These Highlights Do Not Include All The Information Needed To Use Omnipaque Safely And Effectively. See Full Prescribing Information For Omnipaque.
164b27b9-97ae-4279-896f-1c4afa583dd1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Use only the OMNIPAQUE iodine concentrations and presentations recommended for intrathecal procedure [see Dosage and Administration (2.2 , 2.8) ]. Intrathecal administration of OMNIPAQUE of a wrong iodine concentration, even if inadvertent, may cause death, convulsions, seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema [see Warnings and Precautions (5.1) ].
Indications and Usage
OMNIPAQUE is a radiographic contrast agent indicated for intrathecal, intra-arterial, intravenous, oral, rectal, intraarticular, and body cavity imaging procedures in adults and pediatric patients. ( 1 )
Dosage and Administration
For each imaging procedure, specific dosage forms, concentrations, and presentations are recommended. Individualize the concentration and volume according to the specific dosing tables and accounting for factors such as age, body weight, and condition of the patient, and the equipment and imaging technique used. ( 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 , 2.8 , 2.9 ) See full prescribing information for complete dosing and administration information. ( 2 )
Warnings and Precautions
Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency equipment and trained personnel available. ( 5.3 ) Acute Kidney Injury: Acute injury including renal failure can occur. Minimize dose and maintain adequate hydration to minimize risk. ( 5.4 ) Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after administration. ( 5.5 ) Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. ( 5.9 )
Contraindications
OMNIPAQUE for hysterosalpingography is contraindicated during pregnancy or suspected pregnancy, menstruation or when menstruation is imminent, within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract including the external genitalia, and when reproductive tract neoplasia is known or suspected because of the risk of peritoneal spread of neoplasm.
Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravenous or intra-arterial contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agents; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering OMNIPAQUE to patients with a history of a severe cutaneous adverse reaction to OMNIPAQUE.
Medication Information
Warnings and Precautions
Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency equipment and trained personnel available. ( 5.3 ) Acute Kidney Injury: Acute injury including renal failure can occur. Minimize dose and maintain adequate hydration to minimize risk. ( 5.4 ) Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after administration. ( 5.5 ) Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. ( 5.9 )
Indications and Usage
OMNIPAQUE is a radiographic contrast agent indicated for intrathecal, intra-arterial, intravenous, oral, rectal, intraarticular, and body cavity imaging procedures in adults and pediatric patients. ( 1 )
Dosage and Administration
For each imaging procedure, specific dosage forms, concentrations, and presentations are recommended. Individualize the concentration and volume according to the specific dosing tables and accounting for factors such as age, body weight, and condition of the patient, and the equipment and imaging technique used. ( 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 , 2.8 , 2.9 ) See full prescribing information for complete dosing and administration information. ( 2 )
Contraindications
OMNIPAQUE for hysterosalpingography is contraindicated during pregnancy or suspected pregnancy, menstruation or when menstruation is imminent, within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract including the external genitalia, and when reproductive tract neoplasia is known or suspected because of the risk of peritoneal spread of neoplasm.
Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravenous or intra-arterial contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agents; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering OMNIPAQUE to patients with a history of a severe cutaneous adverse reaction to OMNIPAQUE.
Description
Use only the OMNIPAQUE iodine concentrations and presentations recommended for intrathecal procedure [see Dosage and Administration (2.2 , 2.8) ]. Intrathecal administration of OMNIPAQUE of a wrong iodine concentration, even if inadvertent, may cause death, convulsions, seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema [see Warnings and Precautions (5.1) ].
Section 42229-5
Recommended Dosage for Radiographic Examination of the GI Tract in Adults
The recommended dose for radiographic examination of the GI tract in adults is 50 mL to 100 mL of OMNIPAQUE injection 350 mg iodine/mL administered undiluted orally.
Section 43683-2
Section 44425-7
Storage and Handling
- OMNIPAQUE Injection: Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. May be stored in a contrast media warmer for up to one month, not to exceed 37°C (98.6°F).
- OMNIPAQUE Oral Solution: Store between 0°C and 30°C (32°F to 86°F).
Protect from light. Do not freeze. Discard any product that is inadvertently frozen, as freezing may compromise the closure integrity of the immediate container.
10 Overdosage
The adverse effects of overdosage in intra-arterial or intravenous administration are life-threatening and affect mainly the pulmonary and cardiovascular systems. The symptoms include: cyanosis, bradycardia, acidosis, pulmonary hemorrhage, convulsions, coma, and cardiac arrest. Treatment of an overdosage is directed toward the support of all vital functions, and prompt institution of symptomatic therapy. Iohexol can be dialyzed.
11 Description
Iohexol is a nonionic radiographic contrast agent available as:
- OMNIPAQUE (iohexol) injection for intrathecal, intra-arterial, intravenous, oral, rectal, intraarticular, and body cavity use
- OMNIPAQUE (iohexol) oral solution for oral use
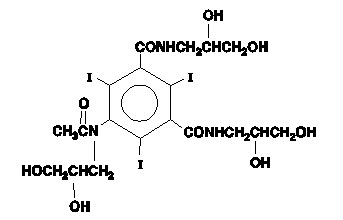
The chemical name of iohexol is Bis(2,3-dihydroxypropyl)-5-[N-(2,3-dihydroxypropyl)-acetamido]-2,4,6- triiodoisophthalamide with a molecular weight of 821.14 (iodine content 46.36%). Iohexol has the following structural formula:
OMNIPAQUE injection is a sterile, pyrogen-free, colorless to pale yellow solution available in five concentrations of iodine:
- OMNIPAQUE 140 mg iodine/mL: Each mL contains 302 mg iohexol (providing 140 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 180 mg iodine/mL: Each mL contains 388 mg iohexol (providing 180 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 240 mg iodine/mL: Each mL contains 518 mg iohexol (providing 240 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 300 mg iodine/mL: Each mL contains 647 mg iohexol (providing 300 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 350 mg iodine/mL: Each mL contains 755 mg iohexol (providing 350 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
OMNIPAQUE oral solution is a sterile, pyrogen-free, colorless to pale yellow solution available in two concentrations of iodine:
- OMNIPAQUE oral solution 9 mg iodine/mL: Each mL contains 19 mg iohexol (providing 9 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE oral solution 12 mg iodine/mL: Each mL contains 26 mg iohexol (providing 12 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium hydroxide. OMNIPAQUE injection and OMNIPAQUE oral solution contain no preservatives and no ingredient made from a gluten-containing grain (wheat, barley, or rye).
OMNIPAQUE injection and OMNIPAQUE oral solution have the following physical properties:
| Dosage Form | Concentration (mg iodine/mL) |
Osmolality
By vapor-pressure osmometry.
(mOsmol/kg water) |
Absolute Viscosity (cP) |
Specific Gravity | |
|---|---|---|---|---|---|
| 20°C | 37°C | 37°C | |||
| Injection | 140 | 322 | 2.3 | 1.5 | 1.164 |
| 180 | 408 | 3.1 | 2.0 | 1.209 | |
| 240 | 520 | 5.8 | 3.4 | 1.280 | |
| 300 | 672 | 11.8 | 6.3 | 1.349 | |
| 350 | 844 | 20.4 | 10.4 | 1.406 | |
| Oral solution | 9 | 38 | 1.1 | 0.8 | 1.011 |
| 12 | 45 | 1.1 | 0.8 | 1.014 |
OMNIPAQUE injection has osmolalities from approximately 1.1 to 3 times that of plasma (285 mOsmol/kg water) or cerebrospinal fluid (301 mOsmol/kg water) as shown in the above table and are hypertonic .
OMNIPAQUE oral solution is hypotonic.
8.5 Geriatric Use
In clinical studies of OMNIPAQUE for CT of the head and body, 52 (17%) of patients were 70 and over. No overall differences in safety were observed between these patients and younger patients. Other reported clinical experience has not identified differences in safety and effectiveness between the elderly and younger patients.
Iohexol is substantially excreted by the kidney, and the risk of adverse reactions to iohexol may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
14 Clinical Studies
The safety and effectiveness of OMNIPAQUE for CT of the head were evaluated in three clinical studies. Each study also used an ionic high-osmolar iodinated contrast agent as a comparator. A total of 280 patients were randomized to administration of either OMNIPAQUE (n=142) or the comparator (n=138). OMNIPAQUE patients had a mean age of 52 years (range 16 to 85), 41% were women, and were administered a mean of 692 mg iodine/kg (range 337 to 1,250 mg iodine/kg) by intravenous injection with OMNIPAQUE 240 mg iodine/mL (1 study) or 300 mg iodine/mL (2 studies).
