pseudoephedrine hydrochloride extended-release tablets, usp 240 mg
1425a847-bf34-1795-e063-6294a90a85b6
34390-5
Human OTC Drug Label
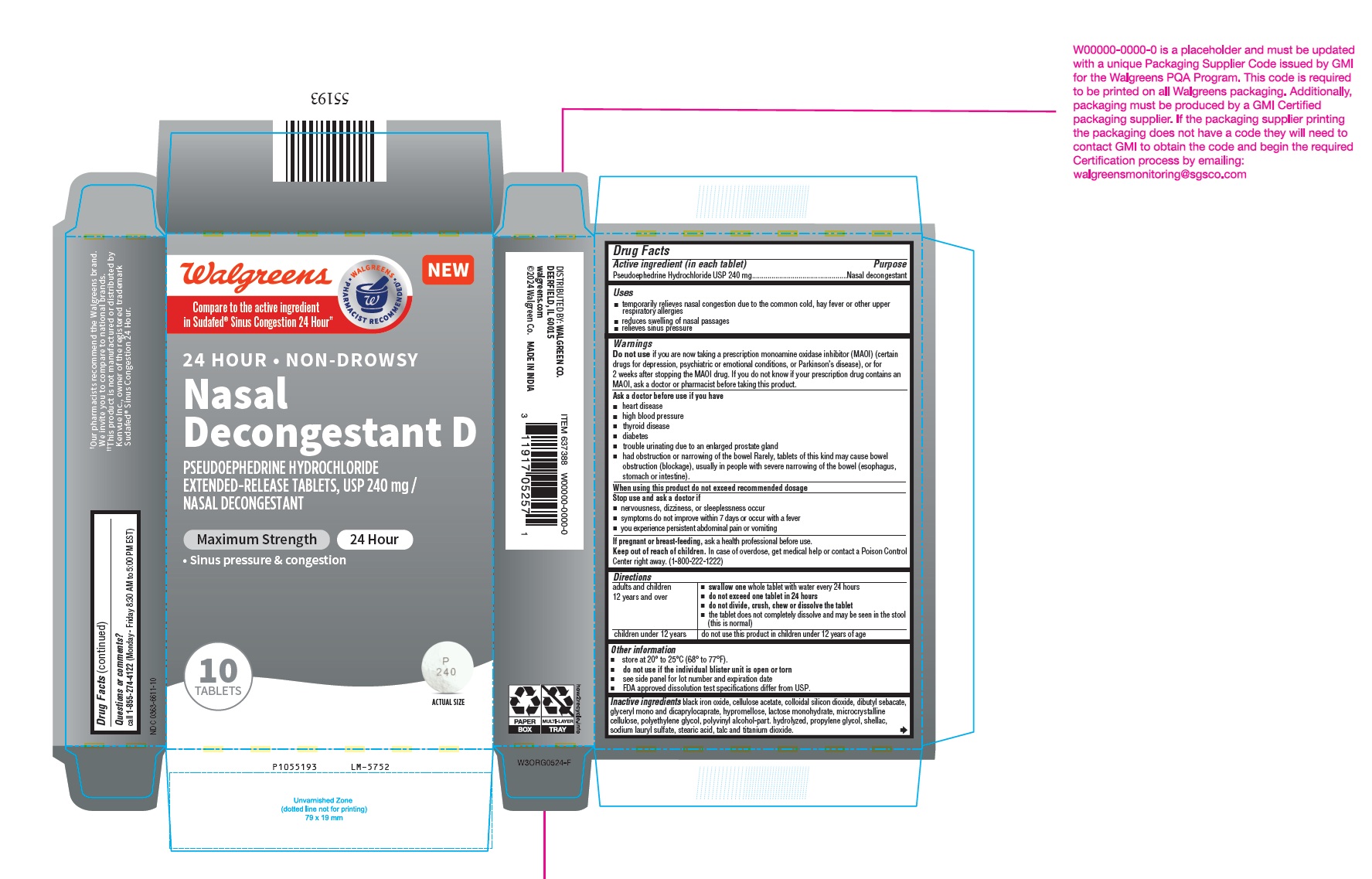
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Nasal decongestant
Description
Active ingredient (in each tablet) Pseudoephedrine Hydrochloride USP 240 mg
Medication Information
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- reduces swelling of nasal passages
- relieves sinus pressure
Purpose
Nasal decongestant
Directions
| adults and children 12 years and over |
|
| children under 12 years
|
do not use this product in children under 12 years of age |
Other Information
- store at 20° to 25°C (68° to 77°F).
