These Highlights Do Not Include All The Information Needed To Use Amlodipine And Atorvastatin Tablets Safely And Effectively. See Full Prescribing Information For Amlodipine And Atorvastatin Tablets.
14124e56-77ba-426d-8f5a-dbe42eda1a20
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Contraindications, Pregnancy and Lactation ( 4 ) Removed 05/2024 Warnings and Precautions, CNS Toxicity ( 5.7 ) Removed 05/2024
Indications and Usage
Amlodipine and atorvastatin tablets are indicated in patients for whom treatment with both amlodipine and atorvastatin is appropriate. Amlodipine Hypertension Amlodipine is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including amlodipine. Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC). Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly. Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal. Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy. Amlodipine may be used alone or in combination with other antihypertensive agents. Coronary Artery Disease (CAD) Chronic Stable Angina Amlodipine is indicated for the symptomatic treatment of chronic stable angina. Amlodipine may be used alone or in combination with other antianginal agents. Vasospastic Angina (Prinzmetal’s or Variant Angina) Amlodipine is indicated for the treatment of confirmed or suspected vasospastic angina. Amlodipine may be used as monotherapy or in combination with other antianginal agents. Angiographically Documented CAD In patients with recently documented CAD by angiography and without heart failure or an ejection fraction < 40%, amlodipine is indicated to reduce the risk of hospitalization for angina and to reduce the risk of a coronary revascularization procedure. Atorvastatin Atorvastatin is indicated: • To reduce the risk of: o Myocardial infarction (MI), stroke, revascularization procedures, and angina in adults with multiple risk factors for coronary heart disease (CHD) but without clinically evident CHD o MI and stroke in adults with type 2 diabetes mellitus with multiple risk factors for CHD but without clinically evident CHD o Non-fatal MI, fatal and non-fatal stroke, revascularization procedures, hospitalization for congestive heart failure, and angina in adults with clinically evident CHD • As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in: o Adults with primary hyperlipidemia. o Adults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH). • As an adjunct to other LDL-C-lowering therapies, or alone if such treatments are unavailable, to reduce LDL-C in adults and pediatric patients aged 10 years and older with homozygous familial hypercholesterolemia (HoFH). • As an adjunct to diet for the treatment of adults with: o Primary dysbetalipoproteinemia o Hypertriglyceridemia
Dosage and Administration
Usual starting dose (mg daily) Maximum dose (mg daily) Amlodipine 5 Start small adults or children, fragile, or elderly patients, or patients with hepatic insufficiency on 2.5 mg once daily ( 2 ). 10 Atorvastatin 10-20 Start patients requiring large LDL-C reduction (> 45%) at 40 mg once daily ( 2 ). 80
Warnings and Precautions
• Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher amlodipine and atorvastatin dosage. Discontinue amlodipine and atorvastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue amlodipine and atorvastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing amlodipine and atorvastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever ( 2 , 5.1 , 7.3 , 8.5 , 8.6 ). • Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported with statin use. Discontinue amlodipine and atorvastatin tablets if IMNM is suspected ( 5.2 ). • Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue amlodipine and atorvastatin ( 5.3 ). • Angina or myocardial infarction may occur with initiation or dose increase ( 5.4 ). • Symptomatic hypotension is possible, particularly in patients with severe aortic stenosis. However, acute hypotension is unlikely ( 5.5 ).
Contraindications
• Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3) ] . • Hypersensitivity to amlodipine, atorvastatin or any excipients in amlodipine and atorvastatin tablets. Hypersensitivity reactions, including anaphylaxis, angioneurotic edema, erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis, have been reported [see Adverse Reactions (6.2) ] .
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: • Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1) ] • Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2) ] • Hepatic Dysfunction [see Warnings and Precautions (5.3) ] • Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.6) ]
Drug Interactions
Atorvastatin is a substrate of CYP3A4 and transporters (e.g., OATP1B1/1B3, P-gp, or BCRP). Atorvastatin plasma levels can be significantly increased with concomitant administration of inhibitors of CYP3A4 and transporters. Table 3 includes a list of drugs that may increase exposure to atorvastatin and may increase the risk of myopathy and rhabdomyolysis when used concomitantly and instructions for preventing or managing them [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3) ]. Table 3. Drug Interactions that may Increase the Risk of Myopathy and Rhabdomyolysis with Atorvastatin Cyclosporine or Gemfibrozil Clinical Impact: Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin and cyclosporine, an inhibitor of CYP3A4 and OATP1B1 [see Clinical Pharmacology (12.3) ]. Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of cyclosporine or gemfibrozil with atorvastatin. Intervention: Concomitant use of cyclosporine or gemfibrozil with atorvastatin is not recommended. Anti-Viral Medications Clinical Impact: Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with many anti-viral medications, which are inhibitors of CYP3A4 and/or transporters (e.g., BCRP, OATP1B1/1B3, P-gp, MRP2, and/or OAT2) [see Clinical Pharmacology (12.3) ]. Cases of myopathy and rhabdomyolysis have been reported with concomitant use of ledipasvir plus sofosbuvir with atorvastatin. Intervention: • Concomitant use of tipranavir plus ritonavir or glecaprevir plus pibrentasvir with atorvastatin is not recommended. • In patients taking lopinavir plus ritonavir, or simeprevir, consider the risk/benefit of concomitant use with atorvastatin. • In patients taking saquinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, fosamprenavir plus ritonavir, elbasvir plus grazoprevir or letermovir do not exceed atorvastatin 20 mg. • In patients taking nelfinavir, do not exceed atorvastatin 40 mg [see Dosage and Administration (2) ]. • Consider the risk/benefit of concomitant use of ledipasvir plus sofosbuvir with atorvastatin. • Monitor all patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Examples: Tipranavir plus ritonavir, glecaprevir plus pibrentasvir, lopinavir plus ritonavir, simeprevir, saquinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, fosamprenavir plus ritonavir, elbasvir plus grazoprevir, letermovir, nelfinavir, and ledipasvir plus sofosbuvir. Select Azole Antifungals or Macrolide Antibiotics Clinical Impact: Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with select azole antifungals or macrolide antibiotics, due to inhibition of CYP3A4 and/or transporters [see Clinical Pharmacology (12.3) ]. Intervention: In patients taking clarithromycin or itraconazole, do not exceed atorvastatin 20 mg [see Dosage and Administration (2) ]. Consider the risk/benefit of concomitant use of other azole antifungals or macrolide antibiotics with atorvastatin. Monitor all patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Examples: Erythromycin, clarithromycin, itraconazole, ketoconazole, posaconazole, and voriconazole. Niacin Clinical Impact: Cases of myopathy and rhabdomyolysis have been observed with concomitant use of lipid modifying dosages of niacin (≥ 1 gram/day niacin) with atorvastatin. Intervention: Consider if the benefit of using lipid modifying dosages of niacin concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Fibrates (other than Gemfibrozil) Clinical Impact: Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with atorvastatin. Intervention: Consider if the benefit of using fibrates concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Colchicine Clinical Impact: Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with atorvastatin. Intervention: Consider the risk/benefit of concomitant use of colchicine with atorvastatin. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Grapefruit Juice Clinical Impact: Grapefruit juice consumption, especially excessive consumption, more than 1.2 liters/daily can raise the plasma levels of atorvastatin and may increase the risk of myopathy and rhabdomyolysis. Intervention: Avoid intake of large quantities of grapefruit juice, more than 1.2 liters daily, when taking atorvastatin.
Storage and Handling
Amlodipine and atorvastatin tablets contain amlodipine besylate and atorvastatin calcium equivalent to amlodipine and atorvastatin in the dose strengths described below. Amlodipine and atorvastatin tablets are differentiated by tablet color/size and are engraved with a unique number on one side. Combinations of atorvastatin with 2.5 mg amlodipine are round and film-coated white, combinations of atorvastatin with 5 mg amlodipine are oval and film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are oval and are film-coated blue tablets. Amlodipine and atorvastatin tablets are supplied for oral administration in the following strengths and package configurations: Table 16. Amlodipine and Atorvastatin Tablets Packaging Configurations Amlodipine and Atorvastatin Tablets Package Configuration Tablet Strength mg (amlodipine/atorvastatin) NDC # Engraving Side 1/Side 2 Tablet Color Tablet Shape Bottle of 30 2.5/10 66993-263-30 CDT 251/Blank White Round Bottle of 30 2.5/20 66993-264-30 CDT 252/Blank White Round Bottle of 30 2.5/40 66993-265-30 CDT 254/Blank White Round Bottle of 30 5/10 66993-266-30 CDT 051/Pfizer White Oval Bottle of 30 5/20 66993-267-30 CDT 052/Pfizer White Oval Bottle of 30 5/40 66993-268-30 CDT 054/Pfizer White Oval Bottle of 30 5/80 66993-269-30 CDT 058/Pfizer White Oval Bottle of 30 10/10 66993-270-30 CDT 101/Pfizer Blue Oval Bottle of 30 10/20 66993-271-30 CDT 102/Pfizer Blue Oval Bottle of 30 10/40 66993-272-30 CDT 104/Pfizer Blue Oval Bottle of 30 10/80 66993-273-30 CDT 108/Pfizer Blue Oval
How Supplied
Amlodipine and atorvastatin tablets contain amlodipine besylate and atorvastatin calcium equivalent to amlodipine and atorvastatin in the dose strengths described below. Amlodipine and atorvastatin tablets are differentiated by tablet color/size and are engraved with a unique number on one side. Combinations of atorvastatin with 2.5 mg amlodipine are round and film-coated white, combinations of atorvastatin with 5 mg amlodipine are oval and film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are oval and are film-coated blue tablets. Amlodipine and atorvastatin tablets are supplied for oral administration in the following strengths and package configurations: Table 16. Amlodipine and Atorvastatin Tablets Packaging Configurations Amlodipine and Atorvastatin Tablets Package Configuration Tablet Strength mg (amlodipine/atorvastatin) NDC # Engraving Side 1/Side 2 Tablet Color Tablet Shape Bottle of 30 2.5/10 66993-263-30 CDT 251/Blank White Round Bottle of 30 2.5/20 66993-264-30 CDT 252/Blank White Round Bottle of 30 2.5/40 66993-265-30 CDT 254/Blank White Round Bottle of 30 5/10 66993-266-30 CDT 051/Pfizer White Oval Bottle of 30 5/20 66993-267-30 CDT 052/Pfizer White Oval Bottle of 30 5/40 66993-268-30 CDT 054/Pfizer White Oval Bottle of 30 5/80 66993-269-30 CDT 058/Pfizer White Oval Bottle of 30 10/10 66993-270-30 CDT 101/Pfizer Blue Oval Bottle of 30 10/20 66993-271-30 CDT 102/Pfizer Blue Oval Bottle of 30 10/40 66993-272-30 CDT 104/Pfizer Blue Oval Bottle of 30 10/80 66993-273-30 CDT 108/Pfizer Blue Oval
Patient Information
Amlodipine and Atorvastatin Tablets Read the patient information that comes with amlodipine and atorvastatin tablets before you start taking them, and each time you get a refill. There may be new information. This information does not replace talking with your healthcare provider about your condition or treatment. If you have any questions about amlodipine and atorvastatin tablets, ask your healthcare provider or pharmacist. What are amlodipine and atorvastatin tablets? Amlodipine and atorvastatin tablets are a prescription drug that combines Norvasc ® (amlodipine besylate) and Lipitor ® (atorvastatin calcium) in one pill. Amlodipine and atorvastatin tablets are used in adults who need both Norvasc and Lipitor. Norvasc is used to treat: • High blood pressure (hypertension) and • Chest pain (angina) and • Blocked arteries of the heart (coronary artery disease) Lipitor is used to lower the levels of “bad” cholesterol and triglycerides in your blood. It can also raise the levels of “good” cholesterol. Lipitor is also used to lower the risk for heart attack, stroke, certain types of heart surgery, and chest pain in patients who have heart disease or risk factors for heart disease such as: • age, smoking, high blood pressure, low levels of “good” cholesterol, heart disease in the family. Lipitor can lower the risk for heart attack or stroke in patients with diabetes and risk factors such as: • diabetic eye or kidney problems, smoking, or high blood pressure. Amlodipine and atorvastatin tablets have not been studied in children. Who should not use amlodipine and atorvastatin tablets? Do not use amlodipine and atorvastatin tablets if you : • Have liver problems (acute liver failure or decompensated cirrhosis). • Are allergic to anything in amlodipine and atorvastatin tablets. The active ingredients are atorvastatin calcium and amlodipine besylate. Stop using amlodipine and atorvastatin tablets and get medical help right away if you have symptoms of a serious allergic reaction including: o swelling of your face, lips, tongue or throat o problems breathing or swallowing o fainting or feeling dizzy o very rapid heartbeat o severe skin rash or itching o flu-like symptoms including fever, sore throat, cough, tiredness, and joint pain See the end of this leaflet for a complete list of ingredients. What should I tell my healthcare provider before taking amlodipine and atorvastatin tablets? Tell your healthcare provider about all of your health conditions, including, if you: • have unexplained muscle aches or weakness • drink more than 2 glasses of alcohol daily • have heart disease • have diabetes • have thyroid problems • have kidney problems • had a stroke • are pregnant or plan to become pregnant. Amlodipine and atorvastatin tablets may harm your unborn baby. If you become pregnant, stop taking amlodipine and atorvastatin tablets and call your healthcare provider right away. • are breastfeeding or plan to breastfeed. You and your healthcare provider should decide if you will take amlodipine and atorvastatin tablets or breastfeed. You should not do both. Talk to your healthcare provider about the best way to feed your baby if you take amlodipine and atorvastatin tablets. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Amlodipine and atorvastatin tablets and certain other medicines can increase the risk of muscle problems or other side effects. Especially tell your healthcare provider if you take medicines for: • your immune system (cyclosporine) • cholesterol (gemfibrozil) • infections (erythromycin, clarithromycin, itraconazole, ketoconazole, posaconazole, and • voriconazole) • birth control pills • heart failure (digoxin) • gout (colchicine) • niacin • fibrates • treating HIV, AIDS, or hepatitis C (anti-virals) ○ tipranavir plus ritonavir ○ ledipasvir plus sofosbuvir ○ saquinavir plus ritonavir ○ fosamprenavir ○ elbasvir plus grazoprevir ○ nelfinavir ○ glecaprevir plus pibrentasvir ○ simeprevir ○ darunavir plus ritonavir ○ fosamprenavir plus ritonavir ○ letermovir You can use nitroglycerin and amlodipine and atorvastatin tablets together. If you take nitroglycerin for chest pain (angina), do not stop taking it while taking amlodipine and atorvastatin tablets. Ask your healthcare provider or pharmacist for a list of medicines if you are not sure. Know all the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist when you get a new medicine. How should I take amlodipine and atorvastatin tablets? • Take amlodipine and atorvastatin tablets exactly as your healthcare provider tells you to take it. • Do not change your dose or stop amlodipine and atorvastatin tablets without talking to your healthcare provider. • Your healthcare provider may do blood tests to check your cholesterol levels during your treatment with amlodipine and atorvastatin tablets. Your dose of amlodipine and atorvastatin tablets may be changed based on these blood test results. • Take amlodipine and atorvastatin tablets each day at any time of day. Amlodipine and atorvastatin tablets can be taken with or without food. • Do not break the tablets before taking them. Talk to your healthcare provider if you have a problem swallowing pills. • Your healthcare provider may start you on a cholesterol-lowering diet before giving you amlodipine and atorvastatin tablets. Stay on this low-fat diet when you take amlodipine and atorvastatin tablets. • If you miss a dose, take it as soon as you remember. Do not take amlodipine and atorvastatin tablets if it has been more than 12 hours since you missed your last dose. Wait and take the next dose at your regular time. Do not take 2 doses of amlodipine and atorvastatin tablets at the same time. If you take too much amlodipine and atorvastatin tablets or overdose, call your healthcare provider or Poison Control Center at 1-800-222-1222 or go to the nearest emergency room right away. What should I avoid while taking amlodipine and atorvastatin tablets? • Avoid drinking more than 1.2 liters of grapefruit juice each day. What are possible side effects of amlodipine and atorvastatin tablets? Amlodipine and atorvastatin tablets can cause serious side effects including: • Muscle pain, tenderness and weakness (myopathy). Muscle problems, including muscle breakdown, can be serious in some people and, rarely, cause kidney damage that can lead to death. Tell your healthcare provider right away if you have: o unexplained muscle pain, tenderness, or weakness, especially if you also have a fever or feel more tired than usual while you take amlodipine and atorvastatin tablets. o muscle problems that do not go away after your healthcare provider has told you to stop taking amlodipine and atorvastatin tablets. Your healthcare provider may do further tests to diagnose the cause of your muscle problems. Your chances of getting muscle problems are higher if you: o are taking certain other medicines while you take amlodipine and atorvastatin tablets o drink large amounts of grapefruit juice o are 65 years of age or older o have thyroid problems (hypothyroidism) that are not controlled o have kidney problems o are taking higher doses of amlodipine and atorvastatin tablets • Liver problems. Your healthcare provider should do blood tests to check your liver before you start taking amlodipine and atorvastatin tablets and if you have symptoms of liver problems while you take amlodipine and atorvastatin tablets. Call your healthcare provider right away if you have the following symptoms of liver problems: o feel tired or weak o nausea or vomiting o loss of appetite o upper belly pain o dark amber colored urine o yellowing of your skin or the whites of your eyes • Low blood pressure or dizziness • Muscle rigidity, tremor and/or abnormal muscle movement • Increase in blood sugar level. Your blood sugar level may increase while you are taking amlodipine and atorvastatin tablets. Exercise regularly and make healthy food choices to maintain healthy body weight. Call your healthcare provider right away if: • allergic reactions including swelling of the face, lips, tongue, and/or throat that may cause difficulty in breathing or swallowing which may require treatment right away • you have allergic skin reactions • Chest pain that does not go away or gets worse. Sometimes when you start amlodipine and atorvastatin tablets or increase your dose, chest pain can get worse or a heart attack can happen. If this happens, call your healthcare provider or go to the emergency room right away. Common side effects of amlodipine and atorvastatin tablets include: • nasal congestion, sore throat, runny nose • diarrhea • urinary tract infection • nausea • muscle spasms • throat pain • muscle and joint pain • pain in extremity • upset stomach • musculoskeletal pain • trouble sleeping • swelling of your legs or ankles Additional side effects have been reported: tiredness, tendon problems, memory loss, and confusion. Talk to your healthcare provider or pharmacist about side effects that bother you or do not go away. There are other side effects of amlodipine and atorvastatin tablets. Ask your healthcare provider or pharmacist for a complete list. How do I store amlodipine and atorvastatin tablets? • Store amlodipine and atorvastatin tablets at room temperature, 68°F to 77°F (20°C to 25°C). • Do not keep medicine that is out-of-date or that you no longer need. • Keep amlodipine and atorvastatin tablets and all medicines out of the reach of children. General information about the safe and effective use of amlodipine and atorvastatin tablets Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use amlodipine and atorvastatin tablets for a condition for which it was not prescribed. Do not give amlodipine and atorvastatin tablets to other people, even if they have the same symptoms that you have. It may harm them. If you want more information about amlodipine and atorvastatin tablets, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about amlodipine and atorvastatin tablets that is written for health professionals. What is high blood pressure (hypertension)? You have high blood pressure when the force of blood against the walls of your arteries stays high. This can damage your heart and other parts of your body. Drugs that lower blood pressure lower your risk of having a stroke or heart attack. What is angina (chest pain)? Angina is a pain that keeps coming back when part of your heart does not get enough blood. It feels like something is pressing or squeezing your chest under the breastbone. Sometimes you can feel it in your shoulders, arms, neck, jaw, or back. What is cholesterol? Cholesterol is a fat-like substance made in your body. It is also found in foods. You need some cholesterol for good health, but too much is not good for you. Cholesterol can clog your blood vessels. What is a heart attack? A heart attack occurs when heart muscle does not get enough blood. Symptoms include chest pain, trouble breathing, nausea, and weakness. Heart muscle cells may be damaged or die. The heart cannot pump well or may stop beating. What is a stroke? A stroke occurs when nerve cells in the brain do not get enough blood. The cells may be damaged or die. The damaged cells may cause weakness or problems speaking or thinking. WHAT ARE THE INGREDIENTS IN AMLODIPINE AND ATORVASTATIN TABLETS? Active ingredients: amlodipine besylate, atorvastatin calcium Inactive ingredients: calcium carbonate, croscarmellose sodium, microcrystalline cellulose, pregelatinized starch, polysorbate 80, hydroxypropyl cellulose, purified water, colloidal silicon dioxide (anhydrous), magnesium stearate Film coating: Opadry ® II White 85F28751 (polyvinyl alcohol, titanium dioxide, PEG 3000, and talc) or Opadry ® II Blue 85F10919 (polyvinyl alcohol, titanium dioxide, PEG 3000, talc, and FD&C blue #2) Distributed by: Prasco Laboratories Mason, OH 45040 USA LAB-1465-5.0 Revised: January 2025
Medication Information
Warnings and Precautions
• Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher amlodipine and atorvastatin dosage. Discontinue amlodipine and atorvastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue amlodipine and atorvastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing amlodipine and atorvastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever ( 2 , 5.1 , 7.3 , 8.5 , 8.6 ). • Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported with statin use. Discontinue amlodipine and atorvastatin tablets if IMNM is suspected ( 5.2 ). • Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue amlodipine and atorvastatin ( 5.3 ). • Angina or myocardial infarction may occur with initiation or dose increase ( 5.4 ). • Symptomatic hypotension is possible, particularly in patients with severe aortic stenosis. However, acute hypotension is unlikely ( 5.5 ).