Efficacy was determined from investigator ratings of quality of contrast enhancement (none, poor, good, or excellent; only scans rated as good or excellent were considered diagnostic).
The percentage of OMNIPAQUE-enhanced scans rated as good or excellent was 100% in the two studies using OMNIPAQUE 300 mg iodine/mL, and 79% in the third study using OMNIPAQUE 240 mg iodine/mL.
4 Contraindications
OMNIPAQUE for hysterosalpingography is contraindicated during pregnancy or suspected pregnancy, menstruation or when menstruation is imminent, within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract including the external genitalia, and when reproductive tract neoplasia is known or suspected because of the risk of peritoneal spread of neoplasm.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks Associated with Intrathecal Administration of OMNIPAQUE Injection 140 mg Iodine/mL and 350 mg Iodine/mL [see Warnings and Precautions (5.1)]
- Risks Associated with Parenteral Administration of OMNIPAQUE Oral Solution [see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Acute Kidney Injury [see Warnings and Precautions (5.4)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.5)]
- Thromboembolic Events [see Warnings and Precautions (5.6)]
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions (5.9)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.12)]
8.6 Renal Impairment
The clearance of iohexol decreases with increasing degree of renal impairment and results in delayed opacification of the urinary system. In addition, preexisting renal impairment increases the risk for acute kidney injury [see Warnings and Precautions (5.4)]. Iohexol can be removed by dialysis.
12.3 Pharmacokinetics
Following the intravenous administration of iohexol (between 500 mg iodine/kg to 1,500 mg iodine/kg) to 16 adult subjects, apparent first-order terminal elimination half-life was 12.6 hours and total body clearance was 131 (98 to 165) mL/min. Clearance was not dose dependent.
1 Indications and Usage
OMNIPAQUE is a radiographic contrast agent indicated for intrathecal, intra-arterial, intravenous, oral, rectal, intraarticular, and body cavity imaging procedures in adults and pediatric patients. (1)
5.4 Acute Kidney Injury
Acute kidney injury, including renal failure, may occur after parenteral administration of OMNIPAQUE. Risk factors include: pre-existing renal impairment, dehydration, diabetes mellitus, congestive heart failure, advanced vascular disease, elderly age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma/paraproteinaceous diseases, repetitive and/or large doses of an iodinated contrast agent.
Use the lowest necessary dose of OMNIPAQUE in patients with renal impairment. Adequately hydrate patients prior to and following parenteral administration of OMNIPAQUE. Do not use laxatives, diuretics, or preparatory dehydration prior to OMNIPAQUE administration.
12.1 Mechanism of Action
The iodine atoms in iohexol provide attenuation of X-rays in direct proportion to the concentration of iohexol. Since concentration changes over time, iohexol provides time-dependent image contrast which may assist in visualizing body structures.
5.6 Thromboembolic Events
Serious, rarely fatal, thromboembolic events causing myocardial infarction and stroke can occur during angiocardiography procedures with iodinated contrast agents including OMNIPAQUE. During these procedures, increased thrombosis and activation of the complement system occurs. Risk factors for thromboembolic events include: length of procedure, catheter and syringe material, underlying disease state, and concomitant medications.
To minimize thromboembolic events, use meticulous angiographic techniques, and minimize the length of the procedure. Avoid blood remaining in contact with syringes containing OMNIPAQUE, which increases the risk of clotting. Avoid angiocardiography in patients with homocystinuria because of the risk of inducing thrombosis and embolism.
5 Warnings and Precautions
- Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency equipment and trained personnel available. (5.3)
- Acute Kidney Injury: Acute injury including renal failure can occur. Minimize dose and maintain adequate hydration to minimize risk. (5.4)
- Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after administration. (5.5)
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. (5.9)
2 Dosage and Administration
- For each imaging procedure, specific dosage forms, concentrations, and presentations are recommended.
- Individualize the concentration and volume according to the specific dosing tables and accounting for factors such as age, body weight, and condition of the patient, and the equipment and imaging technique used. (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9)
- See full prescribing information for complete dosing and administration information. (2)
3 Dosage Forms and Strengths
Injection and Oral Solution: Colorless to pale yellow solution available in the following presentations:
| Dosage Form | Concentration (mg of iodine/mL) |
Package Size | Package Type |
|---|---|---|---|
| Injection | 140 | 50 mL | Single-Dose Bottle |
| 180 | 10 mL | Single-Dose Vial | |
| 240 | 10 mL and 20 mL | Single-Dose Vial | |
| 50 mL and 100 mL | Sigle-Dose Bottle | ||
| 300 | 10 mL | Single-Dose Vial | |
| 30 mL, 50 mL, 100 mL, 125 mL, and 150 mL | Single-Dose Bottle | ||
| 500 mL OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in bottles of 500 mL can be used as either an Imaging Bulk Package or a Pharmacy Bulk Package [see Dosage and Administration (2.10, 2.11)].
|
Imaging or Pharmacy Bulk Package | ||
| 350 | 50 mL, 75 mL, 100 mL, 125 mL, 150 mL, and 200 mL | Single-Dose Bottle | |
| 500 mL | Imaging or Pharmacy Bulk Package | ||
| Oral Solution | 9 | 500 mL | Single-Dose Bottle |
| 12 | 500 mL | Single-Dose Bottle |
1.1 Intrathecal Procedures‡
OMNIPAQUE is indicated for:
- Myelography and computerized tomography (CT) myelography (lumbar, thoracic, cervical, total columnar) in adults and pediatric patients aged 2 weeks and older
- CT cisternography in adults and pediatric patients aged 2 weeks and older
1.3 Intravenous Procedures‡
OMNIPAQUE is indicated for:
- Excretory urography in adults and pediatric patients
- CT of the head and body in adults and pediatric patients
- Peripheral venography (phlebography) in adults
- Intravenous digital subtraction angiography (IV-DSA) of the head, neck, abdominal, renal, and peripheral vessels in adults
1.6 Body Cavity Procedures‡
OMNIPAQUE is indicated for:
- Endoscopic retrograde pancreatography (ERP) and cholangiopancreatography (ERCP) in adults
- Herniography in adults
- Hysterosalpingography in adults
- Voiding cystourethrography (VCU) in pediatric patients
‡ Specific dosage forms, concentrations, and presentations of OMNIPAQUE are recommended for each type of imaging procedure [see Dosage and Administrations (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9) and Warnings and Precautions (5.1, 5.2)].
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of OMNIPAQUE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
- Lactation: A lactating woman may pump and discard breast milk for 10 hours after OMNIPAQUE administration. (8.2)
5.3 Hypersensitivity Reactions
OMNIPAQUE can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock. Most severe reactions develop shortly after the start of the injection (within 1 to 3 minutes), but delayed reactions can also occur. There is an increased risk in patients with a history of a previous reaction to contrast agent and known allergic disorders (i.e., bronchial asthma, drug, or food allergies) or other hypersensitivities. Premedication with antihistamines or corticosteroids does not prevent serious life-threatening reactions but may reduce both their incidence and severity.
Obtain a history of allergy, hypersensitivity, or hypersensitivity reactions to iodinated contrast agents and always have emergency resuscitation equipment and trained personnel available prior to OMNIPAQUE administration. Monitor all patients for hypersensitivity reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.2 Intra Arterial Procedures‡
OMNIPAQUE is indicated for:
- Cardiac ventriculography in adults and pediatric patients
- Aortography including studies of aorta and its branches in adults and pediatric patients
- Selective coronary arteriography in adults
- Cerebral arteriography in adults
- Peripheral arteriography in adults
- Intra-arterial digital subtraction angiography (IA-DSA) of the head, neck, abdominal, renal, and peripheral vessels in adults
- Pulmonary angiography in pediatric patients
1.4 Oral Or Rectal Procedures‡
OMNIPAQUE is indicated for:
- Radiographic examination of the gastrointestinal (GI) tract in adults and pediatric patients
- CT of the abdomen and pelvis in conjunction with intravenous administration of OMNIPAQUE in adults and pediatric patients
1.5 Intraarticular Procedures‡
OMNIPAQUE is indicated for:
- Arthrography in adults
5.5 Cardiovascular Adverse Reactions
Life-threatening or fatal cardiovascular reactions including hypotension, shock, cardiac arrest have occurred with the parenteral administration of OMNIPAQUE. Most deaths occur during injection or five to ten minutes later, with cardiovascular disease as the main aggravating factor. Cardiac decompensation, serious arrhythmias, and myocardial ischemia or infarction can occur during coronary arteriography and ventriculography.
