These Highlights Do Not Include All The Information Needed To Use Isovue-m Safely And Effectively. See Full Prescribing Information For Isovue-m.
11e893d2-0183-4581-b908-c8b7302c7edb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage, CT cerebral ventriculography ( 1 )-Removed 1/2026
Indications and Usage
ISOVUE-M is indicated for: Lumbar and thoracic myelography, and computed tomography (CT) myelography in adults and pediatric patients aged 2 years and older Cervical and total columnar myelography and CT myelography in adults CT cisternography in adults Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration ( 2.2 , 2.3 )].
Dosage and Administration
ISOVUE-M is for intrathecal use only. Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration ( 2.2 , 2.3 )]. Individualize the volume, concentration, and injection rate of ISOVUE-M according to the dosing tables [see Dosage and Administration ( 2.2 , 2.3 )]. Consider factors such as age, body weight, anticipated pathology and degree and extent of opacification required, structure(s) or area to be examined, concomitant medical conditions, and imaging equipment and technique to be employed. Hydrate patients prior to and following ISOVUE-M administration [see Warnings and Precautions ( 5.2) ] . Use aseptic technique for all handling and administration of ISOVUE-M. ISOVUE-M may be administered at either body temperature (37°C, 98.6°F) or room temperature (20°C to 25°C, 68°F to 77°F). Visually inspect ISOVUE-M for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not administer ISOVUE-M if particulate matter or discoloration are observed. Do not mix ISOVUE-M with other drugs. ISOVUE-M is packaged in a single-dose vial and intended for one procedure only. Discard any unused portion.
Warnings and Precautions
Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency resuscitation equipment and trained personnel available. ( 5.1 ) Acute Kidney Injury: Acute injury including renal failure can occur. Use the lowest dose and maintain adequate hydration to minimize risk. ( 5.2 ) Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after ISOVUE-M administration. ( 5.4 ) Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. ( 5.6 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail in other sections: Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Acute Kidney Injury [see Warnings and Precautions ( 5.2 )] Increased Risk of Seizures [see Warnings and Precautions ( 5.3 )] Cardiovascular Adverse Reactions [see Warnings and Precautions ( 5.4 )] Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions ( 5.6 )] Severe Cutaneous Adverse Reactions [see Warnings and Precautions ( 5.9 )]
Drug Interactions
Metformin In patients with renal impairment, metformin can cause lactic acidosis. Iodinated contrast agents appear to increase the risk of metformin-induced lactic acidosis, possibly as a result of worsening renal function. Stop metformin at the time of, or prior to, ISOVUE-M administration in patients with an eGFR between 30 and 60 mL/min/1.73 m 2 and in patients with a history of hepatic impairment, alcoholism, or heart failure. Re-evaluate eGFR 48 hours after administration of ISOVUE-M and resume metformin only after renal function is stable. Radioactive Iodine Administration of iodinated contrast agents may interfere with thyroid uptake of radioactive iodine (I-131 and I-123) and decrease therapeutic and diagnostic efficacy. Avoid thyroid therapy or testing for up to 6 weeks post ISOVUE-M.
Storage and Handling
How Supplied ISOVUE-M (iopamidol) injection is a clear, colorless to pale yellow solution available in the following presentations: Concentration (mg Iodine/mL) Package Size Package Type Sale Unit NDC 200 10 mL Single-Dose Vial Carton of 10 0270-1411-11 300 15 mL Single-Dose Vial Carton of 10 0270-1412-15 Storage and Handling Store at 20°C to 25°C (68°F to 77°F) [See USP controlled room temperature]. Protect from light.
How Supplied
How Supplied ISOVUE-M (iopamidol) injection is a clear, colorless to pale yellow solution available in the following presentations: Concentration (mg Iodine/mL) Package Size Package Type Sale Unit NDC 200 10 mL Single-Dose Vial Carton of 10 0270-1411-11 300 15 mL Single-Dose Vial Carton of 10 0270-1412-15 Storage and Handling Store at 20°C to 25°C (68°F to 77°F) [See USP controlled room temperature]. Protect from light.
Medication Information
Recent Major Changes
| Indications and Usage, CT cerebral ventriculography (1)-Removed | 1/2026 |
Warnings and Precautions
Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency resuscitation equipment and trained personnel available. ( 5.1 ) Acute Kidney Injury: Acute injury including renal failure can occur. Use the lowest dose and maintain adequate hydration to minimize risk. ( 5.2 ) Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after ISOVUE-M administration. ( 5.4 ) Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. ( 5.6 )
Indications and Usage
ISOVUE-M is indicated for: Lumbar and thoracic myelography, and computed tomography (CT) myelography in adults and pediatric patients aged 2 years and older Cervical and total columnar myelography and CT myelography in adults CT cisternography in adults Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration ( 2.2 , 2.3 )].
Dosage and Administration
ISOVUE-M is for intrathecal use only. Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration ( 2.2 , 2.3 )]. Individualize the volume, concentration, and injection rate of ISOVUE-M according to the dosing tables [see Dosage and Administration ( 2.2 , 2.3 )]. Consider factors such as age, body weight, anticipated pathology and degree and extent of opacification required, structure(s) or area to be examined, concomitant medical conditions, and imaging equipment and technique to be employed. Hydrate patients prior to and following ISOVUE-M administration [see Warnings and Precautions ( 5.2) ] . Use aseptic technique for all handling and administration of ISOVUE-M. ISOVUE-M may be administered at either body temperature (37°C, 98.6°F) or room temperature (20°C to 25°C, 68°F to 77°F). Visually inspect ISOVUE-M for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not administer ISOVUE-M if particulate matter or discoloration are observed. Do not mix ISOVUE-M with other drugs. ISOVUE-M is packaged in a single-dose vial and intended for one procedure only. Discard any unused portion.
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail in other sections: Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Acute Kidney Injury [see Warnings and Precautions ( 5.2 )] Increased Risk of Seizures [see Warnings and Precautions ( 5.3 )] Cardiovascular Adverse Reactions [see Warnings and Precautions ( 5.4 )] Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions ( 5.6 )] Severe Cutaneous Adverse Reactions [see Warnings and Precautions ( 5.9 )]
Drug Interactions
Metformin In patients with renal impairment, metformin can cause lactic acidosis. Iodinated contrast agents appear to increase the risk of metformin-induced lactic acidosis, possibly as a result of worsening renal function. Stop metformin at the time of, or prior to, ISOVUE-M administration in patients with an eGFR between 30 and 60 mL/min/1.73 m 2 and in patients with a history of hepatic impairment, alcoholism, or heart failure. Re-evaluate eGFR 48 hours after administration of ISOVUE-M and resume metformin only after renal function is stable. Radioactive Iodine Administration of iodinated contrast agents may interfere with thyroid uptake of radioactive iodine (I-131 and I-123) and decrease therapeutic and diagnostic efficacy. Avoid thyroid therapy or testing for up to 6 weeks post ISOVUE-M.
