Perrigo Tussin Dm Drug Facts
15c8e177-a381-43b2-aed8-d917c7c4372e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
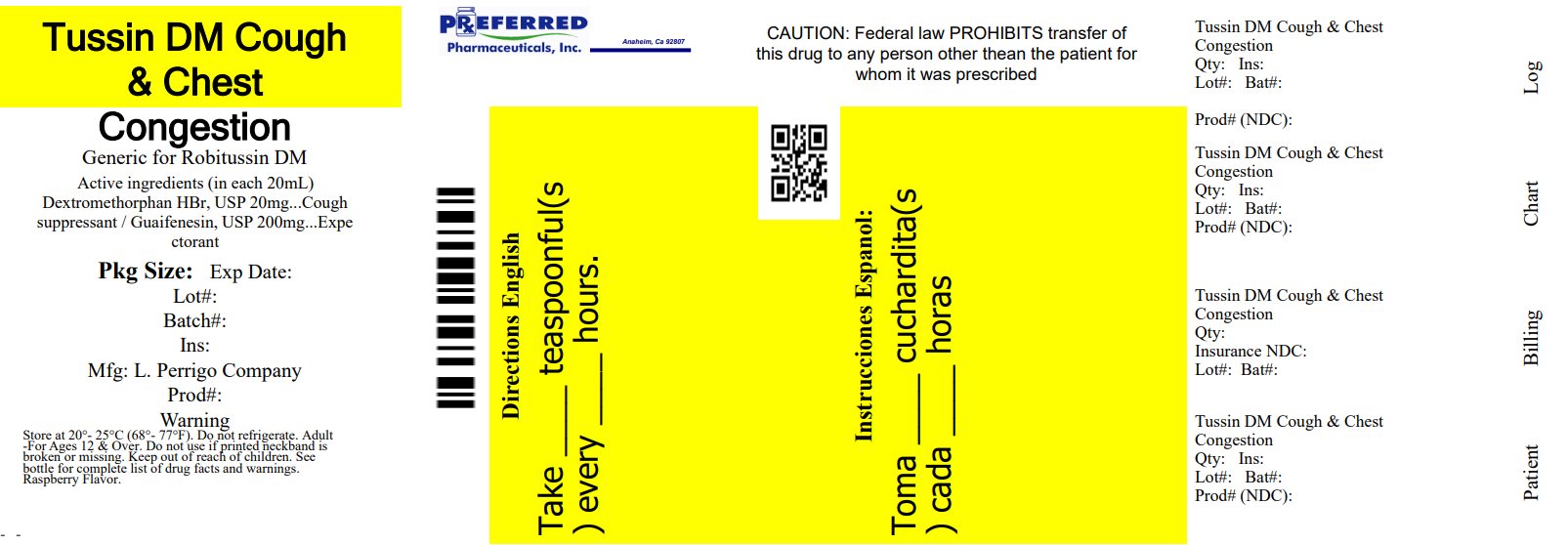
Dextromethorphan HBr, USP 20 mg Guaifenesin, USP 200 mg
Purpose
Cough suppressant Expectorant
Medication Information
Purpose
Cough suppressant Expectorant
Description
Dextromethorphan HBr, USP 20 mg Guaifenesin, USP 200 mg
Uses
-
•temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
-
•helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes
Purposes
Cough suppressant
Expectorant
Directions
-
•do not take more than 6 doses in any 24-hour period
-
•measure only with dosing cup provided
-
•keep dosing cup with product
-
•mL = milliliter
-
•this adult product is not intended for use in children under 12 years of age
|
age |
dose |
|
adults and children 12 years and over |
20 mL every 4 hours |
|
children under 12 years |
do not use |
Do Not Use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Other Information
-
•each 20 mL contains: sodium 14 mg
-
•store at 20-25°C (68-77°F). Do not refrigerate.
Inactive Ingredients
anhydrous citric acid, FD&C red no. 40, flavor, glycerin, propylene glycol, purified water, sodium benzoate, sodium citrate, sorbitol solution, sucralose, xanthan gum
Questions Or Comments?
1-800-719-9260
Stop Use and Ask A Doctor If
cough lasts for more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache. A persistent cough may be a sign of a serious condition.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Active Ingredients (in Each 20 Ml)
Dextromethorphan HBr, USP 20 mg
Guaifenesin, USP 200 mg
Ask A Doctor Before Use If You Have
-
•cough that occurs with too much phlegm (mucus)
-
•cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis, or emphysema
Package/label Principal Display Panel
GoodSense®
Relabeled By: Preferred Pharmaceuticals Inc.
NDC 68788-8300-1
Non-Drowsy
See New Dosing
Tussin DM
Cough Suppressant (Dextromethorphan HBr)
Expectorant (Guaifenesin)
Cough & Chest Congestion
Relieves:
• Cough
• Chest Congestion/Mucus
Adult
For Ages 12 & Over
Raspberry Flavor
Dosage Cup Included
Compare to active ingredients of Robitussin® Cough + Chest Congestion DM
4 FL OZ (118 mL)
Structured Label Content
Uses
-
•temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
-
•helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes
Purposes
Cough suppressant
Expectorant
Directions
-
•do not take more than 6 doses in any 24-hour period
-
•measure only with dosing cup provided
-
•keep dosing cup with product
-
•mL = milliliter
-
•this adult product is not intended for use in children under 12 years of age
|
age |
dose |
|
adults and children 12 years and over |
20 mL every 4 hours |
|
children under 12 years |
do not use |
Do Not Use (Do not use)
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Other Information (Other information)
-
•each 20 mL contains: sodium 14 mg
-
•store at 20-25°C (68-77°F). Do not refrigerate.
Inactive Ingredients (Inactive ingredients)
anhydrous citric acid, FD&C red no. 40, flavor, glycerin, propylene glycol, purified water, sodium benzoate, sodium citrate, sorbitol solution, sucralose, xanthan gum
Questions Or Comments? (Questions or comments?)
1-800-719-9260
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
cough lasts for more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache. A persistent cough may be a sign of a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Active Ingredients (in Each 20 Ml) (Active ingredients (in each 20 mL))
Dextromethorphan HBr, USP 20 mg
Guaifenesin, USP 200 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
-
•cough that occurs with too much phlegm (mucus)
-
•cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis, or emphysema
Package/label Principal Display Panel (Package/Label Principal Display Panel)
GoodSense®
Relabeled By: Preferred Pharmaceuticals Inc.
NDC 68788-8300-1
Non-Drowsy
See New Dosing
Tussin DM
Cough Suppressant (Dextromethorphan HBr)
Expectorant (Guaifenesin)
Cough & Chest Congestion
Relieves:
• Cough
• Chest Congestion/Mucus
Adult
For Ages 12 & Over
Raspberry Flavor
Dosage Cup Included
Compare to active ingredients of Robitussin® Cough + Chest Congestion DM
4 FL OZ (118 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:20.147227 · Updated: 2026-03-14T23:06:59.037711