- do not use if the individual blister unit is open or torn
- see side panel for lot number and expiration date
- FDA approved dissolution test specifications differ from USP.
Inactive Ingredients
black iron oxide, cellulose acetate, colloidal silicon dioxide, dibutyl sebacate, glyceryl mono and dicaprylocaprate, hypromellose, lactose monohydrate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol-part. hydrolyzed, propylene glycol, shellac, sodium lauryl sulfate, stearic acid, talc and titanium dioxide.
Questions or comments?
call 1-855-274-4122 (Monday - Friday 8:30 AM to 5:00 PM EST)
DISTRIBUTED BY: WALGREEN CO.
DEERFIELD, IL 60015
MADE IN INDIA
Description
Active ingredient (in each tablet) Pseudoephedrine Hydrochloride USP 240 mg
Section 50566-9
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with a fever
- you experience persistent abdominal pain or vomiting
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Drug Facts
Active ingredient (in each tablet)
Pseudoephedrine Hydrochloride USP 240 mg
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Ask A Doctor Before Use If You Have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
- had obstruction or narrowing of the bowel Rarely, tablets of this kind may cause bowel obstruction (blockage), usually in people with severe narrowing of the bowel (esophagus, stomach or intestine).
When Using This Product Do Not Exceed Recommended Dosage
Stop use and ask a doctor if
Package Label Principal Display Panel 1 X 10 Blister Carton Label
Walgreens
NEW
Compare to the active ingredient
in Sudafed® Sinus Congestion 24 Hour††
24 HOUR • NON-DROWSY
Nasal
Decongestant D
PSEUDOEPHEDRINE HYDROCHLORIDE
EXTENDED-RELEASE TABLETS, USP 240 mg /
NASAL DECONGESTANT
Maximum Strength 24 Hour
• Sinus pressure & congestion
10
TABLETS ACTUAL SIZE
Structured Label Content
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- reduces swelling of nasal passages
- relieves sinus pressure
Purpose
Nasal decongestant
Directions
| adults and children 12 years and over |
|
| children under 12 years
|
do not use this product in children under 12 years of age |
Other Information (Other information)
- store at 20° to 25°C (68° to 77°F).
- do not use if the individual blister unit is open or torn
- see side panel for lot number and expiration date
- FDA approved dissolution test specifications differ from USP.
Inactive Ingredients (Inactive ingredients)
black iron oxide, cellulose acetate, colloidal silicon dioxide, dibutyl sebacate, glyceryl mono and dicaprylocaprate, hypromellose, lactose monohydrate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol-part. hydrolyzed, propylene glycol, shellac, sodium lauryl sulfate, stearic acid, talc and titanium dioxide.
Questions or comments?
call 1-855-274-4122 (Monday - Friday 8:30 AM to 5:00 PM EST)
DISTRIBUTED BY: WALGREEN CO.
DEERFIELD, IL 60015
MADE IN INDIA
Section 50566-9 (50566-9)
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with a fever
- you experience persistent abdominal pain or vomiting
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Drug Facts
Active ingredient (in each tablet)
Pseudoephedrine Hydrochloride USP 240 mg
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
- had obstruction or narrowing of the bowel Rarely, tablets of this kind may cause bowel obstruction (blockage), usually in people with severe narrowing of the bowel (esophagus, stomach or intestine).
When Using This Product Do Not Exceed Recommended Dosage (When using this product do not exceed recommended dosage)
Stop use and ask a doctor if
Package Label Principal Display Panel 1 X 10 Blister Carton Label (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1 x 10 Blister Carton Label)
Walgreens
NEW
Compare to the active ingredient
in Sudafed® Sinus Congestion 24 Hour††
24 HOUR • NON-DROWSY
Nasal
Decongestant D
PSEUDOEPHEDRINE HYDROCHLORIDE
EXTENDED-RELEASE TABLETS, USP 240 mg /
NASAL DECONGESTANT
Maximum Strength 24 Hour
• Sinus pressure & congestion
10
TABLETS ACTUAL SIZE
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:30.677166 · Updated: 2026-03-14T23:12:45.219834