Storage and Handling
How Supplied ISOVUE-M (iopamidol) injection is a clear, colorless to pale yellow solution available in the following presentations: Concentration (mg Iodine/mL) Package Size Package Type Sale Unit NDC 200 10 mL Single-Dose Vial Carton of 10 0270-1411-11 300 15 mL Single-Dose Vial Carton of 10 0270-1412-15 Storage and Handling Store at 20°C to 25°C (68°F to 77°F) [See USP controlled room temperature]. Protect from light.
How Supplied
How Supplied ISOVUE-M (iopamidol) injection is a clear, colorless to pale yellow solution available in the following presentations: Concentration (mg Iodine/mL) Package Size Package Type Sale Unit NDC 200 10 mL Single-Dose Vial Carton of 10 0270-1411-11 300 15 mL Single-Dose Vial Carton of 10 0270-1412-15 Storage and Handling Store at 20°C to 25°C (68°F to 77°F) [See USP controlled room temperature]. Protect from light.
Description
Indications and Usage, CT cerebral ventriculography ( 1 )-Removed 1/2026
Section 51945-4
Isovue-M 200:10x 10mL Box label
NDC 0270-1411-11
Isovue-M 200: 10 mL Box internal label
NDC 0270-1411-11
10 Overdosage
Doses above 3,000 mg iodine in adults and 2,400 mg iodine in pediatric patients aged 2 years and older may result in an increased frequency and severity of adverse reactions including seizures.
Treatment of an overdose is directed toward the support of all vital functions and prompt institution of symptomatic therapy. Iopamidol can be removed by dialysis.
8.1 Pregnancy
Risk Summary
Available data from published literature and postmarketing cases from decades of use with iopamidol during pregnancy have not identified a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Iopamidol crosses the placenta and reaches fetal tissues in small amounts (see Data). In animal reproduction studies, no adverse developmental outcomes were observed with intravenous administration of iopamidol to pregnant rats and rabbits during organogenesis at doses up to 2.7 and 1.4 times, respectively, the maximum recommended human dose (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Literature reports show that intravenously administered iopamidol crosses the placenta and is visualized in the digestive tract of exposed infants after birth.
Animal Data
Iopamidol did not affect fetal development and did not induce teratogenic changes in the offspring in either rats or rabbits at the following dose levels tested: 600 mg, 1,500 mg, or 4,000 mg iodine/kg in rats, administered intravenously once a day during days 6 through 15 of pregnancy; 300 mg, 800 mg, or 2,000 mg iodine/kg in rabbits, administered intravenously once a day during days 6 through 18 of pregnancy.
8.2 Lactation
Risk Summary
There are no data on the presence of iopamidol in human milk, the effects on the breastfed infant, or the effects on milk production. Iodinated contrast agents are present unchanged in human milk in very low amounts, with poor absorption from the gastrointestinal tract of a breastfed infant. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ISOVUE- M and any potential adverse effects on the breastfed infant from ISOVUE-M or from the underlying maternal condition.
Clinical Considerations
Interruption of breastfeeding after exposure to iodinated contrast agents is not necessary because the potential exposure of the breastfed infant to iodine is small. However, a lactating woman may consider interrupting breastfeeding and pumping and discarding breast milk for 10 hours (approximately 5 half- lives) after ISOVUE-M administration in order to minimize drug exposure to a breastfed infant.
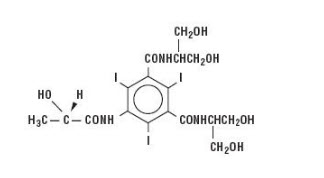
11 Description
ISOVUE-M (iopamidol) injection is a radiographic contrast agent for intrathecal use.
Iopamidol is designated chemically as (S)-N,N’-bis[2-hydroxy-1-(hydroxymethyl)-ethyl]-2,4,6- triiodo-5-lactamidoisophthalamide with a molecular weight of 777.09, an empirical formula of C17H22I3N3O8, and the following structural formula:
ISOVUE-M is a sterile, clear, colorless to pale yellow solution available in two concentrations of iodine:
- ISOVUE-M 200 mg iodine/mL: Each mL contains 408 mg iopamidol (providing 200 mg organically bound iodine) and the following inactive ingredients: 0.26 mg edetate calcium disodium (providing 0.029 mg sodium) and 1 mg tromethamine.
- ISOVUE-M 300 mg iodine/mL: Each mL contains 612 mg iopamidol (providing 300 mg organically bound iodine) and the following inactive ingredients: 0.39 mg edetate calcium disodium (providing 0.043 mg sodium) and 1 mg tromethamine.
The pH of ISOVUE-M has been adjusted to 6.5 to 7.5 with hydrochloric acid and/or sodium hydroxide.
Physicochemical characteristics are shown in Table 4. ISOVUE-M is hypertonic as compared to plasma and cerebrospinal fluid (approximately 285 and 301 mOsm/kg water, respectively).
| Concentration (mg Iodine/mL) | 200 | 300 |
| Osmolality @ 37°C (mOsm/kg water) | 413 | 616 |
| Viscosity (cP) @ 37°C | 2.0 | 4.7 |
| Viscosity (cP) @ 20°C | 3.3 | 8.8 |
| Specific Gravity @ 37°C | 1.227 | 1.339 |
8.4 Pediatric Use
The safety and effectiveness of ISOVUE-M for lumbar and thoracic myelography and CT myelography have been established in pediatric patients aged 2 years and older.
Pediatric patients at higher risk of experiencing adverse reactions during and after any iodinated contrast agent administration may include those having asthma, sensitivity to medication or allergens, cyanotic heart disease, congestive heart failure, or serum creatinine greater than 1.5 mg/dL.
Thyroid function tests indicative of thyroid dysfunction, characterized by hypothyroidism or transient thyroid suppression have been reported following iodinated contrast agent administration in pediatric patients, including term and preterm neonates; Some patients were treated for hypothyroidism. After exposure to iodinated contrast agent, individualize thyroid function monitoring in pediatric patients 0 to 3 years of age based on underlying risk factors, especially in term and preterm neonates [see Warnings and Precautions (5.6) and Adverse Reactions (6.2)].
The safety and effectiveness of ISOVUE-M for lumbar and thoracic myelography and CT myelography have not been established in pediatric patients younger than 2 years of age.