Based on clinical literature, reported deaths from the administration of iodinated contrast agents range from 6.6 per million (0.00066%) to 1 in 10,000 (0.01%). Use the lowest necessary dose of OMNIPAQUE in patients with congestive heart failure and always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
5.13 Interference With Laboratory Test
OMNIPAQUE can interfere with protein-bound iodine test [see Drug Interactions (7.2)].
5.12 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravenous or intra-arterial contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agents; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering OMNIPAQUE to patients with a history of a severe cutaneous adverse reaction to OMNIPAQUE.
5.7 Extravasation and Injection Site Reactions
Extravasation of OMNIPAQUE during intravenous or intra-arterial injection may cause tissue necrosis and/or compartment syndrome, particularly in patients with severe arterial or venous disease. Ensure intravenous or intra-arterial placement of catheters prior to injection. Monitor patients for extravasation and advise patients to seek medical care for progression of symptoms.
Principal Display Panel 300 Mg Bottle Box Label
GE Healthcare
Y-538I
Contains
10 x 500 mL
Bottles
NDC 0407-1413-72
OMNIPAQUE™
(iohexol) Injection
300 mg Iodine/mL
This container can be used either as:
IMAGING BULK PACKAGE
For Intravenous Use Only
or
PHARMACY BULK PACKAGE
Not for Direct Infusion
For Intrathecal, Intra-arterial,
Intravenous, Oral, Rectal,
Intraarticular, or Body Cavity Use
Rx ONLY
See prescribing information for dosage and administration.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C
(59°F to 86°F) [see USP Controlled Room Temperature]. May be stored in a
contrast media warmer for up to one month, not to exceed 37°C (98.6°F).
Protect from light. Do not freeze. Discard unused OMNIPAQUE 8 hours
after initial puncture.
Each mL contains 647 mg of iohexol (300 mg organically bound iodine),
0.1 mg edetate calcium disodium, and 1.21 mg tromethamine.
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium
hydroxide. No preservative added.
+PLUSPAK™ (polymer bottle)
Distributed by GE Healthcare Inc.,
Marlborough, MA 01752 U.S.A.
For inquiries call 1-800-654-0118
Exp.: DD MMM YYYY
Lot: 12345678
0000000 USA
Principal Display Panel 350 Mg Bottle Box Label
GE Healthcare
Y-548I
Contains
10 x 500 mL
Bottles
NDC 0407-1414-72
OMNIPAQUE™
(iohexol) Injection
350 mg Iodine/mL
This container can be used either as:
IMAGING BULK PACKAGE
For Intravenous Use Only
or
PHARMACY BULK PACKAGE
Not for Direct Infusion
For Intra-arterial,
Intravenous, Oral, Rectal,
Intraarticular, or Body Cavity Use
NOT FOR INTRATHECAL USE
Rx ONLY
See prescribing information for dosage and administration.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C
(59°F to 86°F) [see USP Controlled Room Temperature]. May be stored in a
contrast media warmer for up to one month, not to exceed 37°C (98.6°F).
Protect from light. Do not freeze. Discard unused OMNIPAQUE 8 hours
after initial puncture.
Each mL contains 755 mg of iohexol (350 mg organically bound iodine),
0.1 mg edetate calcium disodium, and 1.21 mg tromethamine.
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium
hydroxide. No preservative added.
+PLUSPAK™ (polymer bottle)
Distributed by GE Healthcare Inc.,
Marlborough, MA 01752 U.S.A.
For inquiries call 1-800-654-0118
Exp.: DD MMM YYYY
Lot: 12345678
0000000 USA
5.8 Thyroid Storm in Patients With Hyperthyroidism
Thyroid storm has occurred after the intravenous or intra-arterial use of iodinated contrast agents in patients with hyperthyroidism, or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of OMNIPAQUE.
2.1 Important Dosage and Administration Instructions
- Specific dosage forms, concentrations, and presentations of OMNIPAQUE are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9) and Warnings and Precautions (5.1, 5.2)].
- Individualize the volume, strength, and rate of administration of OMNIPAQUE injection according to the specific dosing tables [see Dosage and Administration (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9)]. Consider factors such as age, body weight, vessel size, blood flow rate within the vessel, anticipated pathology, degree and extent of opacification required, structures or area to be examined, disease processes affecting the patient, and equipment and technique to be employed.
- Hydrate patients before and after administration of OMNIPAQUE injection [see Warnings and Precautions (5.4)].
- Use aseptic technique for all handling and administration of OMNIPAQUE injection.
- Administer OMNIPAQUE injection at either body (37°C, 98.6°F) or room temperature (20°C to 25°C, 68°F to 77°F).
- Do not mix OMNIPAQUE injection with, or inject in intravenous lines containing, other drugs or total nutritional admixtures except when OMNIPAQUE injection is administered in an automated contrast injection system or contrast management system suitable for simultaneous injection of OMNIPAQUE injection and 0.9% Sodium Chloride Injection [see Dosage and Administration (2.10, 2.11)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Each container of OMNIPAQUE injection and OMNIPAQUE oral solution in single-dose containers are intended for one procedure only. Discard any unused portion.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed with iohexol to evaluate carcinogenic potential. Iohexol was not genotoxic in a series of studies, including the Ames test, the mouse lymphoma TK locus forward mutation assay, and a mouse micronucleus assay. Iohexol did not impair the fertility of male or female rats when repeatedly administered at intravenous dosages up to 4 g iodine/kg.
5.10 Hypertensive Crisis in Patients With Pheochromocytoma
Hypertensive crisis has occurred after the use of iodinated contrast agents in patient with pheochromocytoma. Monitor patients when administering OMNIPAQUE intravenously or intra-arterially if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of contrast necessary, assess the blood pressure throughout the procedure, and have measures for treatment of a hypertensive crisis readily available.
2.2 Recommended Dosage for Intrathecal Procedures in Adults
- The recommended doses for intrathecal procedures in adults are shown in Table 1.
- Administer over 1 minute to 2 minutes.
- If sequential or repeat examinations are required, allow at least 48 hours for clearance of the drug from the body before repeat administration; however, whenever possible, 5 days to 7 days is recommended.
- If CT myelography is performed, delay imaging by several hours to reduce the degree of contrast.
| Imaging Procedure | Injection Type | Concentration (mg Iodine/mL) |
Volume to Administer |
|---|---|---|---|
| Lumbar Myelography | Lumbar | 180 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
10 mL to 17 mL |
| 240 | 7 mL to 12.5 mL | ||
| Thoracic Myelography | Lumbar Cervical |
240 | 6 mL to 12.5 mL |
| 300 | 6 mL to 10 mL | ||
| Cervical Myelography | Lumbar | 240 | 6 mL to 12.5 mL |
| 300 | 6 mL to 10 mL | ||
| C1-2 | 180 | 7 mL to 10 mL | |
| 240 | 6 mL to 12.5 mL | ||
| 300 | 4 mL to 10 mL | ||
| Total Columnar Myelography | Lumbar | 240 | 6 mL to 12.5 mL |
| 300 | 6 mL to 10 mL | ||
| CT Cisternography | Lumbar | 180 | 10 mL to 17 mL |
| 240 | 7 mL to 12.5 mL |
2.4 Recommended Dosage for Intravenous Procedures in Adults
The recommended doses for intravenous procedures in adults are shown in Table 3.
| Imaging Procedure | Concentration (mg Iodine/mL) |
Volume to Administer | |
|---|---|---|---|
| Excretory Urography | 300 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
0.6 mL/kg to 1.2 mL/kg body weight (maximum dose is 102 mL) |
|
| 350 | |||
| CT | Head | 240 | 120 mL to 250 mL by infusion |
| 300 Use single-dose containers, 500 mL Imaging Bulk Packages or Pharmacy Bulk Packages, or 150 mL single-dose bottles with an automated contrast injection system or contrast management system [see Dosage and Administration (2.10, 2.11, 2.12)].
|
70 mL to 150 mL by rapid injection | ||
| 350 | 80 mL by rapid injection | ||
| Body | 300 | 50 mL to 200 mL by rapid injection | |
| 350 | 60 mL to 100 mL by rapid injection | ||
| Peripheral Venography (phlebography) | 240 | 20 mL to 150 mL per leg | |
| 300 | 40 mL to 100 mL per leg | ||
| IV-DSA (head, neck, abdominal, renal, and peripheral vessels) | 350 | 30 mL to 50 mL at 7.5 mL/sec to 30 mL/sec using a pressure injector Frequently three or more doses may be required; the maximum cumulative total dose is 250 mL |
2.7 Recommended Dosage for Body Cavity Procedures in Adults
- The recommended doses for body cavity procedures in adults are shown in Table 7.
