Omega-3-acid Ethyl Esters Capsules. These Highlights Do Not Include All The Information Needed To Use Omega-3-acid Ethyl Esters Capsules Safely And Effectively. See Full Prescribing Information For Omega-3-acid Ethyl Esters Capsules.
11d744aa-cc06-1206-e063-6294a90a49c9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Omega-3-acid ethyl esters capsules are indicated as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (greater than or equal to 500 mg per dL) hypertriglyceridemia (HTG).
Indications and Usage
Omega-3-acid ethyl esters capsules are indicated as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (greater than or equal to 500 mg per dL) hypertriglyceridemia (HTG).
Dosage and Administration
Assess triglyceride levels carefully before initiating therapy. Identify other causes (e.g., diabetes mellitus, hypothyroidism, medications) of high triglyceride levels and manage as appropriate [see Indications and Usage (1) ]. Patients should be placed on an appropriate lipid-lowering diet before receiving omega-3-acid ethyl esters, and should continue this diet during treatment with omega-3-acid ethyl esters. In clinical studies, omega-3-acid ethyl esters was administered with meals. The daily dose of omega-3-acid ethyl esters is 4 grams per day. The daily dose may be taken as a single 4-gram dose (4 capsules) or as two 2-gram doses (2 capsules given twice daily). Patients should be advised to swallow omega-3-acid ethyl esters whole. Do not break open, crush, dissolve, or chew omega-3-acid ethyl esters.
Warnings and Precautions
In patients with hepatic impairment, monitor ALT and AST levels periodically during therapy. ( 5.1 ) Omega-3-acid ethyl esters may increase levels of LDL. Monitor LDL levels periodically during therapy. ( 5.1 ) Use with caution in patients with known hypersensitivity to fish and/or shellfish. ( 5.2 ) There is a possible association between omega-3-acid ethyl esters and more frequent recurrences of symptomatic atrial fibrillation or flutter in patients with paroxysmal or persistent atrial fibrillation, particularly within the first months of initiating therapy. ( 5.3 )
Contraindications
Omega-3-acid ethyl esters are contraindicated in patients with known hypersensitivity (e.g., anaphylactic reaction) to omega-3-acid ethyl esters or any of its components.
Adverse Reactions
The most common adverse reactions (incidence >3% and greater than placebo) were eructation, dyspepsia, and taste perversion. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact KD Pharma USA at 1-877-241-7978 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
Omega-3-acids may prolong bleeding time. Patients taking omega-3-acid ethyl esters and an anticoagulant or other drug affecting coagulation (e.g., anti-platelet agents) should be monitored periodically. ( 7.1 )
Storage and Handling
Omega-3-Acid Ethyl Esters Capsules USP are available as follows: 1 gram – slightly yellow to yellow liquid, oblong, clear, soft gel capsules, imprinted with "GLW" in white ink across one side. Bottles of 120: NDC 51407-763-12
How Supplied
Omega-3-Acid Ethyl Esters Capsules USP are available as follows: 1 gram – slightly yellow to yellow liquid, oblong, clear, soft gel capsules, imprinted with "GLW" in white ink across one side. Bottles of 120: NDC 51407-763-12
Patient Information
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP (oh may' ga as' id eth' il es' ters). What are OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP? OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP are a prescription medicine used along with a low-fat and low-cholesterol diet to lower very high triglyceride (fat) levels in adults. It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP change your risk of having inflammation of your pancreas (pancreatitis). It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP prevent you from having a heart attack or stroke. It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP are safe and effective in children. Who should not take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP? Do not take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP if you are allergic to omega-3-acid ethyl esters or any of the ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. See the end of this leaflet for a complete list of ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Before taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, tell your healthcare provider about all of your medical conditions, including if you: have diabetes. have a low thyroid problem (hypothyroidism). have a liver problem. have a pancreas problem. have a certain heart rhythm problem called atrial fibrillation or flutter. are allergic to fish or shellfish. It is not known if people who are allergic to fish or shellfish are also allergic to OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. are pregnant or plan to become pregnant. It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP will harm your unborn baby. are breastfeeding or plan to breastfeed. OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP can pass into your breast milk. Talk to your healthcare provider about the best way to feed your baby if you take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP can interact with certain other medicines that you are taking. Using OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP with medicines that affect blood clotting (anticoagulants or blood thinners) may cause serious side effects. How should I take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP? Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP exactly as your healthcare provider tells you to take them. You should not take more than 4 capsules of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP each day. Either take all 4 capsules at one time or 2 capsules two times a day. Do not change your dose or stop OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP without talking to your healthcare provider. Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP with food. Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP whole. Do not break, open, crush, dissolve, or chew OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP before swallowing. If you cannot swallow OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP whole, tell your healthcare provider. You may need a different medicine. If you miss a dose of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, take the missed dose as soon as you remember. If you miss one day of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, do not double your dose the next time you take it. Your healthcare provider may start you on a cholesterol-lowering diet before giving you OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Stay on this diet while taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Your healthcare provider should do blood tests to check your triglyceride, bad cholesterol (LDL-C) and liver function (ALT and AST) levels while you take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. What are the possible side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP? OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause serious side effects, including: changes in certain blood tests. OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause an increase in the results of blood tests used to check your liver function and your bad cholesterol levels. increased risk of a heart rhythm problem in people who have a heart rhythm problem. OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause an increase in the frequency of a heart rhythm problem (atrial fibrillation or flutter), especially if the first few months of taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, if you already have a heart rhythm problem. The most common side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP include: burping upset stomach a change in your sense of taste These are not all the possible side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP? Store OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP at room temperature between 68°F to 77°F (20°C to 25°C). Do not freeze OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Safely throw away medicine that is out of date or no longer needed. Keep OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP and all medicines out of the reach of children. General information about the safe and effective use of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP for a condition for which it was not prescribed. Do not give OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP that is written for health professionals. What are the ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP? Active Ingredient: omega-3-acid ethyl esters, mostly EPA and DHA Inactive Ingredients: 4 mg alpha-tocopherol, gelatin, glycerin, hypromellose, propylene glycol, and titanium dioxide. Manufactured By: Patheon Softgels Inc. High Point, NC 27265 USA Manufactured For: KD Pharma USA Inc. 12021 SW 144th Street Miami, Florida 33186 USA This patient labeling has been approved by the U.S. Food and Drug Administration. Revised: November 2022 Revision: 1 Marketed by: GSMS, Inc. Camarillo, CA 93012 USA
Medication Information
Warnings and Precautions
In patients with hepatic impairment, monitor ALT and AST levels periodically during therapy. ( 5.1 ) Omega-3-acid ethyl esters may increase levels of LDL. Monitor LDL levels periodically during therapy. ( 5.1 ) Use with caution in patients with known hypersensitivity to fish and/or shellfish. ( 5.2 ) There is a possible association between omega-3-acid ethyl esters and more frequent recurrences of symptomatic atrial fibrillation or flutter in patients with paroxysmal or persistent atrial fibrillation, particularly within the first months of initiating therapy. ( 5.3 )
Indications and Usage
Omega-3-acid ethyl esters capsules are indicated as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (greater than or equal to 500 mg per dL) hypertriglyceridemia (HTG).
Dosage and Administration
Assess triglyceride levels carefully before initiating therapy. Identify other causes (e.g., diabetes mellitus, hypothyroidism, medications) of high triglyceride levels and manage as appropriate [see Indications and Usage (1) ]. Patients should be placed on an appropriate lipid-lowering diet before receiving omega-3-acid ethyl esters, and should continue this diet during treatment with omega-3-acid ethyl esters. In clinical studies, omega-3-acid ethyl esters was administered with meals. The daily dose of omega-3-acid ethyl esters is 4 grams per day. The daily dose may be taken as a single 4-gram dose (4 capsules) or as two 2-gram doses (2 capsules given twice daily). Patients should be advised to swallow omega-3-acid ethyl esters whole. Do not break open, crush, dissolve, or chew omega-3-acid ethyl esters.
Contraindications
Omega-3-acid ethyl esters are contraindicated in patients with known hypersensitivity (e.g., anaphylactic reaction) to omega-3-acid ethyl esters or any of its components.