The safety and effectiveness of ISOVUE-M for cervical and total columnar myelography and CT myelography as well as for CT cisternography have not been established in pediatric patients of any age.
8.5 Geriatric Use
Iopamidol is excreted by the kidney, and the risk of adverse reactions to ISOVUE-M may be greater in patients with renal impairment. Because patients 65 years of age and older are more likely to have renal impairment, care should be taken in dose selection, and it may be useful to monitor renal function [see Warnings and Precautions (5.2) and Use in Specific Populations (8.6)].
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are described in greater detail in other sections:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Acute Kidney Injury [see Warnings and Precautions (5.2)]
- Increased Risk of Seizures [see Warnings and Precautions (5.3)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.4)]
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions (5.6)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.9)]
8.6 Renal Impairment
The clearance of iopamidol decreases with increasing degree of renal impairment. In addition, preexisting renal impairment increases the risk for acute kidney injury [see Warnings and Precautions (5.2)].
12.2 Pharmacodynamics
Following intrathecal injection, iopamidol provides diagnostic contrast for at least 30 minutes for conventional myelography. At about 1 hour, contrast will no longer be sufficient for conventional myelography. However, diagnostic contrast for CT myelography remains at least 6 hours after administration. The exposure-response relationships and time course of pharmacodynamic response of iopamidol have not been fully characterized.
12.3 Pharmacokinetics
Absorption
Iopamidol is absorbed into the bloodstream from cerebrospinal fluid (CSF); following intrathecal administration, iopamidol appears in plasma within 1 hour and virtually all of the drug reaches the systemic circulation within 24 hours.
Distribution
Iopamidol did not bind to serum or plasma proteins at 1 hour after administration.
Elimination
The plasma half-life is approximately 2 hours; the half-life is not dose dependent.
Metabolism
Iopamidol does not undergo significant metabolism, deiodination, or biotransformation.
Excretion
Iopamidol is excreted mainly through the kidneys following intrathecal administration, and the drug is essentially undetectable in the plasma 48 hours later.
In patients with normal renal function, the cumulative urinary excretion for iopamidol, expressed as a percentage of administered intravenous dose is approximately 35% to 40% at 60 minutes, 80% to 90% at 8 hours, and 90% or more in the 72- to 96-hour period after administration. In patients with normal renal function, approximately 1% or less of the administered dose appears in cumulative 72- to 96-hour fecal specimens.
1 Indications and Usage
ISOVUE-M is indicated for:
- Lumbar and thoracic myelography, and computed tomography (CT) myelography in adults and pediatric patients aged 2 years and older
- Cervical and total columnar myelography and CT myelography in adults
- CT cisternography in adults
Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3)].
5.2 Acute Kidney Injury
Acute kidney injury, including renal failure, may occur after administration of iodinated contrast agents. Risk factors include pre-existing renal insufficiency, dehydration, diabetes mellitus, congestive heart failure, advanced vascular disease, elderly age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma or other paraproteinemias, and repetitive or large doses of iodinated contrast agents.
Use the lowest dose of ISOVUE-M, especially in patients with risk factors for acute kidney injury. Adequately hydrate patients prior to and following ISOVUE-M administration.
12.1 Mechanism of Action
Intrathecal administration of iopamidol opacifies the body structures where the contrast agent is present, permitting their radiographic visualization through attenuation of photons.
5 Warnings and Precautions
- Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency resuscitation equipment and trained personnel available. (5.1)
- Acute Kidney Injury: Acute injury including renal failure can occur. Use the lowest dose and maintain adequate hydration to minimize risk. (5.2)
- Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after ISOVUE-M administration. (5.4)
- Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. (5.6)
7.1 Drug Drug Interactions
Metformin
In patients with renal impairment, metformin can cause lactic acidosis. Iodinated contrast agents appear to increase the risk of metformin-induced lactic acidosis, possibly as a result of worsening renal function. Stop metformin at the time of, or prior to, ISOVUE-M administration in patients with an eGFR between 30 and 60 mL/min/1.73 m2 and in patients with a history of hepatic impairment, alcoholism, or heart failure. Re-evaluate eGFR 48 hours after administration of ISOVUE-M and resume metformin only after renal function is stable.
Radioactive Iodine
Administration of iodinated contrast agents may interfere with thyroid uptake of radioactive iodine (I-131 and I-123) and decrease therapeutic and diagnostic efficacy. Avoid thyroid therapy or testing for up to 6 weeks post ISOVUE-M.
2 Dosage and Administration
3 Dosage Forms and Strengths
Injection: clear, colorless to pale yellow solution available in two concentrations of iodine:
|
Concentration
(mg of Iodine/mL) |
Package Size | Package Type |
| 200 | 10 mL | Single-Dose Vial |
| 300 | 15 mL | Single-Dose Vial |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ISOVUE-M and other iopamidol products. Because the reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: thrombocytopenia
Cardiovascular disorders: cardiopulmonary arrest, cardiac decompensation, arrhythmias, myocardial infarction, shock, electrocardiographic changes (e.g., increased QTc, increased R-R, increased T-wave amplitude), decreased systolic pressure, deep vein thrombosis, arterial spasms, transient ischemic attack, flushing, vasodilation, chest pain, pallor
Endocrine disorders: hyperthyroidism, hypothyroidism
Eye disorders: lacrimation increased, conjunctivitis, eye pruritus, transient blindness, visual disturbance, photophobia
Gastrointestinal disorders: retching, abdominal pain, salivary hypersecretion, salivary gland enlargement
General disorders and administration site conditions: chills, malaise
Immune system disorders: anaphylaxis characterized by cardiovascular, respiratory, and cutaneous manifestations (e.g., chest tightness, laryngeal edema, periorbital edema, facial edema); delayed hypersensitivity reactions including generalized maculopapular rash, erythema, pruritus, localized blistering, skin peeling
Infections and infestations: meningitis aseptic, meningitis bacterial as consequence of the procedural hazard
Musculoskeletal disorders: muscle spasm, musculoskeletal pain, muscular weakness
Nervous system: coma, seizure, tremors, syncope, cerebral edema, paralysis, depressed level of consciousness or loss of consciousness, meningism, encephalopathy
Psychiatric disorders: disorientation, agitation, restlessness, confusional state
Respiratory system disorders: respiratory arrest, respiratory failure, acute respiratory distress syndrome, respiratory distress, apnea, asthma, sneezing, choking, laryngeal edema, bronchospasm, rhinitis
Skin and subcutaneous tissue disorders: Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP), erythema multiforme and drug reaction with eosinophilia and systemic symptoms (DRESS), skin necrosis, face edema
8 Use in Specific Populations
Lactation: A lactating woman may pump and discard breast milk for 10 hours after ISOVUE-M administration. (8.2)
5.1 Hypersensitivity Reactions
ISOVUE-M can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock [see Adverse Reactions (6.2)]. Most severe reactions develop shortly after the start of injection (e.g., within 1 to 3 minutes), but delayed reactions can also occur. There is increased risk of hypersensitivity reactions in patients with a history of previous reactions to contrast agents, and known allergic disorders (i.e., bronchial asthma, allergic rhinitis, and food allergies) or other hypersensitivities.