- Volume to administer may vary depending on individual anatomy and/or disease state.
| Imaging Procedure | Concentration (mg Iodine/mL) | Volume to Administer | Route of Administration |
|---|---|---|---|
| Endoscopic retrograde pancreatography (ERP) and cholangiopancreatography (ERCP) | 240 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
10 mL to 50 mL | Intraductal |
| Herniography | 240 | 15 mL to 20 mL | Intraperitoneal |
| 300 | |||
| Hysterosalpingography | 240 | 50 mL | Intrauterine |
5.14 Increased Risk of Seizures With Intrathecal Procedures
Focal and generalized motor seizures have been reported after intrathecal use of iodinated contrast agents. In several of the cases, higher than recommended doses were administered.
Use of medications that may lower the seizure threshold (phenothiazine derivatives, including those used for their antihistaminic properties; tricyclic antidepressants; MAO inhibitors; CNS stimulants; analeptics; antipsychotic agents) should be carefully evaluated. Consider discontinuing these agents at least 48 hours before and for at least 24 hours following intrathecal administration of OMNIPAQUE.
5.11 Sickle Cell Crisis in Patients With Sickle Cell Disease
Iodinated contrast agents when administered intravenously or intra-arterially may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following OMNIPAQUE administration and use OMNIPAQUE only if the necessary imaging information cannot be obtained with alternative imaging modalities.
2.3 Recommended Dosage for Intra Arterial Procedures in Adults
The recommended doses for intra-arterial procedures in adults are shown in Table 2.
| Imaging Procedure | Concentration (mg Iodine/mL) |
Volume to Administer per Single Injection for Selective Injection Sites | Maximum Cumulative Total Dose | |
|---|---|---|---|---|
| Cardiac Ventriculography | 350 Use single-dose containers or a 500 mL Pharmacy Bulk Package [see Dosage and Administration (2.11)].
|
40 mL (Range of 30 mL to 60 mL) may be combined with selective coronary arteriography |
250 mL | |
| Aortography and Selective Visceral Arteriography | 300 |
|
290 mL | |
| 350 | 250 mL | |||
| Aortic root and arch study when used alone | 350 | 50 mL (Range of 20 mL to 75 mL) | 250 mL | |
| Selective Coronary Arteriography | 350 | 5 mL (Range of 3 mL to 14 mL) | 250 mL | |
| Cerebral Arteriography | 300 |
|
290 mL | |
| Peripheral Arteriography | Aortofemoral Runoffs | 300 | 30 mL to 90 mL | 290 mL |
| 350 | 20 mL to 70 mL | 250 mL | ||
| Selective Arteriogram | 300 | 10 mL to 60 mL | 290 mL | |
| 350 | 10 mL to 30 mL | 250 mL | ||
| IA-DSA (head, neck, abdominal, renal, and peripheral vessels) | 140 |
|
250 mL |
2.6 Recommended Dosage for Intraarticular Procedures in Adults
- The recommended doses for intraarticular procedures in adults are shown in Table 6.
- Use passive or active manipulation to disperse the medium throughout the joint space.
| Imaging Procedure | Location | Concentration (mg Iodine/mL) |
Volume to Administer | Double Contrast/Single Contrast |
|---|---|---|---|---|
| Arthrography | Knee | 240 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
5 mL to 15 mL | Lower volumes recommended for double-contrast examinations; higher volumes recommended for single-contrast examinations. |
| 300 | 5 mL to 15 mL | |||
| 350 | 5 mL to 10 mL | |||
| Shoulder | 300 | 10 mL | ||
| Temporomandibular | 300 | 0.5 mL to 1 mL |
5.9 Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age
Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast media (ICM) in pediatric patients 0 to 3 years of age.
Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after ICM exposure. Pediatric patients with congenital cardiac conditions may be at the greatest risk given that they often require high doses of contrast during invasive cardiac procedures.
An underactive thyroid during early life may be harmful for cognitive and neurological development and may require thyroid hormone replacement therapy. After exposure to ICM, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates.
5.2 Risks Associated With Parenteral Administration of Omnipaque Oral Solution
Adverse reactions such as hemolysis may occur if OMNIPAQUE oral solution is administered intravenously or intraarterially due to low osmolality [see Description (11)]. OMNIPAQUE oral solution is for oral use only.
5.1 Risks Associated With Intrathecal Administration of Omnipaque Injection 140 Mg Iodine/ml and 350 Mg Iodine/ml
Use only the iodine concentrations and presentations recommended for intrathecal procedures [see Dosage and Administration (2.2, 2.8)]. Intrathecal administration of OMNIPAQUE of a wrong iodine concentration, even if inadvertent, can cause death, convulsions, seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema.
Warning: Risks Associated With Intrathecal Administration of Omnipaque Injection 140 Mg Iodine/ml and 350 Mg Iodine/ml
Use only the OMNIPAQUE iodine concentrations and presentations recommended for intrathecal procedure [see Dosage and Administration (2.2, 2.8)]. Intrathecal administration of OMNIPAQUE of a wrong iodine concentration, even if inadvertent, may cause death, convulsions, seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema [see Warnings and Precautions (5.1)].
2.10 Directions for Use of the 500 Ml Bottles of Omnipaque Injection 300 Mg Iodine/ml and 350 Mg Iodine/ml As Imaging Bulk Package
- OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in 500 mL bottles may be used as an Imaging Bulk Package (IBP) for intravenous use [see Dosage and Administration (2.4, 2.8)].
- When used as an IBP, check the appropriate box on the container label to indicate that the selected container is to be utilized as an Imaging Bulk Package. This container is for use only with an automated contrast injection system, contrast management system, or contrast media transfer set approved or cleared for use with this contrast agent in this IBP. See device labeling for information on devices indicated for use with this IBP and techniques to help assure safe use.
- Use the OMNIPAQUE IBP and 0.9% Sodium Chloride Injection USP in a room designated for radiological procedures that involve administration of a contrast agent.
- Using aseptic technique, penetrate the container closure only one time with a suitable sterile component of the automated contrast injection system, contrast management system, or contrast media transfer set cleared for use with the OMNIPAQUE IBP.
- If 0.9% Sodium Chloride Injection USP is used with the OMNIPAQUE IBP, prepare the 0.9% Sodium Chloride Injection USP sterile port in accordance with the dosage and administration section of its approved prescribing information and affix the saline tag provided with the OMNIPAQUE IBP on the 0.9% Sodium Chloride Injection USP container.
- Once the OMNIPAQUE IBP and 0.9% Sodium Chloride Injection containers are punctured, do not remove them from the work area during the entire period of use. Maintain the OMNIPAQUE IBP in an inverted position such that container contents are in continuous contact with the dispensing set.
- After the container closure is punctured, if the integrity of the OMNIPAQUE IBP, the 0.9% Sodium Chloride Injection, and the delivery system cannot be assured through direct continuous supervision, discard the IBP, 0.9% Sodium Chloride Injection USP, and all associated disposables for the contrast media transfer set.
- A maximum time from initial puncture is 8 hours. Discard any unused portion.
2.11 Directions for Use of the 500 Ml Bottles of Omnipaque Injection 300 Mg Iodine/ml and 350 Mg Iodine/ml As Pharmacy Bulk Package
- OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in 500 mL bottles contain many single doses and may be used as a Pharmacy Bulk Package (PBP).
- When used as a PBP, check the appropriate box on the container label to indicate that the selected container is to be utilized as a Pharmacy Bulk Package. This container is not for direct infusion; it is for use in pharmacy admixture service to dispense aliquots using a suitable dispensing set under a laminar flow hood or equivalent clean air compounding area using aseptic technique.
- Penetrate the container closure only one time, utilizing a suitable transfer device and aseptic technique.
- The withdrawal of container contents should be accomplished without delay. However, should this not be possible, a maximum time of 8 hours from initial closure entry is permitted to complete fluid transfer operations.
- Do not remove the container from the aseptic area during the entire 8-hour period.
- The temperature of the container should not exceed 37°C (98.6°F), after the closure has been entered.