Indications and Usage
Amlodipine and atorvastatin tablets are indicated in patients for whom treatment with both amlodipine and atorvastatin is appropriate. Amlodipine Hypertension Amlodipine is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including amlodipine. Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC). Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly. Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal. Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy. Amlodipine may be used alone or in combination with other antihypertensive agents. Coronary Artery Disease (CAD) Chronic Stable Angina Amlodipine is indicated for the symptomatic treatment of chronic stable angina. Amlodipine may be used alone or in combination with other antianginal agents. Vasospastic Angina (Prinzmetal’s or Variant Angina) Amlodipine is indicated for the treatment of confirmed or suspected vasospastic angina. Amlodipine may be used as monotherapy or in combination with other antianginal agents. Angiographically Documented CAD In patients with recently documented CAD by angiography and without heart failure or an ejection fraction < 40%, amlodipine is indicated to reduce the risk of hospitalization for angina and to reduce the risk of a coronary revascularization procedure. Atorvastatin Atorvastatin is indicated: • To reduce the risk of: o Myocardial infarction (MI), stroke, revascularization procedures, and angina in adults with multiple risk factors for coronary heart disease (CHD) but without clinically evident CHD o MI and stroke in adults with type 2 diabetes mellitus with multiple risk factors for CHD but without clinically evident CHD o Non-fatal MI, fatal and non-fatal stroke, revascularization procedures, hospitalization for congestive heart failure, and angina in adults with clinically evident CHD • As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in: o Adults with primary hyperlipidemia. o Adults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH). • As an adjunct to other LDL-C-lowering therapies, or alone if such treatments are unavailable, to reduce LDL-C in adults and pediatric patients aged 10 years and older with homozygous familial hypercholesterolemia (HoFH). • As an adjunct to diet for the treatment of adults with: o Primary dysbetalipoproteinemia o Hypertriglyceridemia
Dosage and Administration
Usual starting dose (mg daily) Maximum dose (mg daily) Amlodipine 5 Start small adults or children, fragile, or elderly patients, or patients with hepatic insufficiency on 2.5 mg once daily ( 2 ). 10 Atorvastatin 10-20 Start patients requiring large LDL-C reduction (> 45%) at 40 mg once daily ( 2 ). 80
Contraindications
• Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3) ] . • Hypersensitivity to amlodipine, atorvastatin or any excipients in amlodipine and atorvastatin tablets. Hypersensitivity reactions, including anaphylaxis, angioneurotic edema, erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis, have been reported [see Adverse Reactions (6.2) ] .
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: • Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1) ] • Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2) ] • Hepatic Dysfunction [see Warnings and Precautions (5.3) ] • Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.6) ]
Drug Interactions
Atorvastatin is a substrate of CYP3A4 and transporters (e.g., OATP1B1/1B3, P-gp, or BCRP). Atorvastatin plasma levels can be significantly increased with concomitant administration of inhibitors of CYP3A4 and transporters. Table 3 includes a list of drugs that may increase exposure to atorvastatin and may increase the risk of myopathy and rhabdomyolysis when used concomitantly and instructions for preventing or managing them [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3) ]. Table 3. Drug Interactions that may Increase the Risk of Myopathy and Rhabdomyolysis with Atorvastatin Cyclosporine or Gemfibrozil Clinical Impact: Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin and cyclosporine, an inhibitor of CYP3A4 and OATP1B1 [see Clinical Pharmacology (12.3) ]. Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of cyclosporine or gemfibrozil with atorvastatin. Intervention: Concomitant use of cyclosporine or gemfibrozil with atorvastatin is not recommended. Anti-Viral Medications Clinical Impact: Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with many anti-viral medications, which are inhibitors of CYP3A4 and/or transporters (e.g., BCRP, OATP1B1/1B3, P-gp, MRP2, and/or OAT2) [see Clinical Pharmacology (12.3) ]. Cases of myopathy and rhabdomyolysis have been reported with concomitant use of ledipasvir plus sofosbuvir with atorvastatin. Intervention: • Concomitant use of tipranavir plus ritonavir or glecaprevir plus pibrentasvir with atorvastatin is not recommended. • In patients taking lopinavir plus ritonavir, or simeprevir, consider the risk/benefit of concomitant use with atorvastatin. • In patients taking saquinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, fosamprenavir plus ritonavir, elbasvir plus grazoprevir or letermovir do not exceed atorvastatin 20 mg. • In patients taking nelfinavir, do not exceed atorvastatin 40 mg [see Dosage and Administration (2) ]. • Consider the risk/benefit of concomitant use of ledipasvir plus sofosbuvir with atorvastatin. • Monitor all patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Examples: Tipranavir plus ritonavir, glecaprevir plus pibrentasvir, lopinavir plus ritonavir, simeprevir, saquinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, fosamprenavir plus ritonavir, elbasvir plus grazoprevir, letermovir, nelfinavir, and ledipasvir plus sofosbuvir. Select Azole Antifungals or Macrolide Antibiotics Clinical Impact: Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with select azole antifungals or macrolide antibiotics, due to inhibition of CYP3A4 and/or transporters [see Clinical Pharmacology (12.3) ]. Intervention: In patients taking clarithromycin or itraconazole, do not exceed atorvastatin 20 mg [see Dosage and Administration (2) ]. Consider the risk/benefit of concomitant use of other azole antifungals or macrolide antibiotics with atorvastatin. Monitor all patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Examples: Erythromycin, clarithromycin, itraconazole, ketoconazole, posaconazole, and voriconazole. Niacin Clinical Impact: Cases of myopathy and rhabdomyolysis have been observed with concomitant use of lipid modifying dosages of niacin (≥ 1 gram/day niacin) with atorvastatin. Intervention: Consider if the benefit of using lipid modifying dosages of niacin concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Fibrates (other than Gemfibrozil) Clinical Impact: Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with atorvastatin. Intervention: Consider if the benefit of using fibrates concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Colchicine Clinical Impact: Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with atorvastatin. Intervention: Consider the risk/benefit of concomitant use of colchicine with atorvastatin. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. Grapefruit Juice Clinical Impact: Grapefruit juice consumption, especially excessive consumption, more than 1.2 liters/daily can raise the plasma levels of atorvastatin and may increase the risk of myopathy and rhabdomyolysis. Intervention: Avoid intake of large quantities of grapefruit juice, more than 1.2 liters daily, when taking atorvastatin.
Storage and Handling
Amlodipine and atorvastatin tablets contain amlodipine besylate and atorvastatin calcium equivalent to amlodipine and atorvastatin in the dose strengths described below. Amlodipine and atorvastatin tablets are differentiated by tablet color/size and are engraved with a unique number on one side. Combinations of atorvastatin with 2.5 mg amlodipine are round and film-coated white, combinations of atorvastatin with 5 mg amlodipine are oval and film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are oval and are film-coated blue tablets. Amlodipine and atorvastatin tablets are supplied for oral administration in the following strengths and package configurations: Table 16. Amlodipine and Atorvastatin Tablets Packaging Configurations Amlodipine and Atorvastatin Tablets Package Configuration Tablet Strength mg (amlodipine/atorvastatin) NDC # Engraving Side 1/Side 2 Tablet Color Tablet Shape Bottle of 30 2.5/10 66993-263-30 CDT 251/Blank White Round Bottle of 30 2.5/20 66993-264-30 CDT 252/Blank White Round Bottle of 30 2.5/40 66993-265-30 CDT 254/Blank White Round Bottle of 30 5/10 66993-266-30 CDT 051/Pfizer White Oval Bottle of 30 5/20 66993-267-30 CDT 052/Pfizer White Oval Bottle of 30 5/40 66993-268-30 CDT 054/Pfizer White Oval Bottle of 30 5/80 66993-269-30 CDT 058/Pfizer White Oval Bottle of 30 10/10 66993-270-30 CDT 101/Pfizer Blue Oval Bottle of 30 10/20 66993-271-30 CDT 102/Pfizer Blue Oval Bottle of 30 10/40 66993-272-30 CDT 104/Pfizer Blue Oval Bottle of 30 10/80 66993-273-30 CDT 108/Pfizer Blue Oval
How Supplied
Amlodipine and atorvastatin tablets contain amlodipine besylate and atorvastatin calcium equivalent to amlodipine and atorvastatin in the dose strengths described below. Amlodipine and atorvastatin tablets are differentiated by tablet color/size and are engraved with a unique number on one side. Combinations of atorvastatin with 2.5 mg amlodipine are round and film-coated white, combinations of atorvastatin with 5 mg amlodipine are oval and film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are oval and are film-coated blue tablets. Amlodipine and atorvastatin tablets are supplied for oral administration in the following strengths and package configurations: Table 16. Amlodipine and Atorvastatin Tablets Packaging Configurations Amlodipine and Atorvastatin Tablets Package Configuration Tablet Strength mg (amlodipine/atorvastatin) NDC # Engraving Side 1/Side 2 Tablet Color Tablet Shape Bottle of 30 2.5/10 66993-263-30 CDT 251/Blank White Round Bottle of 30 2.5/20 66993-264-30 CDT 252/Blank White Round Bottle of 30 2.5/40 66993-265-30 CDT 254/Blank White Round Bottle of 30 5/10 66993-266-30 CDT 051/Pfizer White Oval Bottle of 30 5/20 66993-267-30 CDT 052/Pfizer White Oval Bottle of 30 5/40 66993-268-30 CDT 054/Pfizer White Oval Bottle of 30 5/80 66993-269-30 CDT 058/Pfizer White Oval Bottle of 30 10/10 66993-270-30 CDT 101/Pfizer Blue Oval Bottle of 30 10/20 66993-271-30 CDT 102/Pfizer Blue Oval Bottle of 30 10/40 66993-272-30 CDT 104/Pfizer Blue Oval Bottle of 30 10/80 66993-273-30 CDT 108/Pfizer Blue Oval
Patient Information
Amlodipine and Atorvastatin Tablets
Read the patient information that comes with amlodipine and atorvastatin tablets before you start taking them, and each time you get a refill. There may be new information. This information does not replace talking with your healthcare provider about your condition or treatment. If you have any questions about amlodipine and atorvastatin tablets, ask your healthcare provider or pharmacist.
What are amlodipine and atorvastatin tablets?
Amlodipine and atorvastatin tablets are a prescription drug that combines Norvasc® (amlodipine besylate) and Lipitor® (atorvastatin calcium) in one pill.
Amlodipine and atorvastatin tablets are used in adults who need both Norvasc and Lipitor.
Norvasc is used to treat:
-
•High blood pressure (hypertension) and
-
•Chest pain (angina) and
-
•Blocked arteries of the heart (coronary artery disease)
Lipitor is used to lower the levels of “bad” cholesterol and triglycerides in your blood. It can also raise the levels of “good” cholesterol.
Lipitor is also used to lower the risk for heart attack, stroke, certain types of heart surgery, and chest pain in patients who have heart disease or risk factors for heart disease such as:
-
•age, smoking, high blood pressure, low levels of “good” cholesterol, heart disease in the family.
Lipitor can lower the risk for heart attack or stroke in patients with diabetes and risk factors such as:
-
•diabetic eye or kidney problems, smoking, or high blood pressure.
Amlodipine and atorvastatin tablets have not been studied in children.
Who should not use amlodipine and atorvastatin tablets?
Do not use amlodipine and atorvastatin tablets if you:
-
•Have liver problems (acute liver failure or decompensated cirrhosis).
-
•Are allergic to anything in amlodipine and atorvastatin tablets. The active ingredients are atorvastatin calcium and amlodipine besylate. Stop using amlodipine and atorvastatin tablets and get medical help right away if you have symptoms of a serious allergic reaction including:
-
oswelling of your face, lips, tongue or throat
-
oproblems breathing or swallowing
-
ofainting or feeling dizzy
-
overy rapid heartbeat
-
osevere skin rash or itching
-
oflu-like symptoms including fever, sore throat, cough, tiredness, and joint pain
-
See the end of this leaflet for a complete list of ingredients.
What should I tell my healthcare provider before taking amlodipine and atorvastatin tablets?
Tell your healthcare provider about all of your health conditions, including, if you:
-
•have unexplained muscle aches or weakness
-
•drink more than 2 glasses of alcohol daily
-
•have heart disease
-
•have diabetes
-
•have thyroid problems
-
•have kidney problems
-
•had a stroke
-
•are pregnant or plan to become pregnant. Amlodipine and atorvastatin tablets may harm your unborn baby. If you become pregnant, stop taking amlodipine and atorvastatin tablets and call your healthcare provider right away.
-
•are breastfeeding or plan to breastfeed. You and your healthcare provider should decide if you will take amlodipine and atorvastatin tablets or breastfeed. You should not do both. Talk to your healthcare provider about the best way to feed your baby if you take amlodipine and atorvastatin tablets.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Amlodipine and atorvastatin tablets and certain other medicines can increase the risk of muscle problems or other side effects. Especially tell your healthcare provider if you take medicines for:
-
•your immune system (cyclosporine)
-
•cholesterol (gemfibrozil)
-
•infections (erythromycin, clarithromycin, itraconazole, ketoconazole, posaconazole, and
-
•voriconazole)
-
•birth control pills
-
•heart failure (digoxin)
-
•gout (colchicine)
-
•niacin
-
•fibrates
-
•treating HIV, AIDS, or hepatitis C (anti-virals)
|
○ tipranavir plus ritonavir ○ ledipasvir plus sofosbuvir ○ saquinavir plus ritonavir ○ fosamprenavir ○ elbasvir plus grazoprevir ○ nelfinavir |
○ glecaprevir plus pibrentasvir ○ simeprevir ○ darunavir plus ritonavir ○ fosamprenavir plus ritonavir ○ letermovir |
You can use nitroglycerin and amlodipine and atorvastatin tablets together. If you take nitroglycerin for chest pain (angina), do not stop taking it while taking amlodipine and atorvastatin tablets.
Ask your healthcare provider or pharmacist for a list of medicines if you are not sure. Know all the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist when you get a new medicine.
How should I take amlodipine and atorvastatin tablets?
-
•Take amlodipine and atorvastatin tablets exactly as your healthcare provider tells you to take it.
-
•Do not change your dose or stop amlodipine and atorvastatin tablets without talking to your healthcare provider.
-
•Your healthcare provider may do blood tests to check your cholesterol levels during your treatment with amlodipine and atorvastatin tablets. Your dose of amlodipine and atorvastatin tablets may be changed based on these blood test results.
-
•Take amlodipine and atorvastatin tablets each day at any time of day. Amlodipine and atorvastatin tablets can be taken with or without food.
-
•Do not break the tablets before taking them. Talk to your healthcare provider if you have a problem swallowing pills.
-
•Your healthcare provider may start you on a cholesterol-lowering diet before giving you amlodipine and atorvastatin tablets. Stay on this low-fat diet when you take amlodipine and atorvastatin tablets.
-
•If you miss a dose, take it as soon as you remember. Do not take amlodipine and atorvastatin tablets if it has been more than 12 hours since you missed your last dose. Wait and take the next dose at your regular time. Do not take 2 doses of amlodipine and atorvastatin tablets at the same time. If you take too much amlodipine and atorvastatin tablets or overdose, call your healthcare provider or Poison Control Center at 1-800-222-1222 or go to the nearest emergency room right away.
What should I avoid while taking amlodipine and atorvastatin tablets?
-
•Avoid drinking more than 1.2 liters of grapefruit juice each day.
What are possible side effects of amlodipine and atorvastatin tablets?
Amlodipine and atorvastatin tablets can cause serious side effects including:
-
•Muscle pain, tenderness and weakness (myopathy). Muscle problems, including muscle breakdown, can be serious in some people and, rarely, cause kidney damage that can lead to death.
Tell your healthcare provider right away if you have:-
ounexplained muscle pain, tenderness, or weakness, especially if you also have a fever or feel more tired than usual while you take amlodipine and atorvastatin tablets.
-
omuscle problems that do not go away after your healthcare provider has told you to stop taking amlodipine and atorvastatin tablets. Your healthcare provider may do further tests to diagnose the cause of your muscle problems.
Your chances of getting muscle problems are higher if you: -
oare taking certain other medicines while you take amlodipine and atorvastatin tablets
-
odrink large amounts of grapefruit juice
-
oare 65 years of age or older
-
ohave thyroid problems (hypothyroidism) that are not controlled
-
ohave kidney problems
-
oare taking higher doses of amlodipine and atorvastatin tablets
-
-
•Liver problems. Your healthcare provider should do blood tests to check your liver before you start taking amlodipine and atorvastatin tablets and if you have symptoms of liver problems while you take amlodipine and atorvastatin tablets. Call your healthcare provider right away if you have the following symptoms of liver problems:
-
ofeel tired or weak
-
onausea or vomiting
-
oloss of appetite
-
oupper belly pain
-
odark amber colored urine
-
oyellowing of your skin or the whites of your eyes
-
•Low blood pressure or dizziness
-
•Muscle rigidity, tremor and/or abnormal muscle movement
-
•Increase in blood sugar level. Your blood sugar level may increase while you are taking amlodipine and atorvastatin tablets.
Exercise regularly and make healthy food choices to maintain healthy body weight.
Call your healthcare provider right away if:
-
•allergic reactions including swelling of the face, lips, tongue, and/or throat that may cause difficulty in breathing or swallowing which may require treatment right away
-
•you have allergic skin reactions
-
•Chest pain that does not go away or gets worse. Sometimes when you start amlodipine and atorvastatin tablets or increase your dose, chest pain can get worse or a heart attack can happen. If this happens, call your healthcare provider or go to the emergency room right away.
