for HBV reactivation and hepatitis flare during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated. (5.1)
- ALT Elevations: Perform hepatic laboratory testing prior to therapy, at treatment week 8, and as clinically indicated. For patients receiving 16 weeks of therapy, perform additional hepatic laboratory testing at treatment week 12. For ALT elevations on ZEPATIER, follow recommendations in full prescribing information. (5.2)
- Risk of Hepatic Decompensation/Failure in Patients with Evidence of Advanced Liver Disease: Hepatic decompensation/failure, including fatal outcomes, have been reported mostly in patients with cirrhosis and baseline moderate or severe liver impairment (Child-Pugh B or C) treated with HCV NS3/4A protease inhibitor-containing regimens. Monitor for clinical and laboratory evidence of hepatic decompensation. Discontinue ZEPATIER in patients who develop evidence of hepatic decompensation/failure. (5.3)
- Risk Associated with Ribavirin Combination Treatment: If ZEPATIER is administered with ribavirin, the warnings and precautions for ribavirin also apply. (5.4)
2 Dosage and Administration
- Testing Prior to Initiation of Therapy:
- Recommended dosage in adult and pediatric patients 12 years of age and older or weighing at least 30 kg: One tablet taken orally once daily with or without food. (2.2)
| Patient Population | Treatment | Duration |
|---|---|---|
| Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced Peginterferon alfa + ribavirin. without baseline NS5A polymorphismsPolymorphisms at amino acid positions 28, 30, 31, or 93.
|
ZEPATIER | 12 weeks |
| Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced with baseline NS5A polymorphisms |
ZEPATIER + ribavirin | 16 weeks |
| Genotype 1b: Treatment-naïve or PegIFN/RBV-experienced |
ZEPATIER | 12 weeks |
| Genotype 1a or 1b: PegIFN/RBV/PI-experienced Peginterferon alfa + ribavirin + HCV NS3/4A protease inhibitor.
|
ZEPATIER + ribavirin | 12 weeks |
| Genotype 4: Treatment-naïve |
ZEPATIER | 12 weeks |
| Genotype 4: PegIFN/RBV-experienced |
ZEPATIER + ribavirin | 16 weeks |
3 Dosage Forms and Strengths
ZEPATIER is available as a beige-colored, oval-shaped, film-coated tablet debossed with "770" on one side and plain on the other. Each tablet contains 50 mg elbasvir and 100 mg grazoprevir.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of ZEPATIER. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
If ZEPATIER is administered with ribavirin, refer to the prescribing information for ribavirin for a description of ribavirin-associated adverse reactions.
Clinical Trials in Adult Subjects
The safety of ZEPATIER in adult subjects was assessed based on 2 placebo-controlled trials and 7 uncontrolled Phase 2 and 3 clinical trials in approximately 1700 subjects with chronic hepatitis C virus infection with compensated liver disease (with or without cirrhosis) [see Clinical Studies (14)].
14.1 Overview of Clinical Trials
The efficacy of ZEPATIER was assessed in 2 placebo-controlled trials and 4 uncontrolled Phase 2 and 3 clinical trials in 1401 subjects with genotype (GT) 1, 4, or 6 and in 1 clinical trial in 22 pediatric subjects with GT 1 or 4 chronic hepatitis C virus infection with compensated liver disease (with or without cirrhosis). An overview of the 6 trials (n=1373) contributing to the assessment of efficacy in genotype 1 or 4 is provided in Table 13. C-EDGE TN, C-EDGE COINFECTION, C-SCAPE, and C-EDGE TE also included subjects with genotype 6 HCV infection (n=28). Because ZEPATIER is not indicated for genotype 6 infection, results in patients with genotype 6 infection are not included in Clinical Studies (14).
| Trial | Population | Study Groups and Duration (Number of Subjects Treated) |
|---|---|---|
| GT = Genotype | ||
| TN = Treatment-Naïve | ||
| TE = Treatment-Experienced (failed prior treatment with interferon [IFN] or peginterferon alfa [PegIFN] with or without ribavirin [RBV] or were intolerant to prior therapy). | ||
| C-EDGE TN (double-blind) |
GT 1, 4 TN with or without cirrhosis |
|
| C-EDGE COINFECTION (open-label) |
GT 1, 4 TN with or without cirrhosis HCV/HIV-1 co-infection |
|
| C-SURFER (double-blind) |
GT 1 TN or TE with or without cirrhosis Severe Renal Impairment including Hemodialysis |
|
| C-SCAPE (open-label) |
GT 4 TN without cirrhosis |
|
| C-EDGE TE (open-label) |
GT 1, 4 TE with or without cirrhosis with or without HCV/HIV-1 co-infection |
|
| C-SALVAGE (open-label) |
GT 1 TE with HCV protease inhibitor regimen Failed prior treatment with boceprevir, telaprevir, or simeprevir in combination with PegIFN + RBV. with or without cirrhosis |
|
| MK-5172-079 (open-label) |
GT 1, 4 TN or TE pediatric subjects |
|
ZEPATIER was administered once daily by mouth in these trials. For subjects who received ribavirin (RBV), the RBV dosage was weight-based (less than 66 kg = 800 mg per day, 66 to 80 kg = 1000 mg per day, 81 to 105 kg = 1200 mg per day, greater than 105 kg = 1400 mg per day) administered by mouth in two divided doses with food.
Sustained virologic response (SVR) was the primary endpoint in all trials and was defined as HCV RNA less than lower limit of quantification (LLOQ) at 12 weeks after the cessation of treatment (SVR12). Serum HCV RNA values were measured during these clinical trials using the COBAS AmpliPrep/COBAS Taqman HCV test (version 2.0) with an LLOQ of 15 HCV RNA IU per mL, with the exception of C-SCAPE where the assay had an LLOQ of 25 HCV RNA IU per mL.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
For patients receiving ZEPATIER with ribavirin, advise patients to read the FDA-approved patient labeling (Medication Guide) for ribavirin [see Warnings and Precautions (5.4)].
7.1 Potential for Drug Interactions
Grazoprevir is a substrate of OATP1B1/3 transporters. Co-administration of ZEPATIER with OATP1B1/3 inhibitors that are known or expected to significantly increase grazoprevir plasma concentrations is contraindicated [see Contraindications (4), Clinical Pharmacology (12.3), and Table 2].
Elbasvir and grazoprevir are substrates of CYP3A and P-gp, but the role of intestinal P-gp in the absorption of elbasvir and grazoprevir appears to be minimal. Co-administration of moderate or strong inducers of CYP3A with ZEPATIER may decrease elbasvir and grazoprevir plasma concentrations, leading to reduced therapeutic effect of ZEPATIER. Co-administration of ZEPATIER with strong CYP3A inducers or efavirenz is contraindicated [see Contraindications (4), Clinical Pharmacology (12.3), and Table 2]. Co-administration of ZEPATIER with moderate CYP3A inducers is not recommended [see Warnings and Precautions (5.5), Clinical Pharmacology (12.3), and Table 6]. Co-administration of ZEPATIER with strong CYP3A inhibitors may increase elbasvir and grazoprevir concentrations. Co-administration of ZEPATIER with certain strong CYP3A inhibitors is not recommended [see Warnings and Precautions (5.5), Clinical Pharmacology (12.3), and Table 6].
16 How Supplied/storage and Handling
Each ZEPATIER tablet contains 50 mg elbasvir and 100 mg grazoprevir, is beige, oval-shaped, film-coated, debossed with "770" on one side and plain on the other. The tablets are packaged into a carton (NDC 0006-3074-02) containing two (2) 14-count child-resistant dose packs for a total of 28 tablets.
5.2 Increased Risk of Alt Elevations
During clinical trials with ZEPATIER with or without ribavirin, 1% of subjects experienced elevations of ALT from normal levels to greater than 5 times the upper limit of normal (ULN), generally at or after treatment week 8. ALT elevations were typically asymptomatic and most resolved with ongoing or completion of therapy. Higher rates of late ALT elevations occurred in the following subpopulations: female sex (2% [10/608]), Asian race (2% [4/164]), and age 65 years or older (2% [3/177]) [see Adverse Reactions (6.1)].
Hepatic laboratory testing should be performed prior to therapy, at treatment week 8, and as clinically indicated. For patients receiving 16 weeks of therapy, additional hepatic laboratory testing should be performed at treatment week 12.
- Patients should be instructed to consult their healthcare professional without delay if they have onset of fatigue, weakness, lack of appetite, nausea and vomiting, jaundice, or discolored feces.
- Consider discontinuing ZEPATIER if ALT levels remain persistently greater than 10 times the ULN.
- Discontinue ZEPATIER if ALT elevation is accompanied by signs or symptoms of liver inflammation or increasing conjugated bilirubin, alkaline phosphatase, or International Normalized Ratio (INR).