- Discard any unused portion after 8 hours from the initial closure entry.
2.12 Directions for Use of the 150 Ml Bottles of Omnipaque Injection 300 Mg Iodine/ml and 350 Mg Iodine/ml With An Automated Contrast Injection System Or Contrast Management System
- OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in 150 mL bottles may be used with a contrast media management system cleared for use with OMNIPAQUE injection 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles.
- See device labeling for information on device indications, instructions for use, and techniques to help assure safe use.
- Using aseptic technique, penetrate the container closure only one time with a suitable sterile component of the contrast media management system cleared for use with OMNIPAQUE 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles.
- Once the container is punctured, do not remove the bottle from the work area during the entire period of use.
- Maximum use time is 4 hours after initial puncture.
- Each bottle is for one procedure only. Discard unused portion.
Structured Label Content
Section 42229-5 (42229-5)
Recommended Dosage for Radiographic Examination of the GI Tract in Adults
The recommended dose for radiographic examination of the GI tract in adults is 50 mL to 100 mL of OMNIPAQUE injection 350 mg iodine/mL administered undiluted orally.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
- OMNIPAQUE Injection: Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. May be stored in a contrast media warmer for up to one month, not to exceed 37°C (98.6°F).
- OMNIPAQUE Oral Solution: Store between 0°C and 30°C (32°F to 86°F).
Protect from light. Do not freeze. Discard any product that is inadvertently frozen, as freezing may compromise the closure integrity of the immediate container.
10 Overdosage (10 OVERDOSAGE)
The adverse effects of overdosage in intra-arterial or intravenous administration are life-threatening and affect mainly the pulmonary and cardiovascular systems. The symptoms include: cyanosis, bradycardia, acidosis, pulmonary hemorrhage, convulsions, coma, and cardiac arrest. Treatment of an overdosage is directed toward the support of all vital functions, and prompt institution of symptomatic therapy. Iohexol can be dialyzed.
11 Description (11 DESCRIPTION)
Iohexol is a nonionic radiographic contrast agent available as:
- OMNIPAQUE (iohexol) injection for intrathecal, intra-arterial, intravenous, oral, rectal, intraarticular, and body cavity use
- OMNIPAQUE (iohexol) oral solution for oral use
The chemical name of iohexol is Bis(2,3-dihydroxypropyl)-5-[N-(2,3-dihydroxypropyl)-acetamido]-2,4,6- triiodoisophthalamide with a molecular weight of 821.14 (iodine content 46.36%). Iohexol has the following structural formula:
OMNIPAQUE injection is a sterile, pyrogen-free, colorless to pale yellow solution available in five concentrations of iodine:
- OMNIPAQUE 140 mg iodine/mL: Each mL contains 302 mg iohexol (providing 140 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 180 mg iodine/mL: Each mL contains 388 mg iohexol (providing 180 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 240 mg iodine/mL: Each mL contains 518 mg iohexol (providing 240 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 300 mg iodine/mL: Each mL contains 647 mg iohexol (providing 300 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE 350 mg iodine/mL: Each mL contains 755 mg iohexol (providing 350 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
OMNIPAQUE oral solution is a sterile, pyrogen-free, colorless to pale yellow solution available in two concentrations of iodine:
- OMNIPAQUE oral solution 9 mg iodine/mL: Each mL contains 19 mg iohexol (providing 9 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
- OMNIPAQUE oral solution 12 mg iodine/mL: Each mL contains 26 mg iohexol (providing 12 mg organically bound iodine) and the following inactive ingredients: 0.1 mg edetate calcium disodium and 1.21 mg tromethamine.
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium hydroxide. OMNIPAQUE injection and OMNIPAQUE oral solution contain no preservatives and no ingredient made from a gluten-containing grain (wheat, barley, or rye).
OMNIPAQUE injection and OMNIPAQUE oral solution have the following physical properties:
| Dosage Form | Concentration (mg iodine/mL) |
Osmolality
By vapor-pressure osmometry.
(mOsmol/kg water) |
Absolute Viscosity (cP) |
Specific Gravity | |
|---|---|---|---|---|---|
| 20°C | 37°C | 37°C | |||
| Injection | 140 | 322 | 2.3 | 1.5 | 1.164 |
| 180 | 408 | 3.1 | 2.0 | 1.209 | |
| 240 | 520 | 5.8 | 3.4 | 1.280 | |
| 300 | 672 | 11.8 | 6.3 | 1.349 | |
| 350 | 844 | 20.4 | 10.4 | 1.406 | |
| Oral solution | 9 | 38 | 1.1 | 0.8 | 1.011 |
| 12 | 45 | 1.1 | 0.8 | 1.014 |
OMNIPAQUE injection has osmolalities from approximately 1.1 to 3 times that of plasma (285 mOsmol/kg water) or cerebrospinal fluid (301 mOsmol/kg water) as shown in the above table and are hypertonic .
OMNIPAQUE oral solution is hypotonic.
8.5 Geriatric Use
In clinical studies of OMNIPAQUE for CT of the head and body, 52 (17%) of patients were 70 and over. No overall differences in safety were observed between these patients and younger patients. Other reported clinical experience has not identified differences in safety and effectiveness between the elderly and younger patients.
Iohexol is substantially excreted by the kidney, and the risk of adverse reactions to iohexol may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and effectiveness of OMNIPAQUE for CT of the head were evaluated in three clinical studies. Each study also used an ionic high-osmolar iodinated contrast agent as a comparator. A total of 280 patients were randomized to administration of either OMNIPAQUE (n=142) or the comparator (n=138). OMNIPAQUE patients had a mean age of 52 years (range 16 to 85), 41% were women, and were administered a mean of 692 mg iodine/kg (range 337 to 1,250 mg iodine/kg) by intravenous injection with OMNIPAQUE 240 mg iodine/mL (1 study) or 300 mg iodine/mL (2 studies).
Efficacy was determined from investigator ratings of quality of contrast enhancement (none, poor, good, or excellent; only scans rated as good or excellent were considered diagnostic).
The percentage of OMNIPAQUE-enhanced scans rated as good or excellent was 100% in the two studies using OMNIPAQUE 300 mg iodine/mL, and 79% in the third study using OMNIPAQUE 240 mg iodine/mL.
4 Contraindications (4 CONTRAINDICATIONS)
OMNIPAQUE for hysterosalpingography is contraindicated during pregnancy or suspected pregnancy, menstruation or when menstruation is imminent, within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract including the external genitalia, and when reproductive tract neoplasia is known or suspected because of the risk of peritoneal spread of neoplasm.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks Associated with Intrathecal Administration of OMNIPAQUE Injection 140 mg Iodine/mL and 350 mg Iodine/mL [see Warnings and Precautions (5.1)]
- Risks Associated with Parenteral Administration of OMNIPAQUE Oral Solution [see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Acute Kidney Injury [see Warnings and Precautions (5.4)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.5)]
- Thromboembolic Events [see Warnings and Precautions (5.6)]
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions (5.9)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.12)]
8.6 Renal Impairment
The clearance of iohexol decreases with increasing degree of renal impairment and results in delayed opacification of the urinary system. In addition, preexisting renal impairment increases the risk for acute kidney injury [see Warnings and Precautions (5.4)]. Iohexol can be removed by dialysis.
12.3 Pharmacokinetics
Following the intravenous administration of iohexol (between 500 mg iodine/kg to 1,500 mg iodine/kg) to 16 adult subjects, apparent first-order terminal elimination half-life was 12.6 hours and total body clearance was 131 (98 to 165) mL/min. Clearance was not dose dependent.
1 Indications and Usage (1 INDICATIONS AND USAGE)
OMNIPAQUE is a radiographic contrast agent indicated for intrathecal, intra-arterial, intravenous, oral, rectal, intraarticular, and body cavity imaging procedures in adults and pediatric patients. (1)
5.4 Acute Kidney Injury
Acute kidney injury, including renal failure, may occur after parenteral administration of OMNIPAQUE. Risk factors include: pre-existing renal impairment, dehydration, diabetes mellitus, congestive heart failure, advanced vascular disease, elderly age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma/paraproteinaceous diseases, repetitive and/or large doses of an iodinated contrast agent.
Use the lowest necessary dose of OMNIPAQUE in patients with renal impairment. Adequately hydrate patients prior to and following parenteral administration of OMNIPAQUE. Do not use laxatives, diuretics, or preparatory dehydration prior to OMNIPAQUE administration.