Premedication with antihistamines or corticosteroids does not prevent serious life-threatening reactions, but may reduce their incidence and severity. Obtain a history of allergy or hypersensitivity reactions to iodinated contrast agents and always have emergency resuscitation equipment and trained personnel available prior to ISOVUE-M administration. Monitor all patients for hypersensitivity reactions.
5.3 Increased Risk of Seizures
Focal and generalized motor seizures have been reported after intrathecal use of iodinated contrast agents including ISOVUE-M. In several of the cases, higher than recommended doses were administered.
Use of medications that may lower the seizure threshold (phenothiazine derivatives, including those used for their antihistaminic properties; tricyclic antidepressants; MAO inhibitors; CNS stimulants; analeptics; antipsychotic agents) should be carefully evaluated. Consider discontinuing these agents at least 48 hours before and for at least 24 hours following intrathecal administration of ISOVUE-M.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ISOVUE-M was evaluated in 686 adult patients receiving ISOVUE-M intrathecally in clinical studies. Table 3 shows the common adverse reactions (>1%).
These reactions usually occur 1 to 10 hours after injection, almost all occurring within 24 hours.
| Adverse Reaction |
ISOVUE-M
(N=686) % |
| Headache | 16.4 |
| Nausea | 7.3 |
| Vomiting | 3.6 |
| Back pain | 2.2 |
| Leg Pain | 1.4 |
| Neck Pain | 1.1 |
| Hypotension | 1.1 |
The following additional adverse reactions occurred in ≤ 1% of patients receiving intrathecal injection of ISOVUE-M:
Cardiovascular disorder: tachycardia, hypertension, chest pain
Gastrointestinal: diarrhea, heartburn
General disorders and administration site conditions: pyrexia, muscle weakness, hot flashes, malaise, fatigue, weakness, injection site pain
Musculoskeletal: leg cramps, sciatica, cervicobrachial irritation, meningeal irritation, radicular irritation lumbosacral, other musculoskeletal pain, involuntary movement, burning sensation
Nervous system: dizziness, paresthesia, confusion, hallucinations, lightheadedness, syncope, numbness, cold extremities, ataxia, irritability
Urogenital: urinary retention
Respiratory: dyspnea
Skin and subcutaneous tissues: rash
17 Patient Counseling Information
Hypersensitivity Reactions
Advise the patient concerning the risk of hypersensitivity reactions that can occur both during and after ISOVUE-M administration. Advise the patient to report any signs or symptoms of hypersensitivity reactions during the procedure and to seek immediate medical attention for any signs or symptoms experienced after discharge [see Warnings and Precautions (5.1)].
Advise patients to inform their physician if they develop a rash after receiving ISOVUE-M [see Warnings and Precautions (5.9)].
Acute Kidney Injury
Advise the patient concerning appropriate hydration to decrease the risk of contrast induced kidney injury [see Warnings and Precautions (5.2)].
Thyroid Dysfunction
Advise parents/caregivers about the risk of developing thyroid dysfunction after ISOVUE-M administration. Advise parents/caregivers about when to seek medical care for their child to monitor for thyroid function [see Warnings and Precautions (5.6)].
Lactation
Advise lactating women that interruption of breast feeding is not necessary, however, to avoid any exposure a lactating woman may consider pumping and discarding breast milk for 10 hours after ISOVUE-M administration to minimize drug exposure to a breastfed infant [see Use in Specific Populations (8.2)].
Manufactured for:
Bracco Diagnostic Inc.
Princeton, NJ 08540
Manufactured by:
BIPSO GmbH
78224 Singen (Germany)
Revised January 2026
CL63A-07
ISOVUE-M is a registered trademark of Bracco Diagnostics Inc.
16 How Supplied/storage and Handling
How Supplied
ISOVUE-M (iopamidol) injection is a clear, colorless to pale yellow solution available in the following presentations:
|
Concentration
(mg Iodine/mL) |
Package Size | Package Type | Sale Unit | NDC |
| 200 | 10 mL | Single-Dose Vial | Carton of 10 | 0270-1411-11 |
| 300 | 15 mL | Single-Dose Vial | Carton of 10 | 0270-1412-15 |
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F) [See USP controlled room temperature]. Protect from light.
5.4 Cardiovascular Adverse Reactions
Iodinated contrast agents increase the circulatory osmotic load and may induce acute or delayed hemodynamic disturbances in patients with congestive heart failure, severely impaired renal function, combined renal and hepatic disease, and combined renal and cardiac disease, particularly when repetitive or large doses are administered. Fatal cardiovascular reactions have occurred mostly within 10 minutes of injection of iodinated contrast agent by an intravascular route; the main feature was cardiac arrest with cardiovascular disease as the main underlying factor. Hypotensive collapse and shock have occurred.
The administration of iodinated contrast agent may cause pulmonary edema in patients with heart failure. Based upon published reports, deaths associated with the administration of iodinated contrast agents range from 6.6 per 1 million (0.00066 percent) to 1 in 10,000 patients (0.01 percent).
Use the lowest necessary dose of ISOVUE-M in patients with congestive heart failure and always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
7.2 Drug Laboratory Test Interactions
Protein-Bound Iodine Test
Iodinated contrast agents will temporarily increase protein-bound iodine in blood. Avoid protein-bound iodine test for at least 16 days following administration of ISOVUE-M. However, thyroid function tests that do not depend on iodine estimations, e.g., triiodothyronine (T3) resin uptake and total or free thyroxine (T4) assays, are not affected.
5.10 Interference With Laboratory Test
ISOVUE-M can interfere with protein-bound iodine test [see Drug Interactions (7.2)].
5.9 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after administration of iodinated contrast agent. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP), and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agent; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering ISOVUE-M to patients with a history of a severe cutaneous adverse reaction to ISOVUE-M.
5.5 Thyroid Storm in Patients With Hyperthyroidism
Thyroid storm has occurred after the use of iodinated contrast agents in patients with hyperthyroidism, or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of ISOVUE-M.
2.1 Important Dosage and Administration Information
- ISOVUE-M is for intrathecal use only.
- Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3)].
