These Highlights Do Not Include All The Information Needed To Use Zoledronic Acid Injection Safely And Effectively. See Full Prescribing Information For Zoledronic Acid Injection.
164500d7-51cf-438a-8bee-7a7dce76ad7a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Zoledronic Acid Injection is a bisphosphonate indicated for: Treatment and prevention of postmenopausal osteoporosis ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment and prevention of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone in men and women ( 1.5 ) Limitations of Use Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use ( 1.6 )
Indications and Usage
Zoledronic Acid Injection is a bisphosphonate indicated for: Treatment and prevention of postmenopausal osteoporosis ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment and prevention of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone in men and women ( 1.5 ) Limitations of Use Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use ( 1.6 )
Dosage and Administration
Infusion given intravenously over no less than 15 minutes: Treatment of postmenopausal osteoporosis ( 2.2 ); treatment to increase bone mass in men with osteoporosis ( 2.4 ): treatment and prevention of glucocorticoid-induced osteoporosis ( 2.5 ): 5 mg once a year Prevention of postmenopausal osteoporosis: 5 mg once every 2 years ( 2.3 ) Treatment of Paget's disease of bone: a single 5 mg infusion. Patients should receive 1500 mg elemental calcium and 800 international units vitamin D daily ( 2.6 )
Warnings and Precautions
Products Containing Same Active Ingredient: Patients receiving Zometa should not receive Zoledronic Acid Injection ( 5.1 ) Hypocalcemia may worsen during treatment. Patients must be adequately supplemented with calcium and vitamin D ( 5.2 ) Renal Impairment: A single dose should not exceed 5 mg and the duration of infusion should be no less than 15 minutes. Renal toxicity may be greater in patients with underlying renal impairment or with other risk factors, including advanced age or dehydration. Monitor creatinine clearance before each dose ( 2.7 , 5.3 ) Osteonecrosis of the Jaw (ONJ) has been reported. All patients should have a routine oral exam by the prescriber prior to treatment ( 5.4 ) Atypical Femur Fractures have been reported. Patients with thigh or groin pain should be evaluated to rule out a femoral fracture ( 5.5 ) Severe Bone, Joint, and Muscle Pain may occur. Withhold future doses of Zoledronic Acid Injection if severe symptoms occur ( 5.7 )
Contraindications
Zoledronic Acid Injection is contraindicated in patients with the following conditions: Hypocalcemia [see Warnings and Precautions ( 5.2 )]. Creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment due to an increased risk of renal failure [see Warnings and Precautions ( 5.3 )]. Known hypersensitivity to zoledronic acid or any components of Zoledronic Acid Injection. Hypersensitivity reactions including urticaria, angioedema, and anaphylactic reaction/shock have been reported [see Adverse Reactions ( 6.2 )].
Adverse Reactions
The most common adverse reactions (greater than 10%) were pyrexia, myalgia, headache, arthralgia, pain in extremity ( 6.1 ). Other important adverse reactions were flu-like illness, nausea, vomiting, diarrhea ( 6.2 ), and eye inflammation ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
No in vivo drug interaction studies have been performed for Zoledronic Acid Injection. In vitro and ex vivo studies showed low affinity of zoledronic acid for the cellular components of human blood. In vitro mean zoledronic acid protein binding in human plasma ranged from 28% at 200 ng/mL to 53% at 50 ng/mL. In vivo studies showed that zoledronic acid is not metabolized, and is excreted into the urine as the intact drug.
Storage and Handling
Zoledronic Acid Injection is supplied as follows: NDC Zoledronic Acid Injection (0.05 mg per mL) Package Factor 25021-830-82 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 1 bag per carton 25021-830-87 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 5 bags per carton
How Supplied
Zoledronic Acid Injection is supplied as follows: NDC Zoledronic Acid Injection (0.05 mg per mL) Package Factor 25021-830-82 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 1 bag per carton 25021-830-87 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 5 bags per carton
Medication Information
Warnings and Precautions
Products Containing Same Active Ingredient: Patients receiving Zometa should not receive Zoledronic Acid Injection ( 5.1 ) Hypocalcemia may worsen during treatment. Patients must be adequately supplemented with calcium and vitamin D ( 5.2 ) Renal Impairment: A single dose should not exceed 5 mg and the duration of infusion should be no less than 15 minutes. Renal toxicity may be greater in patients with underlying renal impairment or with other risk factors, including advanced age or dehydration. Monitor creatinine clearance before each dose ( 2.7 , 5.3 ) Osteonecrosis of the Jaw (ONJ) has been reported. All patients should have a routine oral exam by the prescriber prior to treatment ( 5.4 ) Atypical Femur Fractures have been reported. Patients with thigh or groin pain should be evaluated to rule out a femoral fracture ( 5.5 ) Severe Bone, Joint, and Muscle Pain may occur. Withhold future doses of Zoledronic Acid Injection if severe symptoms occur ( 5.7 )
Indications and Usage
Zoledronic Acid Injection is a bisphosphonate indicated for: Treatment and prevention of postmenopausal osteoporosis ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment and prevention of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone in men and women ( 1.5 ) Limitations of Use Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use ( 1.6 )
Dosage and Administration
Infusion given intravenously over no less than 15 minutes: Treatment of postmenopausal osteoporosis ( 2.2 ); treatment to increase bone mass in men with osteoporosis ( 2.4 ): treatment and prevention of glucocorticoid-induced osteoporosis ( 2.5 ): 5 mg once a year Prevention of postmenopausal osteoporosis: 5 mg once every 2 years ( 2.3 ) Treatment of Paget's disease of bone: a single 5 mg infusion. Patients should receive 1500 mg elemental calcium and 800 international units vitamin D daily ( 2.6 )
Contraindications
Zoledronic Acid Injection is contraindicated in patients with the following conditions: Hypocalcemia [see Warnings and Precautions ( 5.2 )]. Creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment due to an increased risk of renal failure [see Warnings and Precautions ( 5.3 )]. Known hypersensitivity to zoledronic acid or any components of Zoledronic Acid Injection. Hypersensitivity reactions including urticaria, angioedema, and anaphylactic reaction/shock have been reported [see Adverse Reactions ( 6.2 )].
Adverse Reactions
The most common adverse reactions (greater than 10%) were pyrexia, myalgia, headache, arthralgia, pain in extremity ( 6.1 ). Other important adverse reactions were flu-like illness, nausea, vomiting, diarrhea ( 6.2 ), and eye inflammation ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
No in vivo drug interaction studies have been performed for Zoledronic Acid Injection. In vitro and ex vivo studies showed low affinity of zoledronic acid for the cellular components of human blood. In vitro mean zoledronic acid protein binding in human plasma ranged from 28% at 200 ng/mL to 53% at 50 ng/mL. In vivo studies showed that zoledronic acid is not metabolized, and is excreted into the urine as the intact drug.
Storage and Handling
Zoledronic Acid Injection is supplied as follows: NDC Zoledronic Acid Injection (0.05 mg per mL) Package Factor 25021-830-82 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 1 bag per carton 25021-830-87 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 5 bags per carton
How Supplied
Zoledronic Acid Injection is supplied as follows: NDC Zoledronic Acid Injection (0.05 mg per mL) Package Factor 25021-830-82 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 1 bag per carton 25021-830-87 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) 5 bags per carton
Description
Zoledronic Acid Injection is a bisphosphonate indicated for: Treatment and prevention of postmenopausal osteoporosis ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment and prevention of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone in men and women ( 1.5 ) Limitations of Use Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use ( 1.6 )
Section 42229-5
Re-treatment of Paget's Disease
After a single treatment with Zoledronic Acid Injection in Paget's disease an extended remission period is observed. Specific re-treatment data are not available. However, re-treatment with Zoledronic Acid Injection may be considered in patients who have relapsed, based on increases in serum alkaline phosphatase, or in those patients who failed to achieve normalization of their serum alkaline phosphatase, or in those patients with symptoms, as dictated by medical practice.
Section 42231-1
MEDICATION GUIDE
Zoledronic Acid (ZOE-le-DRON-ik AS-id) Injection
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
| Read the Medication Guide that comes with Zoledronic Acid Injection before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. Talk to your doctor if you have any questions about Zoledronic Acid Injection. |
|
What is the most important information I should know about Zoledronic Acid Injection?
|
| 1. Low calcium levels in your blood (hypocalcemia). Zoledronic Acid Injection may lower the calcium levels in your blood. If you have low blood calcium before you start taking Zoledronic Acid Injection, it may get worse during treatment. Your low blood calcium must be treated before you take Zoledronic Acid Injection. Most people with low blood calcium levels do not have symptoms, but some people may have symptoms. Call your doctor right away if you have symptoms of low blood calcium such as:
|
| Your doctor may prescribe calcium and vitamin D to help prevent low calcium levels in your blood, while you take Zoledronic Acid Injection. Take calcium and vitamin D as your doctor tells you to. 2. Severe kidney problems. Severe kidney problems may happen when you take Zoledronic Acid Injection. Severe kidney problems may lead to hospitalization or kidney dialysis and can be life-threatening. Your risk of kidney problems is higher if you:
|
| You should drink at least 2 glasses of fluid within a few hours before receiving Zoledronic Acid Injection to reduce the risk of kidney problems. 3. Severe jaw bone problems (osteonecrosis). Severe jaw bone problems may happen when you take Zoledronic Acid Injection. Your doctor should examine your mouth before you start Zoledronic Acid Injection. Your doctor may tell you to see your dentist before you start Zoledronic Acid Injection. It is important for you to practice good mouth care during treatment with Zoledronic Acid Injection. 4. Unusual thigh bone fractures. Some people have developed unusual fractures in their thigh bone. Symptoms of a fracture may include new or unusual pain in your hip, groin, or thigh. 5. Bone, joint, or muscle pain. Some people who take bisphosphonates develop severe bone, joint, or muscle pain. Call your doctor right away if you have any of these side effects. |
|
What is Zoledronic Acid Injection?