14.5 Clinical Trials With Genotype 4 Hcv
The efficacy of ZEPATIER in subjects with genotype 4 chronic HCV infection was demonstrated in C-EDGE TN, C-EDGE COINFECTION, C-EDGE TE, and C-SCAPE. C-SCAPE was a randomized, open-label trial which included treatment-naïve subjects with genotype 4 infection without cirrhosis. Subjects were randomized in a 1:1 ratio to EBR 50 mg once daily + GZR 100 mg once daily for 12 weeks or EBR 50 mg once daily + GZR 100 mg once daily + RBV for 12 weeks. In these combined studies in subjects with genotype 4 infection, 64% were treatment-naïve; 66% of the subjects were male; 87% were White; 10% were Black or African American; 22% had cirrhosis; and 30% had HCV/HIV-1 co-infection.
In C-SCAPE, C-EDGE TN, and C-EDGE COINFECTION trials combined, a total of 66 genotype 4 treatment-naïve subjects received ZEPATIER or EBR + GZR for 12 weeks. In these combined trials, SVR12 among subjects treated with ZEPATIER or EBR + GZR for 12 weeks was 97% (64/66).
In C-EDGE TE, a total of 37 genotype 4 treatment-experienced subjects received a 12- or 16-week ZEPATIER with or without RBV regimen. SVR12 among randomized subjects treated with ZEPATIER + RBV for 16 weeks was 100% (8/8).
8.3 Females and Males of Reproductive Potential
If ZEPATIER is administered with ribavirin, the information for ribavirin with regard to pregnancy testing, contraception, and infertility also applies to this combination regimen. Refer to ribavirin prescribing information for additional information.
5.4 Risks Associated With Ribavirin Combination Treatment
If ZEPATIER is administered with ribavirin, the warnings and precautions for ribavirin, including the pregnancy avoidance warning, also apply to this combination regimen. Refer to the ribavirin prescribing information for a full list of warnings and precautions for ribavirin [see Dosage and Administration (2.2)].
Principal Display Panel 50 Mg/100 Mg Tablet Dose Pack Carton
NDC 0006-3074-02
Zepatier
®
(elbasvir and grazoprevir) tablets
50 mg/100 mg
Rx only
28 Tablets
This carton contains a total of 28 tablets
packaged within 2 dose packs.
Each dose pack contains 14 blister units
with one tablet per blister unit.
7.2 Established and Other Potentially Significant Drug Interactions
If dose adjustments of concomitant medications are made due to treatment with ZEPATIER, doses should be readjusted after administration of ZEPATIER is completed.
Clearance of HCV infection with direct-acting antivirals may lead to changes in hepatic function, which may impact the safe and effective use of concomitant medications. For example, altered blood glucose control resulting in serious symptomatic hypoglycemia has been reported in diabetic patients in postmarketing case reports and published epidemiological studies. Management of hypoglycemia in these cases required either discontinuation or dose modification of concomitant medications used for diabetes treatment.
Frequent monitoring of relevant laboratory parameters (e.g., International Normalized Ratio [INR] in patients taking warfarin, blood glucose levels in diabetic patients) or drug concentrations of concomitant medications such as CYP450 substrates with a narrow therapeutic index (e.g., certain immunosuppressants) is recommended to ensure safe and effective use. Dose adjustments of concomitant medications may be necessary.
Table 6 provides a listing of established or potentially clinically significant drug interactions. The drug interactions described are based on studies conducted with either ZEPATIER, the components of ZEPATIER (elbasvir [EBR] and grazoprevir [GZR]) as individual agents, or are predicted drug interactions that may occur with ZEPATIER [see Contraindications (4), Warnings and Precautions (5.5), and Clinical Pharmacology (12.3)].
| Concomitant Drug Class: Drug Name | Effect on Concentration ↓ = decrease, ↑ = increase
|
Clinical Comment |
|---|---|---|
|
Antibiotics: Nafcillin |
↓ EBR ↓ GZR |
Co-administration of ZEPATIER with nafcillin may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
|
Antifungals:
oral Ketoconazole These interactions have been studied in healthy adults.
|
↑ EBR ↑ GZR |
Co-administration of oral ketoconazole is not recommended. |
|
Endothelin Antagonists:
Bosentan |
↓ EBR ↓ GZR |
Co-administration of ZEPATIER with bosentan may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
|
Immunosuppressants:
Tacrolimus |
↑ tacrolimus | Frequent monitoring of tacrolimus whole blood concentrations, changes in renal function, and tacrolimus-associated adverse events upon the initiation of co-administration is recommended. |
| HIV Medications: | ||
| Etravirine | ↓ EBR ↓ GZR |
Co-administration of ZEPATIER with etravirine may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
| Elvitegravir/ cobicistat/ emtricitabine/ tenofovir (disoproxil fumarate or alafenamide) | ↑ EBR ↑ GZR |
Co-administration of cobicistat-containing regimens is not recommended. |
|
HMG-CoA Reductase Inhibitors
See Drug Interactions (7.3)
for a list of HMG Co-A reductase inhibitors without clinically relevant interactions with ZEPATIER. :
|
||
| Atorvastatin | ↑ atorvastatin | The dose of atorvastatin should not exceed a daily dose of 20 mg when co-administered with ZEPATIER. |
| Rosuvastatin | ↑ rosuvastatin | The dose of rosuvastatin should not exceed a daily dose of 10 mg when co-administered with ZEPATIER. |
| Fluvastatin Lovastatin Simvastatin |
↑ fluvastatin ↑ lovastatin ↑ simvastatin |
Statin-associated adverse events such as myopathy should be closely monitored. The lowest necessary dose should be used when co-administered with ZEPATIER. |
|
Wakefulness-Promoting Agents:
Modafinil |
↓ EBR ↓ GZR |
Co-administration of ZEPATIER with modafinil may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
7.3 Drugs Without Clinically Significant Interactions With Zepatier
The interaction between the components of ZEPATIER (elbasvir or grazoprevir) or ZEPATIER and the following drugs were evaluated in clinical studies, and no dose adjustments are needed when ZEPATIER is used with the following drugs individually: acid reducing agents (proton pump inhibitors, H2 blockers, antacids), buprenorphine/naloxone, digoxin, dolutegravir, methadone, mycophenolate mofetil, oral contraceptive pills, phosphate binders, pitavastatin, pravastatin, prednisone, raltegravir, ribavirin, rilpivirine, tenofovir disoproxil fumarate, and sofosbuvir [see Clinical Pharmacology (12.3)].
No clinically relevant drug-drug interaction is expected when ZEPATIER is co-administered with abacavir, emtricitabine, entecavir, and lamivudine.
5.1 Risk of Hepatitis B Virus Reactivation in Patients Coinfected With Hcv and Hbv
Hepatitis B virus (HBV) reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals, and who were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure and death. Cases have been reported in patients who are HBsAg positive and also in patients with serologic evidence of resolved HBV infection (i.e., HBsAg negative and anti-HBc positive). HBV reactivation has also been reported in patients receiving certain immunosuppressant or chemotherapeutic agents; the risk of HBV reactivation associated with treatment with HCV direct-acting antivirals may be increased in these patients.
HBV reactivation is characterized as an abrupt increase in HBV replication manifesting as a rapid increase in serum HBV DNA level. In patients with resolved HBV infection reappearance of HBsAg can occur. Reactivation of HBV replication may be accompanied by hepatitis, i.e., increases in aminotransferase levels and, in severe cases, increases in bilirubin levels, liver failure, and death can occur.
Test all patients for evidence of current or prior HBV infection by measuring HBsAg and anti-HBc before initiating HCV treatment with ZEPATIER. In patients with serologic evidence of HBV infection, monitor for clinical and laboratory signs of hepatitis flare or HBV reactivation during HCV treatment with ZEPATIER and during post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated.
5.5 Risk of Adverse Reactions Or Reduced Therapeutic Effect Due to Drug Interactions
The concomitant use of ZEPATIER and certain drugs may result in known or potentially significant drug interactions, some of which may lead to:
- Possible clinically significant adverse reactions from greater exposure of concomitant drugs or components of ZEPATIER.
- Significant decrease of elbasvir and grazoprevir plasma concentrations which may lead to reduced therapeutic effect of ZEPATIER and possible development of resistance.
See Tables 2 and 6 for steps to prevent or manage these known or potentially significant drug interactions, including dosing recommendations [see Contraindications (4) and Drug Interactions (7.2)].
Warning: Risk of Hepatitis B Virus Reactivation in Patients Coinfected With Hcv and Hbv
Test all patients for evidence of current or prior hepatitis B virus (HBV) infection before initiating treatment with ZEPATIER. HBV reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals and were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure, and death. Monitor HCV/HBV coinfected patients for hepatitis flare or HBV reactivation during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated [see Warnings and Precautions (5.1)].