12.1 Mechanism of Action
The iodine atoms in iohexol provide attenuation of X-rays in direct proportion to the concentration of iohexol. Since concentration changes over time, iohexol provides time-dependent image contrast which may assist in visualizing body structures.
5.6 Thromboembolic Events
Serious, rarely fatal, thromboembolic events causing myocardial infarction and stroke can occur during angiocardiography procedures with iodinated contrast agents including OMNIPAQUE. During these procedures, increased thrombosis and activation of the complement system occurs. Risk factors for thromboembolic events include: length of procedure, catheter and syringe material, underlying disease state, and concomitant medications.
To minimize thromboembolic events, use meticulous angiographic techniques, and minimize the length of the procedure. Avoid blood remaining in contact with syringes containing OMNIPAQUE, which increases the risk of clotting. Avoid angiocardiography in patients with homocystinuria because of the risk of inducing thrombosis and embolism.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency equipment and trained personnel available. (5.3)
- Acute Kidney Injury: Acute injury including renal failure can occur. Minimize dose and maintain adequate hydration to minimize risk. (5.4)
- Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after administration. (5.5)
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For each imaging procedure, specific dosage forms, concentrations, and presentations are recommended.
- Individualize the concentration and volume according to the specific dosing tables and accounting for factors such as age, body weight, and condition of the patient, and the equipment and imaging technique used. (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9)
- See full prescribing information for complete dosing and administration information. (2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection and Oral Solution: Colorless to pale yellow solution available in the following presentations:
| Dosage Form | Concentration (mg of iodine/mL) |
Package Size | Package Type |
|---|---|---|---|
| Injection | 140 | 50 mL | Single-Dose Bottle |
| 180 | 10 mL | Single-Dose Vial | |
| 240 | 10 mL and 20 mL | Single-Dose Vial | |
| 50 mL and 100 mL | Sigle-Dose Bottle | ||
| 300 | 10 mL | Single-Dose Vial | |
| 30 mL, 50 mL, 100 mL, 125 mL, and 150 mL | Single-Dose Bottle | ||
| 500 mL OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in bottles of 500 mL can be used as either an Imaging Bulk Package or a Pharmacy Bulk Package [see Dosage and Administration (2.10, 2.11)].
|
Imaging or Pharmacy Bulk Package | ||
| 350 | 50 mL, 75 mL, 100 mL, 125 mL, 150 mL, and 200 mL | Single-Dose Bottle | |
| 500 mL | Imaging or Pharmacy Bulk Package | ||
| Oral Solution | 9 | 500 mL | Single-Dose Bottle |
| 12 | 500 mL | Single-Dose Bottle |
1.1 Intrathecal Procedures‡
OMNIPAQUE is indicated for:
- Myelography and computerized tomography (CT) myelography (lumbar, thoracic, cervical, total columnar) in adults and pediatric patients aged 2 weeks and older
- CT cisternography in adults and pediatric patients aged 2 weeks and older
1.3 Intravenous Procedures‡
OMNIPAQUE is indicated for:
- Excretory urography in adults and pediatric patients
- CT of the head and body in adults and pediatric patients
- Peripheral venography (phlebography) in adults
- Intravenous digital subtraction angiography (IV-DSA) of the head, neck, abdominal, renal, and peripheral vessels in adults
1.6 Body Cavity Procedures‡
OMNIPAQUE is indicated for:
- Endoscopic retrograde pancreatography (ERP) and cholangiopancreatography (ERCP) in adults
- Herniography in adults
- Hysterosalpingography in adults
- Voiding cystourethrography (VCU) in pediatric patients
‡ Specific dosage forms, concentrations, and presentations of OMNIPAQUE are recommended for each type of imaging procedure [see Dosage and Administrations (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9) and Warnings and Precautions (5.1, 5.2)].
6.2 Post Marketing Experience (6.2 Post-marketing Experience)
The following adverse reactions have been identified during post-approval use of OMNIPAQUE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: A lactating woman may pump and discard breast milk for 10 hours after OMNIPAQUE administration. (8.2)
5.3 Hypersensitivity Reactions
OMNIPAQUE can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock. Most severe reactions develop shortly after the start of the injection (within 1 to 3 minutes), but delayed reactions can also occur. There is an increased risk in patients with a history of a previous reaction to contrast agent and known allergic disorders (i.e., bronchial asthma, drug, or food allergies) or other hypersensitivities. Premedication with antihistamines or corticosteroids does not prevent serious life-threatening reactions but may reduce both their incidence and severity.
Obtain a history of allergy, hypersensitivity, or hypersensitivity reactions to iodinated contrast agents and always have emergency resuscitation equipment and trained personnel available prior to OMNIPAQUE administration. Monitor all patients for hypersensitivity reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.2 Intra Arterial Procedures‡ (1.2 Intra-arterial Procedures‡)
OMNIPAQUE is indicated for:
- Cardiac ventriculography in adults and pediatric patients
- Aortography including studies of aorta and its branches in adults and pediatric patients
- Selective coronary arteriography in adults
- Cerebral arteriography in adults
- Peripheral arteriography in adults
- Intra-arterial digital subtraction angiography (IA-DSA) of the head, neck, abdominal, renal, and peripheral vessels in adults
- Pulmonary angiography in pediatric patients
1.4 Oral Or Rectal Procedures‡ (1.4 Oral or Rectal Procedures‡)
OMNIPAQUE is indicated for:
- Radiographic examination of the gastrointestinal (GI) tract in adults and pediatric patients
- CT of the abdomen and pelvis in conjunction with intravenous administration of OMNIPAQUE in adults and pediatric patients
1.5 Intraarticular Procedures‡
OMNIPAQUE is indicated for:
- Arthrography in adults
5.5 Cardiovascular Adverse Reactions
Life-threatening or fatal cardiovascular reactions including hypotension, shock, cardiac arrest have occurred with the parenteral administration of OMNIPAQUE. Most deaths occur during injection or five to ten minutes later, with cardiovascular disease as the main aggravating factor. Cardiac decompensation, serious arrhythmias, and myocardial ischemia or infarction can occur during coronary arteriography and ventriculography.
Based on clinical literature, reported deaths from the administration of iodinated contrast agents range from 6.6 per million (0.00066%) to 1 in 10,000 (0.01%). Use the lowest necessary dose of OMNIPAQUE in patients with congestive heart failure and always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
5.13 Interference With Laboratory Test (5.13 Interference with Laboratory Test)
OMNIPAQUE can interfere with protein-bound iodine test [see Drug Interactions (7.2)].
5.12 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravenous or intra-arterial contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agents; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering OMNIPAQUE to patients with a history of a severe cutaneous adverse reaction to OMNIPAQUE.
5.7 Extravasation and Injection Site Reactions
Extravasation of OMNIPAQUE during intravenous or intra-arterial injection may cause tissue necrosis and/or compartment syndrome, particularly in patients with severe arterial or venous disease. Ensure intravenous or intra-arterial placement of catheters prior to injection. Monitor patients for extravasation and advise patients to seek medical care for progression of symptoms.
Principal Display Panel 300 Mg Bottle Box Label (PRINCIPAL DISPLAY PANEL - 300 mg Bottle Box Label)
GE Healthcare
Y-538I
Contains
10 x 500 mL
Bottles
NDC 0407-1413-72
OMNIPAQUE™
(iohexol) Injection
300 mg Iodine/mL
This container can be used either as:
IMAGING BULK PACKAGE
For Intravenous Use Only
or
PHARMACY BULK PACKAGE
Not for Direct Infusion
For Intrathecal, Intra-arterial,
Intravenous, Oral, Rectal,
Intraarticular, or Body Cavity Use
Rx ONLY
See prescribing information for dosage and administration.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C
(59°F to 86°F) [see USP Controlled Room Temperature]. May be stored in a
contrast media warmer for up to one month, not to exceed 37°C (98.6°F).
Protect from light. Do not freeze. Discard unused OMNIPAQUE 8 hours
after initial puncture.
Each mL contains 647 mg of iohexol (300 mg organically bound iodine),
0.1 mg edetate calcium disodium, and 1.21 mg tromethamine.
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium
hydroxide. No preservative added.
+PLUSPAK™ (polymer bottle)
Distributed by GE Healthcare Inc.,
Marlborough, MA 01752 U.S.A.