Zoledronic Acid Injection is a prescription medicine used to:
|
| It is not known how long Zoledronic Acid Injection works for the treatment and prevention of osteoporosis. You should see your doctor regularly to determine if Zoledronic Acid Injection is still right for you. Zoledronic Acid Injection is not for use in children. |
|
Who should not take Zoledronic Acid Injection?
Do not take Zoledronic Acid Injection if you:
|
|
What should I tell my doctor before taking Zoledronic Acid Injection?
Before you start Zoledronic Acid Injection, be sure to talk to your doctor if you:
|
Especially tell your doctor if you are taking:
|
| Ask your doctor or pharmacist for a list of these medicines, if you are not sure. Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist each time you get a new medicine. How will I receive Zoledronic Acid Injection?
|
|
What are the possible side effects of Zoledronic Acid Injection?
Zoledronic Acid Injection may cause serious side effects. The most common side effects of Zoledronic Acid Injection included:
|
| Talk to your doctor about things you can do to help decrease some of these side effects that might happen with a Zoledronic Acid Injection infusion. You may get allergic reactions, such as hives, swelling of your face, lips, tongue, or throat. Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Zoledronic Acid Injection. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. General information about safe and effective use of Zoledronic Acid Injection. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. This Medication Guide summarizes the most important information about Zoledronic Acid Injection. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about Zoledronic Acid Injection that is written for health professionals. What are the ingredients in Zoledronic Acid Injection? Active ingredient: zoledronic acid monohydrate. Inactive ingredients: mannitol and sodium citrate. Brands listed are the trademarks of their respective owners. sagent® Mfd. for SAGENT Pharmaceuticals Schaumburg, IL 60173 (USA) Made in Switzerland ©2025 Sagent Pharmaceuticals Revised: January 2025 This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Section 44425-7
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
Single-use only. Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free, DEHP-free, PVC-free.
The container closure is not made with natural rubber latex.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-830-82
100 mL
Zoledronic Acid Injection
5 mg per 100 mL
(0.05 mg per mL)
Rx only
For Intravenous Infusion
Do not mix with calcium-containing solutions
Infusion time must not be less than 15 minutes
Single Use Only – Discard Unused Portion
10 Overdosage
Clinical experience with acute overdosage of zoledronic acid (Zoledronic Acid Injection) solution for intravenous infusion is limited. Patients who have received doses higher than those recommended should be carefully monitored. Overdosage may cause clinically significant renal impairment, hypocalcemia, hypophosphatemia, and hypomagnesemia. Clinically relevant reductions in serum levels of calcium, phosphorus, and magnesium should be corrected by intravenous administration of calcium gluconate, potassium or sodium phosphate, and magnesium sulfate, respectively.
Single doses of Zoledronic Acid Injection should not exceed 5 mg and the duration of the intravenous infusion should be no less than 15 minutes [see Dosage and Administration (2)].
11 Description
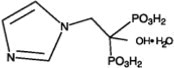
Zoledronic Acid Injection contains zoledronic acid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronic acid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its structural formula is:
Zoledronic acid monohydrate is a white crystalline powder. Its molecular formula is C5H10N2O7P2 • H2O and a molar mass of 290.1 g/Mol. Zoledronic acid monohydrate is highly soluble in 0.1N sodium hydroxide solution, sparingly soluble in water and 0.1N hydrochloric acid, and practically insoluble in organic solvents. The pH of the Zoledronic Acid Injection solution for infusion is approximately 6.0 to 7.0.
Zoledronic Acid Injection is available as a sterile solution in bags for intravenous infusion. One bag with 100 mL solution contains 5.330 mg of zoledronic acid monohydrate, equivalent to 5 mg zoledronic acid on an anhydrous basis.
Inactive Ingredients: 4950 mg of mannitol, USP; and 30 mg of sodium citrate, USP.
8.4 Pediatric Use
Zoledronic Acid Injection is not indicated for use in children.
The safety and effectiveness of zoledronic acid was studied in a one-year active controlled trial of 152 pediatric subjects (74 receiving zoledronic acid). The enrolled population was subjects with severe osteogenesis imperfecta, aged 1 to 17 years, 55% male, 84% Caucasian, with a mean lumbar spine BMD of 0.431 gm/cm2, which is 2.7 standard deviations below the mean for age-matched controls (BMD Z-score of -2.7). At one year, increases in BMD were observed in the zoledronic acid treatment group. However, changes in BMD in individual patients with severe osteogenesis imperfecta did not necessarily correlate with the risk for fracture or the incidence or severity of chronic bone pain. The adverse events observed with zoledronic acid use in children did not raise any new safety findings beyond those previously seen in adults treated for Paget's disease of bone and treatment of osteoporosis including osteonecrosis of the jaw (ONJ) and renal impairment. However, adverse reactions seen more commonly in pediatric patients included pyrexia (61%), arthralgia (26%), hypocalcemia (22%) and headache (22%). These reactions, excluding arthralgia, occurred most frequently within three days after the first infusion and became less common with repeat dosing. No cases of ONJ or renal impairment were observed in this study. Because of long-term retention in bone, Zoledronic Acid Injection should only be used in children if the potential benefit outweighs the potential risk.
Plasma zoledronic acid concentration data was obtained from 10 patients with severe osteogenesis imperfecta (4 in the age group of 3 to 8 years and 6 in the age group of 9 to 17 years) infused with 0.05 mg/kg dose over 30 minutes. Mean Cmax and AUC(0-last) was 167 ng/mL and 220 ng.h/mL respectively. The plasma concentration time profile of zoledronic acid in pediatric patients represent a multi-exponential decline, as observed in adult cancer patients at an approximately equivalent mg/kg dose.
8.5 Geriatric Use
The combined osteoporosis trials included 4863 Zoledronic Acid Injection-treated patients who were at least 65 years of age, while 2101 patients were at least 75 years old. No overall differences in efficacy or safety were observed between patients under 75 years of age with those at least 75 years of age, except that the acute phase reactions occurred less frequently in the older patients.
Of the patients receiving Zoledronic Acid Injection in the osteoporosis study in men, glucocorticoid-induced osteoporosis, and Paget's disease studies, 83, 116, and 132 patients, respectively were 65 years of age or over, while 24, 29, and 68 patients, respectively were at least 75 years of age.
However, because decreased renal function occurs more commonly in the elderly, special care should be taken to monitor renal function.
7.2 Loop Diuretics
Caution should also be exercised when Zoledronic Acid Injection is used in combination with loop diuretics due to an increased risk of hypocalcemia.
4 Contraindications
Zoledronic Acid Injection is contraindicated in patients with the following conditions:
- Hypocalcemia [see Warnings and Precautions (5.2)].
- Creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment due to an increased risk of renal failure [see Warnings and Precautions (5.3)].
- Known hypersensitivity to zoledronic acid or any components of Zoledronic Acid Injection. Hypersensitivity reactions including urticaria, angioedema, and anaphylactic reaction/shock have been reported [see Adverse Reactions (6.2)].
6 Adverse Reactions
The most common adverse reactions (greater than 10%) were pyrexia, myalgia, headache, arthralgia, pain in extremity (6.1). Other important adverse reactions were flu-like illness, nausea, vomiting, diarrhea (6.2), and eye inflammation (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions
No in vivo drug interaction studies have been performed for Zoledronic Acid Injection. In vitro and ex vivo studies showed low affinity of zoledronic acid for the cellular components of human blood. In vitro mean zoledronic acid protein binding in human plasma ranged from 28% at 200 ng/mL to 53% at 50 ng/mL. In vivo studies showed that zoledronic acid is not metabolized, and is excreted into the urine as the intact drug.
7.1 Aminoglycosides
Caution is advised when bisphosphonates, including zoledronic acid, are administered with aminoglycosides, since these agents may have an additive effect to lower serum calcium level for prolonged periods. This effect has not been reported in zoledronic acid clinical trials.
5.3 Renal Impairment
A single dose of Zoledronic Acid Injection should not exceed 5 mg and the duration of infusion should be no less than 15 minutes [see Dosage and Administration (2)].
Zoledronic Acid Injection is contraindicated in patients with creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment [see Contraindications (4)]. If history or physical signs suggest dehydration, Zoledronic Acid Injection therapy should be withheld until normovolemic status has been achieved [see Adverse Reactions (6.2)].
Zoledronic Acid Injection should be used with caution in patients with chronic renal impairment. Acute renal impairment, including renal failure, has been observed following the administration of zoledronic acid, especially in patients with pre-existing renal compromise, advanced age, concomitant nephrotoxic medications, concomitant diuretic therapy, or severe dehydration occurring before or after Zoledronic Acid Injection administration. Acute renal failure (ARF) has been observed in patients after a single administration. Rare reports of hospitalization and/or dialysis or fatal outcome occurred in patients with underlying moderate to severe renal impairment or with any of the risk factors described in this section [see Adverse Reactions (6.2)]. Renal impairment may lead to increased exposure of concomitant medications and/or their metabolites that are primarily renally excreted [see Drug Interactions (7.4)].