Common side effects of amlodipine and atorvastatin tablets include:
|
|
Additional side effects have been reported: tiredness, tendon problems, memory loss, and confusion.
Talk to your healthcare provider or pharmacist about side effects that bother you or do not go away.
There are other side effects of amlodipine and atorvastatin tablets. Ask your healthcare provider or pharmacist for a complete list.
How do I store amlodipine and atorvastatin tablets?
-
•Store amlodipine and atorvastatin tablets at room temperature, 68°F to 77°F (20°C to 25°C).
-
•Do not keep medicine that is out-of-date or that you no longer need.
-
•Keep amlodipine and atorvastatin tablets and all medicines out of the reach of children.
General information about the safe and effective use of amlodipine and atorvastatin tablets
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use amlodipine and atorvastatin tablets for a condition for which it was not prescribed. Do not give amlodipine and atorvastatin tablets to other people, even if they have the same symptoms that you have. It may harm them.
If you want more information about amlodipine and atorvastatin tablets, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about amlodipine and atorvastatin tablets that is written for health professionals.
What is high blood pressure (hypertension)?
You have high blood pressure when the force of blood against the walls of your arteries stays high. This can damage your heart and other parts of your body. Drugs that lower blood pressure lower your risk of having a stroke or heart attack.
What is angina (chest pain)?
Angina is a pain that keeps coming back when part of your heart does not get enough blood. It feels like something is pressing or squeezing your chest under the breastbone. Sometimes you can feel it in your shoulders, arms, neck, jaw, or back.
What is cholesterol?
Cholesterol is a fat-like substance made in your body. It is also found in foods. You need some cholesterol for good health, but too much is not good for you. Cholesterol can clog your blood vessels.
What is a heart attack?
A heart attack occurs when heart muscle does not get enough blood. Symptoms include chest pain, trouble breathing, nausea, and weakness. Heart muscle cells may be damaged or die. The heart cannot pump well or may stop beating.
What is a stroke?
A stroke occurs when nerve cells in the brain do not get enough blood. The cells may be damaged or die. The damaged cells may cause weakness or problems speaking or thinking.
WHAT ARE THE INGREDIENTS IN AMLODIPINE AND ATORVASTATIN TABLETS?
Active ingredients: amlodipine besylate, atorvastatin calcium
Inactive ingredients: calcium carbonate, croscarmellose sodium, microcrystalline cellulose, pregelatinized starch, polysorbate 80, hydroxypropyl cellulose, purified water, colloidal silicon dioxide (anhydrous), magnesium stearate
Film coating: Opadry® II White 85F28751 (polyvinyl alcohol, titanium dioxide, PEG 3000, and talc) or Opadry® II Blue 85F10919 (polyvinyl alcohol, titanium dioxide, PEG 3000, talc, and FD&C blue #2)
Distributed by:
Prasco Laboratories
Mason, OH 45040 USA
LAB-1465-5.0
Revised: January 2025
Description
Contraindications, Pregnancy and Lactation ( 4 ) Removed 05/2024 Warnings and Precautions, CNS Toxicity ( 5.7 ) Removed 05/2024
Section 42229-5
Amlodipine and atorvastatin tablets
Dosage of amlodipine and atorvastatin tablets must be individualized on the basis of both effectiveness and tolerance for each individual component in the treatment of hypertension/angina and hyperlipidemia. Select doses of amlodipine and atorvastatin independently.
Amlodipine and atorvastatin tablets may be substituted for its individually titrated components. Patients may be given the equivalent dose of amlodipine and atorvastatin tablets or a dose of amlodipine and atorvastatin tablets with increased amounts of amlodipine, atorvastatin, or both for additional antianginal effects, blood pressure lowering, or lipid-lowering effect.
Amlodipine and atorvastatin tablets may be used to provide additional therapy for patients already on one of its components. Amlodipine and atorvastatin tablets may be used to initiate treatment in patients with hyperlipidemia and either hypertension or angina.
Section 43683-2
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15°C –30°C (59°F –86°F) [see USP Controlled Room Temperature].
10 Overdosage
There is no information on overdosage with amlodipine and atorvastatin tablets in humans.
11 Description
Amlodipine and atorvastatin tablets combine the calcium channel blocker amlodipine besylate with the HMG-CoA-reductase inhibitor atorvastatin calcium.
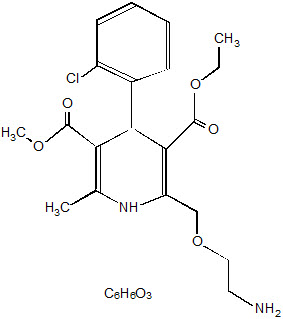
Amlodipine besylate is chemically described as 3-ethyl-5-methyl (±)-2-[(2-aminoethoxy)methyl]-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, monobenzenesulphonate. Its empirical formula is C 20 H 25 ClN 2 O 5 ∙C 6 H 6 O 3 S.
Atorvastatin calcium is chemically described as [R-(R*, R*)]-2-(4-fluorophenyl)-β, δ-dihydroxy-5-(1-methylethyl)-3-phenyl-4-[(phenylamino)carbonyl]-1H-pyrrole-1-heptanoic acid, calcium salt (2:1) trihydrate. Its empirical formula is (C33H34FN2O5)2Ca•3H2O.
The structural formulae for amlodipine besylate and atorvastatin calcium are shown below.
|
Amlodipine besylate |
Atorvastatin calcium |
Amlodipine and atorvastatin tablets contain amlodipine besylate, a white to off-white crystalline powder, and atorvastatin calcium, also a white to off-white crystalline powder. Amlodipine besylate has a molecular weight of 567.1 and atorvastatin calcium has a molecular weight of 1209.42. Amlodipine besylate is slightly soluble in water and sparingly soluble in ethanol. Atorvastatin calcium is insoluble in aqueous solutions of pH 4 and below. Atorvastatin calcium is very slightly soluble in distilled water, pH 7.4 phosphate buffer, and acetonitrile; slightly soluble in ethanol; and freely soluble in methanol.
Amlodipine and atorvastatin tablets are available as film-coated tablets containing:
-
•2.5mg amlodipine equivalent to 3.47mg amlodipine besylate and 10mg atorvastatin equivalent to 10.4mg atorvastatin calcium.
-
•2.5mg amlodipine equivalent to 3.47mg amlodipine besylate and 20mg atorvastatin equivalent to 20.7mg atorvastatin calcium.
-
•2.5mg amlodipine equivalent to 3.47mg amlodipine besylate and 40mg atorvastatin equivalent to 41.4mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 10 mg atorvastatin equivalent to 10.4 mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 20 mg atorvastatin equivalent to 20.7 mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 40 mg atorvastatin equivalent to 41.4 mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 80 mg atorvastatin equivalent to 82.9 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 10 mg atorvastatin equivalent to 10.4 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 20 mg atorvastatin equivalent to 20.7 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 40 mg atorvastatin equivalent to 41.4 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 80 mg atorvastatin equivalent to 82.9 mg atorvastatin calcium.
Each film-coated tablet also contains calcium carbonate, croscarmellose sodium, microcrystalline cellulose, pregelatinized starch, polysorbate 80, hydroxypropyl cellulose, purified water, colloidal silicon dioxide (anhydrous), magnesium stearate, Opadry® II White 85F28751 (polyvinyl alcohol, titanium dioxide, PEG 3000, and talc) or Opadry® II Blue 85F10919 (polyvinyl alcohol, titanium dioxide, PEG 3000, talc, and FD&C blue #2).
5.5 Hypotension
Symptomatic hypotension is possible with use of amlodipine, particularly in patients with severe aortic stenosis. Because of the gradual onset of action, acute hypotension is unlikely.
8.4 Pediatric Use
The safety and effectiveness of amlodipine and atorvastatin tablets have not been established in pediatric populations.
8.5 Geriatric Use
Safety and effectiveness of amlodipine and atorvastatin tablets have not been established in geriatric populations.
4 Contraindications
-
•Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3)].
-
•Hypersensitivity to amlodipine, atorvastatin or any excipients in amlodipine and atorvastatin tablets. Hypersensitivity reactions, including anaphylaxis, angioneurotic edema, erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis, have been reported [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling:
-
•Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1)]
-
•Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2)]
-
•Hepatic Dysfunction [see Warnings and Precautions (5.3)]
-
•Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.6)]
7 Drug Interactions
Data from a drug-drug interaction study involving 10 mg of amlodipine and 80 mg of atorvastatin in healthy subjects indicate that the pharmacokinetics of amlodipine are not altered when the drugs are co-administered. The effect of amlodipine on the pharmacokinetics of atorvastatin showed no effect on the Cmax: 91% (90% confidence interval: 80 to 103%), but the AUC of atorvastatin increased by 18% (90% confidence interval: 109 to 127%) in the presence of amlodipine, which is not clinically meaningful.
No drug interaction studies have been conducted with amlodipine and atorvastatin tablets and other drugs, although studies have been conducted in the individual amlodipine and atorvastatin components, as described below:
Amlodipine
8.6 Renal Impairment
Renal impairment is a risk factor for myopathy and rhabdomyolysis. Monitor all patients with renal impairment for development of myopathy. Renal impairment does not affect the plasma concentrations of atorvastatin, therefore there is no dosage adjustment in patients with renal impairment [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Amlodipine
Following administration of therapeutic doses to patients with hypertension, amlodipine produces vasodilation resulting in a reduction of supine and standing blood pressures. These decreases in blood pressure are not accompanied by a significant change in heart rate or plasma catecholamine levels with chronic dosing. Although the acute intravenous administration of amlodipine decreases arterial blood pressure and increases heart rate in hemodynamic studies of patients with chronic stable angina, chronic oral administration of amlodipine in clinical trials did not lead to clinically significant changes in heart rate or blood pressures in normotensive patients with angina.
With chronic once daily oral administration, antihypertensive effectiveness is maintained for at least 24 hours. Plasma concentrations correlate with effect in both young and elderly patients. The magnitude of reduction in blood pressure with amlodipine is also correlated with the height of pretreatment elevation; thus, individuals with moderate hypertension (diastolic pressure 105–114 mmHg) had about a 50% greater response than patients with mild hypertension (diastolic pressure 90–104 mmHg). Normotensive subjects experienced no clinically significant change in blood pressures (+1/–2 mmHg).
In hypertensive patients with normal renal function, therapeutic doses of amlodipine resulted in a decrease in renal vascular resistance and an increase in glomerular filtration rate and effective renal plasma flow without change in filtration fraction or proteinuria.
As with other calcium channel blockers, hemodynamic measurements of cardiac function at rest and during exercise (or pacing) in patients with normal ventricular function treated with amlodipine have generally demonstrated a small increase in cardiac index without significant influence on dP/dt or on left ventricular end diastolic pressure or volume. In hemodynamic studies, amlodipine has not been associated with a negative inotropic effect when administered in the therapeutic dose range to intact animals and man, even when co-administered with beta-blockers to man. Similar findings, however, have been observed in normal or well-compensated patients with heart failure with agents possessing significant negative inotropic effects.
Amlodipine does not change sinoatrial nodal function or atrioventricular conduction in intact animals or man. In patients with chronic stable angina, intravenous administration of 10 mg did not significantly alter A-H and H-V conduction and sinus node recovery time after pacing. Similar results were obtained in patients receiving amlodipine and concomitant beta-blockers. In clinical studies in which amlodipine was administered in combination with beta-blockers to patients with either hypertension or angina, no adverse effects on electrocardiographic parameters were observed. In clinical trials with angina patients alone, amlodipine therapy did not alter electrocardiographic intervals or produce higher degrees of AV blocks.
8.7 Hepatic Impairment
In patients with chronic alcoholic liver disease, plasma concentrations of atorvastatin are markedly increased. Cmax and AUC are each 4-fold greater in patients with Childs-Pugh A disease. Cmax and AUC are approximately 16-fold and 11-fold increased, respectively, in patients with Childs-Pugh B disease. Amlodipine and atorvastatin tablets are contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)].
1 Indications and Usage
Amlodipine and atorvastatin tablets are indicated in patients for whom treatment with both amlodipine and atorvastatin is appropriate.
Amlodipine
Hypertension
Amlodipine is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including amlodipine.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Amlodipine may be used alone or in combination with other antihypertensive agents.
Coronary Artery Disease (CAD)
Chronic Stable Angina
Amlodipine is indicated for the symptomatic treatment of chronic stable angina. Amlodipine may be used alone or in combination with other antianginal agents.
Vasospastic Angina (Prinzmetal’s or Variant Angina)
Amlodipine is indicated for the treatment of confirmed or suspected vasospastic angina. Amlodipine may be used as monotherapy or in combination with other antianginal agents.
Angiographically Documented CAD
In patients with recently documented CAD by angiography and without heart failure or an ejection fraction < 40%, amlodipine is indicated to reduce the risk of hospitalization for angina and to reduce the risk of a coronary revascularization procedure.
Atorvastatin
Atorvastatin is indicated:
-
•To reduce the risk of:
-
oMyocardial infarction (MI), stroke, revascularization procedures, and angina in adults with multiple risk factors for coronary heart disease (CHD) but without clinically evident CHD
-
oMI and stroke in adults with type 2 diabetes mellitus with multiple risk factors for CHD but without clinically evident CHD
-
oNon-fatal MI, fatal and non-fatal stroke, revascularization procedures, hospitalization for congestive heart failure, and angina in adults with clinically evident CHD
-
-
•As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in:
-
oAdults with primary hyperlipidemia.
-
oAdults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH).
-
-
•As an adjunct to other LDL-C-lowering therapies, or alone if such treatments are unavailable, to reduce LDL-C in adults and pediatric patients aged 10 years and older with homozygous familial hypercholesterolemia (HoFH).
-
•As an adjunct to diet for the treatment of adults with:
-
oPrimary dysbetalipoproteinemia
-
oHypertriglyceridemia
-
5.3 Hepatic Dysfunction
Increases in serum transaminases have been reported with use of atorvastatin [see Adverse Reactions (6.1)]. In most cases, these changes appeared soon after initiation, were transient, were not accompanied by symptoms, and resolved or improved on continued therapy or after a brief interruption in therapy. Persistent increases to more than three times the ULN in serum transaminases have occurred in approximately 0.7% of patients receiving atorvastatin in clinical trials. There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking statins, including atorvastatin.
Patients who consume substantial quantities of alcohol and/or have a history of liver disease may be at increased risk for hepatic injury [see Use in Specific Populations (8.7)].
Consider liver enzyme testing before atorvastatin initiation and when clinically indicated thereafter. Atorvastatin is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)]. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue atorvastatin.
12.1 Mechanism of Action
Amlodipine and atorvastatin tablets are a combination of two drugs, a dihydropyridine calcium channel blocker (amlodipine) and an HMG-CoA reductase inhibitor (atorvastatin). The amlodipine component of amlodipine and atorvastatin tablets inhibits the transmembrane influx of calcium ions into vascular smooth muscle and cardiac muscle. The atorvastatin component of amlodipine and atorvastatin tablets is a selective, competitive inhibitor of HMG-CoA reductase, the rate-limiting enzyme that converts 3-hydroxy-3-methylglutaryl-coenzyme A to mevalonate, a precursor of sterols, including cholesterol.
5 Warnings and Precautions
-
•Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher amlodipine and atorvastatin dosage. Discontinue amlodipine and atorvastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue amlodipine and atorvastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing amlodipine and atorvastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever (2, 5.1, 7.3, 8.5, 8.6).
-
•Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported with statin use. Discontinue amlodipine and atorvastatin tablets if IMNM is suspected (5.2).
-
•Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue amlodipine and atorvastatin (5.3).
-
•Angina or myocardial infarction may occur with initiation or dose increase (5.4).
-
•Symptomatic hypotension is possible, particularly in patients with severe aortic stenosis. However, acute hypotension is unlikely (5.5).
2 Dosage and Administration
3 Dosage Forms and Strengths
Amlodipine and atorvastatin tablets are formulated for oral administration in the following strength combinations:
|
Atorvastatin (mg) |
|||||
|
10 |
20 |
40 |
80 |
||
|
Amlodipine
|
2.5 |
X |
X |
X |
-- |
|
5 |
X |
X |
X |
X |
|
|
10 |
X |
X |
X |
X |
Combinations of atorvastatin with 2.5 mg and 5 mg amlodipine are film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are film-coated blue tablets.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of amlodipine and atorvastatin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Important Dosage Information
Take amlodipine and atorvastatin tablets orally once daily at any time of the day, with or without food.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.1 Myopathy and Rhabdomyolysis
Amlodipine and atorvastatin tablets may cause myopathy (muscle pain, tenderness, or weakness associated with elevated creatine kinase [CK]) and rhabdomyolysis. Acute kidney injury secondary to myoglobinuria and rare fatalities have occurred as a result of rhabdomyolysis in patients treated with statins, including amlodipine and atorvastatin tablets.
14.5 Amlodipine for Heart Failure
Amlodipine has been compared to placebo in four 8–12 week studies of patients with NYHA Class II/III heart failure, involving a total of 697 patients. In these studies, there was no evidence of worsened heart failure based on measures of exercise tolerance, NYHA classification, symptoms, or left ventricular ejection fraction. In a long-term (follow-up at least 6 months, mean 13.8 months) placebo-controlled mortality/morbidity study of amlodipine 5–10 mg in 1153 patients with NYHA Classes III (n=931) or IV (n=222) heart failure on stable doses of diuretics, digoxin, and ACE inhibitors, amlodipine had no effect on the primary endpoint of the study which was the combined endpoint of all-cause mortality and cardiac morbidity (as defined by life-threatening arrhythmia, acute myocardial infarction, or hospitalization for worsened heart failure), or on NYHA classification, or symptoms of heart failure. Total combined all-cause mortality and cardiac morbidity events were 222/571 (39%) for patients on amlodipine and 246/583 (42%) for patients on placebo; the cardiac morbid events represented about 25% of the endpoints in the study.
Another study (PRAISE-2) randomized patients with NYHA Class III (80%) or IV (20%) heart failure without clinical symptoms or objective evidence of underlying ischemic disease, on stable doses of ACE inhibitors (99%), digitalis (99%), and diuretics (99%), to placebo (n=827) or amlodipine (n=827) and followed them for a mean of 33 months. There was no statistically significant difference between amlodipine and placebo in the primary endpoint of all-cause mortality (95% confidence limits from 8% reduction to 29% increase on amlodipine). With amlodipine there were more reports of pulmonary edema.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Amlodipine and atorvastatin tablets contain amlodipine besylate and atorvastatin calcium equivalent to amlodipine and atorvastatin in the dose strengths described below.