- Individualize the volume, concentration, and injection rate of ISOVUE-M according to the dosing tables [see Dosage and Administration (2.2, 2.3)]. Consider factors such as age, body weight, anticipated pathology and degree and extent of opacification required, structure(s) or area to be examined, concomitant medical conditions, and imaging equipment and technique to be employed.
- Hydrate patients prior to and following ISOVUE-M administration [see Warnings and Precautions (5.2)].
- Use aseptic technique for all handling and administration of ISOVUE-M.
- ISOVUE-M may be administered at either body temperature (37°C, 98.6°F) or room temperature (20°C to 25°C, 68°F to 77°F).
- Visually inspect ISOVUE-M for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not administer ISOVUE-M if particulate matter or discoloration are observed.
- Do not mix ISOVUE-M with other drugs.
- ISOVUE-M is packaged in a single-dose vial and intended for one procedure only. Discard any unused portion.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed with iopamidol to evaluate carcinogenic potential. No evidence of genetic toxicity was obtained in in vitro tests. In animal reproduction studies performed on rats, intravenously administered iopamidol did not induce adverse effects on fertility or general reproductive performance.
5.7 Hypertensive Crisis in Patients With Pheochromocytoma
Hypertensive crisis in patients with pheochromocytoma has occurred with iodinated contrast agents. Closely monitor patients when administering ISOVUE-M if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of ISOVUE-M necessary and have measures for treatment of hypertensive crisis readily available.
2.2 Recommended Dosage for Intrathecal Procedures in Adults
- The recommended doses in adults are shown in Table 1.
- Administer over 1 minute to 2 minutes.
- Allow at least 48 hours before repeat administration to avoid overdose; however, whenever possible, 5 days to 7 days is recommended.
- If CT myelography is performed, delay imaging by at least 4 hours to reduce the degree of contrast.
| Imaging Procedure |
Concentration
(mg Iodine/mL) |
Volume to Administer | Injection Type |
| Lumbar and thoracic myelography | 200 | 10 mL to 15 mL | Lumbar |
| Cervical myelography | 200 | 10 mL to 15 mL | Lumbar |
| 10 mL | Lateral cervical | ||
| 300 | 10 mL | Lumbar | |
| Total columnar myelography | 300 | 10 mL | Lumbar |
| CT cisternography | 200 | 4 mL to 6 mL | Lumbar |
5.8 Sickle Cell Crisis in Patients With Sickle Cell Disease
Iodinated contrast agents may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following ISOVUE-M administration and use only if the necessary imaging information cannot be obtained with alternative imaging modalities.
5.6 Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age
Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast agents in pediatric patients 0 years to 3 years of age.
Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after iodinated contrast agent exposure. Pediatric patients with congenital cardiac conditions may be at greatest risk given that they often require high doses of contrast during invasive cardiac procedures.
An underactive thyroid during early life may be harmful for cognitive and neurological development and may require thyroid hormone replacement therapy. After exposure to iodinated contrast agent, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates. ISOVUE-M is not indicated for use in pediatric patients younger than 2 years of age [see Use in Specific Populations (8.4)].
2.3 Recommended Dosage for Intrathecal Procedures in Pediatric Patients Aged 2 Years and Older
- The recommended doses based on age for pediatric patients aged 2 years and older are shown in Table 2.
- Administer over 1 minute to 2 minutes.
- Allow at least 48 hours before repeat administration to avoid overdose; however, whenever possible, 5 days to 7 days is recommended.
- If CT myelography is performed, delay imaging by at least 4 hours to reduce the degree of contrast.
| Imaging Procedure | Age | Concentration (mg Iodine/mL) | Volume to Administer | Injection Type |
| Lumbar and thoracic myelography | 2 years to 7 years |
200 |
7 mL to 9 mL | Lumbar |
| 8 years to 12 years | 8 mL to 11 mL | Lumbar | ||
| 13 years and older | 10 mL to 12 mL | Lumbar |
Structured Label Content
Section 51945-4 (51945-4)
Isovue-M 200:10x 10mL Box label
NDC 0270-1411-11
Isovue-M 200: 10 mL Box internal label
NDC 0270-1411-11
10 Overdosage (10 OVERDOSAGE)
Doses above 3,000 mg iodine in adults and 2,400 mg iodine in pediatric patients aged 2 years and older may result in an increased frequency and severity of adverse reactions including seizures.
Treatment of an overdose is directed toward the support of all vital functions and prompt institution of symptomatic therapy. Iopamidol can be removed by dialysis.
8.1 Pregnancy
Risk Summary
Available data from published literature and postmarketing cases from decades of use with iopamidol during pregnancy have not identified a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Iopamidol crosses the placenta and reaches fetal tissues in small amounts (see Data). In animal reproduction studies, no adverse developmental outcomes were observed with intravenous administration of iopamidol to pregnant rats and rabbits during organogenesis at doses up to 2.7 and 1.4 times, respectively, the maximum recommended human dose (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Literature reports show that intravenously administered iopamidol crosses the placenta and is visualized in the digestive tract of exposed infants after birth.
Animal Data
Iopamidol did not affect fetal development and did not induce teratogenic changes in the offspring in either rats or rabbits at the following dose levels tested: 600 mg, 1,500 mg, or 4,000 mg iodine/kg in rats, administered intravenously once a day during days 6 through 15 of pregnancy; 300 mg, 800 mg, or 2,000 mg iodine/kg in rabbits, administered intravenously once a day during days 6 through 18 of pregnancy.
8.2 Lactation
Risk Summary
There are no data on the presence of iopamidol in human milk, the effects on the breastfed infant, or the effects on milk production. Iodinated contrast agents are present unchanged in human milk in very low amounts, with poor absorption from the gastrointestinal tract of a breastfed infant. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ISOVUE- M and any potential adverse effects on the breastfed infant from ISOVUE-M or from the underlying maternal condition.
Clinical Considerations
Interruption of breastfeeding after exposure to iodinated contrast agents is not necessary because the potential exposure of the breastfed infant to iodine is small. However, a lactating woman may consider interrupting breastfeeding and pumping and discarding breast milk for 10 hours (approximately 5 half- lives) after ISOVUE-M administration in order to minimize drug exposure to a breastfed infant.
11 Description (11 DESCRIPTION)
ISOVUE-M (iopamidol) injection is a radiographic contrast agent for intrathecal use.