Creatinine clearance should be calculated based on actual body weight using Cockcroft-Gault formula before each Zoledronic Acid Injection dose. Transient increase in serum creatinine may be greater in patients with impaired renal function; interim monitoring of creatinine clearance should be performed in at-risk patients. Elderly patients and those receiving diuretic therapy are at increased risk of acute renal failure. These patients should have their fluid status assessed and be appropriately hydrated prior to administration of Zoledronic Acid Injection. Zoledronic Acid Injection should be used with caution with other nephrotoxic drugs [see Drug Interactions (7.3)]. Consider monitoring creatinine clearance in patients at-risk for ARF who are taking concomitant medications that are primarily excreted by the kidney [see Drug Interactions (7.4)].
8.6 Renal Impairment
Zoledronic Acid Injection is contraindicated in patients with creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment. There are no safety or efficacy data to support the adjustment of the Zoledronic Acid Injection dose based on baseline renal function. Therefore, no dosage adjustment is required in patients with a creatinine clearance of greater than or equal to 35 mL/min [see Warnings and Precautions (5.3), Clinical Pharmacology (12.3)]. Risk of acute renal failure may increase with underlying renal disease and dehydration secondary to fever, sepsis, gastrointestinal losses, diuretic therapy, advanced age, etc. [see Adverse Reactions (6.2)].
12.2 Pharmacodynamics
In the osteoporosis treatment trial, the effect of zoledronic acid injection treatment on markers of bone resorption (serum beta-C-telopeptides [b-CTx]) and bone formation (bone specific alkaline phosphatase [BSAP], serum N-terminal propeptide of type I collagen [P1NP]) was evaluated in patients (subsets ranging from 517 to 1246 patients) at periodic intervals. Treatment with a 5 mg annual dose of zoledronic acid injection reduces bone turnover markers to the pre-menopausal range with an approximate 55% reduction in b-CTx, a 29% reduction in BSAP and a 52% reduction in P1NP over 36 months. There was no progressive reduction of bone turnover markers with repeated annual dosing.
12.3 Pharmacokinetics
Pharmacokinetic data in patients with osteoporosis and Paget's disease of bone are not available.
7.3 Nephrotoxic Drugs
Caution is indicated when Zoledronic Acid Injection is used with other potentially nephrotoxic drugs such as nonsteroidal anti-inflammatory drugs.
8.7 Hepatic Impairment
Zoledronic Acid Injection is not metabolized in the liver. No clinical data are available for use of Zoledronic Acid Injection in patients with hepatic impairment.
1 Indications and Usage
Zoledronic Acid Injection is a bisphosphonate indicated for:
- Treatment and prevention of postmenopausal osteoporosis (1.1, 1.2)
- Treatment to increase bone mass in men with osteoporosis (1.3)
- Treatment and prevention of glucocorticoid-induced osteoporosis (1.4)
- Treatment of Paget's disease of bone in men and women (1.5)
Limitations of Use
Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use (1.6)
1.3 Osteoporosis in Men
Zoledronic Acid Injection is indicated for treatment to increase bone mass in men with osteoporosis [see Clinical Studies (14.3)].
2.4 Osteoporosis in Men
The recommended regimen is a 5 mg infusion once a year given intravenously over no less than 15 minutes.
12.1 Mechanism of Action
Zoledronic Acid Injection is a bisphosphonate and acts primarily on bone. It is an inhibitor of osteoclast-mediated bone resorption.
The selective action of bisphosphonates on bone is based on their high affinity for mineralized bone. Intravenously administered zoledronic acid rapidly partitions to bone and localizes preferentially at sites of high bone turnover. The main molecular target of zoledronic acid in the osteoclast is the enzyme farnesyl pyrophosphate synthase. The relatively long duration of action of zoledronic acid is attributable to its high binding affinity to bone mineral.
14.3 Osteoporosis in Men
The efficacy and safety of Zoledronic Acid Injection in men with osteoporosis or significant osteoporosis secondary to hypogonadism, was assessed in a randomized, multicenter, double-blind, active controlled, study of 302 men aged 25 to 86 years (mean age of 64). The duration of the trial was two years. Patients were randomized to either Zoledronic Acid Injection which was administered once annually as a 5 mg dose in 100 mL infused over 15 minutes for a total of up to two doses, or to an oral weekly bisphosphonate (active control) for up to two years. All participants received 1000 mg of elemental calcium plus 800 to 1000 international units of vitamin D supplementation per day.
5.6 Musculoskeletal Pain
In post-marketing experience, severe and occasionally incapacitating bone, joint, and/or muscle pain have been infrequently reported in patients taking bisphosphonates, including Zoledronic Acid Injection. The time to onset of symptoms varied from one day to several months after starting the drug. Consider withholding future Zoledronic Acid Injection treatment if severe symptoms develop. Most patients had relief of symptoms after stopping. A subset had recurrence of symptoms when rechallenged with the same drug or another bisphosphonate [see Adverse Reactions (6.2)].
5.7 Patients With Asthma
While not observed in clinical trials with Zoledronic Acid Injection, there have been reports of bronchoconstriction in aspirin-sensitive patients receiving bisphosphonates. Use Zoledronic Acid Injection with caution in aspirin-sensitive patients.
5 Warnings and Precautions
- Products Containing Same Active Ingredient: Patients receiving Zometa should not receive Zoledronic Acid Injection (5.1)
- Hypocalcemia may worsen during treatment. Patients must be adequately supplemented with calcium and vitamin D (5.2)
- Renal Impairment: A single dose should not exceed 5 mg and the duration of infusion should be no less than 15 minutes. Renal toxicity may be greater in patients with underlying renal impairment or with other risk factors, including advanced age or dehydration. Monitor creatinine clearance before each dose (2.7, 5.3)
- Osteonecrosis of the Jaw (ONJ) has been reported. All patients should have a routine oral exam by the prescriber prior to treatment (5.4)
- Atypical Femur Fractures have been reported. Patients with thigh or groin pain should be evaluated to rule out a femoral fracture (5.5)
- Severe Bone, Joint, and Muscle Pain may occur. Withhold future doses of Zoledronic Acid Injection if severe symptoms occur (5.7)
1.5 Paget's Disease of Bone
Zoledronic Acid Injection is indicated for treatment of Paget's disease of bone in men and women. Treatment is indicated in patients with Paget's disease of bone with elevations in serum alkaline phosphatase of two times or higher than the upper limit of the age-specific normal reference range, or those who are symptomatic, or those at risk for complications from their disease [see Clinical Studies (14.5)].
2 Dosage and Administration
Infusion given intravenously over no less than 15 minutes:
- Treatment of postmenopausal osteoporosis (2.2); treatment to increase bone mass in men with osteoporosis (2.4): treatment and prevention of glucocorticoid-induced osteoporosis (2.5): 5 mg once a year
- Prevention of postmenopausal osteoporosis: 5 mg once every 2 years (2.3)
- Treatment of Paget's disease of bone: a single 5 mg infusion. Patients should receive 1500 mg elemental calcium and 800 international units vitamin D daily (2.6)
2.9 Method of Administration
The Zoledronic Acid Injection infusion time must not be less than 15 minutes given over a constant infusion rate.
The intravenous infusion should be followed by a 10 mL normal saline flush of the intravenous line.
Zoledronic Acid Injection solution for infusion must not be allowed to come in contact with any calcium or other divalent cation-containing solutions, and should be administered as a single intravenous solution through a separate infusion line.
If refrigerated, allow the refrigerated solution to reach room temperature before administration. After opening, the solution is stable for 24 hours at 2°C to 8°C (36°F to 46°F) [see How Supplied/Storage and Handling (16)].
3 Dosage Forms and Strengths
5 mg in a 100 mL ready to infuse solution.
5.4 Osteonecrosis of the Jaw
Osteonecrosis of the jaw (ONJ) has been reported in patients treated with bisphosphonates, including zoledronic acid. Most cases have been in cancer patients treated with intravenous bisphosphonates undergoing dental procedures. Some cases have occurred in patients with postmenopausal osteoporosis treated with either oral or intravenous bisphosphonates. A routine oral examination should be performed by the prescriber prior to initiation of bisphosphonate treatment. A dental examination with appropriate preventive dentistry should be considered prior to treatment with bisphosphonates in patients with a history of concomitant risk factors (e.g., cancer, chemotherapy, angiogenesis inhibitors, radiotherapy, corticosteroids, poor oral hygiene, pre-existing dental disease or infection, anemia, coagulopathy). The risk of ONJ may increase with duration of exposure to bisphosphonates. Concomitant administration of drugs associated with ONJ may increase the risk of developing ONJ.
While on treatment, patients with concomitant risk factors should avoid invasive dental procedures if possible. For patients who develop ONJ while on bisphosphonate therapy, dental surgery may exacerbate the condition. For patients requiring dental procedures, there are no data available to suggest whether discontinuation of bisphosphonate treatment reduces the risk of ONJ. The clinical judgment of the treating physician should guide the management plan of each patient based on individual benefit/risk assessment [see Adverse Reactions (6.1)].
6.2 Post Marketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post approval use of Zoledronic Acid Injection:
Acute Phase Reactions
Fever, headache, flu-like symptoms, nausea, vomiting, diarrhea, arthralgia, and myalgia. Symptoms may be significant and lead to dehydration.
Acute Renal Failure
Acute renal failure requiring hospitalization and/or dialysis or with a fatal outcome have been rarely reported. Increased serum creatinine was reported in patients with 1) underlying renal disease, 2) dehydration secondary to fever, sepsis, gastrointestinal losses, or diuretic therapy, or 3) other risk factors such as advanced age, or concomitant nephrotoxic drugs in the post-infusion period. Transient rise in serum creatinine can be correctable with intravenous fluids.
Allergic Reactions
Allergic reactions with intravenous zoledronic acid including anaphylactic reaction/shock, urticaria, angioedema, Stevens-Johnson syndrome, toxic epidermal necrolysis, and bronchoconstriction have been reported.