For inquiries call 1-800-654-0118
Exp.: DD MMM YYYY
Lot: 12345678
0000000 USA
Principal Display Panel 350 Mg Bottle Box Label (PRINCIPAL DISPLAY PANEL - 350 mg Bottle Box Label)
GE Healthcare
Y-548I
Contains
10 x 500 mL
Bottles
NDC 0407-1414-72
OMNIPAQUE™
(iohexol) Injection
350 mg Iodine/mL
This container can be used either as:
IMAGING BULK PACKAGE
For Intravenous Use Only
or
PHARMACY BULK PACKAGE
Not for Direct Infusion
For Intra-arterial,
Intravenous, Oral, Rectal,
Intraarticular, or Body Cavity Use
NOT FOR INTRATHECAL USE
Rx ONLY
See prescribing information for dosage and administration.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C
(59°F to 86°F) [see USP Controlled Room Temperature]. May be stored in a
contrast media warmer for up to one month, not to exceed 37°C (98.6°F).
Protect from light. Do not freeze. Discard unused OMNIPAQUE 8 hours
after initial puncture.
Each mL contains 755 mg of iohexol (350 mg organically bound iodine),
0.1 mg edetate calcium disodium, and 1.21 mg tromethamine.
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium
hydroxide. No preservative added.
+PLUSPAK™ (polymer bottle)
Distributed by GE Healthcare Inc.,
Marlborough, MA 01752 U.S.A.
For inquiries call 1-800-654-0118
Exp.: DD MMM YYYY
Lot: 12345678
0000000 USA
5.8 Thyroid Storm in Patients With Hyperthyroidism (5.8 Thyroid Storm in Patients with Hyperthyroidism)
Thyroid storm has occurred after the intravenous or intra-arterial use of iodinated contrast agents in patients with hyperthyroidism, or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of OMNIPAQUE.
2.1 Important Dosage and Administration Instructions
- Specific dosage forms, concentrations, and presentations of OMNIPAQUE are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9) and Warnings and Precautions (5.1, 5.2)].
- Individualize the volume, strength, and rate of administration of OMNIPAQUE injection according to the specific dosing tables [see Dosage and Administration (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9)]. Consider factors such as age, body weight, vessel size, blood flow rate within the vessel, anticipated pathology, degree and extent of opacification required, structures or area to be examined, disease processes affecting the patient, and equipment and technique to be employed.
- Hydrate patients before and after administration of OMNIPAQUE injection [see Warnings and Precautions (5.4)].
- Use aseptic technique for all handling and administration of OMNIPAQUE injection.
- Administer OMNIPAQUE injection at either body (37°C, 98.6°F) or room temperature (20°C to 25°C, 68°F to 77°F).
- Do not mix OMNIPAQUE injection with, or inject in intravenous lines containing, other drugs or total nutritional admixtures except when OMNIPAQUE injection is administered in an automated contrast injection system or contrast management system suitable for simultaneous injection of OMNIPAQUE injection and 0.9% Sodium Chloride Injection [see Dosage and Administration (2.10, 2.11)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Each container of OMNIPAQUE injection and OMNIPAQUE oral solution in single-dose containers are intended for one procedure only. Discard any unused portion.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed with iohexol to evaluate carcinogenic potential. Iohexol was not genotoxic in a series of studies, including the Ames test, the mouse lymphoma TK locus forward mutation assay, and a mouse micronucleus assay. Iohexol did not impair the fertility of male or female rats when repeatedly administered at intravenous dosages up to 4 g iodine/kg.
5.10 Hypertensive Crisis in Patients With Pheochromocytoma (5.10 Hypertensive Crisis in Patients with Pheochromocytoma)
Hypertensive crisis has occurred after the use of iodinated contrast agents in patient with pheochromocytoma. Monitor patients when administering OMNIPAQUE intravenously or intra-arterially if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of contrast necessary, assess the blood pressure throughout the procedure, and have measures for treatment of a hypertensive crisis readily available.
2.2 Recommended Dosage for Intrathecal Procedures in Adults
- The recommended doses for intrathecal procedures in adults are shown in Table 1.
- Administer over 1 minute to 2 minutes.
- If sequential or repeat examinations are required, allow at least 48 hours for clearance of the drug from the body before repeat administration; however, whenever possible, 5 days to 7 days is recommended.
- If CT myelography is performed, delay imaging by several hours to reduce the degree of contrast.
| Imaging Procedure | Injection Type | Concentration (mg Iodine/mL) |
Volume to Administer |
|---|---|---|---|
| Lumbar Myelography | Lumbar | 180 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
10 mL to 17 mL |
| 240 | 7 mL to 12.5 mL | ||
| Thoracic Myelography | Lumbar Cervical |
240 | 6 mL to 12.5 mL |
| 300 | 6 mL to 10 mL | ||
| Cervical Myelography | Lumbar | 240 | 6 mL to 12.5 mL |
| 300 | 6 mL to 10 mL | ||
| C1-2 | 180 | 7 mL to 10 mL | |
| 240 | 6 mL to 12.5 mL | ||
| 300 | 4 mL to 10 mL | ||
| Total Columnar Myelography | Lumbar | 240 | 6 mL to 12.5 mL |
| 300 | 6 mL to 10 mL | ||
| CT Cisternography | Lumbar | 180 | 10 mL to 17 mL |
| 240 | 7 mL to 12.5 mL |
2.4 Recommended Dosage for Intravenous Procedures in Adults
The recommended doses for intravenous procedures in adults are shown in Table 3.
| Imaging Procedure | Concentration (mg Iodine/mL) |
Volume to Administer | |
|---|---|---|---|
| Excretory Urography | 300 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
0.6 mL/kg to 1.2 mL/kg body weight (maximum dose is 102 mL) |
|
| 350 | |||
| CT | Head | 240 | 120 mL to 250 mL by infusion |
| 300 Use single-dose containers, 500 mL Imaging Bulk Packages or Pharmacy Bulk Packages, or 150 mL single-dose bottles with an automated contrast injection system or contrast management system [see Dosage and Administration (2.10, 2.11, 2.12)].
|
70 mL to 150 mL by rapid injection | ||
| 350 | 80 mL by rapid injection | ||
| Body | 300 | 50 mL to 200 mL by rapid injection | |
| 350 | 60 mL to 100 mL by rapid injection | ||
| Peripheral Venography (phlebography) | 240 | 20 mL to 150 mL per leg | |
| 300 | 40 mL to 100 mL per leg | ||
| IV-DSA (head, neck, abdominal, renal, and peripheral vessels) | 350 | 30 mL to 50 mL at 7.5 mL/sec to 30 mL/sec using a pressure injector Frequently three or more doses may be required; the maximum cumulative total dose is 250 mL |
2.7 Recommended Dosage for Body Cavity Procedures in Adults
- The recommended doses for body cavity procedures in adults are shown in Table 7.
- Volume to administer may vary depending on individual anatomy and/or disease state.
| Imaging Procedure | Concentration (mg Iodine/mL) | Volume to Administer | Route of Administration |
|---|---|---|---|
| Endoscopic retrograde pancreatography (ERP) and cholangiopancreatography (ERCP) | 240 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
10 mL to 50 mL | Intraductal |
| Herniography | 240 | 15 mL to 20 mL | Intraperitoneal |
| 300 | |||
| Hysterosalpingography | 240 | 50 mL | Intrauterine |
5.14 Increased Risk of Seizures With Intrathecal Procedures (5.14 Increased Risk of Seizures with Intrathecal Procedures)
Focal and generalized motor seizures have been reported after intrathecal use of iodinated contrast agents. In several of the cases, higher than recommended doses were administered.
Use of medications that may lower the seizure threshold (phenothiazine derivatives, including those used for their antihistaminic properties; tricyclic antidepressants; MAO inhibitors; CNS stimulants; analeptics; antipsychotic agents) should be carefully evaluated. Consider discontinuing these agents at least 48 hours before and for at least 24 hours following intrathecal administration of OMNIPAQUE.
5.11 Sickle Cell Crisis in Patients With Sickle Cell Disease (5.11 Sickle Cell Crisis in Patients with Sickle Cell Disease)
Iodinated contrast agents when administered intravenously or intra-arterially may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following OMNIPAQUE administration and use OMNIPAQUE only if the necessary imaging information cannot be obtained with alternative imaging modalities.