Iopamidol is designated chemically as (S)-N,N’-bis[2-hydroxy-1-(hydroxymethyl)-ethyl]-2,4,6- triiodo-5-lactamidoisophthalamide with a molecular weight of 777.09, an empirical formula of C17H22I3N3O8, and the following structural formula:
ISOVUE-M is a sterile, clear, colorless to pale yellow solution available in two concentrations of iodine:
- ISOVUE-M 200 mg iodine/mL: Each mL contains 408 mg iopamidol (providing 200 mg organically bound iodine) and the following inactive ingredients: 0.26 mg edetate calcium disodium (providing 0.029 mg sodium) and 1 mg tromethamine.
- ISOVUE-M 300 mg iodine/mL: Each mL contains 612 mg iopamidol (providing 300 mg organically bound iodine) and the following inactive ingredients: 0.39 mg edetate calcium disodium (providing 0.043 mg sodium) and 1 mg tromethamine.
The pH of ISOVUE-M has been adjusted to 6.5 to 7.5 with hydrochloric acid and/or sodium hydroxide.
Physicochemical characteristics are shown in Table 4. ISOVUE-M is hypertonic as compared to plasma and cerebrospinal fluid (approximately 285 and 301 mOsm/kg water, respectively).
| Concentration (mg Iodine/mL) | 200 | 300 |
| Osmolality @ 37°C (mOsm/kg water) | 413 | 616 |
| Viscosity (cP) @ 37°C | 2.0 | 4.7 |
| Viscosity (cP) @ 20°C | 3.3 | 8.8 |
| Specific Gravity @ 37°C | 1.227 | 1.339 |
8.4 Pediatric Use
The safety and effectiveness of ISOVUE-M for lumbar and thoracic myelography and CT myelography have been established in pediatric patients aged 2 years and older.
Pediatric patients at higher risk of experiencing adverse reactions during and after any iodinated contrast agent administration may include those having asthma, sensitivity to medication or allergens, cyanotic heart disease, congestive heart failure, or serum creatinine greater than 1.5 mg/dL.
Thyroid function tests indicative of thyroid dysfunction, characterized by hypothyroidism or transient thyroid suppression have been reported following iodinated contrast agent administration in pediatric patients, including term and preterm neonates; Some patients were treated for hypothyroidism. After exposure to iodinated contrast agent, individualize thyroid function monitoring in pediatric patients 0 to 3 years of age based on underlying risk factors, especially in term and preterm neonates [see Warnings and Precautions (5.6) and Adverse Reactions (6.2)].
The safety and effectiveness of ISOVUE-M for lumbar and thoracic myelography and CT myelography have not been established in pediatric patients younger than 2 years of age.
The safety and effectiveness of ISOVUE-M for cervical and total columnar myelography and CT myelography as well as for CT cisternography have not been established in pediatric patients of any age.
8.5 Geriatric Use
Iopamidol is excreted by the kidney, and the risk of adverse reactions to ISOVUE-M may be greater in patients with renal impairment. Because patients 65 years of age and older are more likely to have renal impairment, care should be taken in dose selection, and it may be useful to monitor renal function [see Warnings and Precautions (5.2) and Use in Specific Populations (8.6)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in greater detail in other sections:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Acute Kidney Injury [see Warnings and Precautions (5.2)]
- Increased Risk of Seizures [see Warnings and Precautions (5.3)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.4)]
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions (5.6)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.9)]
8.6 Renal Impairment
The clearance of iopamidol decreases with increasing degree of renal impairment. In addition, preexisting renal impairment increases the risk for acute kidney injury [see Warnings and Precautions (5.2)].
Recent Major Changes
| Indications and Usage, CT cerebral ventriculography (1)-Removed | 1/2026 |
12.2 Pharmacodynamics
Following intrathecal injection, iopamidol provides diagnostic contrast for at least 30 minutes for conventional myelography. At about 1 hour, contrast will no longer be sufficient for conventional myelography. However, diagnostic contrast for CT myelography remains at least 6 hours after administration. The exposure-response relationships and time course of pharmacodynamic response of iopamidol have not been fully characterized.
12.3 Pharmacokinetics
Absorption
Iopamidol is absorbed into the bloodstream from cerebrospinal fluid (CSF); following intrathecal administration, iopamidol appears in plasma within 1 hour and virtually all of the drug reaches the systemic circulation within 24 hours.
Distribution
Iopamidol did not bind to serum or plasma proteins at 1 hour after administration.
Elimination
The plasma half-life is approximately 2 hours; the half-life is not dose dependent.
Metabolism
Iopamidol does not undergo significant metabolism, deiodination, or biotransformation.
Excretion
Iopamidol is excreted mainly through the kidneys following intrathecal administration, and the drug is essentially undetectable in the plasma 48 hours later.
In patients with normal renal function, the cumulative urinary excretion for iopamidol, expressed as a percentage of administered intravenous dose is approximately 35% to 40% at 60 minutes, 80% to 90% at 8 hours, and 90% or more in the 72- to 96-hour period after administration. In patients with normal renal function, approximately 1% or less of the administered dose appears in cumulative 72- to 96-hour fecal specimens.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ISOVUE-M is indicated for:
- Lumbar and thoracic myelography, and computed tomography (CT) myelography in adults and pediatric patients aged 2 years and older
- Cervical and total columnar myelography and CT myelography in adults
- CT cisternography in adults
Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3)].
5.2 Acute Kidney Injury
Acute kidney injury, including renal failure, may occur after administration of iodinated contrast agents. Risk factors include pre-existing renal insufficiency, dehydration, diabetes mellitus, congestive heart failure, advanced vascular disease, elderly age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma or other paraproteinemias, and repetitive or large doses of iodinated contrast agents.
Use the lowest dose of ISOVUE-M, especially in patients with risk factors for acute kidney injury. Adequately hydrate patients prior to and following ISOVUE-M administration.
12.1 Mechanism of Action
Intrathecal administration of iopamidol opacifies the body structures where the contrast agent is present, permitting their radiographic visualization through attenuation of photons.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency resuscitation equipment and trained personnel available. (5.1)
- Acute Kidney Injury: Acute injury including renal failure can occur. Use the lowest dose and maintain adequate hydration to minimize risk. (5.2)
- Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after ISOVUE-M administration. (5.4)
- Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. (5.6)
7.1 Drug Drug Interactions (7.1 Drug-Drug Interactions)
Metformin
In patients with renal impairment, metformin can cause lactic acidosis. Iodinated contrast agents appear to increase the risk of metformin-induced lactic acidosis, possibly as a result of worsening renal function. Stop metformin at the time of, or prior to, ISOVUE-M administration in patients with an eGFR between 30 and 60 mL/min/1.73 m2 and in patients with a history of hepatic impairment, alcoholism, or heart failure. Re-evaluate eGFR 48 hours after administration of ISOVUE-M and resume metformin only after renal function is stable.