Asthma Exacerbations
Asthma exacerbations have been reported.
Hypocalcemia
Hypocalcemia has been reported.
Hypophosphatemia
Hypophosphatemia has been reported.
Osteonecrosis of the Jaw
Osteonecrosis of the jaw has been reported.
Osteonecrosis of other bones
Cases of osteonecrosis of other bones (including femur, hip, knee, ankle, wrist and humerus) have been reported; causality has not been determined in the population treated with zoledronic acid.
Ocular Adverse Events
Cases of the following events have been reported: conjunctivitis, iritis, iridocyclitis, uveitis, episcleritis, scleritis and orbital inflammation/edema.
Other
Hypotension in patients with underlying risk factors has been reported.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.6 Important Limitations of Use
The safety and effectiveness of Zoledronic Acid Injection for the treatment of osteoporosis is based on clinical data of three years duration. The optimal duration of use has not been determined. All patients on bisphosphonate therapy should have the need for continued therapy re-evaluated on a periodic basis. Patients at low-risk for fracture should be considered for drug discontinuation after 3 to 5 years of use. Patients who discontinue therapy should have their risk for fracture re-evaluated periodically.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
Zoledronic Acid Injection is supplied as follows:
| NDC | Zoledronic Acid Injection (0.05 mg per mL) | Package Factor |
| 25021-830-82 | 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) | 1 bag per carton |
| 25021-830-87 | 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) | 5 bags per carton |
1.4 Glucocorticoid Induced Osteoporosis
Zoledronic Acid Injection is indicated for the treatment and prevention of glucocorticoid-induced osteoporosis in men and women who are either initiating or continuing systemic glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and who are expected to remain on glucocorticoids for at least 12 months [see Clinical Studies (14.4)].
5.2 Hypocalcemia and Mineral Metabolism
Pre-existing hypocalcemia and disturbances of mineral metabolism (e.g., hypoparathyroidism, thyroid surgery, parathyroid surgery; malabsorption syndromes, excision of small intestine) must be effectively treated before initiating therapy with Zoledronic Acid Injection. Clinical monitoring of calcium and mineral levels (phosphorus and magnesium) is highly recommended for these patients [see Contraindications (4)].
Hypocalcemia following Zoledronic Acid Injection administration is a significant risk in Paget's disease. All patients should be instructed about the symptoms of hypocalcemia and the importance of calcium and vitamin D supplementation in maintaining serum calcium levels [see Dosage and Administration (2.8), Adverse Reactions (6.1), Patient Counseling Information (17)].
All osteoporosis patients should be instructed on the importance of calcium and vitamin D supplementation in maintaining serum calcium levels [see Dosage and Administration (2.8), Adverse Reactions (6.1), Patient Counseling Information (17)].
2.6 Treatment of Paget's Disease of Bone
The recommended dose is a 5 mg infusion. The infusion time must not be less than 15 minutes given over a constant infusion rate.
14.5 Treatment of Paget's Disease of Bone
Zoledronic Acid Injection was studied in male and female patients with moderate to severe Paget's disease of bone, defined as serum alkaline phosphatase level at least twice the upper limit of the age-specific normal reference range at the time of study entry. Diagnosis was confirmed by radiographic evidence.
The efficacy of one infusion of 5 mg Zoledronic Acid Injection vs. oral daily doses of 30 mg risedronate for 2 months was demonstrated in two identically designed 6-month randomized, double-blind trials. The mean age of patients in the two trials was 70. Ninety-three percent (93%) of patients were Caucasian. Therapeutic response was defined as either normalization of serum alkaline phosphatase (SAP) or a reduction of at least 75% from baseline in total SAP excess at the end of 6 months. SAP excess was defined as the difference between the measured level and midpoint of normal range.
In both trials Zoledronic Acid Injection demonstrated a superior and more rapid therapeutic response compared with risedronate and returned more patients to normal levels of bone turnover, as evidenced by biochemical markers of formation (SAP, serum N-terminal propeptide of type I collagen [P1NP]) and resorption (serum CTx 1 [cross-linked C-telopeptides of type I collagen] and urine α-CTx).
The 6-month combined data from both trials showed that 96% (169/176) of Zoledronic Acid Injection-treated patients achieved a therapeutic response as compared with 74% (127/171) of patients treated with risedronate. Most Zoledronic Acid Injection patients achieved a therapeutic response by the Day 63 visit. In addition, at 6 months, 89% (156/176) of Zoledronic Acid Injection-treated patients achieved normalization of SAP levels, compared to 58% (99/171) of patients treated with risedronate (p<0.0001) (see Figure 2).
Figure 2. Therapeutic Response/Serum Alkaline Phosphatase (SAP) Normalization Over Time
The therapeutic response to Zoledronic Acid Injection was similar across demographic and disease-severity groups defined by gender, age, previous bisphosphonate use, and disease severity. At 6 months, the percentage of Zoledronic Acid Injection-treated patients who achieved therapeutic response was 97% and 95%, respectively, in each of the baseline disease severity subgroups (baseline SAP less than 3xULN, greater than or equal to 3xULN) compared to 75% and 74%, respectively, for the same disease severity subgroups of risedronate-treated patients.
In patients who had previously received treatment with oral bisphosphonates, therapeutic response rates were 96% and 55% for Zoledronic Acid Injection and risedronate, respectively. The comparatively low risedronate response was due to the low response rate (7/23, 30%) in patients previously treated with risedronate. In patients naïve to previous treatment, a greater therapeutic response was also observed with Zoledronic Acid Injection (98%) relative to risedronate (86%). In patients with symptomatic pain at screening, therapeutic response rates were 94% and 70% for Zoledronic Acid Injection and risedronate respectively. For patients without pain at screening, therapeutic response rates were 100% and 82% for Zoledronic Acid Injection and risedronate respectively.
Bone histology was evaluated in 7 patients with Paget's disease 6 months after being treated with Zoledronic Acid Injection 5 mg. Bone biopsy results showed bone of normal quality with no evidence of impaired bone remodeling and no evidence of mineralization defect.
2.1 Important Administration Instructions
Zoledronic Acid Injection must be administered as an intravenous infusion over no less than 15 minutes.
- Patients must be appropriately hydrated prior to administration of Zoledronic Acid Injection [see Warnings and Precautions (5.3)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Intravenous infusion should be followed by a 10 mL normal saline flush of the intravenous line.
- Administration of acetaminophen following Zoledronic Acid Injection administration may reduce the incidence of acute-phase reaction symptoms.
Caution: After removing the overwrap check for minute leaks by squeezing the inner bag firmly. If leaks are found, discard container as sterility may be compromised. Use only if solution is clear and the container is undamaged.
Preparation for Administration:
- Close flow control clamp of administration set.
- Remove cover from port at bottom of container.
- Insert piercing pin of administration set into port with a twisting motion until the pin is firmly seated. NOTE: See full directions on administration set carton.
- Suspend container from hanger.
- Squeeze and release drip chamber to establish proper fluid level in chamber during infusion of Zoledronic Acid Injection.
- Open flow control clamp to expel air from set. Close clamp.
- Regulate rate of administration with flow control clamp.
2.8 Calcium and Vitamin D Supplementation
- Instruct patients being treated for Paget's disease of bone on the importance of calcium and vitamin D supplementation in maintaining serum calcium levels, and on the symptoms of hypocalcemia. All patients should take 1500 mg elemental calcium daily in divided doses (750 mg two times a day, or 500 mg three times a day) and 800 international units vitamin D daily, particularly in the 2 weeks following Zoledronic Acid Injection administration [see Warnings and Precautions (5.2)].
- Instruct patients being treated for osteoporosis to take supplemental calcium and vitamin D if their dietary intake is inadequate. An average of at least 1200 mg calcium and 800 to 1000 international units vitamin D daily is recommended.
7.4 Drugs Primarily Excreted By the Kidney
Renal impairment has been observed following the administration of zoledronic acid in patients with pre-existing renal compromise or other risk factors [see Warnings and Precautions (5.3)]. In patients with renal impairment, the exposure to concomitant medications that are primarily renally excreted (e.g., digoxin) may increase. Consider monitoring serum creatinine in patients at risk for renal impairment who are taking concomitant medications that are primarily excreted by the kidney.
5.1 Drug Products With Same Active Ingredient
Zoledronic Acid Injection contains the same active ingredient found in Zometa, used for oncology indications, and a patient being treated with Zometa should not be treated with Zoledronic Acid Injection.
14.2 Prevention of Postmenopausal Osteoporosis
The efficacy and safety of Zoledronic Acid Injection in postmenopausal women with osteopenia (low bone mass) was assessed in a 2-year randomized, multi-center, double-blind, placebo-controlled study of 581 postmenopausal women aged greater than or equal to 45 years, who were stratified by years since menopause: Stratum I women less than 5 years from menopause (n = 224); Stratum II women greater than or equal to 5 years from menopause (n = 357). Patients within Stratum I and II were randomized to one of three treatment groups: (1) Zoledronic Acid Injection given at randomization and at Month 12 (n = 77) in Stratum I and (n = 121) in Stratum II; (2) Zoledronic Acid Injection given at randomization and placebo at Month 12 (n = 70) in Stratum I and (n = 111) in Stratum II; and (3) Placebo given at randomization and Month 12 (n = 202). Zoledronic Acid Injection was administered as a single 5 mg dose in 100 mL solution infused over at least 15 minutes. All women received 500 to 1200 mg elemental calcium plus 400 to 800 international units vitamin D supplementation per day. The primary efficacy variable was the percent change of BMD at 24 Months relative to baseline.