Amlodipine and atorvastatin tablets are differentiated by tablet color/size and are engraved with a unique number on one side. Combinations of atorvastatin with 2.5 mg amlodipine are round and film-coated white, combinations of atorvastatin with 5 mg amlodipine are oval and film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are oval and are film-coated blue tablets. Amlodipine and atorvastatin tablets are supplied for oral administration in the following strengths and package configurations:
|
Amlodipine and Atorvastatin Tablets |
|||||
|
Package Configuration |
Tablet Strength mg (amlodipine/atorvastatin) |
NDC # |
Engraving
|
Tablet Color |
Tablet Shape |
|
Bottle of 30 |
2.5/10 |
66993-263-30 |
CDT 251/Blank |
White |
Round |
|
Bottle of 30 |
2.5/20 |
66993-264-30 |
CDT 252/Blank |
White |
Round |
|
Bottle of 30 |
2.5/40 |
66993-265-30 |
CDT 254/Blank |
White |
Round |
|
Bottle of 30 |
5/10 |
66993-266-30 |
CDT 051/Pfizer |
White |
Oval |
|
Bottle of 30 |
5/20 |
66993-267-30 |
CDT 052/Pfizer |
White |
Oval |
|
Bottle of 30 |
5/40 |
66993-268-30 |
CDT 054/Pfizer |
White |
Oval |
|
Bottle of 30 |
5/80 |
66993-269-30 |
CDT 058/Pfizer |
White |
Oval |
|
Bottle of 30 |
10/10 |
66993-270-30 |
CDT 101/Pfizer |
Blue |
Oval |
|
Bottle of 30 |
10/20 |
66993-271-30 |
CDT 102/Pfizer |
Blue |
Oval |
|
Bottle of 30 |
10/40 |
66993-272-30 |
CDT 104/Pfizer |
Blue |
Oval |
|
Bottle of 30 |
10/80 |
66993-273-30 |
CDT 108/Pfizer |
Blue |
Oval |
14.3 Amlodipine for Vasospastic Angina
In a double-blind, placebo-controlled clinical trial of 4 weeks duration in 50 patients, amlodipine therapy decreased attacks by approximately 4/week compared with a placebo decrease of approximately 1/week (p < 0.01). Two of 23 amlodipine and 7 of 27 placebo patients discontinued from the study for lack of clinical improvement.
7.5 Atorvastatin Effects On Other Drugs
Table 5 presents atorvastatin's effect on other drugs and instructions for preventing or managing them.
|
Oral Contraceptives |
|
|
Clinical Impact: |
Co-administration of atorvastatin and an oral contraceptive increased plasma concentrations of norethindrone and ethinyl estradiol [see Clinical Pharmacology (12.3)]. |
|
Intervention: |
Consider this when selecting an oral contraceptive for patients taking atorvastatin. |
|
Digoxin |
|
|
Clinical Impact: |
When multiple doses of atorvastatin and digoxin were co-administered, steady state plasma digoxin concentrations increased [see Clinical Pharmacology (12.3)]. |
|
Intervention: |
Monitor patients taking digoxin appropriately. |
5.2 Immune Mediated Necrotizing Myopathy
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use, including reports of recurrence when the same or a different statin was administered. IMNM is characterized by: proximal muscle weakness and elevated serum creatine kinase that persists despite discontinuation of statin treatment; positive anti-HMG-CoA reductase antibody; muscle biopsy showing necrotizing myopathy; and improvement with immunosuppressive agents. Additional neuromuscular and serologic testing may be necessary. Treatment with immunosuppressive agents may be required. Discontinue amlodipine and atorvastatin tablets if IMNM is suspected.
14.2 Amlodipine for Chronic Stable Angina
The effectiveness of 5–10 mg/day of amlodipine in exercise-induced angina has been evaluated in 8 placebo-controlled, double-blind clinical trials of up to 6 weeks duration involving 1038 patients (684 amlodipine, 354 placebo) with chronic stable angina. In 5 of the 8 studies, significant increases in exercise time (bicycle or treadmill) were seen with the 10 mg dose. Increases in symptom-limited exercise time averaged 12.8% (63 sec) for amlodipine 10 mg, and averaged 7.9% (38 sec) for amlodipine 5 mg. Amlodipine 10 mg also increased time to 1 mm ST segment deviation in several studies and decreased angina attack rate. The sustained efficacy of amlodipine in angina patients has been demonstrated over long-term dosing. In patients with angina, there were no clinically significant reductions in blood pressures (4/1 mmHg) or changes in heart rate (+0.3 bpm).
14.4 Amlodipine for Coronary Artery Disease
In PREVENT, 825 patients with angiographically documented CAD were randomized to amlodipine (5–10 mg once daily) or placebo and followed for 3 years. Although the study did not show significance on the primary objective of change in coronary luminal diameter as assessed by quantitative coronary angiography, the data suggested a favorable outcome with respect to fewer hospitalizations for angina and revascularization procedures in patients with CAD.
CAMELOT enrolled 1318 patients with CAD recently documented by angiography, without left main coronary disease and without heart failure or an ejection fraction < 40%. Patients (76% males, 89% Caucasian, 93% enrolled at U.S. sites, 89% with a history of angina, 52% without PCI, 4% with PCI and no stent, and 44% with a stent) were randomized to double-blind treatment with either amlodipine (5–10 mg once daily) or placebo in addition to standard care that included aspirin (89%), statins (83%), beta-blockers (74%), nitroglycerin (50%), anticoagulants (40%), and diuretics (32%), but excluded other calcium channel blockers. The mean duration of follow-up was 19 months. The primary endpoint was the time to first occurrence of one of the following events: hospitalization for angina pectoris, coronary revascularization, myocardial infarction, cardiovascular death, resuscitated cardiac arrest, hospitalization for heart failure, stroke/TIA, or peripheral vascular disease. A total of 110 (16.6%) and 151 (23.1%) first events occurred in the amlodipine and placebo groups, respectively, for a hazard ratio of 0.691 (95% CI: 0.540–0.884, p=0.003). The primary endpoint is summarized in Figure 1 below. The outcome of this study was largely derived from the prevention of hospitalizations for angina and the prevention of revascularization procedures (see Table 8). Effects in various subgroups are shown in Figure 2.
In an angiographic substudy (n=274) conducted within CAMELOT, there was no significant difference between amlodipine and placebo on the change of atheroma volume in the coronary artery as assessed by intravascular ultrasound.
Figure 1. Kaplan-Meier Analysis of Composite Clinical Outcomes for Amlodipine versus Placebo
Figure 2. Effects on Primary Endpoint of Amlodipine versus Placebo across Sub-Groups
Table 8 below summarizes the significant composite endpoint and clinical outcomes from the composites of the primary endpoint. The other components of the primary endpoint including cardiovascular death, resuscitated cardiac arrest, myocardial infarction, hospitalization for heart failure, stroke/TIA, or peripheral vascular disease did not demonstrate a significant difference between amlodipine and placebo.
|
Clinical Outcomes N (%) |
Amlodipine (N=663) |
Placebo (N=655) |
Risk Reduction (p-value) |
|
Composite CV Endpoint |
110 (16.6) |
151 (23.1) |
31% (0.003) |
|
Hospitalization for Angina Total patients with these events.
|
51 (7.7) |
84 (12.8) |
42% (0.002) |
|
Coronary Revascularization |
78 (11.8) |
103 (15.7) |
27% (0.033) |
5.4 Increased Angina and Myocardial Infarction
Worsening angina and acute myocardial infarction can develop after starting or increasing the dose of amlodipine, particularly in patients with severe obstructive coronary artery disease.
14.8 Atorvastatin for Hypertriglyceridemia in Adults
The response to atorvastatin in 64 patients with isolated hypertriglyceridemia treated across several clinical trials is shown in the table below (Table 12). For the atorvastatin-treated patients, median (min, max) baseline TG level was 565 (267–1,502).
|
Placebo
|
Atorvastatin 10 mg
|
Atorvastatin 20 mg
|
Atorvastatin 80 mg
|
|
|
Triglycerides |
-12.4 (-36.6, 82.7) |
-41.0 (-76.2, 49.4) |
-38.7 (-62.7, 29.5) |
-51.8 (-82.8, 41.3) |
|
Total-C |
-2.3 (-15.5, 24.4) |
-28.2 (-44.9, -6.8) |
-34.9 (-49.6, -15.2) |
-44.4 (-63.5, -3.8) |
|
LDL-C |
3.6 (-31.3, 31.6) |
-26.5 (-57.7, 9.8) |
-30.4 (-53.9, 0.3) |
-40.5 (-60.6, -13.8) |
|
HDL-C |
3.8 (-18.6, 13.4) |
13.8 (-9.7, 61.5) |
11.0 (-3.2, 25.2) |
7.5 (-10.8, 37.2) |
|
non-HDL-C |
-2.8 (-17.6, 30.0) |
-33.0 (-52.1, -13.3) |
-42.7 (-53.7, -17.4) |
-51.5 (-72.9, -4.3) |
14.7 Atorvastatin for Primary Hyperlipidemia in Adults
Atorvastatin reduces total-C, LDL-C, apo B, and TG, and increases HDL-C in patients with hyperlipidemia (heterozygous familial and nonfamilial) and mixed dyslipidemia. Therapeutic response is seen within 2 weeks, and maximum response is usually achieved within 4 weeks and maintained during chronic therapy.
In two multicenter, placebo-controlled, dose-response trials in patients with hyperlipidemia, atorvastatin given as a single dose over 6 weeks significantly reduced total-C, LDL-C, apo B, and TG. (Pooled results are provided in Table 10).
|
Dose |
N |
TC |
LDL-C |
Apo B |
TG |
HDL-C |
|
Placebo |
21 |
4 |
4 |
3 |
10 |
-3 |
|
10 |
22 |
-29 |
-39 |
-32 |
-19 |
6 |
|
20 |
20 |
-33 |
-43 |
-35 |
-26 |
9 |
|
40 |
21 |
-37 |
-50 |
-42 |
-29 |
6 |
|
80 |
23 |
-45 |
-60 |
-50 |
-37 |
5 |
In three multicenter, double-blind trials in patients with hyperlipidemia, atorvastatin was compared to other statins. After randomization, patients were treated for 16 weeks with either atorvastatin 10 mg per day or a fixed dose of the comparative agent (Table 11).
|
Treatment
|
N |
Total-C |
LDL-C |
Apo B |
TG |
HDL-C |
|
Trial 1 |
||||||
|
Atorvastatin 10 mg |
707 |
-27 Significantly different from lovastatin, ANCOVA, p ≤ 0.05
|
-36 |
-28 |
-17 |
+7 |
|
Lovastatin 20 mg |
191 |
-19 |
-27 |
-20 |
-6 |
+7 |
|
95% CI for Diff A negative value for the 95% CI for the difference between treatments favors atorvastatin for all except HDL-C, for which a positive value favors atorvastatin. If the range does not include 0, this indicates a statistically significant difference.
|
-9.2, -6.5 |
-10.7, -7.1 |
-10.0, -6.5 |
-15.2, -7.1 |
-1.7, 2.0 |
|
|
Trial 2 |
||||||
|
Atorvastatin 10 mg |
222 |
-25 Significantly different from pravastatin, ANCOVA, p ≤ 0.05
|
-35 |
-27 |
-17 |
+6 |
|
Pravastatin 20 mg |
77 |
-17 |
-23 |
-17 |
-9 |
+8 |
|
95% CI for Diff |
-10.8, -6.1 |
-14.5, -8.2 |
-13.4, -7.4 |
-14.1, -0.7 |
-4.9, 1.6 |
|
|
Trial 3 |
||||||
|
Atorvastatin 10 mg |
132 |
-29 Significantly different from simvastatin, ANCOVA, p ≤ 0.05
|
-37 |
-34 |
-23 |
+7 |
|
Simvastatin 10 mg |
45 |
-24 |
-30 |
-30 |
-15 |
+7 |
|
95% CI for Diff |
-8.7, -2.7 |
-10.1, -2.6 |
-8.0, -1.1 |
-15.1, -0.7 |
-4.3, 3.9 |
Table 11 does not contain data comparing the effects of atorvastatin 10 mg and higher dosage of lovastatin, pravastatin, and simvastatin. The drugs compared in the trials summarized in the table are not necessarily exchangeable.
14.9 Atorvastatin for Dysbetalipoproteinemia in Adults
The results of an open-label crossover trial of 16 patients (genotypes: 14 apo E2/E2 and 2 apo E3/E2) with dysbetalipoproteinemia are shown in the table below (Table 13).
|
Median % Change (min, max) |
|||
|
Median (min, max) at Baseline (mg/dL) |
Atorvastatin 10 mg |
Atorvastatin 80 mg |
|
|
Total-C |
442 (225, 1320) |
-37 (-85, 17) |
-58 (-90, -31) |
|
Triglycerides |
678 (273, 5990) |
-39 (-92, -8) |
-53 (-95, -30) |
|
Intermediate-density lipoprotein cholesterol (IDL-C) + VLDL-C |
215 (111, 613) |
-32 (-76, 9) |
-63 (-90, -8) |
|
non-HDL-C |
411 (218, 1272) |
-43 (-87, -19) |
-64 (-92, -36) |
5.6 Increases in Hba1c and Fasting Serum Glucose Levels
Increases in HbA1c and fasting serum glucose levels have been reported with statins, including atorvastatin. Optimize lifestyle measures, including regular exercise, maintaining a healthy body weight, and making healthy food choices.
Principal Display Panel 5 Mg/10 Mg Tablet Bottle Label
NDC 66993-266-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/10 mg*
Rx Only
Principal Display Panel 5 Mg/20 Mg Tablet Bottle Label
NDC 66993-267-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/20 mg*
Rx Only
Principal Display Panel 5 Mg/40 Mg Tablet Bottle Label
NDC 66993-268-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/40 mg*
Rx Only
Principal Display Panel 5 Mg/80 Mg Tablet Bottle Label
NDC 66993-269-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/80 mg*
Rx Only
Principal Display Panel 10 Mg/10 Mg Tablet Bottle Label
NDC 66993-270-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/10 mg*
Rx Only
Principal Display Panel 10 Mg/20 Mg Tablet Bottle Label
NDC 66993-271-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/20 mg*
Rx Only
Principal Display Panel 10 Mg/40 Mg Tablet Bottle Label
NDC 66993-272-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/40 mg*
Rx Only
Principal Display Panel 10 Mg/80 Mg Tablet Bottle Label
NDC 66993-273-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/80 mg*
Rx Only
14.6 Atorvastatin for Prevention of Cardiovascular Disease
In the Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT), the effect of atorvastatin on fatal and non-fatal coronary heart disease was assessed in 10,305 patients with hypertension, 40-80 years of age (mean of 63 years; 19% female; 95% White, 3% Black or African American, 1% South Asian, 1% other), without a previous myocardial infarction and with total cholesterol (TC) levels ≤ 251 mg/dL. Additionally, all patients had at least 3 of the following cardiovascular risk factors: male gender (81%), age > 55 years (85%), smoking (33%), diabetes (24%), history of CHD in a first-degree relative (26%), TC:HDL > 6 (14%), peripheral vascular disease (5%), left ventricular hypertrophy (14%), prior cerebrovascular event (10%), specific ECG abnormality (14%), proteinuria/albuminuria (62%). In this double-blind, placebo-controlled trial, patients were treated with antihypertensive therapy (goal BP < 140/90 mm Hg for patients without diabetes; < 130/80 mm Hg for patients with diabetes) and allocated to either atorvastatin 10 mg daily (n=5168) or placebo (n=5137), using a covariate adaptive method which took into account the distribution of nine baseline characteristics of patients already enrolled and minimized the imbalance of those characteristics across the groups. Patients were followed for a median duration of 3.3 years.
The effect of 10 mg/day of atorvastatin on lipid levels was similar to that seen in previous clinical trials.
Atorvastatin significantly reduced the rate of coronary events [either fatal coronary heart disease (46 events in the placebo group vs. 40 events in the atorvastatin group) or non-fatal MI (108 events in the placebo group vs. 60 events in the atorvastatin group)] with a relative risk reduction of 36% [(based on incidences of 1.9% for atorvastatin vs. 3.0% for placebo), p=0.0005 (see Figure 3)]. The risk reduction was consistent regardless of age, smoking status, obesity, or presence of renal dysfunction. The effect of atorvastatin was seen regardless of baseline LDL levels.
Figure 3. Effect of Atorvastatin 10 mg/day on Cumulative Incidence of Non-Fatal Myocardial Infarction or Coronary Heart Disease Death (in ASCOT-LLA)
Atorvastatin also significantly decreased the relative risk for revascularization procedures by 42% (incidences of 1.4% for atorvastatin and 2.5% for placebo). Although the reduction of fatal and non-fatal strokes did not reach a pre-defined significance level (p=0.01), a favorable trend was observed with a 26% relative risk reduction (incidences of 1.7% for atorvastatin and 2.3% for placebo). There was no significant difference between the treatment groups for death due to cardiovascular causes (p=0.51) or noncardiovascular causes (p=0.17).
In the Collaborative Atorvastatin Diabetes Study (CARDS), the effect of atorvastatin on cardiovascular disease (CVD) endpoints was assessed in 2838 subjects (94% White, 2% Black or African American, 2% South Asian, 1% other, 68% male), ages 40–75 with type 2 diabetes based on WHO criteria, without prior history of cardiovascular disease and with LDL ≤ 160 mg/dL and triglycerides (TG) ≤ 600 mg/dL. In addition to diabetes, subjects had 1 or more of the following risk factors: current smoking (23%), hypertension (80%), retinopathy (30%), or microalbuminuria (9%) or macroalbuminuria (3%). No subjects on hemodialysis were enrolled in the trial. In this multicenter, placebo-controlled, double-blind clinical trial, subjects were randomly allocated to either atorvastatin 10 mg daily (1429) or placebo (1411) in a 1:1 ratio and were followed for a median duration of 3.9 years. The primary endpoint was the occurrence of any of the major cardiovascular events: myocardial infarction, acute CHD death, unstable angina, coronary revascularization, or stroke. The primary analysis was the time to first occurrence of the primary endpoint.
Baseline characteristics of subjects were: mean age of 62 years; mean HbA1c 7.7%; median LDL-C 120 mg/dL; median TC 207 mg/dL; median TG 151 mg/dL; median HDL-C 52 mg/dL.
The effect of atorvastatin 10 mg/day on lipid levels was similar to that seen in previous clinical trials.
Atorvastatin significantly reduced the rate of major cardiovascular events (primary endpoint events) (83 events in the atorvastatin group vs. 127 events in the placebo group) with a relative risk reduction of 37%, HR 0.63, 95% CI (0.48, 0.83) (p=0.001) (see Figure 4). An effect of atorvastatin was seen regardless of age, sex, or baseline lipid levels.
Atorvastatin significantly reduced the risk of stroke by 48% (21 events in the atorvastatin group vs. 39 events in the placebo group), HR 0.52, 95% CI (0.31, 0.89) (p=0.016) and reduced the risk of MI by 42% (38 events in the atorvastatin group vs. 64 events in the placebo group), HR 0.58, 95.1% CI (0.39, 0.86) (p=0.007). There was no significant difference between the treatment groups for angina, revascularization procedures, and acute CHD death.
There were 61 deaths in the atorvastatin group vs. 82 deaths in the placebo group (HR 0.73, p=0.059).
Figure 4. Effect of Atorvastatin 10 mg/day on Time to Occurrence of Major Cardiovascular Event (myocardial infarction, acute CHD death, unstable angina, coronary revascularization, or stroke) in CARDS
In the Treating to New Targets Study (TNT), the effect of atorvastatin 80 mg/day vs. atorvastatin 10 mg/day on the reduction in cardiovascular events was assessed in 10,001 subjects (94% White, 81% male, 38% ≥ 65 years) with clinically evident coronary heart disease who had achieved a target LDL-C level < 130 mg/dL after completing an 8-week, open-label, run-in period with atorvastatin 10 mg/day. Subjects were randomly assigned to either 10 mg/day or 80 mg/day of atorvastatin and followed for a median duration of 4.9 years. The primary endpoint was the time to first occurrence of any of the following major cardiovascular events (MCVE): death due to CHD, non-fatal myocardial infarction, resuscitated cardiac arrest, and fatal and non-fatal stroke. The mean LDL-C, TC, TG, non-HDL, and HDL-cholesterol levels at 12 weeks were 73, 145, 128, 98, and 47 mg/dL during treatment with 80 mg of atorvastatin and 99, 177, 152, 129, and 48 mg/dL during treatment with 10 mg of atorvastatin.
