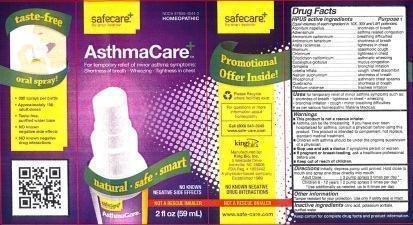
Asthmacare
15cf3795-12f0-4aaa-a311-7a1cd139497f
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
HPUS active ingredients _________________________________________________________________________________________________________________ Equal volumes of each ingredient in 10X, 30X, and LM1 potencies. Aconitum napellus, Adrenalinum, Ammonium carbonicum, Antimonium tartaricum, Aralia racemosa, Bromium, Chlorinum, Eriodictyon Californicum, Eucalyptus globulus, Grindelia, Lobelia inflata, Natrum sulphuricum, Phosphorus, Quebracho, Trifolium pratense.
Medication Information
Warnings and Precautions
Warnings
- This product is not a rescue inhaler.
- Asthma can be life threatening. If you have ever been hospitalized for asthma, consult a physician before using this product. This product is intended to complement, not replace, standard medical treatment.
- Children with asthma should be under the ongoing supervision of a physician.
- Stop use and ask a doctor if symptoms persist or worsen.
- If pregnant or breast-feeding, ask a healthcare professional before use.
Indications and Usage
Uses
For temporary relief or minor asthma symptoms such as:
- shortness of breath
- tightness in chest
- wheezing
- bronchial irritation
- cough
- minor breathing difficulties
† as per various homeopathic Materia Medicas.
Dosage and Administration
Directions
Initially, depress pump until primed. Hold close to mouth and spray one dose directly into mouth.
Adult Dose 3 pump sprays 3 times per day.
Children 6-12 years 2 pump sprays 3 times per day.
Use additionally as needed, up to 6 times per day.
Description
HPUS active ingredients _________________________________________________________________________________________________________________ Equal volumes of each ingredient in 10X, 30X, and LM1 potencies. Aconitum napellus, Adrenalinum, Ammonium carbonicum, Antimonium tartaricum, Aralia racemosa, Bromium, Chlorinum, Eriodictyon Californicum, Eucalyptus globulus, Grindelia, Lobelia inflata, Natrum sulphuricum, Phosphorus, Quebracho, Trifolium pratense.
Section 50565-1
- Keep out of reach of children.
Section 51727-6
Inactive Ingredients: Citric acid, Potassium sorbate and Purified water.
Section 51945-4
Section 55105-1
HPUS active ingredients Purpose †
Equal volumes of each ingredient in 10X, 30X, and LM1 potencies.
Aconitum napellus....................................................shortness of breath
Adrenalinum.............................................................asthma related congestion
Ammonium carbonicum.............................................breathing difficulties
Antimonium tararicum...............................................shortmess of breath
Aralia racemosa........................................................tightness in chest
Bromium..................................................................spasmodic cough
Chlorinum................................................................tightness in chest
Eriodictyon californicum.............................................asthmatic wheezing
Eucalyptus globulus..................................................mucus congestion
Grindelia.................................................................bronchial irritation
Lobelia inflata..........................................................cough, chest discomfort
Natrum sulphuricum.................................................shortness of breath
Phosphorus.............................................................asthmatic chest spasms
Quebracho..............................................................shortness of breath
Trifolium pratense....................................................tracheal irritation
Section 55106-9
HPUS active ingredients
_________________________________________________________________________________________________________________
Equal volumes of each ingredient in 10X, 30X, and LM1 potencies.
Aconitum napellus, Adrenalinum, Ammonium carbonicum, Antimonium tartaricum, Aralia racemosa, Bromium, Chlorinum, Eriodictyon Californicum, Eucalyptus globulus, Grindelia, Lobelia inflata, Natrum sulphuricum, Phosphorus, Quebracho, Trifolium pratense.
Section 60561-8
Other information:
Tamper resistant for your protection. Use only if safety seal is intact.
Structured Label Content
Indications and Usage (34067-9)
Uses
For temporary relief or minor asthma symptoms such as:
- shortness of breath
- tightness in chest
- wheezing
- bronchial irritation
- cough
- minor breathing difficulties
† as per various homeopathic Materia Medicas.
Dosage and Administration (34068-7)
Directions
Initially, depress pump until primed. Hold close to mouth and spray one dose directly into mouth.
Adult Dose 3 pump sprays 3 times per day.
Children 6-12 years 2 pump sprays 3 times per day.
Use additionally as needed, up to 6 times per day.
Warnings and Precautions (34071-1)
Warnings
- This product is not a rescue inhaler.
- Asthma can be life threatening. If you have ever been hospitalized for asthma, consult a physician before using this product. This product is intended to complement, not replace, standard medical treatment.
- Children with asthma should be under the ongoing supervision of a physician.
- Stop use and ask a doctor if symptoms persist or worsen.
- If pregnant or breast-feeding, ask a healthcare professional before use.
Section 50565-1 (50565-1)
- Keep out of reach of children.
Section 51727-6 (51727-6)
Inactive Ingredients: Citric acid, Potassium sorbate and Purified water.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
HPUS active ingredients Purpose †
Equal volumes of each ingredient in 10X, 30X, and LM1 potencies.
Aconitum napellus....................................................shortness of breath
Adrenalinum.............................................................asthma related congestion
Ammonium carbonicum.............................................breathing difficulties
Antimonium tararicum...............................................shortmess of breath
Aralia racemosa........................................................tightness in chest
Bromium..................................................................spasmodic cough
Chlorinum................................................................tightness in chest
Eriodictyon californicum.............................................asthmatic wheezing
Eucalyptus globulus..................................................mucus congestion
Grindelia.................................................................bronchial irritation
Lobelia inflata..........................................................cough, chest discomfort
Natrum sulphuricum.................................................shortness of breath
Phosphorus.............................................................asthmatic chest spasms
Quebracho..............................................................shortness of breath
Trifolium pratense....................................................tracheal irritation
Section 55106-9 (55106-9)
HPUS active ingredients
_________________________________________________________________________________________________________________
Equal volumes of each ingredient in 10X, 30X, and LM1 potencies.
Aconitum napellus, Adrenalinum, Ammonium carbonicum, Antimonium tartaricum, Aralia racemosa, Bromium, Chlorinum, Eriodictyon Californicum, Eucalyptus globulus, Grindelia, Lobelia inflata, Natrum sulphuricum, Phosphorus, Quebracho, Trifolium pratense.
Section 60561-8 (60561-8)
Other information:
Tamper resistant for your protection. Use only if safety seal is intact.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:17.587928 · Updated: 2026-03-14T23:14:29.546572