Radioactive Iodine
Administration of iodinated contrast agents may interfere with thyroid uptake of radioactive iodine (I-131 and I-123) and decrease therapeutic and diagnostic efficacy. Avoid thyroid therapy or testing for up to 6 weeks post ISOVUE-M.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: clear, colorless to pale yellow solution available in two concentrations of iodine:
|
Concentration
(mg of Iodine/mL) |
Package Size | Package Type |
| 200 | 10 mL | Single-Dose Vial |
| 300 | 15 mL | Single-Dose Vial |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ISOVUE-M and other iopamidol products. Because the reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: thrombocytopenia
Cardiovascular disorders: cardiopulmonary arrest, cardiac decompensation, arrhythmias, myocardial infarction, shock, electrocardiographic changes (e.g., increased QTc, increased R-R, increased T-wave amplitude), decreased systolic pressure, deep vein thrombosis, arterial spasms, transient ischemic attack, flushing, vasodilation, chest pain, pallor
Endocrine disorders: hyperthyroidism, hypothyroidism
Eye disorders: lacrimation increased, conjunctivitis, eye pruritus, transient blindness, visual disturbance, photophobia
Gastrointestinal disorders: retching, abdominal pain, salivary hypersecretion, salivary gland enlargement
General disorders and administration site conditions: chills, malaise
Immune system disorders: anaphylaxis characterized by cardiovascular, respiratory, and cutaneous manifestations (e.g., chest tightness, laryngeal edema, periorbital edema, facial edema); delayed hypersensitivity reactions including generalized maculopapular rash, erythema, pruritus, localized blistering, skin peeling
Infections and infestations: meningitis aseptic, meningitis bacterial as consequence of the procedural hazard
Musculoskeletal disorders: muscle spasm, musculoskeletal pain, muscular weakness
Nervous system: coma, seizure, tremors, syncope, cerebral edema, paralysis, depressed level of consciousness or loss of consciousness, meningism, encephalopathy
Psychiatric disorders: disorientation, agitation, restlessness, confusional state
Respiratory system disorders: respiratory arrest, respiratory failure, acute respiratory distress syndrome, respiratory distress, apnea, asthma, sneezing, choking, laryngeal edema, bronchospasm, rhinitis
Skin and subcutaneous tissue disorders: Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP), erythema multiforme and drug reaction with eosinophilia and systemic symptoms (DRESS), skin necrosis, face edema
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: A lactating woman may pump and discard breast milk for 10 hours after ISOVUE-M administration. (8.2)
5.1 Hypersensitivity Reactions
ISOVUE-M can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock [see Adverse Reactions (6.2)]. Most severe reactions develop shortly after the start of injection (e.g., within 1 to 3 minutes), but delayed reactions can also occur. There is increased risk of hypersensitivity reactions in patients with a history of previous reactions to contrast agents, and known allergic disorders (i.e., bronchial asthma, allergic rhinitis, and food allergies) or other hypersensitivities.
Premedication with antihistamines or corticosteroids does not prevent serious life-threatening reactions, but may reduce their incidence and severity. Obtain a history of allergy or hypersensitivity reactions to iodinated contrast agents and always have emergency resuscitation equipment and trained personnel available prior to ISOVUE-M administration. Monitor all patients for hypersensitivity reactions.
5.3 Increased Risk of Seizures
Focal and generalized motor seizures have been reported after intrathecal use of iodinated contrast agents including ISOVUE-M. In several of the cases, higher than recommended doses were administered.
Use of medications that may lower the seizure threshold (phenothiazine derivatives, including those used for their antihistaminic properties; tricyclic antidepressants; MAO inhibitors; CNS stimulants; analeptics; antipsychotic agents) should be carefully evaluated. Consider discontinuing these agents at least 48 hours before and for at least 24 hours following intrathecal administration of ISOVUE-M.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ISOVUE-M was evaluated in 686 adult patients receiving ISOVUE-M intrathecally in clinical studies. Table 3 shows the common adverse reactions (>1%).
These reactions usually occur 1 to 10 hours after injection, almost all occurring within 24 hours.
| Adverse Reaction |
ISOVUE-M
(N=686) % |
| Headache | 16.4 |
| Nausea | 7.3 |
| Vomiting | 3.6 |
| Back pain | 2.2 |
| Leg Pain | 1.4 |
| Neck Pain | 1.1 |
| Hypotension | 1.1 |
The following additional adverse reactions occurred in ≤ 1% of patients receiving intrathecal injection of ISOVUE-M:
Cardiovascular disorder: tachycardia, hypertension, chest pain
Gastrointestinal: diarrhea, heartburn
General disorders and administration site conditions: pyrexia, muscle weakness, hot flashes, malaise, fatigue, weakness, injection site pain
Musculoskeletal: leg cramps, sciatica, cervicobrachial irritation, meningeal irritation, radicular irritation lumbosacral, other musculoskeletal pain, involuntary movement, burning sensation
Nervous system: dizziness, paresthesia, confusion, hallucinations, lightheadedness, syncope, numbness, cold extremities, ataxia, irritability
Urogenital: urinary retention
Respiratory: dyspnea
Skin and subcutaneous tissues: rash
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Hypersensitivity Reactions
Advise the patient concerning the risk of hypersensitivity reactions that can occur both during and after ISOVUE-M administration. Advise the patient to report any signs or symptoms of hypersensitivity reactions during the procedure and to seek immediate medical attention for any signs or symptoms experienced after discharge [see Warnings and Precautions (5.1)].
Advise patients to inform their physician if they develop a rash after receiving ISOVUE-M [see Warnings and Precautions (5.9)].
Acute Kidney Injury
Advise the patient concerning appropriate hydration to decrease the risk of contrast induced kidney injury [see Warnings and Precautions (5.2)].
Thyroid Dysfunction
Advise parents/caregivers about the risk of developing thyroid dysfunction after ISOVUE-M administration. Advise parents/caregivers about when to seek medical care for their child to monitor for thyroid function [see Warnings and Precautions (5.6)].
Lactation
Advise lactating women that interruption of breast feeding is not necessary, however, to avoid any exposure a lactating woman may consider pumping and discarding breast milk for 10 hours after ISOVUE-M administration to minimize drug exposure to a breastfed infant [see Use in Specific Populations (8.2)].
Manufactured for:
Bracco Diagnostic Inc.
