Equaline 44-226
15e4d040-ed21-46e7-98c6-56267f388f57
34390-5
HUMAN OTC DRUG LABEL
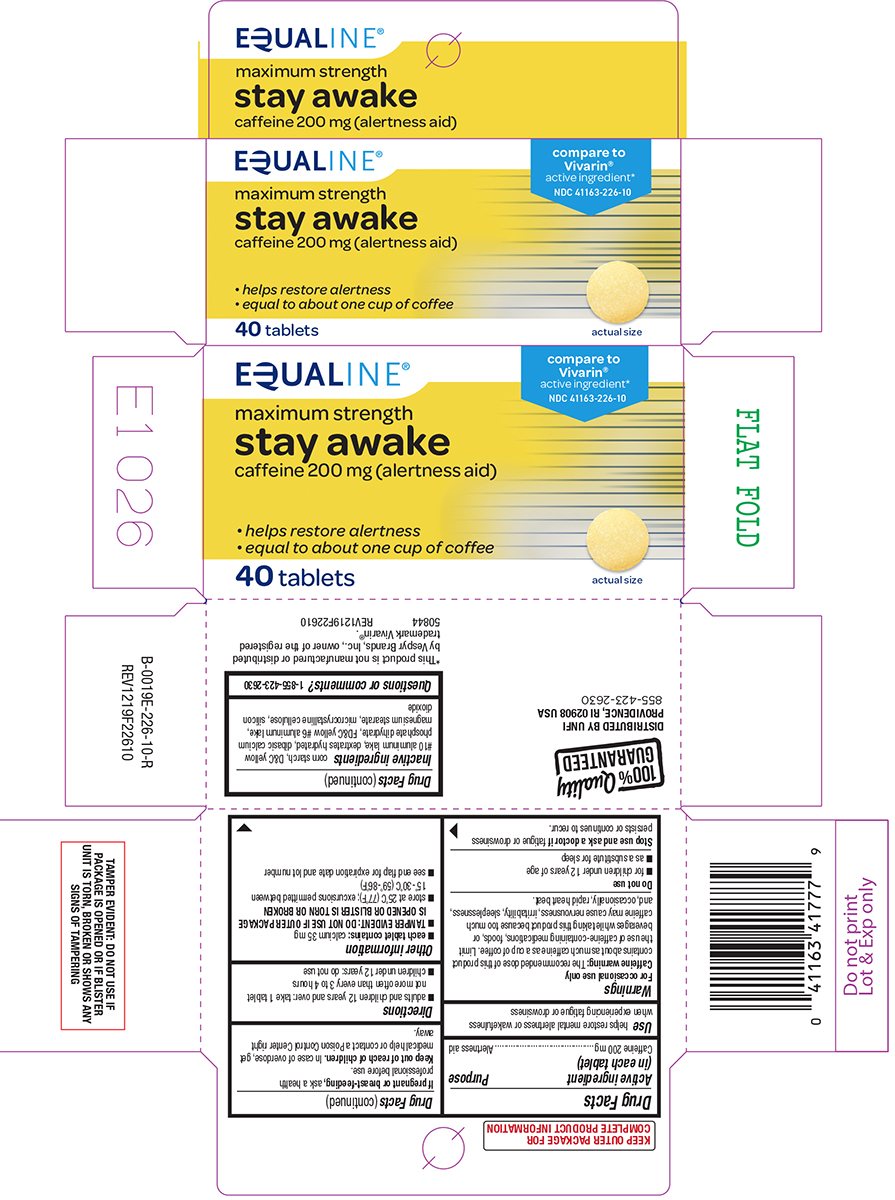
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Caffeine 200 mg
Purpose
Alertness aid
Medication Information
Purpose
Alertness aid
Description
Caffeine 200 mg
Use
helps restore mental alertness or wakefulness when experiencing fatigue or drowsiness
Warnings
For occasional use only
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heart beat.
Directions
- adults and children 12 years and over: take 1 tablet not more often than every 3 to 4 hours
- children under 12 years: do not use
Do Not Use
- for children under 12 years of age
- as a substitute for sleep
Other Information
- each tablet contains: calcium 35 mg
-
TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive Ingredients
corn starch, D&C yellow #10 aluminum lake, dextrates hydrated, dibasic calcium phosphate dihydrate, FD&C yellow #6 aluminum lake, magnesium stearate, microcrystalline cellulose, silicon dioxide
Questions Or Comments?
1-855-423-2630
Principal Display Panel
EQUALINE®
compare to
Vivarin®
active ingredient*
NDC 41163-226-10
maximum strength
stay awake
caffeine 200 mg (alertness aid)
• helps restore alertness
• equal to about one cup of coffee
40 tablets
actual size
TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY
SIGNS OF TAMPERING
*This product is not manufactured or distributed
by Vespyr Brands, Inc., owner of the registered
trademark Vivarin®.
50844 REV1219F22610
DISTRIBUTED BY UNFI
PROVIDENCE, RI 02908 USA
855-423-2630
Stop Use and Ask A Doctor If
fatigue or drowsiness persists or continues to recur.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet)
Caffeine 200 mg
Structured Label Content
Use
helps restore mental alertness or wakefulness when experiencing fatigue or drowsiness
Purpose
Alertness aid
Warnings
For occasional use only
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heart beat.
Directions
- adults and children 12 years and over: take 1 tablet not more often than every 3 to 4 hours
- children under 12 years: do not use
Do Not Use (Do not use)
- for children under 12 years of age
- as a substitute for sleep
Other Information (Other information)
- each tablet contains: calcium 35 mg
-
TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive Ingredients (Inactive ingredients)
corn starch, D&C yellow #10 aluminum lake, dextrates hydrated, dibasic calcium phosphate dihydrate, FD&C yellow #6 aluminum lake, magnesium stearate, microcrystalline cellulose, silicon dioxide
Questions Or Comments? (Questions or comments?)
1-855-423-2630
Principal Display Panel
EQUALINE®
compare to
Vivarin®
active ingredient*
NDC 41163-226-10
maximum strength
stay awake
caffeine 200 mg (alertness aid)
• helps restore alertness
• equal to about one cup of coffee
40 tablets
actual size
TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY
SIGNS OF TAMPERING
*This product is not manufactured or distributed
by Vespyr Brands, Inc., owner of the registered
trademark Vivarin®.
50844 REV1219F22610
DISTRIBUTED BY UNFI
PROVIDENCE, RI 02908 USA
855-423-2630
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
fatigue or drowsiness persists or continues to recur.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Caffeine 200 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:47.905528 · Updated: 2026-03-14T23:09:45.619768