2.3 Recommended Dosage for Intra Arterial Procedures in Adults (2.3 Recommended Dosage for Intra-arterial Procedures in Adults)
The recommended doses for intra-arterial procedures in adults are shown in Table 2.
| Imaging Procedure | Concentration (mg Iodine/mL) |
Volume to Administer per Single Injection for Selective Injection Sites | Maximum Cumulative Total Dose | |
|---|---|---|---|---|
| Cardiac Ventriculography | 350 Use single-dose containers or a 500 mL Pharmacy Bulk Package [see Dosage and Administration (2.11)].
|
40 mL (Range of 30 mL to 60 mL) may be combined with selective coronary arteriography |
250 mL | |
| Aortography and Selective Visceral Arteriography | 300 |
|
290 mL | |
| 350 | 250 mL | |||
| Aortic root and arch study when used alone | 350 | 50 mL (Range of 20 mL to 75 mL) | 250 mL | |
| Selective Coronary Arteriography | 350 | 5 mL (Range of 3 mL to 14 mL) | 250 mL | |
| Cerebral Arteriography | 300 |
|
290 mL | |
| Peripheral Arteriography | Aortofemoral Runoffs | 300 | 30 mL to 90 mL | 290 mL |
| 350 | 20 mL to 70 mL | 250 mL | ||
| Selective Arteriogram | 300 | 10 mL to 60 mL | 290 mL | |
| 350 | 10 mL to 30 mL | 250 mL | ||
| IA-DSA (head, neck, abdominal, renal, and peripheral vessels) | 140 |
|
250 mL |
2.6 Recommended Dosage for Intraarticular Procedures in Adults
- The recommended doses for intraarticular procedures in adults are shown in Table 6.
- Use passive or active manipulation to disperse the medium throughout the joint space.
| Imaging Procedure | Location | Concentration (mg Iodine/mL) |
Volume to Administer | Double Contrast/Single Contrast |
|---|---|---|---|---|
| Arthrography | Knee | 240 Use single-dose containers or 500 mL Pharmacy Bulk Packages [see Dosage and Administration (2.11)].
|
5 mL to 15 mL | Lower volumes recommended for double-contrast examinations; higher volumes recommended for single-contrast examinations. |
| 300 | 5 mL to 15 mL | |||
| 350 | 5 mL to 10 mL | |||
| Shoulder | 300 | 10 mL | ||
| Temporomandibular | 300 | 0.5 mL to 1 mL |
5.9 Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age
Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast media (ICM) in pediatric patients 0 to 3 years of age.
Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after ICM exposure. Pediatric patients with congenital cardiac conditions may be at the greatest risk given that they often require high doses of contrast during invasive cardiac procedures.
An underactive thyroid during early life may be harmful for cognitive and neurological development and may require thyroid hormone replacement therapy. After exposure to ICM, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates.
5.2 Risks Associated With Parenteral Administration of Omnipaque Oral Solution (5.2 Risks Associated with Parenteral Administration of OMNIPAQUE Oral Solution)
Adverse reactions such as hemolysis may occur if OMNIPAQUE oral solution is administered intravenously or intraarterially due to low osmolality [see Description (11)]. OMNIPAQUE oral solution is for oral use only.
5.1 Risks Associated With Intrathecal Administration of Omnipaque Injection 140 Mg Iodine/ml and 350 Mg Iodine/ml (5.1 Risks Associated with Intrathecal Administration of OMNIPAQUE Injection 140 mg Iodine/mL and 350 mg Iodine/mL)
Use only the iodine concentrations and presentations recommended for intrathecal procedures [see Dosage and Administration (2.2, 2.8)]. Intrathecal administration of OMNIPAQUE of a wrong iodine concentration, even if inadvertent, can cause death, convulsions, seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema.
Warning: Risks Associated With Intrathecal Administration of Omnipaque Injection 140 Mg Iodine/ml and 350 Mg Iodine/ml (WARNING: RISKS ASSOCIATED WITH INTRATHECAL ADMINISTRATION OF OMNIPAQUE INJECTION 140 mg IODINE/mL and 350 mg IODINE/mL)
Use only the OMNIPAQUE iodine concentrations and presentations recommended for intrathecal procedure [see Dosage and Administration (2.2, 2.8)]. Intrathecal administration of OMNIPAQUE of a wrong iodine concentration, even if inadvertent, may cause death, convulsions, seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema [see Warnings and Precautions (5.1)].
2.10 Directions for Use of the 500 Ml Bottles of Omnipaque Injection 300 Mg Iodine/ml and 350 Mg Iodine/ml As Imaging Bulk Package (2.10 Directions for Use of the 500 mL Bottles of OMNIPAQUE Injection 300 mg Iodine/mL and 350 mg Iodine/mL as Imaging Bulk Package)
- OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in 500 mL bottles may be used as an Imaging Bulk Package (IBP) for intravenous use [see Dosage and Administration (2.4, 2.8)].
- When used as an IBP, check the appropriate box on the container label to indicate that the selected container is to be utilized as an Imaging Bulk Package. This container is for use only with an automated contrast injection system, contrast management system, or contrast media transfer set approved or cleared for use with this contrast agent in this IBP. See device labeling for information on devices indicated for use with this IBP and techniques to help assure safe use.
- Use the OMNIPAQUE IBP and 0.9% Sodium Chloride Injection USP in a room designated for radiological procedures that involve administration of a contrast agent.
- Using aseptic technique, penetrate the container closure only one time with a suitable sterile component of the automated contrast injection system, contrast management system, or contrast media transfer set cleared for use with the OMNIPAQUE IBP.
- If 0.9% Sodium Chloride Injection USP is used with the OMNIPAQUE IBP, prepare the 0.9% Sodium Chloride Injection USP sterile port in accordance with the dosage and administration section of its approved prescribing information and affix the saline tag provided with the OMNIPAQUE IBP on the 0.9% Sodium Chloride Injection USP container.
- Once the OMNIPAQUE IBP and 0.9% Sodium Chloride Injection containers are punctured, do not remove them from the work area during the entire period of use. Maintain the OMNIPAQUE IBP in an inverted position such that container contents are in continuous contact with the dispensing set.
- After the container closure is punctured, if the integrity of the OMNIPAQUE IBP, the 0.9% Sodium Chloride Injection, and the delivery system cannot be assured through direct continuous supervision, discard the IBP, 0.9% Sodium Chloride Injection USP, and all associated disposables for the contrast media transfer set.
- A maximum time from initial puncture is 8 hours. Discard any unused portion.
2.11 Directions for Use of the 500 Ml Bottles of Omnipaque Injection 300 Mg Iodine/ml and 350 Mg Iodine/ml As Pharmacy Bulk Package (2.11 Directions for Use of the 500 mL Bottles of OMNIPAQUE Injection 300 mg Iodine/mL and 350 mg Iodine/mL as Pharmacy Bulk Package)
- OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in 500 mL bottles contain many single doses and may be used as a Pharmacy Bulk Package (PBP).
- When used as a PBP, check the appropriate box on the container label to indicate that the selected container is to be utilized as a Pharmacy Bulk Package. This container is not for direct infusion; it is for use in pharmacy admixture service to dispense aliquots using a suitable dispensing set under a laminar flow hood or equivalent clean air compounding area using aseptic technique.
- Penetrate the container closure only one time, utilizing a suitable transfer device and aseptic technique.
- The withdrawal of container contents should be accomplished without delay. However, should this not be possible, a maximum time of 8 hours from initial closure entry is permitted to complete fluid transfer operations.
- Do not remove the container from the aseptic area during the entire 8-hour period.
- The temperature of the container should not exceed 37°C (98.6°F), after the closure has been entered.
- Discard any unused portion after 8 hours from the initial closure entry.
2.12 Directions for Use of the 150 Ml Bottles of Omnipaque Injection 300 Mg Iodine/ml and 350 Mg Iodine/ml With An Automated Contrast Injection System Or Contrast Management System (2.12 Directions for Use of the 150 mL Bottles of OMNIPAQUE Injection 300 mg Iodine/mL and 350 mg Iodine/mL with an Automated Contrast Injection System or Contrast Management System)
- OMNIPAQUE injection 300 mg iodine/mL and OMNIPAQUE injection 350 mg iodine/mL in 150 mL bottles may be used with a contrast media management system cleared for use with OMNIPAQUE injection 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles.
- See device labeling for information on device indications, instructions for use, and techniques to help assure safe use.
- Using aseptic technique, penetrate the container closure only one time with a suitable sterile component of the contrast media management system cleared for use with OMNIPAQUE 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles.
- Once the container is punctured, do not remove the bottle from the work area during the entire period of use.
- Maximum use time is 4 hours after initial puncture.
- Each bottle is for one procedure only. Discard unused portion.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:22.744587 · Updated: 2026-03-14T22:14:57.540523