Treatment with atorvastatin 80 mg/day significantly reduced the rate of MCVE (434 events in the 80 mg/day group vs. 548 events in the 10 mg/day group) with a relative risk reduction of 22%, HR 0.78, 95% CI (0.69, 0.89), p=0.0002 (see Figure 5 and Table 9). The overall risk reduction was consistent regardless of age (< 65, ≥ 65) or sex.
Figure 5. Effect of Atorvastatin 80 mg/day vs. 10 mg/day on Time to Occurrence of Major Cardiovascular Events (TNT)
| HR=hazard ratio; CHD=coronary heart disease; CI=confidence interval; MI=myocardial infarction; CHF=congestive heart failure; CV=cardiovascular; PVD=peripheral vascular disease; CABG=coronary artery bypass graft | |||||
| Confidence intervals for the Secondary Endpoints were not adjusted for multiple comparisons. | |||||
|
Endpoint |
Atorvastatin 10 mg (N=5006) |
Atorvastatin 80 mg (N=4995) |
HR Atorvastatin 80 mg: atorvastatin 10 mg
(95% CI)
|
||
|
PRIMARY ENDPOINT |
n |
(%) |
n |
(%) |
|
|
First major cardiovascular endpoint |
548 |
(10.9) |
434 |
(8.7) |
0.78 (0.69, 0.89) |
|
Components of the Primary Endpoint |
|||||
|
CHD death |
127 |
(2.5) |
101 |
(2.0) |
0.80 (0.61, 1.03) |
|
Non-fatal, non-procedure related MI |
308 |
(6.2) |
243 |
(4.9) |
0.78 (0.66, 0.93) |
|
Resuscitated cardiac arrest |
26 |
(0.5) |
25 |
(0.5) |
0.96 (0.56, 1.67) |
|
Stroke (fatal and non-fatal) |
155 |
(3.1) |
117 |
(2.3) |
0.75 (0.59, 0.96) |
|
SECONDARY ENDPOINTS Secondary endpoints not included in primary endpoint.
|
|||||
|
First CHF with hospitalization |
164 |
(3.3) |
122 |
(2.4) |
0.74 (0.59, 0.94) |
|
First PVD endpoint |
282 |
(5.6) |
275 |
(5.5) |
0.97 (0.83, 1.15) |
|
First CABG or other coronary revascularization procedure Component of other secondary endpoints
|
904 |
(18.1) |
667 |
(13.4) |
0.72 (0.65, 0.80) |
|
First documented angina endpoint |
615 |
(12.3) |
545 |
(10.9) |
0.88 (0.79, 0.99) |
|
All-cause mortality |
282 |
(5.6) |
284 |
(5.7) |
1.01 (0.85, 1.19) |
|
Components of All-Cause Mortality |
|||||
|
Cardiovascular death |
155 |
(3.1) |
126 |
(2.5) |
0.81 (0.64, 1.03) |
|
Noncardiovascular death |
127 |
(2.5) |
158 |
(3.2) |
1.25 (0.99, 1.57) |
|
Cancer death |
75 |
(1.5) |
85 |
(1.7) |
1.13 (0.83, 1.55) |
|
Other non-CV death |
43 |
(0.9) |
58 |
(1.2) |
1.35 (0.91, 2.00) |
|
Suicide, homicide, and other traumatic non-CV death |
9 |
(0.2) |
15 |
(0.3) |
1.67 (0.73, 3.82) |
Of the events that comprised the primary efficacy endpoint, treatment with atorvastatin 80 mg/day significantly reduced the rate of non-fatal, non-procedure related MI and fatal and non-fatal stroke, but not CHD death or resuscitated cardiac arrest (Table 9). Of the predefined secondary endpoints, treatment with atorvastatin 80 mg/day significantly reduced the rate of coronary revascularization, angina, and hospitalization for heart failure, but not peripheral vascular disease. The reduction in the rate of CHF with hospitalization was only observed in the 8% of patients with a prior history of CHF.
There was no significant difference between the treatment groups for all-cause mortality (Table 9). The proportions of subjects who experienced cardiovascular death, including the components of CHD death and fatal stroke, were numerically smaller in the atorvastatin 80 mg group than in the atorvastatin 10 mg treatment group. The proportions of subjects who experienced noncardiovascular death were numerically larger in the atorvastatin 80 mg group than in the atorvastatin 10 mg treatment group.
Principal Display Panel 2.5 Mg/10 Mg Tablet Bottle Label
NDC 66993-263-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
2.5 mg/10 mg*
Rx Only
Principal Display Panel 2.5 Mg/20 Mg Tablet Bottle Label
NDC 66993-264-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
2.5 mg/20 mg*
Rx Only
Principal Display Panel 2.5 Mg/40 Mg Tablet Bottle Label
NDC 66993-265-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
2.5 mg/40 mg*
Rx Only
7.4 Drug Interactions That May Decrease Exposure to Atorvastatin
Table 4 presents drug interactions that may decrease exposure to atorvastatin and instructions for preventing or managing them.
|
Rifampin |
|
|
Clinical Impact: |
Concomitant administration of atorvastatin with rifampin, an inducer of cytochrome P450 3A4 and inhibitor of OATP1B1, can lead to variable reductions in plasma concentrations of atorvastatin. Due to the dual interaction mechanism of rifampin, delayed administration of atorvastatin after administration of rifampin has been associated with a significant reduction in atorvastatin plasma concentrations. |
|
Intervention: |
Administer atorvastatin and rifampin simultaneously. |
14.12 Amlodipine and Atorvastatin Tablets for Hypertension and Dyslipidemia
In a double-blind, placebo-controlled study, a total of 1660 patients with co-morbid hypertension and dyslipidemia received once daily treatment with eight dose combinations of amlodipine and atorvastatin (5/10, 10/10, 5/20, 10/20, 5/40, 10/40, 5/80, or 10/80 mg), amlodipine alone (5 mg or 10 mg), atorvastatin alone (10 mg, 20 mg, 40 mg, or 80 mg), or placebo. In addition to concomitant hypertension and dyslipidemia, 15% of the patients had diabetes mellitus, 22% were smokers, and 14% had a positive family history of cardiovascular disease. At eight weeks, all eight combination-treatment groups of amlodipine and atorvastatin demonstrated statistically significant dose-related reductions in systolic blood pressure (SBP), diastolic blood pressure (DBP), and LDL-C compared to placebo, with no overall modification of effect of either component on SBP, DBP, and LDL-C (Table 15).
|
BP |
Atorvastatin |
||||
|
Amlodipine |
0 mg |
10 mg |
20 mg |
40 mg |
80 mg |
|
0 mg |
— |
-1.5/-0.8 |
-3.2/-0.6 |
-3.2/-1.8 |
-3.4/-0.8 |
|
5 mg |
-9.8/-4.3 |
-10.7/-4.9 |
-12.3/-6.1 |
-9.7/-4.0 |
-9.2/-5.1 |
|
10 mg |
-13.2/-7.1 |
-12.9/-5.8 |
-13.1/-7.3 |
-13.3/-6.5 |
-14.6/-7.8 |
|
LDL-C
|
Atorvastatin |
||||
|
Amlodipine |
0 mg |
10 mg |
20 mg |
40 mg |
80 mg |
|
0 mg |
— |
-32.3 |
-38.4 |
-42.0 |
-46.1 |
|
5 mg |
1.0 |
-37.6 |
-41.2 |
-43.8 |
-47.3 |
|
10 mg |
-1.4 |
-35.5 |
-37.5 |
-42.1 |
-48.0 |
14.11 Atorvastatin for Heterozygous Familial Hypercholesterolemia in Pediatric Patients
In a double-blind, placebo-controlled trial followed by an open-label phase, 187 males and post-menarchal females 10 years to 17 years of age (mean age 14.1 years; 31% female; 92% White, 1.6% Black or African American, 1.6% Asian, 4.8% other) with HeFH or severe hypercholesterolemia, were randomized to atorvastatin (n=140) or placebo (n=47) for 26 weeks and then all received atorvastatin for 26 weeks. Inclusion in the trial required 1) a baseline LDL-C level ≥ 190 mg/dL or 2) a baseline LDL-C level ≥ 160 mg/dL and positive family history of FH or documented premature cardiovascular disease in a first or second-degree relative. The mean baseline LDL-C value was 219 mg/dL (range: 139-385 mg/dL) in the atorvastatin group compared to 230 mg/dL (range: 160–325 mg/dL) in the placebo group. The dosage of atorvastatin (once daily) was 10 mg for the first 4 weeks and uptitrated to 20 mg if the LDL-C level was > 130 mg/dL. The number of atorvastatin-treated patients who required uptitration to 20 mg after Week 4 during the double-blind phase was 78 (56%).
Atorvastatin significantly decreased plasma levels of total-C, LDL-C, triglycerides, and apolipoprotein B during the 26-week double-blind phase (see Table 14).
|
DOSAGE |
N |
Total-C |
LDL-C |
HDL-C |
TG |
Apo B |
|
Placebo |
47 |
-1.5 |
-0.4 |
-1.9 |
1.0 |
0.7 |
|
Atorvastatin |
140 |
-31.4 |
-39.6 |
2.8 |
-12.0 |
-34.0 |
The mean achieved LDL-C value was 130.7 mg/dL (range: 70–242 mg/dL) in the atorvastatin group compared to 228.5 mg/dL (range: 152–385 mg/dL) in the placebo group during the 26-week double-blind phase.
Atorvastatin was also studied in a three year open-label, uncontrolled trial that included 163 patients with HeFH who were 10 years to 15 years old (82 males and 81 females). All patients had a clinical diagnosis of HeFH confirmed by genetic analysis (if not already confirmed by family history). Approximately 98% were White, and less than 1% were Black, African American or Asian. Mean LDL-C at baseline was 232 mg/dL. The starting atorvastatin dosage was 10 mg once daily and doses were adjusted to achieve a target of <130 mg/dL LDL-C. The reductions in LDL-C from baseline were generally consistent across age groups within the trial as well as with previous clinical trials in both adult and pediatric placebo-controlled trials.
14.10 Atorvastatin for Homozygous Familial Hypercholesterolemia in Adults and Pediatric Patients
In a trial without a concurrent control group, 29 patients (mean age of 22 years, median age of 24 years, 31% < 18 years) with HoFH received maximum daily doses of 20 to 80 mg of atorvastatin. The mean LDL-C reduction in this trial was 18%. Twenty-five patients with a reduction in LDL-C had a mean response of 20% (range of 7% to 53%, median of 24%); the remaining 4 patients had 7% to 24% increases in LDL-C. Five of the 29 patients had absent LDL-receptor function. Of these, 2 patients also had a portacaval shunt and had no significant reduction in LDL-C. The remaining 3 receptor-negative patients had a mean LDL-C reduction of 22%.
7.3 Drug Interactions That May Increase the Risk of Myopathy and Rhabdomyolysis With Atorvastatin
Atorvastatin is a substrate of CYP3A4 and transporters (e.g., OATP1B1/1B3, P-gp, or BCRP). Atorvastatin plasma levels can be significantly increased with concomitant administration of inhibitors of CYP3A4 and transporters. Table 3 includes a list of drugs that may increase exposure to atorvastatin and may increase the risk of myopathy and rhabdomyolysis when used concomitantly and instructions for preventing or managing them [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
|
Cyclosporine or Gemfibrozil |
|
|
Clinical Impact: |
Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin and cyclosporine, an inhibitor of CYP3A4 and OATP1B1 [see Clinical Pharmacology (12.3)]. Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of cyclosporine or gemfibrozil with atorvastatin. |
|
Intervention: |
Concomitant use of cyclosporine or gemfibrozil with atorvastatin is not recommended. |
|
Anti-Viral Medications |
|
|
Clinical Impact: |
Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with many anti-viral medications, which are inhibitors of CYP3A4 and/or transporters (e.g., BCRP, OATP1B1/1B3, P-gp, MRP2, and/or OAT2) [see Clinical Pharmacology (12.3)]. Cases of myopathy and rhabdomyolysis have been reported with concomitant use of ledipasvir plus sofosbuvir with atorvastatin. |
|
Intervention: |
|
|
Examples: |
Tipranavir plus ritonavir, glecaprevir plus pibrentasvir, lopinavir plus ritonavir, simeprevir, saquinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, fosamprenavir plus ritonavir, elbasvir plus grazoprevir, letermovir, nelfinavir, and ledipasvir plus sofosbuvir. |
|
Select Azole Antifungals or Macrolide Antibiotics |
|
|
Clinical Impact: |
Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with select azole antifungals or macrolide antibiotics, due to inhibition of CYP3A4 and/or transporters [see Clinical Pharmacology (12.3)]. |
|
Intervention: |
In patients taking clarithromycin or itraconazole, do not exceed atorvastatin 20 mg [see Dosage and Administration (2)]. Consider the risk/benefit of concomitant use of other azole antifungals or macrolide antibiotics with atorvastatin. Monitor all patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Examples: |
Erythromycin, clarithromycin, itraconazole, ketoconazole, posaconazole, and voriconazole. |
|
Niacin |
|
|
Clinical Impact: |
Cases of myopathy and rhabdomyolysis have been observed with concomitant use of lipid modifying dosages of niacin (≥ 1 gram/day niacin) with atorvastatin. |
|
Intervention: |
Consider if the benefit of using lipid modifying dosages of niacin concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Fibrates (other than Gemfibrozil) |
|
|
Clinical Impact: |
Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with atorvastatin. |
|
Intervention: |
Consider if the benefit of using fibrates concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Colchicine |
|
|
Clinical Impact: |
Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with atorvastatin. |
|
Intervention: |
Consider the risk/benefit of concomitant use of colchicine with atorvastatin. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Grapefruit Juice |
|
|
Clinical Impact: |
Grapefruit juice consumption, especially excessive consumption, more than 1.2 liters/daily can raise the plasma levels of atorvastatin and may increase the risk of myopathy and rhabdomyolysis. |
|
Intervention: |
Avoid intake of large quantities of grapefruit juice, more than 1.2 liters daily, when taking atorvastatin. |
5.7 Increased Risk of Hemorrhagic Stroke in Patients On Atorvastatin 80 Mg With Recent Hemorrhagic Stroke
In a post-hoc analysis of the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial where 2365 adult patients, without CHD who had a stroke or Transient Ischemic Attack (TIA) within the preceding 6 months, were treated with atorvastatin 80 mg, a higher incidence of hemorrhagic stroke was seen in the atorvastatin 80 mg group compared to placebo (55, 2.3% atorvastatin vs. 33, 1.4% placebo; HR: 1.68, 95% CI: 1.09, 2.59; p=0.0168). The incidence of fatal hemorrhagic stroke was similar across treatment groups (17 vs. 18 for the atorvastatin and placebo groups, respectively). The incidence of non-fatal hemorrhagic stroke was significantly higher in the atorvastatin group (38, 1.6%) as compared to the placebo group (16, 0.7%). Some baseline characteristics, including hemorrhagic and lacunar stroke on study entry, were associated with a higher incidence of hemorrhagic stroke in the atorvastatin group [see Adverse Reactions (6.1)].
Consider the risk/benefit of use of atorvastatin 80 mg in patients with recent hemorrhagic stroke.
Structured Label Content
Section 42229-5 (42229-5)
Amlodipine and atorvastatin tablets
Dosage of amlodipine and atorvastatin tablets must be individualized on the basis of both effectiveness and tolerance for each individual component in the treatment of hypertension/angina and hyperlipidemia. Select doses of amlodipine and atorvastatin independently.
Amlodipine and atorvastatin tablets may be substituted for its individually titrated components. Patients may be given the equivalent dose of amlodipine and atorvastatin tablets or a dose of amlodipine and atorvastatin tablets with increased amounts of amlodipine, atorvastatin, or both for additional antianginal effects, blood pressure lowering, or lipid-lowering effect.
Amlodipine and atorvastatin tablets may be used to provide additional therapy for patients already on one of its components. Amlodipine and atorvastatin tablets may be used to initiate treatment in patients with hyperlipidemia and either hypertension or angina.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15°C –30°C (59°F –86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
There is no information on overdosage with amlodipine and atorvastatin tablets in humans.
11 Description (11 DESCRIPTION)
Amlodipine and atorvastatin tablets combine the calcium channel blocker amlodipine besylate with the HMG-CoA-reductase inhibitor atorvastatin calcium.
Amlodipine besylate is chemically described as 3-ethyl-5-methyl (±)-2-[(2-aminoethoxy)methyl]-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, monobenzenesulphonate. Its empirical formula is C 20 H 25 ClN 2 O 5 ∙C 6 H 6 O 3 S.
Atorvastatin calcium is chemically described as [R-(R*, R*)]-2-(4-fluorophenyl)-β, δ-dihydroxy-5-(1-methylethyl)-3-phenyl-4-[(phenylamino)carbonyl]-1H-pyrrole-1-heptanoic acid, calcium salt (2:1) trihydrate. Its empirical formula is (C33H34FN2O5)2Ca•3H2O.
The structural formulae for amlodipine besylate and atorvastatin calcium are shown below.
|
Amlodipine besylate |
Atorvastatin calcium |
Amlodipine and atorvastatin tablets contain amlodipine besylate, a white to off-white crystalline powder, and atorvastatin calcium, also a white to off-white crystalline powder. Amlodipine besylate has a molecular weight of 567.1 and atorvastatin calcium has a molecular weight of 1209.42. Amlodipine besylate is slightly soluble in water and sparingly soluble in ethanol. Atorvastatin calcium is insoluble in aqueous solutions of pH 4 and below. Atorvastatin calcium is very slightly soluble in distilled water, pH 7.4 phosphate buffer, and acetonitrile; slightly soluble in ethanol; and freely soluble in methanol.
Amlodipine and atorvastatin tablets are available as film-coated tablets containing:
-
•2.5mg amlodipine equivalent to 3.47mg amlodipine besylate and 10mg atorvastatin equivalent to 10.4mg atorvastatin calcium.
-
•2.5mg amlodipine equivalent to 3.47mg amlodipine besylate and 20mg atorvastatin equivalent to 20.7mg atorvastatin calcium.
-
•2.5mg amlodipine equivalent to 3.47mg amlodipine besylate and 40mg atorvastatin equivalent to 41.4mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 10 mg atorvastatin equivalent to 10.4 mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 20 mg atorvastatin equivalent to 20.7 mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 40 mg atorvastatin equivalent to 41.4 mg atorvastatin calcium.
-
•5 mg amlodipine equivalent to 6.9 mg amlodipine besylate and 80 mg atorvastatin equivalent to 82.9 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 10 mg atorvastatin equivalent to 10.4 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 20 mg atorvastatin equivalent to 20.7 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 40 mg atorvastatin equivalent to 41.4 mg atorvastatin calcium.
-
•10 mg amlodipine equivalent to 13.9 mg amlodipine besylate and 80 mg atorvastatin equivalent to 82.9 mg atorvastatin calcium.
Each film-coated tablet also contains calcium carbonate, croscarmellose sodium, microcrystalline cellulose, pregelatinized starch, polysorbate 80, hydroxypropyl cellulose, purified water, colloidal silicon dioxide (anhydrous), magnesium stearate, Opadry® II White 85F28751 (polyvinyl alcohol, titanium dioxide, PEG 3000, and talc) or Opadry® II Blue 85F10919 (polyvinyl alcohol, titanium dioxide, PEG 3000, talc, and FD&C blue #2).
5.5 Hypotension
Symptomatic hypotension is possible with use of amlodipine, particularly in patients with severe aortic stenosis. Because of the gradual onset of action, acute hypotension is unlikely.
