Lidocaine 4%
14edf7dc-c5dc-4b74-b583-0f87529f4536
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%
Medication Information
Warnings and Precautions
For external use only
Indications and Usage
For the temporary relief of pain
Dosage and Administration
Directions
Adults and children 12 years of age and over:
- Clean and dry the affected area
- Open pouch and remove one patch
- Apply 1 patch at a time to affected area; not more than 3 to 4 times daily
- Reseal pouch containing unused patches after each use
- Remove patch from the skin after at most 8-hour application
Children under 12 years of age: Consult a doctor
Description
Lidocaine 4%
Section 50565-1
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- Localized skin reactions occur, such as rash, itching, redness, irritation, pain, swelling and blistering
- Conditions worsen
- Symptoms persist for more than 7 days
- Symptoms clear up and occur again within a few days
Section 50567-7
When using this product
- Use only as directed
- Avoid contact with the eyes, mucous membranes or rashes
- Do not bandage tightly
Section 50570-1
Do not use
- More than 1 pathch at a time
- On wounds or damaged skin
- With a heating pad
- If you are allergic to any ingredients of this product
Section 51727-6
Other ingredients: Acrylic Adhesive, Flaxseed oil, and Polysorbate 80
Questions or comments? 877-367-3250
Section 51945-4
Packaged For:
Forreal Pharmaceuticals LLC
Royal Oak, MI 48067
Questions or Comments please call 877-367-3250
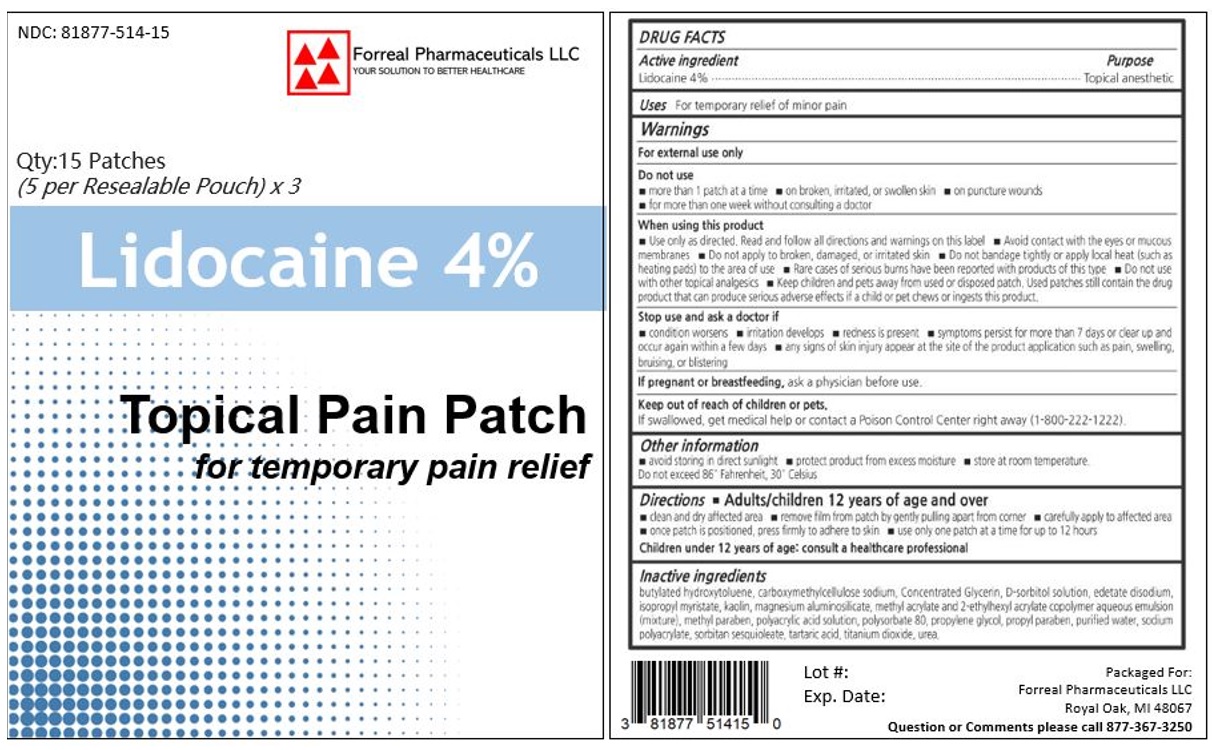
NDC 81877-514-15
Qty:15 Patches
(5 per Resealable Pouch) x 3
Lidocaine 4%
Topical Pain Patch
for temporary pain relief
Section 53414-9
If pregnant or breast-feeding
Ask a health professional before use.
Section 55105-1
Topical Anesthetic
Section 55106-9
Lidocaine 4%
Other Information:
- Avoid storing product in direct sunlight
- Protect product from excessive moisture
Structured Label Content
Indications and Usage (34067-9)
For the temporary relief of pain
Dosage and Administration (34068-7)
Directions
Adults and children 12 years of age and over:
- Clean and dry the affected area
- Open pouch and remove one patch
- Apply 1 patch at a time to affected area; not more than 3 to 4 times daily
- Reseal pouch containing unused patches after each use
- Remove patch from the skin after at most 8-hour application
Children under 12 years of age: Consult a doctor
Warnings and Precautions (34071-1)
For external use only
Section 50565-1 (50565-1)
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- Localized skin reactions occur, such as rash, itching, redness, irritation, pain, swelling and blistering
- Conditions worsen
- Symptoms persist for more than 7 days
- Symptoms clear up and occur again within a few days
Section 50567-7 (50567-7)
When using this product
- Use only as directed
- Avoid contact with the eyes, mucous membranes or rashes
- Do not bandage tightly
Section 50570-1 (50570-1)
Do not use
- More than 1 pathch at a time
- On wounds or damaged skin
- With a heating pad
- If you are allergic to any ingredients of this product
Section 51727-6 (51727-6)
Other ingredients: Acrylic Adhesive, Flaxseed oil, and Polysorbate 80
Questions or comments? 877-367-3250
Section 51945-4 (51945-4)
Packaged For:
Forreal Pharmaceuticals LLC
Royal Oak, MI 48067
Questions or Comments please call 877-367-3250
NDC 81877-514-15
Qty:15 Patches
(5 per Resealable Pouch) x 3
Lidocaine 4%
Topical Pain Patch
for temporary pain relief
Section 53414-9 (53414-9)
If pregnant or breast-feeding
Ask a health professional before use.
Section 55105-1 (55105-1)
Topical Anesthetic
Section 55106-9 (55106-9)
Lidocaine 4%
Other Information: (Other information:)
- Avoid storing product in direct sunlight
- Protect product from excessive moisture
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:08.687262 · Updated: 2026-03-14T23:03:02.507799