14.6 Clinical Trial in Pediatric Subjects With Genotype 1 Or 4 Chronic Hepatitis C Infection
The efficacy of ZEPATIER was evaluated in an open-label study (MK-5172-079, NCT03379506) that evaluated pediatric subjects 12 years to less than 18 years of age who received ZEPATIER for 12 weeks. HCV GT1a infected subjects with one or more baseline NS5A RAS were excluded from study participation. In the MK-5172-079 study, 22 treatment-naïve or treatment-experienced subjects 12 years to less than 18 years of age with genotype 1 (n=21) or 4 CHC (n=1), without cirrhosis, were treated with ZEPATIER for 12 weeks. The median age was 13.5 years (range: 12 to 17); 50% were female; 95% were White; the weight range was 28.1 kg to 96.5 kg; 95.5% had genotype 1 and 4.5% had genotype 4; 64% were treatment-naïve, 36% were treatment-experienced; 46% had baseline HCV RNA levels greater than 800,000 IU/mL. The overall SVR12 rate was 100% (22/22).
5.3 Risk of Hepatic Decompensation/failure in Patients With Evidence of Advanced Liver Disease
Postmarketing cases of hepatic decompensation/failure, including those with fatal outcomes, have been reported in patients treated with HCV NS3/4A protease inhibitor-containing regimens, including ZEPATIER.
Reported cases occurred in patients treated with HCV NS3/4A protease inhibitor-containing regimens with baseline cirrhosis with and without moderate or severe liver impairment (Child-Pugh B or C) as well as some patients without cirrhosis. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatic laboratory testing should be performed in all patients [see Warnings and Precautions (5.2)]. In patients with compensated cirrhosis (Child-Pugh A) or evidence of advanced liver disease, such as portal hypertension, more frequent hepatic laboratory testing may be warranted; and patients should be monitored for signs and symptoms of hepatic decompensation such as the presence of jaundice, ascites, hepatic encephalopathy, and variceal hemorrhage. Discontinue ZEPATIER in patients who develop evidence of hepatic decompensation/failure.
ZEPATIER is contraindicated in patients with moderate to severe hepatic impairment (Child-Pugh B or C) or those with any history of prior hepatic decompensation [see Contraindications (4), Adverse Reactions (6.1), Use in Specific Populations (8.9), and Clinical Pharmacology (12.3)].
14.2 Clinical Trials in Treatment Naïve Subjects With Genotype 1 Hcv (c Edge Tn and C Edge Coinfection)
The efficacy of ZEPATIER in treatment-naïve subjects with genotype 1 chronic hepatitis C virus infection with or without cirrhosis was demonstrated in the C-EDGE TN and C-EDGE COINFECTION trials.
C-EDGE TN was a randomized, double-blind, placebo-controlled trial in treatment-naïve subjects with genotype 1 or 4 infection with or without cirrhosis. Subjects were randomized in a 3:1 ratio to: ZEPATIER for 12 weeks (immediate treatment group) or placebo for 12 weeks followed by open-label treatment with ZEPATIER for 12 weeks (deferred treatment group). Among subjects with genotype 1 infection randomized to the immediate treatment group, the median age was 55 years (range: 20 to 78); 56% of the subjects were male; 61% were White; 20% were Black or African American; 8% were Hispanic or Latino; mean body mass index was 26 kg/m2; 72% had baseline HCV RNA levels greater than 800,000 IU per mL; 24% had cirrhosis; 67% had non-C/C IL28B alleles (CT or TT); and 55% had genotype 1a and 45% had genotype 1b chronic HCV infection.
C-EDGE COINFECTION was an open-label, single-arm trial in treatment-naïve HCV/HIV-1 co-infected subjects with genotype 1 or 4 infection with or without cirrhosis. Subjects received ZEPATIER for 12 weeks. Among subjects with genotype 1 infection, the median age was 50 years (range: 21 to 71); 85% of the subjects were male; 75% were White; 19% were Black or African American; 6% were Hispanic or Latino; mean body mass index was 25 kg/m2; 59% had baseline HCV RNA levels greater than 800,000 IU per mL; 16% had cirrhosis; 65% had non-C/C IL28B alleles (CT or TT); and 76% had genotype 1a, 23% had genotype 1b, and 1% had genotype 1-Other chronic HCV infection.
Table 14 presents treatment outcomes for ZEPATIER in treatment-naïve subjects with genotype 1 infection from C-EDGE TN (immediate treatment group) and C-EDGE COINFECTION. For treatment outcomes for ZEPATIER in genotype 4 infection, [see Clinical Studies (14.5)].
| Trial | C-EDGE TN (Immediate Treatment Group) |
C-EDGE COINFECTION (HCV/HIV-1 Co-Infection) |
|---|---|---|
| Regimen | ZEPATIER 12 Weeks N=288 |
ZEPATIER 12 Weeks N=189 |
| SVR in Genotype 1 | 95% (273/288) | 95% (179/189) |
| Outcome for subjects without SVR | ||
| On-treatment Virologic Failure Includes subjects with virologic breakthrough.
|
<1% (1/288) | 0% (0/189) |
| Relapse | 3% (10/288) | 3% (6/189) |
| Other Other includes subjects who discontinued due to adverse event, lost to follow-up, or subject withdrawal.
|
1% (4/288) | 2% (4/189) |
| SVR by Genotype 1 Subtypes | ||
| GT 1a For the impact of baseline NS5A polymorphisms on SVR12, [see Microbiology (12.4), Table 12].
|
92% (144/157) | 94% (136/144) |
| GT 1b Includes genotype 1 subtypes other than 1a or 1b.
|
98% (129/131) | 96% (43/45) |
| SVR by Cirrhosis status | ||
| Non-cirrhotic | 94% (207/220) | 94% (148/158) |
| Cirrhotic | 97% (66/68) | 100% (31/31) |
2.2 Recommended Dosage in Adult and Pediatric Patients 12 Years of Age and Older Or Weighing At Least 30 Kg
ZEPATIER is a two-drug, fixed-dose combination product containing 50 mg of elbasvir and 100 mg of grazoprevir in a single tablet. The recommended dosage of ZEPATIER is one tablet taken orally once daily with or without food [see Clinical Pharmacology (12.3)]. ZEPATIER is used in combination with ribavirin in certain patient populations (see Table 1). When administered with ZEPATIER, the recommended dosage of ribavirin in patients without renal impairment is weight-based administered in two divided doses with food. For further information on ribavirin dosing and dosage modifications, refer to the ribavirin prescribing information.
14.4 Clinical Trial in Subjects With Genotype 1 Hcv and Severe Renal Impairment Including Subjects On Hemodialysis (c Surfer)
C-SURFER was a randomized, double-blind, placebo-controlled trial in subjects with genotype 1 infection, with or without cirrhosis, with chronic kidney disease (CKD) Stage 4 (eGFR 15-29 mL/min/1.73 m2) or CKD Stage 5 (eGFR <15 mL/min/1.73 m2), including subjects on hemodialysis, who were treatment-naïve or who had failed prior therapy with IFN or PegIFN ± RBV therapy. Subjects were randomized in a 1:1 ratio to one of the following treatment groups: EBR 50 mg once daily + GZR 100 mg once daily for 12 weeks (immediate treatment group) or placebo for 12 weeks followed by open-label treatment with EBR + GZR for 12 weeks (deferred treatment group). In addition, 11 subjects received open-label EBR + GZR for 12 weeks (intensive pharmacokinetic [PK] group). Subjects randomized to the immediate treatment group and intensive PK group had a median age of 58 years (range: 31 to 76); 75% of the subjects were male; 50% were White; 45% were Black or African American; 11% were Hispanic or Latino; 57% had baseline HCV RNA levels greater than 800,000 IU/mL; 6% had cirrhosis; and 72% had non-C/C IL28B alleles (CT or TT).
Treatment outcomes in subjects treated with ZEPATIER for 12 weeks in the pooled immediate treatment group and intensive PK group are presented in Table 16.
| Regimen | EBR + GZR 12 weeks (Immediate Treatment Group) N=122 Includes subjects (n=11) in the intensive PK group.
|
|---|---|
| Overall SVR | 94% (115/122) SVR was achieved in 99% (115/116) of subjects in the pre-specified primary analysis population, which excluded subjects not receiving at least one dose of study treatment and those with missing data due to death or early study discontinuation for reasons unrelated to treatment response.
|
| Outcome for subjects without SVR | |
| On-treatment Virologic Failure | 0% (0/122) |
| Relapse | <1% (1/122) |
| Other Other includes subjects who discontinued due to adverse event, lost to follow-up, or subject withdrawal.
|
5% (6/122) |
| SVR by Genotype | |
| GT 1a | 97% (61/63) |
| GT 1b Includes genotype 1 subtypes other than 1a or 1b.
|
92% (54/59) |
| SVR by Cirrhosis status | |
| No | 95% (109/115) |
| Yes | 86% (6/7) |
| SVR by Prior HCV Treatment Status | |
| Treatment-naïve | 95% (96/101) |
| Treatment-experienced | 90% (19/21) |
| SVR by Dialysis Status | |
| No | 97% (29/30) |
| Yes | 93% (86/92) |
| SVR by Chronic Kidney Disease Stage | |
| Stage 4 | 100% (22/22) |
| Stage 5 | 93% (93/100) |
Structured Label Content
Section 34083-6 (34083-6)
Carcinogenesis and Mutagenesis
Elbasvir and grazoprevir were not genotoxic in a battery of in vitro or in vivo assays, including microbial mutagenesis, chromosomal aberration in Chinese Hamster Ovary cells, and in in vivo rat micronucleus assays.