1.1 Treatment of Osteoporosis in Postmenopausal Women
Zoledronic Acid Injection is indicated for treatment of osteoporosis in postmenopausal women. In postmenopausal women with osteoporosis, diagnosed by bone mineral density (BMD) or prevalent vertebral fracture, Zoledronic Acid Injection reduces the incidence of fractures (hip, vertebral and non-vertebral osteoporosis-related fractures). In patients at high risk of fracture, defined as a recent low-trauma hip fracture, zoledronic acid injection reduces the incidence of new clinical fractures [see Clinical Studies (14.1)].
2.2 Treatment of Osteoporosis in Postmenopausal Women
The recommended regimen is a 5 mg infusion once a year given intravenously over no less than 15 minutes.
1.2 Prevention of Osteoporosis in Postmenopausal Women
Zoledronic Acid Injection is indicated for prevention of osteoporosis in postmenopausal women [see Clinical Studies (14.2)].
2.3 Prevention of Osteoporosis in Postmenopausal Women
The recommended regimen is a 5 mg infusion given once every 2 years intravenously over no less than 15 minutes.
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fractures
Atypical, low-energy, or low trauma fractures of the femoral shaft have been reported in bisphosphonate-treated patients. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution. Causality has not been established as these fractures also occur in osteoporotic patients who have not been treated with bisphosphonates.
Atypical femur fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g., prednisone) at the time of fracture.
Any patient with a history of bisphosphonate exposure who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patients presenting with an atypical femur fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of bisphosphonate therapy should be considered, pending a risk/benefit assessment, on an individual basis.
2.5 Treatment and Prevention of Glucocorticoid Induced Osteoporosis
The recommended regimen is a 5 mg infusion once a year given intravenously over no less than 15 minutes.
2.7 Laboratory Testing and Oral Examination Prior to Administration
- Prior to administration of each dose of Zoledronic Acid Injection, obtain a serum creatinine and creatinine clearance should be calculated based on actual body weight using Cockcroft-Gault formula before each Zoledronic Acid Injection dose. Zoledronic Acid Injection is contraindicated in patients with creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment. A 5 mg dose of Zoledronic Acid Injection administered intravenously is recommended for patients with creatinine clearance greater than or equal to 35 mL/min. There are no safety or efficacy data to support the adjustment of the Zoledronic Acid Injection dose based on baseline renal function. Therefore, no dose adjustment is required in patients with CrCl greater than or equal to 35 mL/min [see Contraindications (4), Warnings and Precautions (5.3)].
- A routine oral examination should be performed by the prescriber prior to initiation of Zoledronic Acid Injection treatment [see Warnings and Precautions (5.4)].
14.4 Treatment and Prevention of Glucocorticoid Induced Osteoporosis
The efficacy and safety of Zoledronic Acid Injection to prevent and treat glucocorticoid-induced osteoporosis (GIO) was assessed in a randomized, multicenter, double-blind, stratified, active controlled study of 833 men and women aged 18 to 85 years (mean age of 54.4 years) treated with greater than or equal to 7.5 mg/day oral prednisone (or equivalent). Patients were stratified according to the duration of their pre-study corticosteroid therapy: less than or equal to 3 months prior to randomization (prevention subpopulation), and greater than 3 months prior to randomization (treatment subpopulation). The duration of the trial was one year. Patients were randomized to either Zoledronic Acid Injection which was administered once as a 5 mg dose in 100 mL infused over 15 minutes, or to an oral daily bisphosphonate (active control) for one year. All participants received 1000 mg of elemental calcium plus 400 to 1000 international units of vitamin D supplementation per day.
Structured Label Content
Section 42229-5 (42229-5)
Re-treatment of Paget's Disease
After a single treatment with Zoledronic Acid Injection in Paget's disease an extended remission period is observed. Specific re-treatment data are not available. However, re-treatment with Zoledronic Acid Injection may be considered in patients who have relapsed, based on increases in serum alkaline phosphatase, or in those patients who failed to achieve normalization of their serum alkaline phosphatase, or in those patients with symptoms, as dictated by medical practice.
Section 42231-1 (42231-1)
MEDICATION GUIDE
Zoledronic Acid (ZOE-le-DRON-ik AS-id) Injection
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
| Read the Medication Guide that comes with Zoledronic Acid Injection before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. Talk to your doctor if you have any questions about Zoledronic Acid Injection. |
|
What is the most important information I should know about Zoledronic Acid Injection?
|
| 1. Low calcium levels in your blood (hypocalcemia). Zoledronic Acid Injection may lower the calcium levels in your blood. If you have low blood calcium before you start taking Zoledronic Acid Injection, it may get worse during treatment. Your low blood calcium must be treated before you take Zoledronic Acid Injection. Most people with low blood calcium levels do not have symptoms, but some people may have symptoms. Call your doctor right away if you have symptoms of low blood calcium such as:
|
| Your doctor may prescribe calcium and vitamin D to help prevent low calcium levels in your blood, while you take Zoledronic Acid Injection. Take calcium and vitamin D as your doctor tells you to. 2. Severe kidney problems. Severe kidney problems may happen when you take Zoledronic Acid Injection. Severe kidney problems may lead to hospitalization or kidney dialysis and can be life-threatening. Your risk of kidney problems is higher if you:
|
| You should drink at least 2 glasses of fluid within a few hours before receiving Zoledronic Acid Injection to reduce the risk of kidney problems. 3. Severe jaw bone problems (osteonecrosis). Severe jaw bone problems may happen when you take Zoledronic Acid Injection. Your doctor should examine your mouth before you start Zoledronic Acid Injection. Your doctor may tell you to see your dentist before you start Zoledronic Acid Injection. It is important for you to practice good mouth care during treatment with Zoledronic Acid Injection. 4. Unusual thigh bone fractures. Some people have developed unusual fractures in their thigh bone. Symptoms of a fracture may include new or unusual pain in your hip, groin, or thigh. 5. Bone, joint, or muscle pain. Some people who take bisphosphonates develop severe bone, joint, or muscle pain. Call your doctor right away if you have any of these side effects. |
|
What is Zoledronic Acid Injection?
Zoledronic Acid Injection is a prescription medicine used to:
|
| It is not known how long Zoledronic Acid Injection works for the treatment and prevention of osteoporosis. You should see your doctor regularly to determine if Zoledronic Acid Injection is still right for you. Zoledronic Acid Injection is not for use in children. |
|
Who should not take Zoledronic Acid Injection?
Do not take Zoledronic Acid Injection if you:
|
|
What should I tell my doctor before taking Zoledronic Acid Injection?
Before you start Zoledronic Acid Injection, be sure to talk to your doctor if you:
|
Especially tell your doctor if you are taking:
|
| Ask your doctor or pharmacist for a list of these medicines, if you are not sure. Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist each time you get a new medicine. How will I receive Zoledronic Acid Injection?
|
|
What are the possible side effects of Zoledronic Acid Injection?
Zoledronic Acid Injection may cause serious side effects. The most common side effects of Zoledronic Acid Injection included:
|
| Talk to your doctor about things you can do to help decrease some of these side effects that might happen with a Zoledronic Acid Injection infusion. You may get allergic reactions, such as hives, swelling of your face, lips, tongue, or throat. Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Zoledronic Acid Injection. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. General information about safe and effective use of Zoledronic Acid Injection. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. This Medication Guide summarizes the most important information about Zoledronic Acid Injection. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about Zoledronic Acid Injection that is written for health professionals. What are the ingredients in Zoledronic Acid Injection? Active ingredient: zoledronic acid monohydrate. Inactive ingredients: mannitol and sodium citrate. Brands listed are the trademarks of their respective owners. sagent® Mfd. for SAGENT Pharmaceuticals Schaumburg, IL 60173 (USA) Made in Switzerland ©2025 Sagent Pharmaceuticals Revised: January 2025 This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Section 44425-7 (44425-7)
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
Single-use only. Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free, DEHP-free, PVC-free.
The container closure is not made with natural rubber latex.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-830-82
100 mL
Zoledronic Acid Injection
5 mg per 100 mL
(0.05 mg per mL)
Rx only
For Intravenous Infusion
Do not mix with calcium-containing solutions
Infusion time must not be less than 15 minutes
Single Use Only – Discard Unused Portion
10 Overdosage (10 OVERDOSAGE)
Clinical experience with acute overdosage of zoledronic acid (Zoledronic Acid Injection) solution for intravenous infusion is limited. Patients who have received doses higher than those recommended should be carefully monitored. Overdosage may cause clinically significant renal impairment, hypocalcemia, hypophosphatemia, and hypomagnesemia. Clinically relevant reductions in serum levels of calcium, phosphorus, and magnesium should be corrected by intravenous administration of calcium gluconate, potassium or sodium phosphate, and magnesium sulfate, respectively.
Single doses of Zoledronic Acid Injection should not exceed 5 mg and the duration of the intravenous infusion should be no less than 15 minutes [see Dosage and Administration (2)].
11 Description (11 DESCRIPTION)
Zoledronic Acid Injection contains zoledronic acid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronic acid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its structural formula is:
Zoledronic acid monohydrate is a white crystalline powder. Its molecular formula is C5H10N2O7P2 • H2O and a molar mass of 290.1 g/Mol. Zoledronic acid monohydrate is highly soluble in 0.1N sodium hydroxide solution, sparingly soluble in water and 0.1N hydrochloric acid, and practically insoluble in organic solvents. The pH of the Zoledronic Acid Injection solution for infusion is approximately 6.0 to 7.0.
Zoledronic Acid Injection is available as a sterile solution in bags for intravenous infusion. One bag with 100 mL solution contains 5.330 mg of zoledronic acid monohydrate, equivalent to 5 mg zoledronic acid on an anhydrous basis.