Princeton, NJ 08540
Manufactured by:
BIPSO GmbH
78224 Singen (Germany)
Revised January 2026
CL63A-07
ISOVUE-M is a registered trademark of Bracco Diagnostics Inc.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
ISOVUE-M (iopamidol) injection is a clear, colorless to pale yellow solution available in the following presentations:
|
Concentration
(mg Iodine/mL) |
Package Size | Package Type | Sale Unit | NDC |
| 200 | 10 mL | Single-Dose Vial | Carton of 10 | 0270-1411-11 |
| 300 | 15 mL | Single-Dose Vial | Carton of 10 | 0270-1412-15 |
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F) [See USP controlled room temperature]. Protect from light.
5.4 Cardiovascular Adverse Reactions
Iodinated contrast agents increase the circulatory osmotic load and may induce acute or delayed hemodynamic disturbances in patients with congestive heart failure, severely impaired renal function, combined renal and hepatic disease, and combined renal and cardiac disease, particularly when repetitive or large doses are administered. Fatal cardiovascular reactions have occurred mostly within 10 minutes of injection of iodinated contrast agent by an intravascular route; the main feature was cardiac arrest with cardiovascular disease as the main underlying factor. Hypotensive collapse and shock have occurred.
The administration of iodinated contrast agent may cause pulmonary edema in patients with heart failure. Based upon published reports, deaths associated with the administration of iodinated contrast agents range from 6.6 per 1 million (0.00066 percent) to 1 in 10,000 patients (0.01 percent).
Use the lowest necessary dose of ISOVUE-M in patients with congestive heart failure and always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
7.2 Drug Laboratory Test Interactions (7.2 Drug-Laboratory Test Interactions)
Protein-Bound Iodine Test
Iodinated contrast agents will temporarily increase protein-bound iodine in blood. Avoid protein-bound iodine test for at least 16 days following administration of ISOVUE-M. However, thyroid function tests that do not depend on iodine estimations, e.g., triiodothyronine (T3) resin uptake and total or free thyroxine (T4) assays, are not affected.
5.10 Interference With Laboratory Test (5.10 Interference with Laboratory Test)
ISOVUE-M can interfere with protein-bound iodine test [see Drug Interactions (7.2)].
5.9 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after administration of iodinated contrast agent. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP), and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agent; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering ISOVUE-M to patients with a history of a severe cutaneous adverse reaction to ISOVUE-M.
5.5 Thyroid Storm in Patients With Hyperthyroidism (5.5 Thyroid Storm in Patients with Hyperthyroidism)
Thyroid storm has occurred after the use of iodinated contrast agents in patients with hyperthyroidism, or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of ISOVUE-M.
2.1 Important Dosage and Administration Information
- ISOVUE-M is for intrathecal use only.
- Specific concentrations of ISOVUE-M are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3)].
- Individualize the volume, concentration, and injection rate of ISOVUE-M according to the dosing tables [see Dosage and Administration (2.2, 2.3)]. Consider factors such as age, body weight, anticipated pathology and degree and extent of opacification required, structure(s) or area to be examined, concomitant medical conditions, and imaging equipment and technique to be employed.
- Hydrate patients prior to and following ISOVUE-M administration [see Warnings and Precautions (5.2)].
- Use aseptic technique for all handling and administration of ISOVUE-M.
- ISOVUE-M may be administered at either body temperature (37°C, 98.6°F) or room temperature (20°C to 25°C, 68°F to 77°F).
- Visually inspect ISOVUE-M for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not administer ISOVUE-M if particulate matter or discoloration are observed.
- Do not mix ISOVUE-M with other drugs.
- ISOVUE-M is packaged in a single-dose vial and intended for one procedure only. Discard any unused portion.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed with iopamidol to evaluate carcinogenic potential. No evidence of genetic toxicity was obtained in in vitro tests. In animal reproduction studies performed on rats, intravenously administered iopamidol did not induce adverse effects on fertility or general reproductive performance.
5.7 Hypertensive Crisis in Patients With Pheochromocytoma (5.7 Hypertensive Crisis in Patients with Pheochromocytoma)
Hypertensive crisis in patients with pheochromocytoma has occurred with iodinated contrast agents. Closely monitor patients when administering ISOVUE-M if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of ISOVUE-M necessary and have measures for treatment of hypertensive crisis readily available.
2.2 Recommended Dosage for Intrathecal Procedures in Adults
- The recommended doses in adults are shown in Table 1.
- Administer over 1 minute to 2 minutes.
- Allow at least 48 hours before repeat administration to avoid overdose; however, whenever possible, 5 days to 7 days is recommended.
- If CT myelography is performed, delay imaging by at least 4 hours to reduce the degree of contrast.
| Imaging Procedure |
Concentration
(mg Iodine/mL) |
Volume to Administer | Injection Type |
| Lumbar and thoracic myelography | 200 | 10 mL to 15 mL | Lumbar |
| Cervical myelography | 200 | 10 mL to 15 mL | Lumbar |
| 10 mL | Lateral cervical | ||
| 300 | 10 mL | Lumbar | |
| Total columnar myelography | 300 | 10 mL | Lumbar |
| CT cisternography | 200 | 4 mL to 6 mL | Lumbar |
5.8 Sickle Cell Crisis in Patients With Sickle Cell Disease (5.8 Sickle Cell Crisis in Patients with Sickle Cell Disease)
Iodinated contrast agents may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following ISOVUE-M administration and use only if the necessary imaging information cannot be obtained with alternative imaging modalities.
5.6 Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age
Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast agents in pediatric patients 0 years to 3 years of age.
Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after iodinated contrast agent exposure. Pediatric patients with congenital cardiac conditions may be at greatest risk given that they often require high doses of contrast during invasive cardiac procedures.
An underactive thyroid during early life may be harmful for cognitive and neurological development and may require thyroid hormone replacement therapy. After exposure to iodinated contrast agent, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates. ISOVUE-M is not indicated for use in pediatric patients younger than 2 years of age [see Use in Specific Populations (8.4)].
2.3 Recommended Dosage for Intrathecal Procedures in Pediatric Patients Aged 2 Years and Older
- The recommended doses based on age for pediatric patients aged 2 years and older are shown in Table 2.
- Administer over 1 minute to 2 minutes.
- Allow at least 48 hours before repeat administration to avoid overdose; however, whenever possible, 5 days to 7 days is recommended.
- If CT myelography is performed, delay imaging by at least 4 hours to reduce the degree of contrast.
| Imaging Procedure | Age | Concentration (mg Iodine/mL) | Volume to Administer | Injection Type |
| Lumbar and thoracic myelography | 2 years to 7 years |
200 |
7 mL to 9 mL | Lumbar |
| 8 years to 12 years | 8 mL to 11 mL | Lumbar | ||
| 13 years and older | 10 mL to 12 mL | Lumbar |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T02:18:22.680654 · Updated: 2026-03-14T22:49:49.558846