Carcinogenicity studies with elbasvir or grazoprevir have not been conducted.
If ZEPATIER is administered in a regimen containing ribavirin, the information for ribavirin on carcinogenesis and mutagenesis also applies to this combination regimen. Refer to the ribavirin prescribing information for information on carcinogenesis and mutagenesis.
Section 42229-5 (42229-5)
Testing for HBV Infection
Test all patients for evidence of current or prior HBV infection by measuring hepatitis B surface antigen (HBsAg) and hepatitis B core antibody (anti-HBc) before initiating HCV treatment with ZEPATIER [see Warnings and Precautions (5.1)].
Section 42230-3 (42230-3)
|
Patient Information ZEPATIER® (ZEP-ah-teer) (elbasvir and grazoprevir) tablets |
What you need to know about ZEPATIER
|
|
What is the most important information I should know about ZEPATIER? |
ZEPATIER can cause serious side effects, including,
Hepatitis B virus reactivation: Before starting treatment with ZEPATIER, your healthcare provider will do blood tests to check for hepatitis B virus infection. If you have ever had hepatitis B virus infection, the hepatitis B virus could become active again during or after treatment of hepatitis C virus infection with ZEPATIER. Hepatitis B virus becoming active again (called reactivation) may cause serious liver problems including liver failure and death. Your healthcare provider will monitor you if you are at risk for hepatitis B virus reactivation during treatment and after you stop taking ZEPATIER.
For more information about side effects, see the section "What are the possible side effects of ZEPATIER?"
| What is ZEPATIER®? |
ZEPATIER is a prescription medicine used with or without ribavirin to treat chronic (long-lasting) hepatitis C virus (HCV) genotypes 1 or 4 infection in adults and children 12 years of age and older or weighing at least 66 pounds (30 kilograms).
ZEPATIER should not be used in children younger than 12 years of age who weigh less than 66 pounds (30 kilograms).
It is not known if ZEPATIER is safe or effective in people awaiting a liver transplant or people who have had a liver transplant.
| Who should not take ZEPATIER? |
Do not take ZEPATIER if you have certain liver problems.
| What should I tell my healthcare provider before taking ZEPATIER? |
Before taking ZEPATIER, tell your healthcare provider about all of your medical conditions, including if you:
- have ever had hepatitis B virus infection
- have liver problems other than hepatitis C
- have ever taken any medicine for hepatitis C
- have HIV
- have had or are waiting for a liver transplant
- are pregnant or trying to get pregnant. ZEPATIER has not been studied in pregnant women. We do not know if ZEPATIER will harm your baby while you are pregnant.
- Males and females who take ZEPATIER and ribavirin should also read the ribavirin Medication Guide for important pregnancy, contraception, and infertility information.
- are breastfeeding or plan to breastfeed. We do not know if ZEPATIER gets in your breast milk and gets passed to your baby.
- Talk to your healthcare provider about the best way to feed your baby during treatment with ZEPATIER.
| Are you taking other medicines? |
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. ZEPATIER may affect the way other medicines work, and other medicines may affect how ZEPATIER works. Some medicines cannot be taken with ZEPATIER. Your healthcare provider can tell you if it is safe to take ZEPATIER with other medicines.
- Know the medicines you take. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with this medicine.
- Do not start taking a new medicine without telling your healthcare provider.
| How do I take ZEPATIER? |
- Take 1 ZEPATIER tablet at the same time every day.
- ZEPATIER comes in a blister package of individually-packaged tablets. Keep the tablets in this package until you are ready to take your dose.
- Take ZEPATIER exactly as your healthcare provider tells you to take it.
- Take ZEPATIER with or without food.
- Do not stop taking ZEPATIER without first talking with your healthcare provider.
- If you take more than your prescribed dose, call your healthcare provider right away.
| What if I forget to take ZEPATIER? |
- Do not take two doses of ZEPATIER at the same time to make up for a missed dose.
- If you are not sure what to do, call your healthcare provider or pharmacist. It is important that you do not miss or skip doses of ZEPATIER during treatment.
|
What are the possible side effects of ZEPATIER? |
ZEPATIER can cause serious side effects, including:
Hepatitis B virus reactivation. See "What is the most important information I should know about ZEPATIER?"
Signs of liver problems. ZEPATIER may cause increases in your liver-related blood tests. This could be a sign of serious liver problems. Your healthcare provider will do blood tests to check your liver before and during treatment with ZEPATIER. Tell your healthcare provider right away if you get any of the following symptoms or if they get worse during treatment with ZEPATIER:
|
|
Common side effects of ZEPATIER when used without ribavirin include:
|
|
Common side effects of ZEPATIER when used with ribavirin include:
|
|
If you have any side effect that bothers you or that does not go away, tell your healthcare provider.
There may be other side effects to ZEPATIER that are not listed.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. For more information or medical advice call your doctor.
| Where should I keep ZEPATIER? |
- Keep ZEPATIER in its original packaging (blister package) until you are ready to take it. Do not take the tablets out of the original blister package to store in another container such as a pill box. This is important because the tablets are sensitive to moisture. The package is designed to protect them.
- Keep ZEPATIER at room temperature.
- Keep ZEPATIER and all medicines out of the reach of children.
| General information about ZEPATIER |
- Medicines are sometimes prescribed for purposes other than those listed in the patient information. Do not use ZEPATIER for a condition for which it was not prescribed. Do not give ZEPATIER to other people, even if they have the same condition. It may harm them.
- If you would like more information, talk with your healthcare provider or pharmacist. You can ask them for information about ZEPATIER that was written for health professionals.
- For more information, call the company that makes ZEPATIER at 1-877-888-4231 or go to www.ZEPATIER.com.
| What are the ingredients in ZEPATIER? |
The active ingredients are: elbasvir and grazoprevir.
The inactive ingredients are: colloidal silicon dioxide, copovidone, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, mannitol, microcrystalline cellulose, sodium chloride, sodium lauryl sulfate, and vitamin E polyethylene glycol succinate.
The tablets are film-coated with a coating material containing the following inactive ingredients: carnauba wax, ferrosoferric oxide, hypromellose, iron oxide red, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store ZEPATIER in the original blister package until use to protect from moisture.
Store ZEPATIER at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (between 59°F to 86°F) [see USP Controlled Room Temperature].
8.7 Race
Higher elbasvir and grazoprevir plasma concentrations were observed in Asians compared to Caucasians. Asians experienced a higher rate of late ALT elevations in clinical trials [see Warnings and Precautions (5.2)]. However, no dose adjustment of ZEPATIER is recommended based on race/ethnicity [see Clinical Pharmacology (12.3)].
8.6 Gender
Higher elbasvir and grazoprevir plasma concentrations were observed in females compared to males. Females experienced a higher rate of late ALT elevations in clinical trials [see Warnings and Precautions (5.2)]. However, no dose adjustment of ZEPATIER is recommended based on gender [see Clinical Pharmacology (12.3)].
10 Overdosage (10 OVERDOSAGE)
Human experience of overdose with ZEPATIER is limited. No specific antidote is available for overdose with ZEPATIER. In case of overdose, it is recommended that the patient be monitored for any signs or symptoms of adverse reactions and appropriate symptomatic treatment instituted.
Hemodialysis does not remove elbasvir or grazoprevir since elbasvir and grazoprevir are highly bound to plasma protein [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
ZEPATIER is a fixed-dose combination tablet containing elbasvir and grazoprevir for oral administration.
Elbasvir is an HCV NS5A inhibitor, and grazoprevir is an HCV NS3/4A protease inhibitor.
Each tablet contains 50 mg elbasvir and 100 mg grazoprevir. The tablets include the following inactive ingredients: colloidal silicon dioxide, copovidone, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, mannitol, microcrystalline cellulose, sodium chloride, sodium lauryl sulfate, and vitamin E polyethylene glycol succinate. The tablets are film-coated with a coating material containing the following inactive ingredients: carnauba wax, ferrosoferric oxide, hypromellose, iron oxide red, iron oxide yellow, lactose monohydrate, titanium dioxide, and triacetin.
8.4 Pediatric Use
The safety, efficacy, and pharmacokinetics of ZEPATIER was evaluated in an open-label clinical trial (MK-5172-079), which included 22 subjects (n=21, genotype 1; n=1, genotype 4) 12 years of age and older who received ZEPATIER for 12 weeks. The safety, pharmacokinetics, and efficacy observed in this trial were comparable to those observed in adults [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.6)].
Safety and effectiveness of ZEPATIER have not been established in pediatric patients younger than 12 years of age who weigh less than 30 kg.