8.4 Pediatric Use
The safety and effectiveness of amlodipine and atorvastatin tablets have not been established in pediatric populations.
8.5 Geriatric Use
Safety and effectiveness of amlodipine and atorvastatin tablets have not been established in geriatric populations.
4 Contraindications (4 CONTRAINDICATIONS)
-
•Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3)].
-
•Hypersensitivity to amlodipine, atorvastatin or any excipients in amlodipine and atorvastatin tablets. Hypersensitivity reactions, including anaphylaxis, angioneurotic edema, erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis, have been reported [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described below and elsewhere in the labeling:
-
•Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1)]
-
•Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2)]
-
•Hepatic Dysfunction [see Warnings and Precautions (5.3)]
-
•Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Data from a drug-drug interaction study involving 10 mg of amlodipine and 80 mg of atorvastatin in healthy subjects indicate that the pharmacokinetics of amlodipine are not altered when the drugs are co-administered. The effect of amlodipine on the pharmacokinetics of atorvastatin showed no effect on the Cmax: 91% (90% confidence interval: 80 to 103%), but the AUC of atorvastatin increased by 18% (90% confidence interval: 109 to 127%) in the presence of amlodipine, which is not clinically meaningful.
No drug interaction studies have been conducted with amlodipine and atorvastatin tablets and other drugs, although studies have been conducted in the individual amlodipine and atorvastatin components, as described below:
Amlodipine
Patient Information (PATIENT INFORMATION)
Amlodipine and Atorvastatin Tablets
Read the patient information that comes with amlodipine and atorvastatin tablets before you start taking them, and each time you get a refill. There may be new information. This information does not replace talking with your healthcare provider about your condition or treatment. If you have any questions about amlodipine and atorvastatin tablets, ask your healthcare provider or pharmacist.
What are amlodipine and atorvastatin tablets?
Amlodipine and atorvastatin tablets are a prescription drug that combines Norvasc® (amlodipine besylate) and Lipitor® (atorvastatin calcium) in one pill.
Amlodipine and atorvastatin tablets are used in adults who need both Norvasc and Lipitor.
Norvasc is used to treat:
-
•High blood pressure (hypertension) and
-
•Chest pain (angina) and
-
•Blocked arteries of the heart (coronary artery disease)
Lipitor is used to lower the levels of “bad” cholesterol and triglycerides in your blood. It can also raise the levels of “good” cholesterol.
Lipitor is also used to lower the risk for heart attack, stroke, certain types of heart surgery, and chest pain in patients who have heart disease or risk factors for heart disease such as:
-
•age, smoking, high blood pressure, low levels of “good” cholesterol, heart disease in the family.
Lipitor can lower the risk for heart attack or stroke in patients with diabetes and risk factors such as:
-
•diabetic eye or kidney problems, smoking, or high blood pressure.
Amlodipine and atorvastatin tablets have not been studied in children.
Who should not use amlodipine and atorvastatin tablets?
Do not use amlodipine and atorvastatin tablets if you:
-
•Have liver problems (acute liver failure or decompensated cirrhosis).
-
•Are allergic to anything in amlodipine and atorvastatin tablets. The active ingredients are atorvastatin calcium and amlodipine besylate. Stop using amlodipine and atorvastatin tablets and get medical help right away if you have symptoms of a serious allergic reaction including:
-
oswelling of your face, lips, tongue or throat
-
oproblems breathing or swallowing
-
ofainting or feeling dizzy
-
overy rapid heartbeat
-
osevere skin rash or itching
-
oflu-like symptoms including fever, sore throat, cough, tiredness, and joint pain
-
See the end of this leaflet for a complete list of ingredients.
What should I tell my healthcare provider before taking amlodipine and atorvastatin tablets?
Tell your healthcare provider about all of your health conditions, including, if you:
-
•have unexplained muscle aches or weakness
-
•drink more than 2 glasses of alcohol daily
-
•have heart disease
-
•have diabetes
-
•have thyroid problems
-
•have kidney problems
-
•had a stroke
-
•are pregnant or plan to become pregnant. Amlodipine and atorvastatin tablets may harm your unborn baby. If you become pregnant, stop taking amlodipine and atorvastatin tablets and call your healthcare provider right away.
-
•are breastfeeding or plan to breastfeed. You and your healthcare provider should decide if you will take amlodipine and atorvastatin tablets or breastfeed. You should not do both. Talk to your healthcare provider about the best way to feed your baby if you take amlodipine and atorvastatin tablets.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Amlodipine and atorvastatin tablets and certain other medicines can increase the risk of muscle problems or other side effects. Especially tell your healthcare provider if you take medicines for:
-
•your immune system (cyclosporine)
-
•cholesterol (gemfibrozil)
-
•infections (erythromycin, clarithromycin, itraconazole, ketoconazole, posaconazole, and
-
•voriconazole)
-
•birth control pills
-
•heart failure (digoxin)
-
•gout (colchicine)
-
•niacin
-
•fibrates
-
•treating HIV, AIDS, or hepatitis C (anti-virals)
|
○ tipranavir plus ritonavir ○ ledipasvir plus sofosbuvir ○ saquinavir plus ritonavir ○ fosamprenavir ○ elbasvir plus grazoprevir ○ nelfinavir |
○ glecaprevir plus pibrentasvir ○ simeprevir ○ darunavir plus ritonavir ○ fosamprenavir plus ritonavir ○ letermovir |
You can use nitroglycerin and amlodipine and atorvastatin tablets together. If you take nitroglycerin for chest pain (angina), do not stop taking it while taking amlodipine and atorvastatin tablets.
Ask your healthcare provider or pharmacist for a list of medicines if you are not sure. Know all the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist when you get a new medicine.
How should I take amlodipine and atorvastatin tablets?
-
•Take amlodipine and atorvastatin tablets exactly as your healthcare provider tells you to take it.
-
•Do not change your dose or stop amlodipine and atorvastatin tablets without talking to your healthcare provider.
-
•Your healthcare provider may do blood tests to check your cholesterol levels during your treatment with amlodipine and atorvastatin tablets. Your dose of amlodipine and atorvastatin tablets may be changed based on these blood test results.
-
•Take amlodipine and atorvastatin tablets each day at any time of day. Amlodipine and atorvastatin tablets can be taken with or without food.
-
•Do not break the tablets before taking them. Talk to your healthcare provider if you have a problem swallowing pills.
-
•Your healthcare provider may start you on a cholesterol-lowering diet before giving you amlodipine and atorvastatin tablets. Stay on this low-fat diet when you take amlodipine and atorvastatin tablets.
-
•If you miss a dose, take it as soon as you remember. Do not take amlodipine and atorvastatin tablets if it has been more than 12 hours since you missed your last dose. Wait and take the next dose at your regular time. Do not take 2 doses of amlodipine and atorvastatin tablets at the same time. If you take too much amlodipine and atorvastatin tablets or overdose, call your healthcare provider or Poison Control Center at 1-800-222-1222 or go to the nearest emergency room right away.
What should I avoid while taking amlodipine and atorvastatin tablets?
-
•Avoid drinking more than 1.2 liters of grapefruit juice each day.
What are possible side effects of amlodipine and atorvastatin tablets?
Amlodipine and atorvastatin tablets can cause serious side effects including:
-
•Muscle pain, tenderness and weakness (myopathy). Muscle problems, including muscle breakdown, can be serious in some people and, rarely, cause kidney damage that can lead to death.
Tell your healthcare provider right away if you have:-
ounexplained muscle pain, tenderness, or weakness, especially if you also have a fever or feel more tired than usual while you take amlodipine and atorvastatin tablets.
-
omuscle problems that do not go away after your healthcare provider has told you to stop taking amlodipine and atorvastatin tablets. Your healthcare provider may do further tests to diagnose the cause of your muscle problems.
Your chances of getting muscle problems are higher if you: -
oare taking certain other medicines while you take amlodipine and atorvastatin tablets
-
odrink large amounts of grapefruit juice
-
oare 65 years of age or older
-
ohave thyroid problems (hypothyroidism) that are not controlled
-
ohave kidney problems
-
oare taking higher doses of amlodipine and atorvastatin tablets
-
-
•Liver problems. Your healthcare provider should do blood tests to check your liver before you start taking amlodipine and atorvastatin tablets and if you have symptoms of liver problems while you take amlodipine and atorvastatin tablets. Call your healthcare provider right away if you have the following symptoms of liver problems:
-
ofeel tired or weak
-
onausea or vomiting
-
oloss of appetite
-
oupper belly pain
-
odark amber colored urine
-
oyellowing of your skin or the whites of your eyes
-
•Low blood pressure or dizziness
-
•Muscle rigidity, tremor and/or abnormal muscle movement
-
•Increase in blood sugar level. Your blood sugar level may increase while you are taking amlodipine and atorvastatin tablets.
Exercise regularly and make healthy food choices to maintain healthy body weight.
Call your healthcare provider right away if:
-
•allergic reactions including swelling of the face, lips, tongue, and/or throat that may cause difficulty in breathing or swallowing which may require treatment right away
-
•you have allergic skin reactions
-
•Chest pain that does not go away or gets worse. Sometimes when you start amlodipine and atorvastatin tablets or increase your dose, chest pain can get worse or a heart attack can happen. If this happens, call your healthcare provider or go to the emergency room right away.
Common side effects of amlodipine and atorvastatin tablets include:
|
|
Additional side effects have been reported: tiredness, tendon problems, memory loss, and confusion.
Talk to your healthcare provider or pharmacist about side effects that bother you or do not go away.
There are other side effects of amlodipine and atorvastatin tablets. Ask your healthcare provider or pharmacist for a complete list.
How do I store amlodipine and atorvastatin tablets?
-
•Store amlodipine and atorvastatin tablets at room temperature, 68°F to 77°F (20°C to 25°C).
-
•Do not keep medicine that is out-of-date or that you no longer need.
-
•Keep amlodipine and atorvastatin tablets and all medicines out of the reach of children.
General information about the safe and effective use of amlodipine and atorvastatin tablets
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use amlodipine and atorvastatin tablets for a condition for which it was not prescribed. Do not give amlodipine and atorvastatin tablets to other people, even if they have the same symptoms that you have. It may harm them.
If you want more information about amlodipine and atorvastatin tablets, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about amlodipine and atorvastatin tablets that is written for health professionals.
What is high blood pressure (hypertension)?
You have high blood pressure when the force of blood against the walls of your arteries stays high. This can damage your heart and other parts of your body. Drugs that lower blood pressure lower your risk of having a stroke or heart attack.
What is angina (chest pain)?
Angina is a pain that keeps coming back when part of your heart does not get enough blood. It feels like something is pressing or squeezing your chest under the breastbone. Sometimes you can feel it in your shoulders, arms, neck, jaw, or back.
What is cholesterol?
Cholesterol is a fat-like substance made in your body. It is also found in foods. You need some cholesterol for good health, but too much is not good for you. Cholesterol can clog your blood vessels.
What is a heart attack?
A heart attack occurs when heart muscle does not get enough blood. Symptoms include chest pain, trouble breathing, nausea, and weakness. Heart muscle cells may be damaged or die. The heart cannot pump well or may stop beating.
What is a stroke?
A stroke occurs when nerve cells in the brain do not get enough blood. The cells may be damaged or die. The damaged cells may cause weakness or problems speaking or thinking.
WHAT ARE THE INGREDIENTS IN AMLODIPINE AND ATORVASTATIN TABLETS?
Active ingredients: amlodipine besylate, atorvastatin calcium
Inactive ingredients: calcium carbonate, croscarmellose sodium, microcrystalline cellulose, pregelatinized starch, polysorbate 80, hydroxypropyl cellulose, purified water, colloidal silicon dioxide (anhydrous), magnesium stearate
Film coating: Opadry® II White 85F28751 (polyvinyl alcohol, titanium dioxide, PEG 3000, and talc) or Opadry® II Blue 85F10919 (polyvinyl alcohol, titanium dioxide, PEG 3000, talc, and FD&C blue #2)
Distributed by:
Prasco Laboratories
Mason, OH 45040 USA
LAB-1465-5.0
Revised: January 2025
8.6 Renal Impairment
Renal impairment is a risk factor for myopathy and rhabdomyolysis. Monitor all patients with renal impairment for development of myopathy. Renal impairment does not affect the plasma concentrations of atorvastatin, therefore there is no dosage adjustment in patients with renal impairment [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Amlodipine
Following administration of therapeutic doses to patients with hypertension, amlodipine produces vasodilation resulting in a reduction of supine and standing blood pressures. These decreases in blood pressure are not accompanied by a significant change in heart rate or plasma catecholamine levels with chronic dosing. Although the acute intravenous administration of amlodipine decreases arterial blood pressure and increases heart rate in hemodynamic studies of patients with chronic stable angina, chronic oral administration of amlodipine in clinical trials did not lead to clinically significant changes in heart rate or blood pressures in normotensive patients with angina.
With chronic once daily oral administration, antihypertensive effectiveness is maintained for at least 24 hours. Plasma concentrations correlate with effect in both young and elderly patients. The magnitude of reduction in blood pressure with amlodipine is also correlated with the height of pretreatment elevation; thus, individuals with moderate hypertension (diastolic pressure 105–114 mmHg) had about a 50% greater response than patients with mild hypertension (diastolic pressure 90–104 mmHg). Normotensive subjects experienced no clinically significant change in blood pressures (+1/–2 mmHg).
In hypertensive patients with normal renal function, therapeutic doses of amlodipine resulted in a decrease in renal vascular resistance and an increase in glomerular filtration rate and effective renal plasma flow without change in filtration fraction or proteinuria.
As with other calcium channel blockers, hemodynamic measurements of cardiac function at rest and during exercise (or pacing) in patients with normal ventricular function treated with amlodipine have generally demonstrated a small increase in cardiac index without significant influence on dP/dt or on left ventricular end diastolic pressure or volume. In hemodynamic studies, amlodipine has not been associated with a negative inotropic effect when administered in the therapeutic dose range to intact animals and man, even when co-administered with beta-blockers to man. Similar findings, however, have been observed in normal or well-compensated patients with heart failure with agents possessing significant negative inotropic effects.
Amlodipine does not change sinoatrial nodal function or atrioventricular conduction in intact animals or man. In patients with chronic stable angina, intravenous administration of 10 mg did not significantly alter A-H and H-V conduction and sinus node recovery time after pacing. Similar results were obtained in patients receiving amlodipine and concomitant beta-blockers. In clinical studies in which amlodipine was administered in combination with beta-blockers to patients with either hypertension or angina, no adverse effects on electrocardiographic parameters were observed. In clinical trials with angina patients alone, amlodipine therapy did not alter electrocardiographic intervals or produce higher degrees of AV blocks.
8.7 Hepatic Impairment
In patients with chronic alcoholic liver disease, plasma concentrations of atorvastatin are markedly increased. Cmax and AUC are each 4-fold greater in patients with Childs-Pugh A disease. Cmax and AUC are approximately 16-fold and 11-fold increased, respectively, in patients with Childs-Pugh B disease. Amlodipine and atorvastatin tablets are contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Amlodipine and atorvastatin tablets are indicated in patients for whom treatment with both amlodipine and atorvastatin is appropriate.
Amlodipine
Hypertension
Amlodipine is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including amlodipine.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Amlodipine may be used alone or in combination with other antihypertensive agents.
Coronary Artery Disease (CAD)
Chronic Stable Angina
Amlodipine is indicated for the symptomatic treatment of chronic stable angina. Amlodipine may be used alone or in combination with other antianginal agents.
Vasospastic Angina (Prinzmetal’s or Variant Angina)
Amlodipine is indicated for the treatment of confirmed or suspected vasospastic angina. Amlodipine may be used as monotherapy or in combination with other antianginal agents.
Angiographically Documented CAD
In patients with recently documented CAD by angiography and without heart failure or an ejection fraction < 40%, amlodipine is indicated to reduce the risk of hospitalization for angina and to reduce the risk of a coronary revascularization procedure.
Atorvastatin
Atorvastatin is indicated:
-
•To reduce the risk of:
-
oMyocardial infarction (MI), stroke, revascularization procedures, and angina in adults with multiple risk factors for coronary heart disease (CHD) but without clinically evident CHD
-
oMI and stroke in adults with type 2 diabetes mellitus with multiple risk factors for CHD but without clinically evident CHD
-
oNon-fatal MI, fatal and non-fatal stroke, revascularization procedures, hospitalization for congestive heart failure, and angina in adults with clinically evident CHD
-
-
•As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) in:
-
oAdults with primary hyperlipidemia.
-
oAdults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH).
-
-
•As an adjunct to other LDL-C-lowering therapies, or alone if such treatments are unavailable, to reduce LDL-C in adults and pediatric patients aged 10 years and older with homozygous familial hypercholesterolemia (HoFH).
-
•As an adjunct to diet for the treatment of adults with:
-
oPrimary dysbetalipoproteinemia
-
oHypertriglyceridemia
-
5.3 Hepatic Dysfunction
Increases in serum transaminases have been reported with use of atorvastatin [see Adverse Reactions (6.1)]. In most cases, these changes appeared soon after initiation, were transient, were not accompanied by symptoms, and resolved or improved on continued therapy or after a brief interruption in therapy. Persistent increases to more than three times the ULN in serum transaminases have occurred in approximately 0.7% of patients receiving atorvastatin in clinical trials. There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking statins, including atorvastatin.
Patients who consume substantial quantities of alcohol and/or have a history of liver disease may be at increased risk for hepatic injury [see Use in Specific Populations (8.7)].
Consider liver enzyme testing before atorvastatin initiation and when clinically indicated thereafter. Atorvastatin is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)]. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue atorvastatin.
12.1 Mechanism of Action
Amlodipine and atorvastatin tablets are a combination of two drugs, a dihydropyridine calcium channel blocker (amlodipine) and an HMG-CoA reductase inhibitor (atorvastatin). The amlodipine component of amlodipine and atorvastatin tablets inhibits the transmembrane influx of calcium ions into vascular smooth muscle and cardiac muscle. The atorvastatin component of amlodipine and atorvastatin tablets is a selective, competitive inhibitor of HMG-CoA reductase, the rate-limiting enzyme that converts 3-hydroxy-3-methylglutaryl-coenzyme A to mevalonate, a precursor of sterols, including cholesterol.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Myopathy and Rhabdomyolysis: Risk factors include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs, and higher amlodipine and atorvastatin dosage. Discontinue amlodipine and atorvastatin tablets if markedly elevated CK levels occur or myopathy is diagnosed or suspected. Temporarily discontinue amlodipine and atorvastatin tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing amlodipine and atorvastatin tablets dosage. Instruct patients to promptly report unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever (2, 5.1, 7.3, 8.5, 8.6).
-
•Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported with statin use. Discontinue amlodipine and atorvastatin tablets if IMNM is suspected (5.2).
-
•Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzymes before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue amlodipine and atorvastatin (5.3).
-
•Angina or myocardial infarction may occur with initiation or dose increase (5.4).
-
•Symptomatic hypotension is possible, particularly in patients with severe aortic stenosis. However, acute hypotension is unlikely (5.5).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Amlodipine and atorvastatin tablets are formulated for oral administration in the following strength combinations:
|
Atorvastatin (mg) |
|||||
|
10 |
20 |
40 |
80 |
||
|
Amlodipine
|
2.5 |
X |
X |
X |
-- |
|
5 |
X |
X |
X |
X |
|
|
10 |
X |
X |
X |
X |
Combinations of atorvastatin with 2.5 mg and 5 mg amlodipine are film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are film-coated blue tablets.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of amlodipine and atorvastatin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Important Dosage Information
Take amlodipine and atorvastatin tablets orally once daily at any time of the day, with or without food.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.1 Myopathy and Rhabdomyolysis
Amlodipine and atorvastatin tablets may cause myopathy (muscle pain, tenderness, or weakness associated with elevated creatine kinase [CK]) and rhabdomyolysis. Acute kidney injury secondary to myoglobinuria and rare fatalities have occurred as a result of rhabdomyolysis in patients treated with statins, including amlodipine and atorvastatin tablets.
