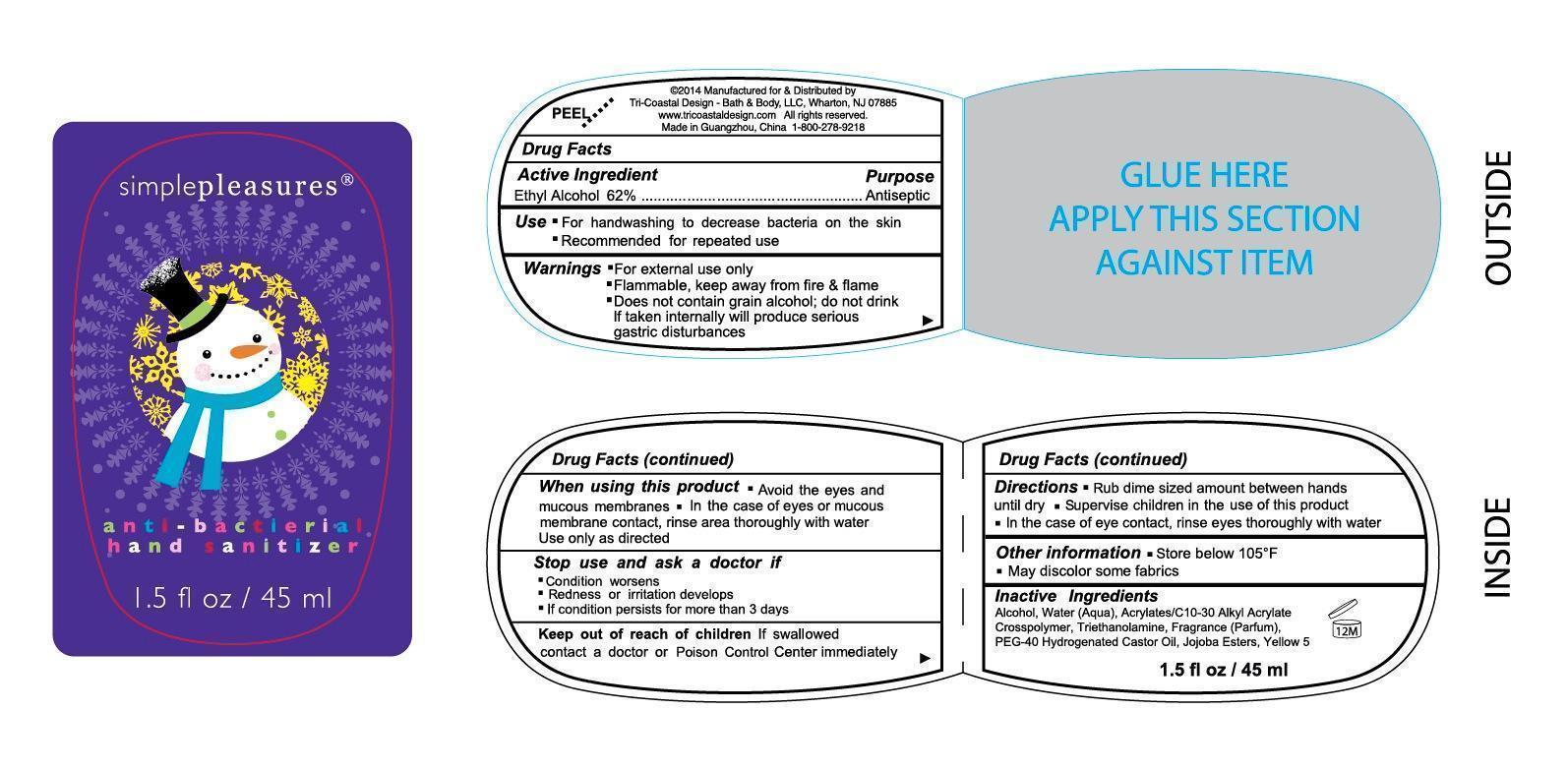
drug facts
1210276c-ec17-4c3c-89b8-8d76c7dbe94d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antiseptic
Description
Ethyl Alcohol 62%
Medication Information
Warnings
- For external use only
- Flammable, keep away from fire and flame
- Does not contain grain alcohol; do not drink, if taken internally will produce serious gastric disturbances
Uses
- For handwashing to decrease bacteria on the skin
- Recommended for repeated use
Purpose
Antiseptic
Directions
- Rub dime sized amount between hands until dry
- Supervise children in the use of this product
- in the case of eye contact, rinse eyes thoroughly with water
Other Information
- Store below 105F
- May discolor some fabrics
Inactive Ingredients
Water (Aqua), Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Triethanolamine, Fragrance (Parfum), PEG-40 Hydrogenated Castor Oil, Jojoba Esters, Yellow 5
Description
Ethyl Alcohol 62%
Package Label
Active Ingredient
Ethyl Alcohol 62%
When Using This Product
- Avoid the eyes and mucous membranes
- In the case of eyes or mucous membrane contact; rinse area thoroughly with water Use only as directed
Stop Use and Ask A Doctor If
- Condition worsens
- Redness or irritation develops
- If condition persists for more than 3 days
Keep Out of Reach of Children
If swallowed contact a doctor or Poison Control Center immediately.
Structured Label Content
Warnings
- For external use only
- Flammable, keep away from fire and flame
- Does not contain grain alcohol; do not drink, if taken internally will produce serious gastric disturbances
Uses
- For handwashing to decrease bacteria on the skin
- Recommended for repeated use
Purpose
Antiseptic
Directions
- Rub dime sized amount between hands until dry
- Supervise children in the use of this product
- in the case of eye contact, rinse eyes thoroughly with water
Other Information (Other information)
- Store below 105F
- May discolor some fabrics
Inactive Ingredients
Water (Aqua), Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Triethanolamine, Fragrance (Parfum), PEG-40 Hydrogenated Castor Oil, Jojoba Esters, Yellow 5
Package Label
Active Ingredient
Ethyl Alcohol 62%
When Using This Product (When using this product)
- Avoid the eyes and mucous membranes
- In the case of eyes or mucous membrane contact; rinse area thoroughly with water Use only as directed
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- Condition worsens
- Redness or irritation develops
- If condition persists for more than 3 days
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed contact a doctor or Poison Control Center immediately.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:27.228325 · Updated: 2026-03-14T22:54:56.160161