8.5 Geriatric Use
Clinical trials of ZEPATIER with or without ribavirin included 187 subjects aged 65 years and over. Higher elbasvir and grazoprevir plasma concentrations were observed in subjects aged 65 years and over. A higher rate of late ALT elevations was observed in subjects aged 65 years and over in clinical trials [see Warnings and Precautions (5.2)]. However, no dosage adjustment of ZEPATIER is recommended in geriatric patients [see Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
- ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) due to the expected significantly increased grazoprevir plasma concentration and the increased risk of alanine aminotransferase (ALT) elevations [see Warnings and Precautions (5.2), Use in Specific Populations (8.9), and Clinical Pharmacology (12.3)].
- ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) or those with any history of hepatic decompensation due to the risk of hepatic decompensation [see Warnings and Precautions (5.3), Use in Specific Populations (8.9)].
- ZEPATIER is contraindicated with inhibitors of organic anion transporting polypeptides 1B1/3 (OATP1B1/3) that are known or expected to significantly increase grazoprevir plasma concentrations, strong inducers of cytochrome P450 3A (CYP3A), and efavirenz [see Warnings and Precautions (5.5), Drug Interactions (7), and Clinical Pharmacology (12.3)].
- If ZEPATIER is administered with ribavirin, the contraindications to ribavirin also apply to this combination regimen. Refer to the ribavirin prescribing information for a list of contraindications for ribavirin.
Table 2 lists drugs that are contraindicated with ZEPATIER.
| Drug Class | Drug(s) within Class that are Contraindicated | Clinical Comment This table is not a comprehensive list of all drugs that strongly induce CYP3A. This table may not include all OATP1B1/3 inhibitors that significantly increase grazoprevir plasma concentrations.
|
|---|---|---|
| Anticonvulsants | Phenytoin Carbamazepine |
May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. |
| Antimycobacterials | Rifampin | May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. |
| Herbal Products | St. John's Wort (Hypericum perforatum) | May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by strong CYP3A induction. |
| HIV Medications | Efavirenz Efavirenz is included as a strong CYP3A inducer in this table, since co-administration reduced grazoprevir exposure by ≥80% [see Table 9].
|
May lead to loss of virologic response to ZEPATIER due to significant decreases in elbasvir and grazoprevir plasma concentrations caused by CYP3A induction. |
| HIV Medications | Atazanavir Darunavir Lopinavir Saquinavir Tipranavir |
May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition. |
| Immunosuppressants | Cyclosporine | May increase the risk of ALT elevations due to a significant increase in grazoprevir plasma concentrations caused by OATP1B1/3 inhibition. |
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reaction is described below and elsewhere in the labeling:
- Increased Risk of ALT Elevations [see Warnings and Precautions (5.2)].
7 Drug Interactions (7 DRUG INTERACTIONS)
- Co-administration of ZEPATIER with moderate CYP3A inducers is not recommended as they may decrease the plasma concentration of ZEPATIER. (7)
- Co-administration of ZEPATIER with certain strong CYP3A inhibitors is not recommended as they may increase the plasma concentration of ZEPATIER. (7)
- Clearance of HCV infection with direct-acting antivirals may lead to changes in hepatic function, which may impact safe and effective use of concomitant medications. Frequent monitoring of relevant laboratory parameters (INR or blood glucose) and dose adjustments of certain concomitant medications may be necessary. (7.2)
- Consult the full prescribing information prior to and during treatment for potential drug interactions. (4, 5.5, 7, 12.3)
2.3 Renal Impairment
No dosage adjustment of ZEPATIER is recommended in patients with any degree of renal impairment including patients on hemodialysis. Administer ZEPATIER with or without ribavirin according to the recommendations in Table 1 [see Use in Specific Populations (8.8) and Clinical Studies (14.4)]. Refer to the ribavirin tablet prescribing information for the correct ribavirin dosage for patients with CrCl less than or equal to 50 mL per minute.
8.8 Renal Impairment
No dosage adjustment of ZEPATIER is recommended in patients with any degree of renal impairment including patients receiving hemodialysis [see Clinical Pharmacology (12.3)]. Administer ZEPATIER with or without ribavirin according to recommendations in Table 1 [see Dosage and Administration (2.2, 2.3)]. Refer to the prescribing information for ribavirin tablets for renal dosage adjustment of ribavirin in patients with CrCl less than or equal to 50 mL per minute.
12.3 Pharmacokinetics
The pharmacokinetic properties of elbasvir and grazoprevir have been evaluated in non-HCV-infected adult subjects and in HCV-infected adult subjects. Elbasvir pharmacokinetics were similar in healthy subjects and HCV-infected subjects and were approximately dose-proportional over the range of 5-100 mg once daily. Grazoprevir oral exposures are approximately 2-fold greater in HCV-infected subjects as compared to healthy subjects. Grazoprevir pharmacokinetics increased in a greater than dose-proportional manner over the range of 10-800 mg once daily in HCV-infected subjects. Ribavirin co-administration with ZEPATIER had no clinically relevant impact on plasma AUC and Cmax of elbasvir and grazoprevir compared to administration of ZEPATIER alone. The geometric mean steady-state pharmacokinetic parameter values for elbasvir and grazoprevir in non-cirrhotic HCV-infected subjects are provided in Table 7. Following once daily administration of ZEPATIER to HCV-infected subjects, elbasvir and grazoprevir reached steady state within approximately 6 days.
| Geometric Mean (90% Confidence Interval) | |||
|---|---|---|---|
| AUC0-24 (ng•hr/mL) | Cmax (ng/mL) | C24 (ng/mL) | |
| Elbasvir | 1920 (1880, 1960) | 121 (118, 123) | 48.4 (47.3, 49.6) |
| Grazoprevir | 1420 (1400, 1530) | 165 (161, 176) | 18.0 (17.8, 19.9) |
2.4 Hepatic Impairment
No dosage adjustment of ZEPATIER is recommended in patients with mild hepatic impairment (Child-Pugh A). ZEPATIER is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) or those with any history of prior hepatic decompensation [see Contraindications (4), Warnings and Precautions (5.3), Use in Specific Populations (8.9), and Clinical Pharmacology (12.3)].
8.9 Hepatic Impairment
No dosage adjustment of ZEPATIER is recommended in patients with mild hepatic impairment (Child-Pugh A). ZEPATIER is contraindicated in patients with moderate hepatic impairment (Child-Pugh B) due to the lack of clinical safety and efficacy experience in HCV-infected Child-Pugh B patients, and in patients with severe hepatic impairment (Child-Pugh C) due to a 12-fold increase in grazoprevir exposure in non-HCV infected Child-Pugh C subjects. In addition, postmarketing cases of hepatic decompensation/failure have been reported in patients with advanced liver disease [see Dosage and Administration (2.4), Contraindications (4), Warnings and Precautions (5.3), and Clinical Pharmacology (12.3)].
The safety and efficacy of ZEPATIER have not been established in patients awaiting liver transplant or in liver transplant recipients.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZEPATIER® is indicated for the treatment of chronic hepatitis C virus (HCV) genotype 1 or 4 infection in adult and pediatric patients 12 years of age and older or weighing at least 30 kg.
ZEPATIER is indicated for use with ribavirin in certain patient populations [see Dosage and Administration (2.2)].
12.1 Mechanism of Action
ZEPATIER is a fixed-dose combination of elbasvir and grazoprevir which are direct-acting antiviral agents against the hepatitis C virus [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk of Hepatitis B Virus Reactivation: Test all patients for evidence of current or prior HBV infection before initiation of HCV treatment. Monitor HCV/HBV coinfected patients for HBV reactivation and hepatitis flare during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated. (5.1)
- ALT Elevations: Perform hepatic laboratory testing prior to therapy, at treatment week 8, and as clinically indicated. For patients receiving 16 weeks of therapy, perform additional hepatic laboratory testing at treatment week 12. For ALT elevations on ZEPATIER, follow recommendations in full prescribing information. (5.2)
- Risk of Hepatic Decompensation/Failure in Patients with Evidence of Advanced Liver Disease: Hepatic decompensation/failure, including fatal outcomes, have been reported mostly in patients with cirrhosis and baseline moderate or severe liver impairment (Child-Pugh B or C) treated with HCV NS3/4A protease inhibitor-containing regimens. Monitor for clinical and laboratory evidence of hepatic decompensation. Discontinue ZEPATIER in patients who develop evidence of hepatic decompensation/failure. (5.3)
- Risk Associated with Ribavirin Combination Treatment: If ZEPATIER is administered with ribavirin, the warnings and precautions for ribavirin also apply. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Testing Prior to Initiation of Therapy:
- Recommended dosage in adult and pediatric patients 12 years of age and older or weighing at least 30 kg: One tablet taken orally once daily with or without food. (2.2)
| Patient Population | Treatment | Duration |
|---|---|---|
| Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced Peginterferon alfa + ribavirin. without baseline NS5A polymorphismsPolymorphisms at amino acid positions 28, 30, 31, or 93.
|
ZEPATIER | 12 weeks |
| Genotype 1a: Treatment-naïve or PegIFN/RBV-experienced with baseline NS5A polymorphisms |
ZEPATIER + ribavirin | 16 weeks |
| Genotype 1b: Treatment-naïve or PegIFN/RBV-experienced |
ZEPATIER | 12 weeks |
| Genotype 1a or 1b: PegIFN/RBV/PI-experienced Peginterferon alfa + ribavirin + HCV NS3/4A protease inhibitor.
|
ZEPATIER + ribavirin | 12 weeks |
| Genotype 4: Treatment-naïve |
ZEPATIER | 12 weeks |
| Genotype 4: PegIFN/RBV-experienced |
ZEPATIER + ribavirin | 16 weeks |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
ZEPATIER is available as a beige-colored, oval-shaped, film-coated tablet debossed with "770" on one side and plain on the other. Each tablet contains 50 mg elbasvir and 100 mg grazoprevir.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of ZEPATIER. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
If ZEPATIER is administered with ribavirin, refer to the prescribing information for ribavirin for a description of ribavirin-associated adverse reactions.
