13d67b51-d9ed-4fa6-8c84-5c21cf31675a
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
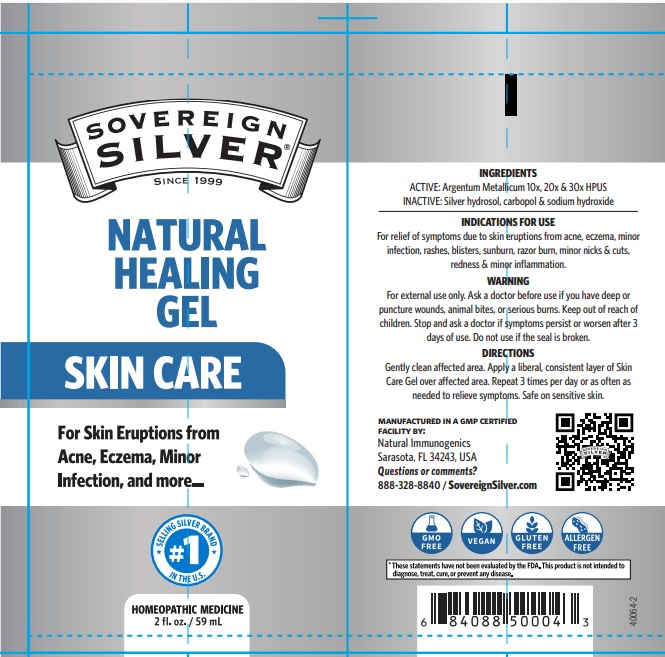
Active Ingredient ARGENTUM METALLICUM 10X, 20X, 30X
Medication Information
Warnings and Precautions
Warnings
FOR EXTERNAL USE ONLY. ASK A DOCTOR BEFORE SE IF YOU HAVE DEEP OR PUNCTURE WOUNDS, ANIMAL BITES OR SERIOUS BURNS. KEEP OUT OF REACH OF CHILDREN. STOP AND ASK A DOCTOR IF SYMPTOMS PERSIST OR WORSEN AFTER 3 DAYS OF USE. DO NOT USE IF THE SEAL IS BROKEN.
Dosage and Administration
Directions
GENTLY CLEAN AFFECTED AREA. APPLY A LIBERAL, CONSISTENT LAYER OF SKIN CARE GEL OVER AFFECTED AREA. REPEAT 3 TIMES PER DAY OR AS OFTEN AS NEEDED TO RELIEVE SYMPTOMS. SAFE ON SENSITIVE SKIN.
Description
Active Ingredient ARGENTUM METALLICUM 10X, 20X, 30X
Uses
FOR RELIEF OF SYMPTOMS DUE TO SKIN ERUPTIONS FROM ACNE, ECZEMA, MINOR INFECTION, RASHES, BLISTERS, SUNBURN, RAZOR BURN, MINOR NICKS & CUTS, REDNESS & MINOR INFLAMMATION
Section 50565-1
Section 55105-1
Purpose
TOPICAL ANALGESIC
Section 55106-9
Active Ingredient
ARGENTUM METALLICUM 10X, 20X, 30X
Product Label
Inactive Ingredients
CARBOPOL, SODIUM HYDROXIDE
Structured Label Content
Uses
FOR RELIEF OF SYMPTOMS DUE TO SKIN ERUPTIONS FROM ACNE, ECZEMA, MINOR INFECTION, RASHES, BLISTERS, SUNBURN, RAZOR BURN, MINOR NICKS & CUTS, REDNESS & MINOR INFLAMMATION
Dosage and Administration (34068-7)
Directions
GENTLY CLEAN AFFECTED AREA. APPLY A LIBERAL, CONSISTENT LAYER OF SKIN CARE GEL OVER AFFECTED AREA. REPEAT 3 TIMES PER DAY OR AS OFTEN AS NEEDED TO RELIEVE SYMPTOMS. SAFE ON SENSITIVE SKIN.
Warnings and Precautions (34071-1)
Warnings
FOR EXTERNAL USE ONLY. ASK A DOCTOR BEFORE SE IF YOU HAVE DEEP OR PUNCTURE WOUNDS, ANIMAL BITES OR SERIOUS BURNS. KEEP OUT OF REACH OF CHILDREN. STOP AND ASK A DOCTOR IF SYMPTOMS PERSIST OR WORSEN AFTER 3 DAYS OF USE. DO NOT USE IF THE SEAL IS BROKEN.
Section 50565-1 (50565-1)
Section 55105-1 (55105-1)
Purpose
TOPICAL ANALGESIC
Section 55106-9 (55106-9)
Active Ingredient
ARGENTUM METALLICUM 10X, 20X, 30X
Product Label (Product label)
Inactive Ingredients
CARBOPOL, SODIUM HYDROXIDE
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:03.334817 · Updated: 2026-03-14T23:17:22.861881