Adverse Reactions
The most common adverse reactions (incidence >3% and greater than placebo) were eructation, dyspepsia, and taste perversion. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact KD Pharma USA at 1-877-241-7978 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
Omega-3-acids may prolong bleeding time. Patients taking omega-3-acid ethyl esters and an anticoagulant or other drug affecting coagulation (e.g., anti-platelet agents) should be monitored periodically. ( 7.1 )
Storage and Handling
Omega-3-Acid Ethyl Esters Capsules USP are available as follows: 1 gram – slightly yellow to yellow liquid, oblong, clear, soft gel capsules, imprinted with "GLW" in white ink across one side. Bottles of 120: NDC 51407-763-12
How Supplied
Omega-3-Acid Ethyl Esters Capsules USP are available as follows: 1 gram – slightly yellow to yellow liquid, oblong, clear, soft gel capsules, imprinted with "GLW" in white ink across one side. Bottles of 120: NDC 51407-763-12
Patient Information
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP
(oh may' ga as' id eth' il es' ters).
What are OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP are a prescription medicine used along with a low-fat and low-cholesterol diet to lower very high triglyceride (fat) levels in adults.
It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP change your risk of having inflammation of your pancreas (pancreatitis).
It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP prevent you from having a heart attack or stroke.
It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP are safe and effective in children.
Who should not take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
Do not take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP if you are allergic to omega-3-acid ethyl esters or any of the ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. See the end of this leaflet for a complete list of ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
Before taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, tell your healthcare provider about all of your medical conditions, including if you:
- have diabetes.
- have a low thyroid problem (hypothyroidism).
- have a liver problem.
- have a pancreas problem.
- have a certain heart rhythm problem called atrial fibrillation or flutter.
- are allergic to fish or shellfish. It is not known if people who are allergic to fish or shellfish are also allergic to OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
- are pregnant or plan to become pregnant. It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP will harm your unborn baby.
- are breastfeeding or plan to breastfeed. OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP can pass into your breast milk. Talk to your healthcare provider about the best way to feed your baby if you take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements.
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP can interact with certain other medicines that you are taking. Using OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP with medicines that affect blood clotting (anticoagulants or blood thinners) may cause serious side effects.
How should I take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
- Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP exactly as your healthcare provider tells you to take them.
- You should not take more than 4 capsules of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP each day. Either take all 4 capsules at one time or 2 capsules two times a day.
- Do not change your dose or stop OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP without talking to your healthcare provider.
- Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP with food.
- Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP whole. Do not break, open, crush, dissolve, or chew OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP before swallowing. If you cannot swallow OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP whole, tell your healthcare provider. You may need a different medicine.
- If you miss a dose of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, take the missed dose as soon as you remember. If you miss one day of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, do not double your dose the next time you take it.
- Your healthcare provider may start you on a cholesterol-lowering diet before giving you OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Stay on this diet while taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
- Your healthcare provider should do blood tests to check your triglyceride, bad cholesterol (LDL-C) and liver function (ALT and AST) levels while you take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
What are the possible side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause serious side effects, including:
- changes in certain blood tests.OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause an increase in the results of blood tests used to check your liver function and your bad cholesterol levels.
- increased risk of a heart rhythm problem in people who have a heart rhythm problem.OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause an increase in the frequency of a heart rhythm problem (atrial fibrillation or flutter), especially if the first few months of taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, if you already have a heart rhythm problem.
The most common side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP include:
- burping
- upset stomach
- a change in your sense of taste
These are not all the possible side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
- Store OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not freeze OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
- Safely throw away medicine that is out of date or no longer needed.
Keep OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP and all medicines out of the reach of children.
General information about the safe and effective use of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP for a condition for which it was not prescribed. Do not give OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP to other people, even if they have the same symptoms you have. It may harm them.
You can ask your healthcare provider or pharmacist for information about OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP that is written for health professionals.
What are the ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
Active Ingredient: omega-3-acid ethyl esters, mostly EPA and DHA
Inactive Ingredients: 4 mg alpha-tocopherol, gelatin, glycerin, hypromellose, propylene glycol, and titanium dioxide.
Manufactured By:
Patheon Softgels Inc.
High Point, NC 27265
USA
Manufactured For:
KD Pharma USA Inc.
12021 SW 144th Street
Miami, Florida 33186
USA
This patient labeling has been approved by the U.S. Food and Drug Administration.
Revised: November 2022
Revision: 1
Marketed by:
GSMS, Inc.
Camarillo, CA 93012 USA
Description
Omega-3-acid ethyl esters capsules are indicated as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (greater than or equal to 500 mg per dL) hypertriglyceridemia (HTG).
Section 42229-5
Usage Considerations:Patients should be placed on an appropriate lipid-lowering diet before receiving omega-3-acid ethyl esters and should continue this diet during treatment with omega-3-acid ethyl esters.