Clinical Trials in Adult Subjects
The safety of ZEPATIER in adult subjects was assessed based on 2 placebo-controlled trials and 7 uncontrolled Phase 2 and 3 clinical trials in approximately 1700 subjects with chronic hepatitis C virus infection with compensated liver disease (with or without cirrhosis) [see Clinical Studies (14)].
14.1 Overview of Clinical Trials
The efficacy of ZEPATIER was assessed in 2 placebo-controlled trials and 4 uncontrolled Phase 2 and 3 clinical trials in 1401 subjects with genotype (GT) 1, 4, or 6 and in 1 clinical trial in 22 pediatric subjects with GT 1 or 4 chronic hepatitis C virus infection with compensated liver disease (with or without cirrhosis). An overview of the 6 trials (n=1373) contributing to the assessment of efficacy in genotype 1 or 4 is provided in Table 13. C-EDGE TN, C-EDGE COINFECTION, C-SCAPE, and C-EDGE TE also included subjects with genotype 6 HCV infection (n=28). Because ZEPATIER is not indicated for genotype 6 infection, results in patients with genotype 6 infection are not included in Clinical Studies (14).
| Trial | Population | Study Groups and Duration (Number of Subjects Treated) |
|---|---|---|
| GT = Genotype | ||
| TN = Treatment-Naïve | ||
| TE = Treatment-Experienced (failed prior treatment with interferon [IFN] or peginterferon alfa [PegIFN] with or without ribavirin [RBV] or were intolerant to prior therapy). | ||
| C-EDGE TN (double-blind) |
GT 1, 4 TN with or without cirrhosis |
|
| C-EDGE COINFECTION (open-label) |
GT 1, 4 TN with or without cirrhosis HCV/HIV-1 co-infection |
|
| C-SURFER (double-blind) |
GT 1 TN or TE with or without cirrhosis Severe Renal Impairment including Hemodialysis |
|
| C-SCAPE (open-label) |
GT 4 TN without cirrhosis |
|
| C-EDGE TE (open-label) |
GT 1, 4 TE with or without cirrhosis with or without HCV/HIV-1 co-infection |
|
| C-SALVAGE (open-label) |
GT 1 TE with HCV protease inhibitor regimen Failed prior treatment with boceprevir, telaprevir, or simeprevir in combination with PegIFN + RBV. with or without cirrhosis |
|
| MK-5172-079 (open-label) |
GT 1, 4 TN or TE pediatric subjects |
|
ZEPATIER was administered once daily by mouth in these trials. For subjects who received ribavirin (RBV), the RBV dosage was weight-based (less than 66 kg = 800 mg per day, 66 to 80 kg = 1000 mg per day, 81 to 105 kg = 1200 mg per day, greater than 105 kg = 1400 mg per day) administered by mouth in two divided doses with food.
Sustained virologic response (SVR) was the primary endpoint in all trials and was defined as HCV RNA less than lower limit of quantification (LLOQ) at 12 weeks after the cessation of treatment (SVR12). Serum HCV RNA values were measured during these clinical trials using the COBAS AmpliPrep/COBAS Taqman HCV test (version 2.0) with an LLOQ of 15 HCV RNA IU per mL, with the exception of C-SCAPE where the assay had an LLOQ of 25 HCV RNA IU per mL.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
For patients receiving ZEPATIER with ribavirin, advise patients to read the FDA-approved patient labeling (Medication Guide) for ribavirin [see Warnings and Precautions (5.4)].
7.1 Potential for Drug Interactions
Grazoprevir is a substrate of OATP1B1/3 transporters. Co-administration of ZEPATIER with OATP1B1/3 inhibitors that are known or expected to significantly increase grazoprevir plasma concentrations is contraindicated [see Contraindications (4), Clinical Pharmacology (12.3), and Table 2].
Elbasvir and grazoprevir are substrates of CYP3A and P-gp, but the role of intestinal P-gp in the absorption of elbasvir and grazoprevir appears to be minimal. Co-administration of moderate or strong inducers of CYP3A with ZEPATIER may decrease elbasvir and grazoprevir plasma concentrations, leading to reduced therapeutic effect of ZEPATIER. Co-administration of ZEPATIER with strong CYP3A inducers or efavirenz is contraindicated [see Contraindications (4), Clinical Pharmacology (12.3), and Table 2]. Co-administration of ZEPATIER with moderate CYP3A inducers is not recommended [see Warnings and Precautions (5.5), Clinical Pharmacology (12.3), and Table 6]. Co-administration of ZEPATIER with strong CYP3A inhibitors may increase elbasvir and grazoprevir concentrations. Co-administration of ZEPATIER with certain strong CYP3A inhibitors is not recommended [see Warnings and Precautions (5.5), Clinical Pharmacology (12.3), and Table 6].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Each ZEPATIER tablet contains 50 mg elbasvir and 100 mg grazoprevir, is beige, oval-shaped, film-coated, debossed with "770" on one side and plain on the other. The tablets are packaged into a carton (NDC 0006-3074-02) containing two (2) 14-count child-resistant dose packs for a total of 28 tablets.
5.2 Increased Risk of Alt Elevations (5.2 Increased Risk of ALT Elevations)
During clinical trials with ZEPATIER with or without ribavirin, 1% of subjects experienced elevations of ALT from normal levels to greater than 5 times the upper limit of normal (ULN), generally at or after treatment week 8. ALT elevations were typically asymptomatic and most resolved with ongoing or completion of therapy. Higher rates of late ALT elevations occurred in the following subpopulations: female sex (2% [10/608]), Asian race (2% [4/164]), and age 65 years or older (2% [3/177]) [see Adverse Reactions (6.1)].
Hepatic laboratory testing should be performed prior to therapy, at treatment week 8, and as clinically indicated. For patients receiving 16 weeks of therapy, additional hepatic laboratory testing should be performed at treatment week 12.
- Patients should be instructed to consult their healthcare professional without delay if they have onset of fatigue, weakness, lack of appetite, nausea and vomiting, jaundice, or discolored feces.
- Consider discontinuing ZEPATIER if ALT levels remain persistently greater than 10 times the ULN.
- Discontinue ZEPATIER if ALT elevation is accompanied by signs or symptoms of liver inflammation or increasing conjugated bilirubin, alkaline phosphatase, or International Normalized Ratio (INR).
14.5 Clinical Trials With Genotype 4 Hcv (14.5 Clinical Trials with Genotype 4 HCV)
The efficacy of ZEPATIER in subjects with genotype 4 chronic HCV infection was demonstrated in C-EDGE TN, C-EDGE COINFECTION, C-EDGE TE, and C-SCAPE. C-SCAPE was a randomized, open-label trial which included treatment-naïve subjects with genotype 4 infection without cirrhosis. Subjects were randomized in a 1:1 ratio to EBR 50 mg once daily + GZR 100 mg once daily for 12 weeks or EBR 50 mg once daily + GZR 100 mg once daily + RBV for 12 weeks. In these combined studies in subjects with genotype 4 infection, 64% were treatment-naïve; 66% of the subjects were male; 87% were White; 10% were Black or African American; 22% had cirrhosis; and 30% had HCV/HIV-1 co-infection.
In C-SCAPE, C-EDGE TN, and C-EDGE COINFECTION trials combined, a total of 66 genotype 4 treatment-naïve subjects received ZEPATIER or EBR + GZR for 12 weeks. In these combined trials, SVR12 among subjects treated with ZEPATIER or EBR + GZR for 12 weeks was 97% (64/66).
In C-EDGE TE, a total of 37 genotype 4 treatment-experienced subjects received a 12- or 16-week ZEPATIER with or without RBV regimen. SVR12 among randomized subjects treated with ZEPATIER + RBV for 16 weeks was 100% (8/8).
8.3 Females and Males of Reproductive Potential
If ZEPATIER is administered with ribavirin, the information for ribavirin with regard to pregnancy testing, contraception, and infertility also applies to this combination regimen. Refer to ribavirin prescribing information for additional information.