14.5 Amlodipine for Heart Failure
Amlodipine has been compared to placebo in four 8–12 week studies of patients with NYHA Class II/III heart failure, involving a total of 697 patients. In these studies, there was no evidence of worsened heart failure based on measures of exercise tolerance, NYHA classification, symptoms, or left ventricular ejection fraction. In a long-term (follow-up at least 6 months, mean 13.8 months) placebo-controlled mortality/morbidity study of amlodipine 5–10 mg in 1153 patients with NYHA Classes III (n=931) or IV (n=222) heart failure on stable doses of diuretics, digoxin, and ACE inhibitors, amlodipine had no effect on the primary endpoint of the study which was the combined endpoint of all-cause mortality and cardiac morbidity (as defined by life-threatening arrhythmia, acute myocardial infarction, or hospitalization for worsened heart failure), or on NYHA classification, or symptoms of heart failure. Total combined all-cause mortality and cardiac morbidity events were 222/571 (39%) for patients on amlodipine and 246/583 (42%) for patients on placebo; the cardiac morbid events represented about 25% of the endpoints in the study.
Another study (PRAISE-2) randomized patients with NYHA Class III (80%) or IV (20%) heart failure without clinical symptoms or objective evidence of underlying ischemic disease, on stable doses of ACE inhibitors (99%), digitalis (99%), and diuretics (99%), to placebo (n=827) or amlodipine (n=827) and followed them for a mean of 33 months. There was no statistically significant difference between amlodipine and placebo in the primary endpoint of all-cause mortality (95% confidence limits from 8% reduction to 29% increase on amlodipine). With amlodipine there were more reports of pulmonary edema.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Amlodipine and atorvastatin tablets contain amlodipine besylate and atorvastatin calcium equivalent to amlodipine and atorvastatin in the dose strengths described below.
Amlodipine and atorvastatin tablets are differentiated by tablet color/size and are engraved with a unique number on one side. Combinations of atorvastatin with 2.5 mg amlodipine are round and film-coated white, combinations of atorvastatin with 5 mg amlodipine are oval and film-coated white tablets and combinations of atorvastatin with 10 mg amlodipine are oval and are film-coated blue tablets. Amlodipine and atorvastatin tablets are supplied for oral administration in the following strengths and package configurations:
|
Amlodipine and Atorvastatin Tablets |
|||||
|
Package Configuration |
Tablet Strength mg (amlodipine/atorvastatin) |
NDC # |
Engraving
|
Tablet Color |
Tablet Shape |
|
Bottle of 30 |
2.5/10 |
66993-263-30 |
CDT 251/Blank |
White |
Round |
|
Bottle of 30 |
2.5/20 |
66993-264-30 |
CDT 252/Blank |
White |
Round |
|
Bottle of 30 |
2.5/40 |
66993-265-30 |
CDT 254/Blank |
White |
Round |
|
Bottle of 30 |
5/10 |
66993-266-30 |
CDT 051/Pfizer |
White |
Oval |
|
Bottle of 30 |
5/20 |
66993-267-30 |
CDT 052/Pfizer |
White |
Oval |
|
Bottle of 30 |
5/40 |
66993-268-30 |
CDT 054/Pfizer |
White |
Oval |
|
Bottle of 30 |
5/80 |
66993-269-30 |
CDT 058/Pfizer |
White |
Oval |
|
Bottle of 30 |
10/10 |
66993-270-30 |
CDT 101/Pfizer |
Blue |
Oval |
|
Bottle of 30 |
10/20 |
66993-271-30 |
CDT 102/Pfizer |
Blue |
Oval |
|
Bottle of 30 |
10/40 |
66993-272-30 |
CDT 104/Pfizer |
Blue |
Oval |
|
Bottle of 30 |
10/80 |
66993-273-30 |
CDT 108/Pfizer |
Blue |
Oval |
14.3 Amlodipine for Vasospastic Angina
In a double-blind, placebo-controlled clinical trial of 4 weeks duration in 50 patients, amlodipine therapy decreased attacks by approximately 4/week compared with a placebo decrease of approximately 1/week (p < 0.01). Two of 23 amlodipine and 7 of 27 placebo patients discontinued from the study for lack of clinical improvement.
7.5 Atorvastatin Effects On Other Drugs (7.5 Atorvastatin Effects on Other Drugs)
Table 5 presents atorvastatin's effect on other drugs and instructions for preventing or managing them.
|
Oral Contraceptives |
|
|
Clinical Impact: |
Co-administration of atorvastatin and an oral contraceptive increased plasma concentrations of norethindrone and ethinyl estradiol [see Clinical Pharmacology (12.3)]. |
|
Intervention: |
Consider this when selecting an oral contraceptive for patients taking atorvastatin. |
|
Digoxin |
|
|
Clinical Impact: |
When multiple doses of atorvastatin and digoxin were co-administered, steady state plasma digoxin concentrations increased [see Clinical Pharmacology (12.3)]. |
|
Intervention: |
Monitor patients taking digoxin appropriately. |
5.2 Immune Mediated Necrotizing Myopathy (5.2 Immune-Mediated Necrotizing Myopathy)
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use, including reports of recurrence when the same or a different statin was administered. IMNM is characterized by: proximal muscle weakness and elevated serum creatine kinase that persists despite discontinuation of statin treatment; positive anti-HMG-CoA reductase antibody; muscle biopsy showing necrotizing myopathy; and improvement with immunosuppressive agents. Additional neuromuscular and serologic testing may be necessary. Treatment with immunosuppressive agents may be required. Discontinue amlodipine and atorvastatin tablets if IMNM is suspected.
14.2 Amlodipine for Chronic Stable Angina
The effectiveness of 5–10 mg/day of amlodipine in exercise-induced angina has been evaluated in 8 placebo-controlled, double-blind clinical trials of up to 6 weeks duration involving 1038 patients (684 amlodipine, 354 placebo) with chronic stable angina. In 5 of the 8 studies, significant increases in exercise time (bicycle or treadmill) were seen with the 10 mg dose. Increases in symptom-limited exercise time averaged 12.8% (63 sec) for amlodipine 10 mg, and averaged 7.9% (38 sec) for amlodipine 5 mg. Amlodipine 10 mg also increased time to 1 mm ST segment deviation in several studies and decreased angina attack rate. The sustained efficacy of amlodipine in angina patients has been demonstrated over long-term dosing. In patients with angina, there were no clinically significant reductions in blood pressures (4/1 mmHg) or changes in heart rate (+0.3 bpm).
14.4 Amlodipine for Coronary Artery Disease
In PREVENT, 825 patients with angiographically documented CAD were randomized to amlodipine (5–10 mg once daily) or placebo and followed for 3 years. Although the study did not show significance on the primary objective of change in coronary luminal diameter as assessed by quantitative coronary angiography, the data suggested a favorable outcome with respect to fewer hospitalizations for angina and revascularization procedures in patients with CAD.
CAMELOT enrolled 1318 patients with CAD recently documented by angiography, without left main coronary disease and without heart failure or an ejection fraction < 40%. Patients (76% males, 89% Caucasian, 93% enrolled at U.S. sites, 89% with a history of angina, 52% without PCI, 4% with PCI and no stent, and 44% with a stent) were randomized to double-blind treatment with either amlodipine (5–10 mg once daily) or placebo in addition to standard care that included aspirin (89%), statins (83%), beta-blockers (74%), nitroglycerin (50%), anticoagulants (40%), and diuretics (32%), but excluded other calcium channel blockers. The mean duration of follow-up was 19 months. The primary endpoint was the time to first occurrence of one of the following events: hospitalization for angina pectoris, coronary revascularization, myocardial infarction, cardiovascular death, resuscitated cardiac arrest, hospitalization for heart failure, stroke/TIA, or peripheral vascular disease. A total of 110 (16.6%) and 151 (23.1%) first events occurred in the amlodipine and placebo groups, respectively, for a hazard ratio of 0.691 (95% CI: 0.540–0.884, p=0.003). The primary endpoint is summarized in Figure 1 below. The outcome of this study was largely derived from the prevention of hospitalizations for angina and the prevention of revascularization procedures (see Table 8). Effects in various subgroups are shown in Figure 2.
In an angiographic substudy (n=274) conducted within CAMELOT, there was no significant difference between amlodipine and placebo on the change of atheroma volume in the coronary artery as assessed by intravascular ultrasound.
Figure 1. Kaplan-Meier Analysis of Composite Clinical Outcomes for Amlodipine versus Placebo
Figure 2. Effects on Primary Endpoint of Amlodipine versus Placebo across Sub-Groups
Table 8 below summarizes the significant composite endpoint and clinical outcomes from the composites of the primary endpoint. The other components of the primary endpoint including cardiovascular death, resuscitated cardiac arrest, myocardial infarction, hospitalization for heart failure, stroke/TIA, or peripheral vascular disease did not demonstrate a significant difference between amlodipine and placebo.
|
Clinical Outcomes N (%) |
Amlodipine (N=663) |
Placebo (N=655) |
Risk Reduction (p-value) |
|
Composite CV Endpoint |
110 (16.6) |
151 (23.1) |
31% (0.003) |
|
Hospitalization for Angina Total patients with these events.
|
51 (7.7) |
84 (12.8) |
42% (0.002) |
|
Coronary Revascularization |
78 (11.8) |
103 (15.7) |
27% (0.033) |
5.4 Increased Angina and Myocardial Infarction
Worsening angina and acute myocardial infarction can develop after starting or increasing the dose of amlodipine, particularly in patients with severe obstructive coronary artery disease.
14.8 Atorvastatin for Hypertriglyceridemia in Adults
The response to atorvastatin in 64 patients with isolated hypertriglyceridemia treated across several clinical trials is shown in the table below (Table 12). For the atorvastatin-treated patients, median (min, max) baseline TG level was 565 (267–1,502).
|
Placebo
|
Atorvastatin 10 mg
|
Atorvastatin 20 mg
|
Atorvastatin 80 mg
|
|
|
Triglycerides |
-12.4 (-36.6, 82.7) |
-41.0 (-76.2, 49.4) |
-38.7 (-62.7, 29.5) |
-51.8 (-82.8, 41.3) |
|
Total-C |
-2.3 (-15.5, 24.4) |
-28.2 (-44.9, -6.8) |
-34.9 (-49.6, -15.2) |
-44.4 (-63.5, -3.8) |
|
LDL-C |
3.6 (-31.3, 31.6) |
-26.5 (-57.7, 9.8) |
-30.4 (-53.9, 0.3) |
-40.5 (-60.6, -13.8) |
|
HDL-C |
3.8 (-18.6, 13.4) |
13.8 (-9.7, 61.5) |
11.0 (-3.2, 25.2) |
7.5 (-10.8, 37.2) |
|
non-HDL-C |
-2.8 (-17.6, 30.0) |
-33.0 (-52.1, -13.3) |
-42.7 (-53.7, -17.4) |
-51.5 (-72.9, -4.3) |
14.7 Atorvastatin for Primary Hyperlipidemia in Adults
Atorvastatin reduces total-C, LDL-C, apo B, and TG, and increases HDL-C in patients with hyperlipidemia (heterozygous familial and nonfamilial) and mixed dyslipidemia. Therapeutic response is seen within 2 weeks, and maximum response is usually achieved within 4 weeks and maintained during chronic therapy.
In two multicenter, placebo-controlled, dose-response trials in patients with hyperlipidemia, atorvastatin given as a single dose over 6 weeks significantly reduced total-C, LDL-C, apo B, and TG. (Pooled results are provided in Table 10).
|
Dose |
N |
TC |
LDL-C |
Apo B |
TG |
HDL-C |
|
Placebo |
21 |
4 |
4 |
3 |
10 |
-3 |
|
10 |
22 |
-29 |
-39 |
-32 |
-19 |
6 |
|
20 |
20 |
-33 |
-43 |
-35 |
-26 |
9 |
|
40 |
21 |
-37 |
-50 |
-42 |
-29 |
6 |
|
80 |
23 |
-45 |
-60 |
-50 |
-37 |
5 |
In three multicenter, double-blind trials in patients with hyperlipidemia, atorvastatin was compared to other statins. After randomization, patients were treated for 16 weeks with either atorvastatin 10 mg per day or a fixed dose of the comparative agent (Table 11).
|
Treatment
|
N |
Total-C |
LDL-C |
Apo B |
TG |
HDL-C |
|
Trial 1 |
||||||
|
Atorvastatin 10 mg |
707 |
-27 Significantly different from lovastatin, ANCOVA, p ≤ 0.05
|
-36 |
-28 |
-17 |
+7 |
|
Lovastatin 20 mg |
191 |
-19 |
-27 |
-20 |
-6 |
+7 |
|
95% CI for Diff A negative value for the 95% CI for the difference between treatments favors atorvastatin for all except HDL-C, for which a positive value favors atorvastatin. If the range does not include 0, this indicates a statistically significant difference.
|
-9.2, -6.5 |
-10.7, -7.1 |
-10.0, -6.5 |
-15.2, -7.1 |
-1.7, 2.0 |
|
|
Trial 2 |
||||||
|
Atorvastatin 10 mg |
222 |
-25 Significantly different from pravastatin, ANCOVA, p ≤ 0.05
|
-35 |
-27 |
-17 |
+6 |
|
Pravastatin 20 mg |
77 |
-17 |
-23 |
-17 |
-9 |
+8 |
|
95% CI for Diff |
-10.8, -6.1 |
-14.5, -8.2 |
-13.4, -7.4 |
-14.1, -0.7 |
-4.9, 1.6 |
|
|
Trial 3 |
||||||
|
Atorvastatin 10 mg |
132 |
-29 Significantly different from simvastatin, ANCOVA, p ≤ 0.05
|
-37 |
-34 |
-23 |
+7 |
|
Simvastatin 10 mg |
45 |
-24 |
-30 |
-30 |
-15 |
+7 |
|
95% CI for Diff |
-8.7, -2.7 |
-10.1, -2.6 |
-8.0, -1.1 |
-15.1, -0.7 |
-4.3, 3.9 |
Table 11 does not contain data comparing the effects of atorvastatin 10 mg and higher dosage of lovastatin, pravastatin, and simvastatin. The drugs compared in the trials summarized in the table are not necessarily exchangeable.
14.9 Atorvastatin for Dysbetalipoproteinemia in Adults
The results of an open-label crossover trial of 16 patients (genotypes: 14 apo E2/E2 and 2 apo E3/E2) with dysbetalipoproteinemia are shown in the table below (Table 13).
|
Median % Change (min, max) |
|||
|
Median (min, max) at Baseline (mg/dL) |
Atorvastatin 10 mg |
Atorvastatin 80 mg |
|
|
Total-C |
442 (225, 1320) |
-37 (-85, 17) |
-58 (-90, -31) |
|
Triglycerides |
678 (273, 5990) |
-39 (-92, -8) |
-53 (-95, -30) |
|
Intermediate-density lipoprotein cholesterol (IDL-C) + VLDL-C |
215 (111, 613) |
-32 (-76, 9) |
-63 (-90, -8) |
|
non-HDL-C |
411 (218, 1272) |
-43 (-87, -19) |
-64 (-92, -36) |
5.6 Increases in Hba1c and Fasting Serum Glucose Levels (5.6 Increases in HbA1c and Fasting Serum Glucose Levels)
Increases in HbA1c and fasting serum glucose levels have been reported with statins, including atorvastatin. Optimize lifestyle measures, including regular exercise, maintaining a healthy body weight, and making healthy food choices.
Principal Display Panel 5 Mg/10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg/10 mg Tablet Bottle Label)
NDC 66993-266-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/10 mg*
Rx Only
Principal Display Panel 5 Mg/20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg/20 mg Tablet Bottle Label)
NDC 66993-267-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/20 mg*
Rx Only
Principal Display Panel 5 Mg/40 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg/40 mg Tablet Bottle Label)
NDC 66993-268-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/40 mg*
Rx Only
Principal Display Panel 5 Mg/80 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg/80 mg Tablet Bottle Label)
NDC 66993-269-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
5 mg/80 mg*
Rx Only
Principal Display Panel 10 Mg/10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg/10 mg Tablet Bottle Label)
NDC 66993-270-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/10 mg*
Rx Only
Principal Display Panel 10 Mg/20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg/20 mg Tablet Bottle Label)
NDC 66993-271-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/20 mg*
Rx Only
Principal Display Panel 10 Mg/40 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg/40 mg Tablet Bottle Label)
NDC 66993-272-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/40 mg*
Rx Only
Principal Display Panel 10 Mg/80 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg/80 mg Tablet Bottle Label)
NDC 66993-273-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
10 mg/80 mg*
Rx Only
14.6 Atorvastatin for Prevention of Cardiovascular Disease
In the Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT), the effect of atorvastatin on fatal and non-fatal coronary heart disease was assessed in 10,305 patients with hypertension, 40-80 years of age (mean of 63 years; 19% female; 95% White, 3% Black or African American, 1% South Asian, 1% other), without a previous myocardial infarction and with total cholesterol (TC) levels ≤ 251 mg/dL. Additionally, all patients had at least 3 of the following cardiovascular risk factors: male gender (81%), age > 55 years (85%), smoking (33%), diabetes (24%), history of CHD in a first-degree relative (26%), TC:HDL > 6 (14%), peripheral vascular disease (5%), left ventricular hypertrophy (14%), prior cerebrovascular event (10%), specific ECG abnormality (14%), proteinuria/albuminuria (62%). In this double-blind, placebo-controlled trial, patients were treated with antihypertensive therapy (goal BP < 140/90 mm Hg for patients without diabetes; < 130/80 mm Hg for patients with diabetes) and allocated to either atorvastatin 10 mg daily (n=5168) or placebo (n=5137), using a covariate adaptive method which took into account the distribution of nine baseline characteristics of patients already enrolled and minimized the imbalance of those characteristics across the groups. Patients were followed for a median duration of 3.3 years.
The effect of 10 mg/day of atorvastatin on lipid levels was similar to that seen in previous clinical trials.
Atorvastatin significantly reduced the rate of coronary events [either fatal coronary heart disease (46 events in the placebo group vs. 40 events in the atorvastatin group) or non-fatal MI (108 events in the placebo group vs. 60 events in the atorvastatin group)] with a relative risk reduction of 36% [(based on incidences of 1.9% for atorvastatin vs. 3.0% for placebo), p=0.0005 (see Figure 3)]. The risk reduction was consistent regardless of age, smoking status, obesity, or presence of renal dysfunction. The effect of atorvastatin was seen regardless of baseline LDL levels.
Figure 3. Effect of Atorvastatin 10 mg/day on Cumulative Incidence of Non-Fatal Myocardial Infarction or Coronary Heart Disease Death (in ASCOT-LLA)
Atorvastatin also significantly decreased the relative risk for revascularization procedures by 42% (incidences of 1.4% for atorvastatin and 2.5% for placebo). Although the reduction of fatal and non-fatal strokes did not reach a pre-defined significance level (p=0.01), a favorable trend was observed with a 26% relative risk reduction (incidences of 1.7% for atorvastatin and 2.3% for placebo). There was no significant difference between the treatment groups for death due to cardiovascular causes (p=0.51) or noncardiovascular causes (p=0.17).