Inactive Ingredients: 4950 mg of mannitol, USP; and 30 mg of sodium citrate, USP.
8.4 Pediatric Use
Zoledronic Acid Injection is not indicated for use in children.
The safety and effectiveness of zoledronic acid was studied in a one-year active controlled trial of 152 pediatric subjects (74 receiving zoledronic acid). The enrolled population was subjects with severe osteogenesis imperfecta, aged 1 to 17 years, 55% male, 84% Caucasian, with a mean lumbar spine BMD of 0.431 gm/cm2, which is 2.7 standard deviations below the mean for age-matched controls (BMD Z-score of -2.7). At one year, increases in BMD were observed in the zoledronic acid treatment group. However, changes in BMD in individual patients with severe osteogenesis imperfecta did not necessarily correlate with the risk for fracture or the incidence or severity of chronic bone pain. The adverse events observed with zoledronic acid use in children did not raise any new safety findings beyond those previously seen in adults treated for Paget's disease of bone and treatment of osteoporosis including osteonecrosis of the jaw (ONJ) and renal impairment. However, adverse reactions seen more commonly in pediatric patients included pyrexia (61%), arthralgia (26%), hypocalcemia (22%) and headache (22%). These reactions, excluding arthralgia, occurred most frequently within three days after the first infusion and became less common with repeat dosing. No cases of ONJ or renal impairment were observed in this study. Because of long-term retention in bone, Zoledronic Acid Injection should only be used in children if the potential benefit outweighs the potential risk.
Plasma zoledronic acid concentration data was obtained from 10 patients with severe osteogenesis imperfecta (4 in the age group of 3 to 8 years and 6 in the age group of 9 to 17 years) infused with 0.05 mg/kg dose over 30 minutes. Mean Cmax and AUC(0-last) was 167 ng/mL and 220 ng.h/mL respectively. The plasma concentration time profile of zoledronic acid in pediatric patients represent a multi-exponential decline, as observed in adult cancer patients at an approximately equivalent mg/kg dose.
8.5 Geriatric Use
The combined osteoporosis trials included 4863 Zoledronic Acid Injection-treated patients who were at least 65 years of age, while 2101 patients were at least 75 years old. No overall differences in efficacy or safety were observed between patients under 75 years of age with those at least 75 years of age, except that the acute phase reactions occurred less frequently in the older patients.
Of the patients receiving Zoledronic Acid Injection in the osteoporosis study in men, glucocorticoid-induced osteoporosis, and Paget's disease studies, 83, 116, and 132 patients, respectively were 65 years of age or over, while 24, 29, and 68 patients, respectively were at least 75 years of age.
However, because decreased renal function occurs more commonly in the elderly, special care should be taken to monitor renal function.
7.2 Loop Diuretics
Caution should also be exercised when Zoledronic Acid Injection is used in combination with loop diuretics due to an increased risk of hypocalcemia.
4 Contraindications (4 CONTRAINDICATIONS)
Zoledronic Acid Injection is contraindicated in patients with the following conditions:
- Hypocalcemia [see Warnings and Precautions (5.2)].
- Creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment due to an increased risk of renal failure [see Warnings and Precautions (5.3)].
- Known hypersensitivity to zoledronic acid or any components of Zoledronic Acid Injection. Hypersensitivity reactions including urticaria, angioedema, and anaphylactic reaction/shock have been reported [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (greater than 10%) were pyrexia, myalgia, headache, arthralgia, pain in extremity (6.1). Other important adverse reactions were flu-like illness, nausea, vomiting, diarrhea (6.2), and eye inflammation (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions (7 DRUG INTERACTIONS)
No in vivo drug interaction studies have been performed for Zoledronic Acid Injection. In vitro and ex vivo studies showed low affinity of zoledronic acid for the cellular components of human blood. In vitro mean zoledronic acid protein binding in human plasma ranged from 28% at 200 ng/mL to 53% at 50 ng/mL. In vivo studies showed that zoledronic acid is not metabolized, and is excreted into the urine as the intact drug.
7.1 Aminoglycosides
Caution is advised when bisphosphonates, including zoledronic acid, are administered with aminoglycosides, since these agents may have an additive effect to lower serum calcium level for prolonged periods. This effect has not been reported in zoledronic acid clinical trials.
5.3 Renal Impairment
A single dose of Zoledronic Acid Injection should not exceed 5 mg and the duration of infusion should be no less than 15 minutes [see Dosage and Administration (2)].
Zoledronic Acid Injection is contraindicated in patients with creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment [see Contraindications (4)]. If history or physical signs suggest dehydration, Zoledronic Acid Injection therapy should be withheld until normovolemic status has been achieved [see Adverse Reactions (6.2)].
Zoledronic Acid Injection should be used with caution in patients with chronic renal impairment. Acute renal impairment, including renal failure, has been observed following the administration of zoledronic acid, especially in patients with pre-existing renal compromise, advanced age, concomitant nephrotoxic medications, concomitant diuretic therapy, or severe dehydration occurring before or after Zoledronic Acid Injection administration. Acute renal failure (ARF) has been observed in patients after a single administration. Rare reports of hospitalization and/or dialysis or fatal outcome occurred in patients with underlying moderate to severe renal impairment or with any of the risk factors described in this section [see Adverse Reactions (6.2)]. Renal impairment may lead to increased exposure of concomitant medications and/or their metabolites that are primarily renally excreted [see Drug Interactions (7.4)].
Creatinine clearance should be calculated based on actual body weight using Cockcroft-Gault formula before each Zoledronic Acid Injection dose. Transient increase in serum creatinine may be greater in patients with impaired renal function; interim monitoring of creatinine clearance should be performed in at-risk patients. Elderly patients and those receiving diuretic therapy are at increased risk of acute renal failure. These patients should have their fluid status assessed and be appropriately hydrated prior to administration of Zoledronic Acid Injection. Zoledronic Acid Injection should be used with caution with other nephrotoxic drugs [see Drug Interactions (7.3)]. Consider monitoring creatinine clearance in patients at-risk for ARF who are taking concomitant medications that are primarily excreted by the kidney [see Drug Interactions (7.4)].
8.6 Renal Impairment
Zoledronic Acid Injection is contraindicated in patients with creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment. There are no safety or efficacy data to support the adjustment of the Zoledronic Acid Injection dose based on baseline renal function. Therefore, no dosage adjustment is required in patients with a creatinine clearance of greater than or equal to 35 mL/min [see Warnings and Precautions (5.3), Clinical Pharmacology (12.3)]. Risk of acute renal failure may increase with underlying renal disease and dehydration secondary to fever, sepsis, gastrointestinal losses, diuretic therapy, advanced age, etc. [see Adverse Reactions (6.2)].
12.2 Pharmacodynamics
In the osteoporosis treatment trial, the effect of zoledronic acid injection treatment on markers of bone resorption (serum beta-C-telopeptides [b-CTx]) and bone formation (bone specific alkaline phosphatase [BSAP], serum N-terminal propeptide of type I collagen [P1NP]) was evaluated in patients (subsets ranging from 517 to 1246 patients) at periodic intervals. Treatment with a 5 mg annual dose of zoledronic acid injection reduces bone turnover markers to the pre-menopausal range with an approximate 55% reduction in b-CTx, a 29% reduction in BSAP and a 52% reduction in P1NP over 36 months. There was no progressive reduction of bone turnover markers with repeated annual dosing.
12.3 Pharmacokinetics
Pharmacokinetic data in patients with osteoporosis and Paget's disease of bone are not available.
7.3 Nephrotoxic Drugs
Caution is indicated when Zoledronic Acid Injection is used with other potentially nephrotoxic drugs such as nonsteroidal anti-inflammatory drugs.
8.7 Hepatic Impairment
Zoledronic Acid Injection is not metabolized in the liver. No clinical data are available for use of Zoledronic Acid Injection in patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Zoledronic Acid Injection is a bisphosphonate indicated for:
- Treatment and prevention of postmenopausal osteoporosis (1.1, 1.2)
- Treatment to increase bone mass in men with osteoporosis (1.3)
- Treatment and prevention of glucocorticoid-induced osteoporosis (1.4)
- Treatment of Paget's disease of bone in men and women (1.5)
Limitations of Use
Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use (1.6)
1.3 Osteoporosis in Men
Zoledronic Acid Injection is indicated for treatment to increase bone mass in men with osteoporosis [see Clinical Studies (14.3)].
2.4 Osteoporosis in Men
The recommended regimen is a 5 mg infusion once a year given intravenously over no less than 15 minutes.
12.1 Mechanism of Action
Zoledronic Acid Injection is a bisphosphonate and acts primarily on bone. It is an inhibitor of osteoclast-mediated bone resorption.
The selective action of bisphosphonates on bone is based on their high affinity for mineralized bone. Intravenously administered zoledronic acid rapidly partitions to bone and localizes preferentially at sites of high bone turnover. The main molecular target of zoledronic acid in the osteoclast is the enzyme farnesyl pyrophosphate synthase. The relatively long duration of action of zoledronic acid is attributable to its high binding affinity to bone mineral.
14.3 Osteoporosis in Men
The efficacy and safety of Zoledronic Acid Injection in men with osteoporosis or significant osteoporosis secondary to hypogonadism, was assessed in a randomized, multicenter, double-blind, active controlled, study of 302 men aged 25 to 86 years (mean age of 64). The duration of the trial was two years. Patients were randomized to either Zoledronic Acid Injection which was administered once annually as a 5 mg dose in 100 mL infused over 15 minutes for a total of up to two doses, or to an oral weekly bisphosphonate (active control) for up to two years. All participants received 1000 mg of elemental calcium plus 800 to 1000 international units of vitamin D supplementation per day.