5.4 Risks Associated With Ribavirin Combination Treatment (5.4 Risks Associated with Ribavirin Combination Treatment)
If ZEPATIER is administered with ribavirin, the warnings and precautions for ribavirin, including the pregnancy avoidance warning, also apply to this combination regimen. Refer to the ribavirin prescribing information for a full list of warnings and precautions for ribavirin [see Dosage and Administration (2.2)].
Principal Display Panel 50 Mg/100 Mg Tablet Dose Pack Carton (PRINCIPAL DISPLAY PANEL - 50 mg/100 mg Tablet Dose Pack Carton)
NDC 0006-3074-02
Zepatier
®
(elbasvir and grazoprevir) tablets
50 mg/100 mg
Rx only
28 Tablets
This carton contains a total of 28 tablets
packaged within 2 dose packs.
Each dose pack contains 14 blister units
with one tablet per blister unit.
7.2 Established and Other Potentially Significant Drug Interactions (7.2 Established and other Potentially Significant Drug Interactions)
If dose adjustments of concomitant medications are made due to treatment with ZEPATIER, doses should be readjusted after administration of ZEPATIER is completed.
Clearance of HCV infection with direct-acting antivirals may lead to changes in hepatic function, which may impact the safe and effective use of concomitant medications. For example, altered blood glucose control resulting in serious symptomatic hypoglycemia has been reported in diabetic patients in postmarketing case reports and published epidemiological studies. Management of hypoglycemia in these cases required either discontinuation or dose modification of concomitant medications used for diabetes treatment.
Frequent monitoring of relevant laboratory parameters (e.g., International Normalized Ratio [INR] in patients taking warfarin, blood glucose levels in diabetic patients) or drug concentrations of concomitant medications such as CYP450 substrates with a narrow therapeutic index (e.g., certain immunosuppressants) is recommended to ensure safe and effective use. Dose adjustments of concomitant medications may be necessary.
Table 6 provides a listing of established or potentially clinically significant drug interactions. The drug interactions described are based on studies conducted with either ZEPATIER, the components of ZEPATIER (elbasvir [EBR] and grazoprevir [GZR]) as individual agents, or are predicted drug interactions that may occur with ZEPATIER [see Contraindications (4), Warnings and Precautions (5.5), and Clinical Pharmacology (12.3)].
| Concomitant Drug Class: Drug Name | Effect on Concentration ↓ = decrease, ↑ = increase
|
Clinical Comment |
|---|---|---|
|
Antibiotics: Nafcillin |
↓ EBR ↓ GZR |
Co-administration of ZEPATIER with nafcillin may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
|
Antifungals:
oral Ketoconazole These interactions have been studied in healthy adults.
|
↑ EBR ↑ GZR |
Co-administration of oral ketoconazole is not recommended. |
|
Endothelin Antagonists:
Bosentan |
↓ EBR ↓ GZR |
Co-administration of ZEPATIER with bosentan may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
|
Immunosuppressants:
Tacrolimus |
↑ tacrolimus | Frequent monitoring of tacrolimus whole blood concentrations, changes in renal function, and tacrolimus-associated adverse events upon the initiation of co-administration is recommended. |
| HIV Medications: | ||
| Etravirine | ↓ EBR ↓ GZR |
Co-administration of ZEPATIER with etravirine may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
| Elvitegravir/ cobicistat/ emtricitabine/ tenofovir (disoproxil fumarate or alafenamide) | ↑ EBR ↑ GZR |
Co-administration of cobicistat-containing regimens is not recommended. |
|
HMG-CoA Reductase Inhibitors
See Drug Interactions (7.3)
for a list of HMG Co-A reductase inhibitors without clinically relevant interactions with ZEPATIER. :
|
||
| Atorvastatin | ↑ atorvastatin | The dose of atorvastatin should not exceed a daily dose of 20 mg when co-administered with ZEPATIER. |
| Rosuvastatin | ↑ rosuvastatin | The dose of rosuvastatin should not exceed a daily dose of 10 mg when co-administered with ZEPATIER. |
| Fluvastatin Lovastatin Simvastatin |
↑ fluvastatin ↑ lovastatin ↑ simvastatin |
Statin-associated adverse events such as myopathy should be closely monitored. The lowest necessary dose should be used when co-administered with ZEPATIER. |
|
Wakefulness-Promoting Agents:
Modafinil |
↓ EBR ↓ GZR |
Co-administration of ZEPATIER with modafinil may lead to reduced therapeutic effect of ZEPATIER. Co-administration is not recommended. |
7.3 Drugs Without Clinically Significant Interactions With Zepatier (7.3 Drugs without Clinically Significant Interactions with ZEPATIER)
The interaction between the components of ZEPATIER (elbasvir or grazoprevir) or ZEPATIER and the following drugs were evaluated in clinical studies, and no dose adjustments are needed when ZEPATIER is used with the following drugs individually: acid reducing agents (proton pump inhibitors, H2 blockers, antacids), buprenorphine/naloxone, digoxin, dolutegravir, methadone, mycophenolate mofetil, oral contraceptive pills, phosphate binders, pitavastatin, pravastatin, prednisone, raltegravir, ribavirin, rilpivirine, tenofovir disoproxil fumarate, and sofosbuvir [see Clinical Pharmacology (12.3)].
No clinically relevant drug-drug interaction is expected when ZEPATIER is co-administered with abacavir, emtricitabine, entecavir, and lamivudine.
5.1 Risk of Hepatitis B Virus Reactivation in Patients Coinfected With Hcv and Hbv (5.1 Risk of Hepatitis B Virus Reactivation in Patients Coinfected with HCV and HBV)
Hepatitis B virus (HBV) reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals, and who were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure and death. Cases have been reported in patients who are HBsAg positive and also in patients with serologic evidence of resolved HBV infection (i.e., HBsAg negative and anti-HBc positive). HBV reactivation has also been reported in patients receiving certain immunosuppressant or chemotherapeutic agents; the risk of HBV reactivation associated with treatment with HCV direct-acting antivirals may be increased in these patients.
HBV reactivation is characterized as an abrupt increase in HBV replication manifesting as a rapid increase in serum HBV DNA level. In patients with resolved HBV infection reappearance of HBsAg can occur. Reactivation of HBV replication may be accompanied by hepatitis, i.e., increases in aminotransferase levels and, in severe cases, increases in bilirubin levels, liver failure, and death can occur.
Test all patients for evidence of current or prior HBV infection by measuring HBsAg and anti-HBc before initiating HCV treatment with ZEPATIER. In patients with serologic evidence of HBV infection, monitor for clinical and laboratory signs of hepatitis flare or HBV reactivation during HCV treatment with ZEPATIER and during post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated.
5.5 Risk of Adverse Reactions Or Reduced Therapeutic Effect Due to Drug Interactions (5.5 Risk of Adverse Reactions or Reduced Therapeutic Effect Due to Drug Interactions)
The concomitant use of ZEPATIER and certain drugs may result in known or potentially significant drug interactions, some of which may lead to:
- Possible clinically significant adverse reactions from greater exposure of concomitant drugs or components of ZEPATIER.
- Significant decrease of elbasvir and grazoprevir plasma concentrations which may lead to reduced therapeutic effect of ZEPATIER and possible development of resistance.
See Tables 2 and 6 for steps to prevent or manage these known or potentially significant drug interactions, including dosing recommendations [see Contraindications (4) and Drug Interactions (7.2)].
Warning: Risk of Hepatitis B Virus Reactivation in Patients Coinfected With Hcv and Hbv (WARNING: RISK OF HEPATITIS B VIRUS REACTIVATION IN PATIENTS COINFECTED WITH HCV AND HBV)
Test all patients for evidence of current or prior hepatitis B virus (HBV) infection before initiating treatment with ZEPATIER. HBV reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals and were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure, and death. Monitor HCV/HBV coinfected patients for hepatitis flare or HBV reactivation during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated [see Warnings and Precautions (5.1)].
14.6 Clinical Trial in Pediatric Subjects With Genotype 1 Or 4 Chronic Hepatitis C Infection (14.6 Clinical Trial in Pediatric Subjects with Genotype 1 or 4 Chronic Hepatitis C Infection)
The efficacy of ZEPATIER was evaluated in an open-label study (MK-5172-079, NCT03379506) that evaluated pediatric subjects 12 years to less than 18 years of age who received ZEPATIER for 12 weeks. HCV GT1a infected subjects with one or more baseline NS5A RAS were excluded from study participation. In the MK-5172-079 study, 22 treatment-naïve or treatment-experienced subjects 12 years to less than 18 years of age with genotype 1 (n=21) or 4 CHC (n=1), without cirrhosis, were treated with ZEPATIER for 12 weeks. The median age was 13.5 years (range: 12 to 17); 50% were female; 95% were White; the weight range was 28.1 kg to 96.5 kg; 95.5% had genotype 1 and 4.5% had genotype 4; 64% were treatment-naïve, 36% were treatment-experienced; 46% had baseline HCV RNA levels greater than 800,000 IU/mL. The overall SVR12 rate was 100% (22/22).