Laboratory studies should be done to ascertain that the lipid levels are consistently abnormal before instituting therapy with omega-3-acid ethyl esters. Every attempt should be made to control serum lipids with appropriate diet, exercise, weight loss in obese patients, and control of any medical problems such as diabetes mellitus and hypothyroidism that are contributing to the lipid abnormalities. Medications known to exacerbate hypertriglyceridemia (such as beta blockers, thiazides, estrogens) should be discontinued or changed if possible prior to consideration of triglyceride-lowering drug therapy.
Section 44425-7
Store at 20°-25°C (68°-77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Do not freeze. Protect from light. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
11 Description
Omega-3-acid ethyl esters, USP, lipid-regulating agents, are supplied as a liquid-filled gel capsule for oral administration. Each 1-gram capsule of omega-3-acid ethyl esters, USP contains at least 900 mg of the ethyl esters of omega-3 fatty acids sourced from fish oils. These are predominantly a combination of ethyl esters of eicosapentaenoic acid (EP — approximately 465 mg) and docosahexaenoic acid (DHA — approximately 375 mg).
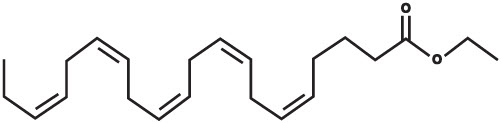
The empirical formula of EPA ethyl ester is C 22H 34O 2, and the molecular weight of EPA ethyl ester is 330.51. The structural formula of EPA ethyl ester is:
The empirical formula of DHA ethyl ester is C 24H 36O 2, and the molecular weight of DHA ethyl ester is 356.55. The structural formula of DHA ethyl ester is:
Omega-3-Acid Ethyl Esters Capsules USP also contain the following inactive ingredients: 4 mg alpha-tocopherol, gelatin, glycerin, hypromellose, propylene glycol, and titanium dioxide.
5.2 Fish Allergy
Omega-3-acid ethyl esters contain ethyl esters of omega-3 fatty acids (EPA and DHA) obtained from the oil of several fish sources. It is not known whether patients with allergies to fish and/or shellfish, are at increased risk of an allergic reaction to omega-3-acid ethyl esters. Omega-3-acid ethyl esters should be used with caution in patients with known hypersensitivity to fish and/or shellfish.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
A limited number of subjects older than 65 years were enrolled in the clinical trials of omega-3-acid ethyl esters. Safety and efficacy findings in subjects older than 60 years did not appear to differ from those of subjects younger than 60 years.
4 Contraindications
Omega-3-acid ethyl esters are contraindicated in patients with known hypersensitivity (e.g., anaphylactic reaction) to omega-3-acid ethyl esters or any of its components.
6 Adverse Reactions
The most common adverse reactions (incidence >3% and greater than placebo) were eructation, dyspepsia, and taste perversion. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact KD Pharma USA at 1-877-241-7978 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions
Omega-3-acids may prolong bleeding time. Patients taking omega-3-acid ethyl esters and an anticoagulant or other drug affecting coagulation (e.g., anti-platelet agents) should be monitored periodically. ( 7.1)
8.3 Nursing Mothers
Studies with omega-3-acid ethyl esters have demonstrated excretion in human milk. The effect of this excretion on the infant of a nursing mother is unknown; caution should be exercised when omega-3-acid ethyl esters are administered to a nursing mother. An animal study in lactating rats given oral gavage 14C-ethyl EPA demonstrated that drug levels were 6 to 14 times higher in milk than in plasma.
1 Indications and Usage
Omega-3-acid ethyl esters capsules are indicated as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (greater than or equal to 500 mg per dL) hypertriglyceridemia (HTG).
12.1 Mechanism of Action
The mechanism of action of omega-3-acid ethyl esters is not completely understood. Potential mechanisms of action include inhibition of acyl-CoA:1,2-diacylglycerol acyltransferase, increased mitochondrial and peroxisomal β-oxidation in the liver, decreased lipogenesis in the liver, and increased plasma lipoprotein lipase activity. Omega-3-acid ethyl esters may reduce the synthesis of triglycerides in the liver because EPA and DHA are poor substrates for the enzymes responsible for TG synthesis, and EPA and DHA inhibit esterification of other fatty acids.