In the Collaborative Atorvastatin Diabetes Study (CARDS), the effect of atorvastatin on cardiovascular disease (CVD) endpoints was assessed in 2838 subjects (94% White, 2% Black or African American, 2% South Asian, 1% other, 68% male), ages 40–75 with type 2 diabetes based on WHO criteria, without prior history of cardiovascular disease and with LDL ≤ 160 mg/dL and triglycerides (TG) ≤ 600 mg/dL. In addition to diabetes, subjects had 1 or more of the following risk factors: current smoking (23%), hypertension (80%), retinopathy (30%), or microalbuminuria (9%) or macroalbuminuria (3%). No subjects on hemodialysis were enrolled in the trial. In this multicenter, placebo-controlled, double-blind clinical trial, subjects were randomly allocated to either atorvastatin 10 mg daily (1429) or placebo (1411) in a 1:1 ratio and were followed for a median duration of 3.9 years. The primary endpoint was the occurrence of any of the major cardiovascular events: myocardial infarction, acute CHD death, unstable angina, coronary revascularization, or stroke. The primary analysis was the time to first occurrence of the primary endpoint.
Baseline characteristics of subjects were: mean age of 62 years; mean HbA1c 7.7%; median LDL-C 120 mg/dL; median TC 207 mg/dL; median TG 151 mg/dL; median HDL-C 52 mg/dL.
The effect of atorvastatin 10 mg/day on lipid levels was similar to that seen in previous clinical trials.
Atorvastatin significantly reduced the rate of major cardiovascular events (primary endpoint events) (83 events in the atorvastatin group vs. 127 events in the placebo group) with a relative risk reduction of 37%, HR 0.63, 95% CI (0.48, 0.83) (p=0.001) (see Figure 4). An effect of atorvastatin was seen regardless of age, sex, or baseline lipid levels.
Atorvastatin significantly reduced the risk of stroke by 48% (21 events in the atorvastatin group vs. 39 events in the placebo group), HR 0.52, 95% CI (0.31, 0.89) (p=0.016) and reduced the risk of MI by 42% (38 events in the atorvastatin group vs. 64 events in the placebo group), HR 0.58, 95.1% CI (0.39, 0.86) (p=0.007). There was no significant difference between the treatment groups for angina, revascularization procedures, and acute CHD death.
There were 61 deaths in the atorvastatin group vs. 82 deaths in the placebo group (HR 0.73, p=0.059).
Figure 4. Effect of Atorvastatin 10 mg/day on Time to Occurrence of Major Cardiovascular Event (myocardial infarction, acute CHD death, unstable angina, coronary revascularization, or stroke) in CARDS
In the Treating to New Targets Study (TNT), the effect of atorvastatin 80 mg/day vs. atorvastatin 10 mg/day on the reduction in cardiovascular events was assessed in 10,001 subjects (94% White, 81% male, 38% ≥ 65 years) with clinically evident coronary heart disease who had achieved a target LDL-C level < 130 mg/dL after completing an 8-week, open-label, run-in period with atorvastatin 10 mg/day. Subjects were randomly assigned to either 10 mg/day or 80 mg/day of atorvastatin and followed for a median duration of 4.9 years. The primary endpoint was the time to first occurrence of any of the following major cardiovascular events (MCVE): death due to CHD, non-fatal myocardial infarction, resuscitated cardiac arrest, and fatal and non-fatal stroke. The mean LDL-C, TC, TG, non-HDL, and HDL-cholesterol levels at 12 weeks were 73, 145, 128, 98, and 47 mg/dL during treatment with 80 mg of atorvastatin and 99, 177, 152, 129, and 48 mg/dL during treatment with 10 mg of atorvastatin.
Treatment with atorvastatin 80 mg/day significantly reduced the rate of MCVE (434 events in the 80 mg/day group vs. 548 events in the 10 mg/day group) with a relative risk reduction of 22%, HR 0.78, 95% CI (0.69, 0.89), p=0.0002 (see Figure 5 and Table 9). The overall risk reduction was consistent regardless of age (< 65, ≥ 65) or sex.
Figure 5. Effect of Atorvastatin 80 mg/day vs. 10 mg/day on Time to Occurrence of Major Cardiovascular Events (TNT)
| HR=hazard ratio; CHD=coronary heart disease; CI=confidence interval; MI=myocardial infarction; CHF=congestive heart failure; CV=cardiovascular; PVD=peripheral vascular disease; CABG=coronary artery bypass graft | |||||
| Confidence intervals for the Secondary Endpoints were not adjusted for multiple comparisons. | |||||
|
Endpoint |
Atorvastatin 10 mg (N=5006) |
Atorvastatin 80 mg (N=4995) |
HR Atorvastatin 80 mg: atorvastatin 10 mg
(95% CI)
|
||
|
PRIMARY ENDPOINT |
n |
(%) |
n |
(%) |
|
|
First major cardiovascular endpoint |
548 |
(10.9) |
434 |
(8.7) |
0.78 (0.69, 0.89) |
|
Components of the Primary Endpoint |
|||||
|
CHD death |
127 |
(2.5) |
101 |
(2.0) |
0.80 (0.61, 1.03) |
|
Non-fatal, non-procedure related MI |
308 |
(6.2) |
243 |
(4.9) |
0.78 (0.66, 0.93) |
|
Resuscitated cardiac arrest |
26 |
(0.5) |
25 |
(0.5) |
0.96 (0.56, 1.67) |
|
Stroke (fatal and non-fatal) |
155 |
(3.1) |
117 |
(2.3) |
0.75 (0.59, 0.96) |
|
SECONDARY ENDPOINTS Secondary endpoints not included in primary endpoint.
|
|||||
|
First CHF with hospitalization |
164 |
(3.3) |
122 |
(2.4) |
0.74 (0.59, 0.94) |
|
First PVD endpoint |
282 |
(5.6) |
275 |
(5.5) |
0.97 (0.83, 1.15) |
|
First CABG or other coronary revascularization procedure Component of other secondary endpoints
|
904 |
(18.1) |
667 |
(13.4) |
0.72 (0.65, 0.80) |
|
First documented angina endpoint |
615 |
(12.3) |
545 |
(10.9) |
0.88 (0.79, 0.99) |
|
All-cause mortality |
282 |
(5.6) |
284 |
(5.7) |
1.01 (0.85, 1.19) |
|
Components of All-Cause Mortality |
|||||
|
Cardiovascular death |
155 |
(3.1) |
126 |
(2.5) |
0.81 (0.64, 1.03) |
|
Noncardiovascular death |
127 |
(2.5) |
158 |
(3.2) |
1.25 (0.99, 1.57) |
|
Cancer death |
75 |
(1.5) |
85 |
(1.7) |
1.13 (0.83, 1.55) |
|
Other non-CV death |
43 |
(0.9) |
58 |
(1.2) |
1.35 (0.91, 2.00) |
|
Suicide, homicide, and other traumatic non-CV death |
9 |
(0.2) |
15 |
(0.3) |
1.67 (0.73, 3.82) |
Of the events that comprised the primary efficacy endpoint, treatment with atorvastatin 80 mg/day significantly reduced the rate of non-fatal, non-procedure related MI and fatal and non-fatal stroke, but not CHD death or resuscitated cardiac arrest (Table 9). Of the predefined secondary endpoints, treatment with atorvastatin 80 mg/day significantly reduced the rate of coronary revascularization, angina, and hospitalization for heart failure, but not peripheral vascular disease. The reduction in the rate of CHF with hospitalization was only observed in the 8% of patients with a prior history of CHF.
There was no significant difference between the treatment groups for all-cause mortality (Table 9). The proportions of subjects who experienced cardiovascular death, including the components of CHD death and fatal stroke, were numerically smaller in the atorvastatin 80 mg group than in the atorvastatin 10 mg treatment group. The proportions of subjects who experienced noncardiovascular death were numerically larger in the atorvastatin 80 mg group than in the atorvastatin 10 mg treatment group.
Principal Display Panel 2.5 Mg/10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 2.5 mg/10 mg Tablet Bottle Label)
NDC 66993-263-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
2.5 mg/10 mg*
Rx Only
Principal Display Panel 2.5 Mg/20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 2.5 mg/20 mg Tablet Bottle Label)
NDC 66993-264-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
2.5 mg/20 mg*
Rx Only
Principal Display Panel 2.5 Mg/40 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 2.5 mg/40 mg Tablet Bottle Label)
NDC 66993-265-30
30 Tablets
PRASCO
Amlodipine and
Atorvastatin
Tablets
2.5 mg/40 mg*
Rx Only
7.4 Drug Interactions That May Decrease Exposure to Atorvastatin (7.4 Drug Interactions that may Decrease Exposure to Atorvastatin)
Table 4 presents drug interactions that may decrease exposure to atorvastatin and instructions for preventing or managing them.
|
Rifampin |
|
|
Clinical Impact: |
Concomitant administration of atorvastatin with rifampin, an inducer of cytochrome P450 3A4 and inhibitor of OATP1B1, can lead to variable reductions in plasma concentrations of atorvastatin. Due to the dual interaction mechanism of rifampin, delayed administration of atorvastatin after administration of rifampin has been associated with a significant reduction in atorvastatin plasma concentrations. |
|
Intervention: |
Administer atorvastatin and rifampin simultaneously. |
14.12 Amlodipine and Atorvastatin Tablets for Hypertension and Dyslipidemia
In a double-blind, placebo-controlled study, a total of 1660 patients with co-morbid hypertension and dyslipidemia received once daily treatment with eight dose combinations of amlodipine and atorvastatin (5/10, 10/10, 5/20, 10/20, 5/40, 10/40, 5/80, or 10/80 mg), amlodipine alone (5 mg or 10 mg), atorvastatin alone (10 mg, 20 mg, 40 mg, or 80 mg), or placebo. In addition to concomitant hypertension and dyslipidemia, 15% of the patients had diabetes mellitus, 22% were smokers, and 14% had a positive family history of cardiovascular disease. At eight weeks, all eight combination-treatment groups of amlodipine and atorvastatin demonstrated statistically significant dose-related reductions in systolic blood pressure (SBP), diastolic blood pressure (DBP), and LDL-C compared to placebo, with no overall modification of effect of either component on SBP, DBP, and LDL-C (Table 15).
|
BP |
Atorvastatin |
||||
|
Amlodipine |
0 mg |
10 mg |
20 mg |
40 mg |
80 mg |
|
0 mg |
— |
-1.5/-0.8 |
-3.2/-0.6 |
-3.2/-1.8 |
-3.4/-0.8 |
|
5 mg |
-9.8/-4.3 |
-10.7/-4.9 |
-12.3/-6.1 |
-9.7/-4.0 |
-9.2/-5.1 |
|
10 mg |
-13.2/-7.1 |
-12.9/-5.8 |
-13.1/-7.3 |
-13.3/-6.5 |
-14.6/-7.8 |
|
LDL-C
|
Atorvastatin |
||||
|
Amlodipine |
0 mg |
10 mg |
20 mg |
40 mg |
80 mg |
|
0 mg |
— |
-32.3 |
-38.4 |
-42.0 |
-46.1 |
|
5 mg |
1.0 |
-37.6 |
-41.2 |
-43.8 |
-47.3 |
|
10 mg |
-1.4 |
-35.5 |
-37.5 |
-42.1 |
-48.0 |
14.11 Atorvastatin for Heterozygous Familial Hypercholesterolemia in Pediatric Patients
In a double-blind, placebo-controlled trial followed by an open-label phase, 187 males and post-menarchal females 10 years to 17 years of age (mean age 14.1 years; 31% female; 92% White, 1.6% Black or African American, 1.6% Asian, 4.8% other) with HeFH or severe hypercholesterolemia, were randomized to atorvastatin (n=140) or placebo (n=47) for 26 weeks and then all received atorvastatin for 26 weeks. Inclusion in the trial required 1) a baseline LDL-C level ≥ 190 mg/dL or 2) a baseline LDL-C level ≥ 160 mg/dL and positive family history of FH or documented premature cardiovascular disease in a first or second-degree relative. The mean baseline LDL-C value was 219 mg/dL (range: 139-385 mg/dL) in the atorvastatin group compared to 230 mg/dL (range: 160–325 mg/dL) in the placebo group. The dosage of atorvastatin (once daily) was 10 mg for the first 4 weeks and uptitrated to 20 mg if the LDL-C level was > 130 mg/dL. The number of atorvastatin-treated patients who required uptitration to 20 mg after Week 4 during the double-blind phase was 78 (56%).
Atorvastatin significantly decreased plasma levels of total-C, LDL-C, triglycerides, and apolipoprotein B during the 26-week double-blind phase (see Table 14).
|
DOSAGE |
N |
Total-C |
LDL-C |
HDL-C |
TG |
Apo B |
|
Placebo |
47 |
-1.5 |
-0.4 |
-1.9 |
1.0 |
0.7 |
|
Atorvastatin |
140 |
-31.4 |
-39.6 |
2.8 |
-12.0 |
-34.0 |
The mean achieved LDL-C value was 130.7 mg/dL (range: 70–242 mg/dL) in the atorvastatin group compared to 228.5 mg/dL (range: 152–385 mg/dL) in the placebo group during the 26-week double-blind phase.
Atorvastatin was also studied in a three year open-label, uncontrolled trial that included 163 patients with HeFH who were 10 years to 15 years old (82 males and 81 females). All patients had a clinical diagnosis of HeFH confirmed by genetic analysis (if not already confirmed by family history). Approximately 98% were White, and less than 1% were Black, African American or Asian. Mean LDL-C at baseline was 232 mg/dL. The starting atorvastatin dosage was 10 mg once daily and doses were adjusted to achieve a target of <130 mg/dL LDL-C. The reductions in LDL-C from baseline were generally consistent across age groups within the trial as well as with previous clinical trials in both adult and pediatric placebo-controlled trials.
14.10 Atorvastatin for Homozygous Familial Hypercholesterolemia in Adults and Pediatric Patients
In a trial without a concurrent control group, 29 patients (mean age of 22 years, median age of 24 years, 31% < 18 years) with HoFH received maximum daily doses of 20 to 80 mg of atorvastatin. The mean LDL-C reduction in this trial was 18%. Twenty-five patients with a reduction in LDL-C had a mean response of 20% (range of 7% to 53%, median of 24%); the remaining 4 patients had 7% to 24% increases in LDL-C. Five of the 29 patients had absent LDL-receptor function. Of these, 2 patients also had a portacaval shunt and had no significant reduction in LDL-C. The remaining 3 receptor-negative patients had a mean LDL-C reduction of 22%.
7.3 Drug Interactions That May Increase the Risk of Myopathy and Rhabdomyolysis With Atorvastatin (7.3 Drug Interactions that may Increase the Risk of Myopathy and Rhabdomyolysis with Atorvastatin)
Atorvastatin is a substrate of CYP3A4 and transporters (e.g., OATP1B1/1B3, P-gp, or BCRP). Atorvastatin plasma levels can be significantly increased with concomitant administration of inhibitors of CYP3A4 and transporters. Table 3 includes a list of drugs that may increase exposure to atorvastatin and may increase the risk of myopathy and rhabdomyolysis when used concomitantly and instructions for preventing or managing them [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
|
Cyclosporine or Gemfibrozil |
|
|
Clinical Impact: |
Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin and cyclosporine, an inhibitor of CYP3A4 and OATP1B1 [see Clinical Pharmacology (12.3)]. Gemfibrozil may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of cyclosporine or gemfibrozil with atorvastatin. |
|
Intervention: |
Concomitant use of cyclosporine or gemfibrozil with atorvastatin is not recommended. |
|
Anti-Viral Medications |
|
|
Clinical Impact: |
Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with many anti-viral medications, which are inhibitors of CYP3A4 and/or transporters (e.g., BCRP, OATP1B1/1B3, P-gp, MRP2, and/or OAT2) [see Clinical Pharmacology (12.3)]. Cases of myopathy and rhabdomyolysis have been reported with concomitant use of ledipasvir plus sofosbuvir with atorvastatin. |
|
Intervention: |
|
|
Examples: |
Tipranavir plus ritonavir, glecaprevir plus pibrentasvir, lopinavir plus ritonavir, simeprevir, saquinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, fosamprenavir plus ritonavir, elbasvir plus grazoprevir, letermovir, nelfinavir, and ledipasvir plus sofosbuvir. |
|
Select Azole Antifungals or Macrolide Antibiotics |
|
|
Clinical Impact: |
Atorvastatin plasma levels were significantly increased with concomitant administration of atorvastatin with select azole antifungals or macrolide antibiotics, due to inhibition of CYP3A4 and/or transporters [see Clinical Pharmacology (12.3)]. |
|
Intervention: |
In patients taking clarithromycin or itraconazole, do not exceed atorvastatin 20 mg [see Dosage and Administration (2)]. Consider the risk/benefit of concomitant use of other azole antifungals or macrolide antibiotics with atorvastatin. Monitor all patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Examples: |
Erythromycin, clarithromycin, itraconazole, ketoconazole, posaconazole, and voriconazole. |
|
Niacin |
|
|
Clinical Impact: |
Cases of myopathy and rhabdomyolysis have been observed with concomitant use of lipid modifying dosages of niacin (≥ 1 gram/day niacin) with atorvastatin. |
|
Intervention: |
Consider if the benefit of using lipid modifying dosages of niacin concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Fibrates (other than Gemfibrozil) |
|
|
Clinical Impact: |
Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with atorvastatin. |
|
Intervention: |
Consider if the benefit of using fibrates concomitantly with atorvastatin outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Colchicine |
|
|
Clinical Impact: |
Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with atorvastatin. |
|
Intervention: |
Consider the risk/benefit of concomitant use of colchicine with atorvastatin. If concomitant use is decided, monitor patients for signs and symptoms of myopathy particularly during initiation of therapy and during upward dose titration of either drug. |
|
Grapefruit Juice |
|
|
Clinical Impact: |
Grapefruit juice consumption, especially excessive consumption, more than 1.2 liters/daily can raise the plasma levels of atorvastatin and may increase the risk of myopathy and rhabdomyolysis. |
|
Intervention: |
Avoid intake of large quantities of grapefruit juice, more than 1.2 liters daily, when taking atorvastatin. |
5.7 Increased Risk of Hemorrhagic Stroke in Patients On Atorvastatin 80 Mg With Recent Hemorrhagic Stroke (5.7 Increased Risk of Hemorrhagic Stroke in Patients on Atorvastatin 80 mg with Recent Hemorrhagic Stroke)
In a post-hoc analysis of the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial where 2365 adult patients, without CHD who had a stroke or Transient Ischemic Attack (TIA) within the preceding 6 months, were treated with atorvastatin 80 mg, a higher incidence of hemorrhagic stroke was seen in the atorvastatin 80 mg group compared to placebo (55, 2.3% atorvastatin vs. 33, 1.4% placebo; HR: 1.68, 95% CI: 1.09, 2.59; p=0.0168). The incidence of fatal hemorrhagic stroke was similar across treatment groups (17 vs. 18 for the atorvastatin and placebo groups, respectively). The incidence of non-fatal hemorrhagic stroke was significantly higher in the atorvastatin group (38, 1.6%) as compared to the placebo group (16, 0.7%). Some baseline characteristics, including hemorrhagic and lacunar stroke on study entry, were associated with a higher incidence of hemorrhagic stroke in the atorvastatin group [see Adverse Reactions (6.1)].
Consider the risk/benefit of use of atorvastatin 80 mg in patients with recent hemorrhagic stroke.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:19.175614 · Updated: 2026-03-14T22:48:26.188326