5.6 Musculoskeletal Pain
In post-marketing experience, severe and occasionally incapacitating bone, joint, and/or muscle pain have been infrequently reported in patients taking bisphosphonates, including Zoledronic Acid Injection. The time to onset of symptoms varied from one day to several months after starting the drug. Consider withholding future Zoledronic Acid Injection treatment if severe symptoms develop. Most patients had relief of symptoms after stopping. A subset had recurrence of symptoms when rechallenged with the same drug or another bisphosphonate [see Adverse Reactions (6.2)].
5.7 Patients With Asthma (5.7 Patients with Asthma)
While not observed in clinical trials with Zoledronic Acid Injection, there have been reports of bronchoconstriction in aspirin-sensitive patients receiving bisphosphonates. Use Zoledronic Acid Injection with caution in aspirin-sensitive patients.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Products Containing Same Active Ingredient: Patients receiving Zometa should not receive Zoledronic Acid Injection (5.1)
- Hypocalcemia may worsen during treatment. Patients must be adequately supplemented with calcium and vitamin D (5.2)
- Renal Impairment: A single dose should not exceed 5 mg and the duration of infusion should be no less than 15 minutes. Renal toxicity may be greater in patients with underlying renal impairment or with other risk factors, including advanced age or dehydration. Monitor creatinine clearance before each dose (2.7, 5.3)
- Osteonecrosis of the Jaw (ONJ) has been reported. All patients should have a routine oral exam by the prescriber prior to treatment (5.4)
- Atypical Femur Fractures have been reported. Patients with thigh or groin pain should be evaluated to rule out a femoral fracture (5.5)
- Severe Bone, Joint, and Muscle Pain may occur. Withhold future doses of Zoledronic Acid Injection if severe symptoms occur (5.7)
1.5 Paget's Disease of Bone
Zoledronic Acid Injection is indicated for treatment of Paget's disease of bone in men and women. Treatment is indicated in patients with Paget's disease of bone with elevations in serum alkaline phosphatase of two times or higher than the upper limit of the age-specific normal reference range, or those who are symptomatic, or those at risk for complications from their disease [see Clinical Studies (14.5)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Infusion given intravenously over no less than 15 minutes:
- Treatment of postmenopausal osteoporosis (2.2); treatment to increase bone mass in men with osteoporosis (2.4): treatment and prevention of glucocorticoid-induced osteoporosis (2.5): 5 mg once a year
- Prevention of postmenopausal osteoporosis: 5 mg once every 2 years (2.3)
- Treatment of Paget's disease of bone: a single 5 mg infusion. Patients should receive 1500 mg elemental calcium and 800 international units vitamin D daily (2.6)
2.9 Method of Administration
The Zoledronic Acid Injection infusion time must not be less than 15 minutes given over a constant infusion rate.
The intravenous infusion should be followed by a 10 mL normal saline flush of the intravenous line.
Zoledronic Acid Injection solution for infusion must not be allowed to come in contact with any calcium or other divalent cation-containing solutions, and should be administered as a single intravenous solution through a separate infusion line.
If refrigerated, allow the refrigerated solution to reach room temperature before administration. After opening, the solution is stable for 24 hours at 2°C to 8°C (36°F to 46°F) [see How Supplied/Storage and Handling (16)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
5 mg in a 100 mL ready to infuse solution.
5.4 Osteonecrosis of the Jaw
Osteonecrosis of the jaw (ONJ) has been reported in patients treated with bisphosphonates, including zoledronic acid. Most cases have been in cancer patients treated with intravenous bisphosphonates undergoing dental procedures. Some cases have occurred in patients with postmenopausal osteoporosis treated with either oral or intravenous bisphosphonates. A routine oral examination should be performed by the prescriber prior to initiation of bisphosphonate treatment. A dental examination with appropriate preventive dentistry should be considered prior to treatment with bisphosphonates in patients with a history of concomitant risk factors (e.g., cancer, chemotherapy, angiogenesis inhibitors, radiotherapy, corticosteroids, poor oral hygiene, pre-existing dental disease or infection, anemia, coagulopathy). The risk of ONJ may increase with duration of exposure to bisphosphonates. Concomitant administration of drugs associated with ONJ may increase the risk of developing ONJ.
While on treatment, patients with concomitant risk factors should avoid invasive dental procedures if possible. For patients who develop ONJ while on bisphosphonate therapy, dental surgery may exacerbate the condition. For patients requiring dental procedures, there are no data available to suggest whether discontinuation of bisphosphonate treatment reduces the risk of ONJ. The clinical judgment of the treating physician should guide the management plan of each patient based on individual benefit/risk assessment [see Adverse Reactions (6.1)].
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post approval use of Zoledronic Acid Injection:
Acute Phase Reactions
Fever, headache, flu-like symptoms, nausea, vomiting, diarrhea, arthralgia, and myalgia. Symptoms may be significant and lead to dehydration.
Acute Renal Failure
Acute renal failure requiring hospitalization and/or dialysis or with a fatal outcome have been rarely reported. Increased serum creatinine was reported in patients with 1) underlying renal disease, 2) dehydration secondary to fever, sepsis, gastrointestinal losses, or diuretic therapy, or 3) other risk factors such as advanced age, or concomitant nephrotoxic drugs in the post-infusion period. Transient rise in serum creatinine can be correctable with intravenous fluids.
Allergic Reactions
Allergic reactions with intravenous zoledronic acid including anaphylactic reaction/shock, urticaria, angioedema, Stevens-Johnson syndrome, toxic epidermal necrolysis, and bronchoconstriction have been reported.
Asthma Exacerbations
Asthma exacerbations have been reported.
Hypocalcemia
Hypocalcemia has been reported.
Hypophosphatemia
Hypophosphatemia has been reported.
Osteonecrosis of the Jaw
Osteonecrosis of the jaw has been reported.
Osteonecrosis of other bones
Cases of osteonecrosis of other bones (including femur, hip, knee, ankle, wrist and humerus) have been reported; causality has not been determined in the population treated with zoledronic acid.
Ocular Adverse Events
Cases of the following events have been reported: conjunctivitis, iritis, iridocyclitis, uveitis, episcleritis, scleritis and orbital inflammation/edema.
Other
Hypotension in patients with underlying risk factors has been reported.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.6 Important Limitations of Use
The safety and effectiveness of Zoledronic Acid Injection for the treatment of osteoporosis is based on clinical data of three years duration. The optimal duration of use has not been determined. All patients on bisphosphonate therapy should have the need for continued therapy re-evaluated on a periodic basis. Patients at low-risk for fracture should be considered for drug discontinuation after 3 to 5 years of use. Patients who discontinue therapy should have their risk for fracture re-evaluated periodically.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Zoledronic Acid Injection is supplied as follows:
| NDC | Zoledronic Acid Injection (0.05 mg per mL) | Package Factor |
| 25021-830-82 | 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) | 1 bag per carton |
| 25021-830-87 | 5 mg per 100 mL ready-to-infuse solution in a flexible plastic container (bag) | 5 bags per carton |
1.4 Glucocorticoid Induced Osteoporosis (1.4 Glucocorticoid-Induced Osteoporosis)
Zoledronic Acid Injection is indicated for the treatment and prevention of glucocorticoid-induced osteoporosis in men and women who are either initiating or continuing systemic glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and who are expected to remain on glucocorticoids for at least 12 months [see Clinical Studies (14.4)].
5.2 Hypocalcemia and Mineral Metabolism
Pre-existing hypocalcemia and disturbances of mineral metabolism (e.g., hypoparathyroidism, thyroid surgery, parathyroid surgery; malabsorption syndromes, excision of small intestine) must be effectively treated before initiating therapy with Zoledronic Acid Injection. Clinical monitoring of calcium and mineral levels (phosphorus and magnesium) is highly recommended for these patients [see Contraindications (4)].
Hypocalcemia following Zoledronic Acid Injection administration is a significant risk in Paget's disease. All patients should be instructed about the symptoms of hypocalcemia and the importance of calcium and vitamin D supplementation in maintaining serum calcium levels [see Dosage and Administration (2.8), Adverse Reactions (6.1), Patient Counseling Information (17)].
All osteoporosis patients should be instructed on the importance of calcium and vitamin D supplementation in maintaining serum calcium levels [see Dosage and Administration (2.8), Adverse Reactions (6.1), Patient Counseling Information (17)].
2.6 Treatment of Paget's Disease of Bone
The recommended dose is a 5 mg infusion. The infusion time must not be less than 15 minutes given over a constant infusion rate.
14.5 Treatment of Paget's Disease of Bone
Zoledronic Acid Injection was studied in male and female patients with moderate to severe Paget's disease of bone, defined as serum alkaline phosphatase level at least twice the upper limit of the age-specific normal reference range at the time of study entry. Diagnosis was confirmed by radiographic evidence.
The efficacy of one infusion of 5 mg Zoledronic Acid Injection vs. oral daily doses of 30 mg risedronate for 2 months was demonstrated in two identically designed 6-month randomized, double-blind trials. The mean age of patients in the two trials was 70. Ninety-three percent (93%) of patients were Caucasian. Therapeutic response was defined as either normalization of serum alkaline phosphatase (SAP) or a reduction of at least 75% from baseline in total SAP excess at the end of 6 months. SAP excess was defined as the difference between the measured level and midpoint of normal range.
In both trials Zoledronic Acid Injection demonstrated a superior and more rapid therapeutic response compared with risedronate and returned more patients to normal levels of bone turnover, as evidenced by biochemical markers of formation (SAP, serum N-terminal propeptide of type I collagen [P1NP]) and resorption (serum CTx 1 [cross-linked C-telopeptides of type I collagen] and urine α-CTx).