5.3 Risk of Hepatic Decompensation/failure in Patients With Evidence of Advanced Liver Disease (5.3 Risk of Hepatic Decompensation/Failure in Patients with Evidence of Advanced Liver Disease)
Postmarketing cases of hepatic decompensation/failure, including those with fatal outcomes, have been reported in patients treated with HCV NS3/4A protease inhibitor-containing regimens, including ZEPATIER.
Reported cases occurred in patients treated with HCV NS3/4A protease inhibitor-containing regimens with baseline cirrhosis with and without moderate or severe liver impairment (Child-Pugh B or C) as well as some patients without cirrhosis. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatic laboratory testing should be performed in all patients [see Warnings and Precautions (5.2)]. In patients with compensated cirrhosis (Child-Pugh A) or evidence of advanced liver disease, such as portal hypertension, more frequent hepatic laboratory testing may be warranted; and patients should be monitored for signs and symptoms of hepatic decompensation such as the presence of jaundice, ascites, hepatic encephalopathy, and variceal hemorrhage. Discontinue ZEPATIER in patients who develop evidence of hepatic decompensation/failure.
ZEPATIER is contraindicated in patients with moderate to severe hepatic impairment (Child-Pugh B or C) or those with any history of prior hepatic decompensation [see Contraindications (4), Adverse Reactions (6.1), Use in Specific Populations (8.9), and Clinical Pharmacology (12.3)].
14.2 Clinical Trials in Treatment Naïve Subjects With Genotype 1 Hcv (c Edge Tn and C Edge Coinfection) (14.2 Clinical Trials in Treatment-Naïve Subjects with Genotype 1 HCV (C-EDGE TN and C-EDGE COINFECTION))
The efficacy of ZEPATIER in treatment-naïve subjects with genotype 1 chronic hepatitis C virus infection with or without cirrhosis was demonstrated in the C-EDGE TN and C-EDGE COINFECTION trials.
C-EDGE TN was a randomized, double-blind, placebo-controlled trial in treatment-naïve subjects with genotype 1 or 4 infection with or without cirrhosis. Subjects were randomized in a 3:1 ratio to: ZEPATIER for 12 weeks (immediate treatment group) or placebo for 12 weeks followed by open-label treatment with ZEPATIER for 12 weeks (deferred treatment group). Among subjects with genotype 1 infection randomized to the immediate treatment group, the median age was 55 years (range: 20 to 78); 56% of the subjects were male; 61% were White; 20% were Black or African American; 8% were Hispanic or Latino; mean body mass index was 26 kg/m2; 72% had baseline HCV RNA levels greater than 800,000 IU per mL; 24% had cirrhosis; 67% had non-C/C IL28B alleles (CT or TT); and 55% had genotype 1a and 45% had genotype 1b chronic HCV infection.
C-EDGE COINFECTION was an open-label, single-arm trial in treatment-naïve HCV/HIV-1 co-infected subjects with genotype 1 or 4 infection with or without cirrhosis. Subjects received ZEPATIER for 12 weeks. Among subjects with genotype 1 infection, the median age was 50 years (range: 21 to 71); 85% of the subjects were male; 75% were White; 19% were Black or African American; 6% were Hispanic or Latino; mean body mass index was 25 kg/m2; 59% had baseline HCV RNA levels greater than 800,000 IU per mL; 16% had cirrhosis; 65% had non-C/C IL28B alleles (CT or TT); and 76% had genotype 1a, 23% had genotype 1b, and 1% had genotype 1-Other chronic HCV infection.
Table 14 presents treatment outcomes for ZEPATIER in treatment-naïve subjects with genotype 1 infection from C-EDGE TN (immediate treatment group) and C-EDGE COINFECTION. For treatment outcomes for ZEPATIER in genotype 4 infection, [see Clinical Studies (14.5)].
| Trial | C-EDGE TN (Immediate Treatment Group) |
C-EDGE COINFECTION (HCV/HIV-1 Co-Infection) |
|---|---|---|
| Regimen | ZEPATIER 12 Weeks N=288 |
ZEPATIER 12 Weeks N=189 |
| SVR in Genotype 1 | 95% (273/288) | 95% (179/189) |
| Outcome for subjects without SVR | ||
| On-treatment Virologic Failure Includes subjects with virologic breakthrough.
|
<1% (1/288) | 0% (0/189) |
| Relapse | 3% (10/288) | 3% (6/189) |
| Other Other includes subjects who discontinued due to adverse event, lost to follow-up, or subject withdrawal.
|
1% (4/288) | 2% (4/189) |
| SVR by Genotype 1 Subtypes | ||
| GT 1a For the impact of baseline NS5A polymorphisms on SVR12, [see Microbiology (12.4), Table 12].
|
92% (144/157) | 94% (136/144) |
| GT 1b Includes genotype 1 subtypes other than 1a or 1b.
|
98% (129/131) | 96% (43/45) |
| SVR by Cirrhosis status | ||
| Non-cirrhotic | 94% (207/220) | 94% (148/158) |
| Cirrhotic | 97% (66/68) | 100% (31/31) |
2.2 Recommended Dosage in Adult and Pediatric Patients 12 Years of Age and Older Or Weighing At Least 30 Kg (2.2 Recommended Dosage in Adult and Pediatric Patients 12 Years of Age and Older or Weighing at Least 30 kg)
ZEPATIER is a two-drug, fixed-dose combination product containing 50 mg of elbasvir and 100 mg of grazoprevir in a single tablet. The recommended dosage of ZEPATIER is one tablet taken orally once daily with or without food [see Clinical Pharmacology (12.3)]. ZEPATIER is used in combination with ribavirin in certain patient populations (see Table 1). When administered with ZEPATIER, the recommended dosage of ribavirin in patients without renal impairment is weight-based administered in two divided doses with food. For further information on ribavirin dosing and dosage modifications, refer to the ribavirin prescribing information.
14.4 Clinical Trial in Subjects With Genotype 1 Hcv and Severe Renal Impairment Including Subjects On Hemodialysis (c Surfer) (14.4 Clinical Trial in Subjects with Genotype 1 HCV and Severe Renal Impairment including Subjects on Hemodialysis (C-SURFER))
C-SURFER was a randomized, double-blind, placebo-controlled trial in subjects with genotype 1 infection, with or without cirrhosis, with chronic kidney disease (CKD) Stage 4 (eGFR 15-29 mL/min/1.73 m2) or CKD Stage 5 (eGFR <15 mL/min/1.73 m2), including subjects on hemodialysis, who were treatment-naïve or who had failed prior therapy with IFN or PegIFN ± RBV therapy. Subjects were randomized in a 1:1 ratio to one of the following treatment groups: EBR 50 mg once daily + GZR 100 mg once daily for 12 weeks (immediate treatment group) or placebo for 12 weeks followed by open-label treatment with EBR + GZR for 12 weeks (deferred treatment group). In addition, 11 subjects received open-label EBR + GZR for 12 weeks (intensive pharmacokinetic [PK] group). Subjects randomized to the immediate treatment group and intensive PK group had a median age of 58 years (range: 31 to 76); 75% of the subjects were male; 50% were White; 45% were Black or African American; 11% were Hispanic or Latino; 57% had baseline HCV RNA levels greater than 800,000 IU/mL; 6% had cirrhosis; and 72% had non-C/C IL28B alleles (CT or TT).
Treatment outcomes in subjects treated with ZEPATIER for 12 weeks in the pooled immediate treatment group and intensive PK group are presented in Table 16.
| Regimen | EBR + GZR 12 weeks (Immediate Treatment Group) N=122 Includes subjects (n=11) in the intensive PK group.
|
|---|---|
| Overall SVR | 94% (115/122) SVR was achieved in 99% (115/116) of subjects in the pre-specified primary analysis population, which excluded subjects not receiving at least one dose of study treatment and those with missing data due to death or early study discontinuation for reasons unrelated to treatment response.
|
| Outcome for subjects without SVR | |
| On-treatment Virologic Failure | 0% (0/122) |
| Relapse | <1% (1/122) |
| Other Other includes subjects who discontinued due to adverse event, lost to follow-up, or subject withdrawal.
|
5% (6/122) |
| SVR by Genotype | |
| GT 1a | 97% (61/63) |
| GT 1b Includes genotype 1 subtypes other than 1a or 1b.
|
92% (54/59) |
| SVR by Cirrhosis status | |
| No | 95% (109/115) |
| Yes | 86% (6/7) |
| SVR by Prior HCV Treatment Status | |
| Treatment-naïve | 95% (96/101) |
| Treatment-experienced | 90% (19/21) |
| SVR by Dialysis Status | |
| No | 97% (29/30) |
| Yes | 93% (86/92) |
| SVR by Chronic Kidney Disease Stage | |
| Stage 4 | 100% (22/22) |
| Stage 5 | 93% (93/100) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:50.772659 · Updated: 2026-03-14T22:48:00.481820