The 6-month combined data from both trials showed that 96% (169/176) of Zoledronic Acid Injection-treated patients achieved a therapeutic response as compared with 74% (127/171) of patients treated with risedronate. Most Zoledronic Acid Injection patients achieved a therapeutic response by the Day 63 visit. In addition, at 6 months, 89% (156/176) of Zoledronic Acid Injection-treated patients achieved normalization of SAP levels, compared to 58% (99/171) of patients treated with risedronate (p<0.0001) (see Figure 2).
Figure 2. Therapeutic Response/Serum Alkaline Phosphatase (SAP) Normalization Over Time
The therapeutic response to Zoledronic Acid Injection was similar across demographic and disease-severity groups defined by gender, age, previous bisphosphonate use, and disease severity. At 6 months, the percentage of Zoledronic Acid Injection-treated patients who achieved therapeutic response was 97% and 95%, respectively, in each of the baseline disease severity subgroups (baseline SAP less than 3xULN, greater than or equal to 3xULN) compared to 75% and 74%, respectively, for the same disease severity subgroups of risedronate-treated patients.
In patients who had previously received treatment with oral bisphosphonates, therapeutic response rates were 96% and 55% for Zoledronic Acid Injection and risedronate, respectively. The comparatively low risedronate response was due to the low response rate (7/23, 30%) in patients previously treated with risedronate. In patients naïve to previous treatment, a greater therapeutic response was also observed with Zoledronic Acid Injection (98%) relative to risedronate (86%). In patients with symptomatic pain at screening, therapeutic response rates were 94% and 70% for Zoledronic Acid Injection and risedronate respectively. For patients without pain at screening, therapeutic response rates were 100% and 82% for Zoledronic Acid Injection and risedronate respectively.
Bone histology was evaluated in 7 patients with Paget's disease 6 months after being treated with Zoledronic Acid Injection 5 mg. Bone biopsy results showed bone of normal quality with no evidence of impaired bone remodeling and no evidence of mineralization defect.
2.1 Important Administration Instructions
Zoledronic Acid Injection must be administered as an intravenous infusion over no less than 15 minutes.
- Patients must be appropriately hydrated prior to administration of Zoledronic Acid Injection [see Warnings and Precautions (5.3)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Intravenous infusion should be followed by a 10 mL normal saline flush of the intravenous line.
- Administration of acetaminophen following Zoledronic Acid Injection administration may reduce the incidence of acute-phase reaction symptoms.
Caution: After removing the overwrap check for minute leaks by squeezing the inner bag firmly. If leaks are found, discard container as sterility may be compromised. Use only if solution is clear and the container is undamaged.
Preparation for Administration:
- Close flow control clamp of administration set.
- Remove cover from port at bottom of container.
- Insert piercing pin of administration set into port with a twisting motion until the pin is firmly seated. NOTE: See full directions on administration set carton.
- Suspend container from hanger.
- Squeeze and release drip chamber to establish proper fluid level in chamber during infusion of Zoledronic Acid Injection.
- Open flow control clamp to expel air from set. Close clamp.
- Regulate rate of administration with flow control clamp.
2.8 Calcium and Vitamin D Supplementation
- Instruct patients being treated for Paget's disease of bone on the importance of calcium and vitamin D supplementation in maintaining serum calcium levels, and on the symptoms of hypocalcemia. All patients should take 1500 mg elemental calcium daily in divided doses (750 mg two times a day, or 500 mg three times a day) and 800 international units vitamin D daily, particularly in the 2 weeks following Zoledronic Acid Injection administration [see Warnings and Precautions (5.2)].
- Instruct patients being treated for osteoporosis to take supplemental calcium and vitamin D if their dietary intake is inadequate. An average of at least 1200 mg calcium and 800 to 1000 international units vitamin D daily is recommended.
7.4 Drugs Primarily Excreted By the Kidney (7.4 Drugs Primarily Excreted by the Kidney)
Renal impairment has been observed following the administration of zoledronic acid in patients with pre-existing renal compromise or other risk factors [see Warnings and Precautions (5.3)]. In patients with renal impairment, the exposure to concomitant medications that are primarily renally excreted (e.g., digoxin) may increase. Consider monitoring serum creatinine in patients at risk for renal impairment who are taking concomitant medications that are primarily excreted by the kidney.
5.1 Drug Products With Same Active Ingredient (5.1 Drug Products with Same Active Ingredient)
Zoledronic Acid Injection contains the same active ingredient found in Zometa, used for oncology indications, and a patient being treated with Zometa should not be treated with Zoledronic Acid Injection.
14.2 Prevention of Postmenopausal Osteoporosis
The efficacy and safety of Zoledronic Acid Injection in postmenopausal women with osteopenia (low bone mass) was assessed in a 2-year randomized, multi-center, double-blind, placebo-controlled study of 581 postmenopausal women aged greater than or equal to 45 years, who were stratified by years since menopause: Stratum I women less than 5 years from menopause (n = 224); Stratum II women greater than or equal to 5 years from menopause (n = 357). Patients within Stratum I and II were randomized to one of three treatment groups: (1) Zoledronic Acid Injection given at randomization and at Month 12 (n = 77) in Stratum I and (n = 121) in Stratum II; (2) Zoledronic Acid Injection given at randomization and placebo at Month 12 (n = 70) in Stratum I and (n = 111) in Stratum II; and (3) Placebo given at randomization and Month 12 (n = 202). Zoledronic Acid Injection was administered as a single 5 mg dose in 100 mL solution infused over at least 15 minutes. All women received 500 to 1200 mg elemental calcium plus 400 to 800 international units vitamin D supplementation per day. The primary efficacy variable was the percent change of BMD at 24 Months relative to baseline.
1.1 Treatment of Osteoporosis in Postmenopausal Women
Zoledronic Acid Injection is indicated for treatment of osteoporosis in postmenopausal women. In postmenopausal women with osteoporosis, diagnosed by bone mineral density (BMD) or prevalent vertebral fracture, Zoledronic Acid Injection reduces the incidence of fractures (hip, vertebral and non-vertebral osteoporosis-related fractures). In patients at high risk of fracture, defined as a recent low-trauma hip fracture, zoledronic acid injection reduces the incidence of new clinical fractures [see Clinical Studies (14.1)].
2.2 Treatment of Osteoporosis in Postmenopausal Women
The recommended regimen is a 5 mg infusion once a year given intravenously over no less than 15 minutes.
1.2 Prevention of Osteoporosis in Postmenopausal Women
Zoledronic Acid Injection is indicated for prevention of osteoporosis in postmenopausal women [see Clinical Studies (14.2)].
2.3 Prevention of Osteoporosis in Postmenopausal Women
The recommended regimen is a 5 mg infusion given once every 2 years intravenously over no less than 15 minutes.
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fractures
Atypical, low-energy, or low trauma fractures of the femoral shaft have been reported in bisphosphonate-treated patients. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution. Causality has not been established as these fractures also occur in osteoporotic patients who have not been treated with bisphosphonates.
Atypical femur fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g., prednisone) at the time of fracture.
Any patient with a history of bisphosphonate exposure who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patients presenting with an atypical femur fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of bisphosphonate therapy should be considered, pending a risk/benefit assessment, on an individual basis.
2.5 Treatment and Prevention of Glucocorticoid Induced Osteoporosis (2.5 Treatment and Prevention of Glucocorticoid-Induced Osteoporosis)
The recommended regimen is a 5 mg infusion once a year given intravenously over no less than 15 minutes.
2.7 Laboratory Testing and Oral Examination Prior to Administration
- Prior to administration of each dose of Zoledronic Acid Injection, obtain a serum creatinine and creatinine clearance should be calculated based on actual body weight using Cockcroft-Gault formula before each Zoledronic Acid Injection dose. Zoledronic Acid Injection is contraindicated in patients with creatinine clearance less than 35 mL/min and in those with evidence of acute renal impairment. A 5 mg dose of Zoledronic Acid Injection administered intravenously is recommended for patients with creatinine clearance greater than or equal to 35 mL/min. There are no safety or efficacy data to support the adjustment of the Zoledronic Acid Injection dose based on baseline renal function. Therefore, no dose adjustment is required in patients with CrCl greater than or equal to 35 mL/min [see Contraindications (4), Warnings and Precautions (5.3)].
- A routine oral examination should be performed by the prescriber prior to initiation of Zoledronic Acid Injection treatment [see Warnings and Precautions (5.4)].
14.4 Treatment and Prevention of Glucocorticoid Induced Osteoporosis (14.4 Treatment and Prevention of Glucocorticoid-Induced Osteoporosis)
The efficacy and safety of Zoledronic Acid Injection to prevent and treat glucocorticoid-induced osteoporosis (GIO) was assessed in a randomized, multicenter, double-blind, stratified, active controlled study of 833 men and women aged 18 to 85 years (mean age of 54.4 years) treated with greater than or equal to 7.5 mg/day oral prednisone (or equivalent). Patients were stratified according to the duration of their pre-study corticosteroid therapy: less than or equal to 3 months prior to randomization (prevention subpopulation), and greater than 3 months prior to randomization (treatment subpopulation). The duration of the trial was one year. Patients were randomized to either Zoledronic Acid Injection which was administered once as a 5 mg dose in 100 mL infused over 15 minutes, or to an oral daily bisphosphonate (active control) for one year. All participants received 1000 mg of elemental calcium plus 400 to 1000 international units of vitamin D supplementation per day.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:17.220275 · Updated: 2026-03-14T22:24:28.166164