5 Warnings and Precautions
- In patients with hepatic impairment, monitor ALT and AST levels periodically during therapy. ( 5.1)
- Omega-3-acid ethyl esters may increase levels of LDL. Monitor LDL levels periodically during therapy. ( 5.1)
- Use with caution in patients with known hypersensitivity to fish and/or shellfish. ( 5.2)
- There is a possible association between omega-3-acid ethyl esters and more frequent recurrences of symptomatic atrial fibrillation or flutter in patients with paroxysmal or persistent atrial fibrillation, particularly within the first months of initiating therapy. ( 5.3)
2 Dosage and Administration
- Assess triglyceride levels carefully before initiating therapy. Identify other causes (e.g., diabetes mellitus, hypothyroidism, medications) of high triglyceride levels and manage as appropriate [see Indications and Usage (1)].
- Patients should be placed on an appropriate lipid-lowering diet before receiving omega-3-acid ethyl esters, and should continue this diet during treatment with omega-3-acid ethyl esters. In clinical studies, omega-3-acid ethyl esters was administered with meals.
The daily dose of omega-3-acid ethyl esters is 4 grams per day. The daily dose may be taken as a single 4-gram dose (4 capsules) or as two 2-gram doses (2 capsules given twice daily).
Patients should be advised to swallow omega-3-acid ethyl esters whole. Do not break open, crush, dissolve, or chew omega-3-acid ethyl esters.
9 Drug Abuse and Dependence
Omega-3-acid ethyl esters do not have any known drug abuse or withdrawal effects.
3 Dosage Forms and Strengths
Omega-3-acid ethyl esters capsules USP are supplied as 1-gram oblong, clear, soft gel capsules filled with slightly yellow to yellow liquid and imprinted with "GLW" in white ink across one side.
6.2 Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the events described below have been identified during post-approval use of omega-3-acid ethyl esters. Because these events are reported voluntarily from a population of unknown size, it is not possible to reliably estimate their frequency or to always establish a causal relationship to drug exposure.
The following events have been reported: anaphylactic reaction, hemorrhagic diathesis, urticaria.
8 Use in Specific Populations
- Pregnancy: Use during pregnancy only if the potential benefit justifies the potential risk to the fetus. ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse reactions reported in at least 3% of subjects treated with Omega-3-acid ethyl esters and at with omega-3-acid ethyl esters based on pooled data across 23 clinical trials are listed in Table 1.
| Adverse Reaction
Trials included subjects with HTG and severe HTG.
|
Omega-3-acid ethyl esters (n = 655) | Placebo (n = 370) | ||
|---|---|---|---|---|
| n | % | n | % | |
| Eructation | 29 | 4 | 5 | 1 |
| Dyspepsia | 22 | 3 | 6 | 2 |
| Taste perversion | 27 | 4 | 1 | <1 |
Additional adverse reactions from clinical trials are listed below:
14.1 Severe Hypertriglyceridemia
The effects of omega-3-acid ethyl esters 4 grams per day were assessed in 2 randomized, placebo-controlled, doubleblind, parallel-group trials of 84 adult subjects (42 on omega-3-acid ethyl esters, 42 on placebo) with very high triglyceride levels. Subjects whose baseline triglyceride levels were between 500 and 2,000 mg per dL were enrolled in these 2 trials of 6 and 16 weeks' duration. The median triglyceride and LDL-C levels in these subjects were 792 mg per dL and 100 mg per dL, respectively. Median high-density lipoprotein cholesterol (HDL-C) level was 23.0 mg per dL.
The changes in the major lipoprotein lipid parameters for the groups receiving omega-3-acid ethyl esters or placebo are shown in Table 2.
| Parameter | Omega-3-acid ethyl esters
n=42 |
Placebo
n=42 |
Difference | ||
|---|---|---|---|---|---|
| BL | % Change | BL | % Change | ||
| BL = Baseline (mg per dL); % Change = Median Percent Change from Baseline; Difference = Omega-3-acid ethyl esters Median % Change – Placebo Median % Change; VLDL-C = Very-low-density lipoprotein (VLDL) cholesterol. | |||||
| TG | 816 | -44.9 | 788 | +6.7 | -51.6 |
| Non-HDL-C | 271 | -13.8 | 292 | -3.6 | -10.2 |
| TC | 296 | -9.7 | 314 | -1.7 | -8.0 |
| VLDL-C | 175 | -41.7 | 175 | -0.9 | -40.8 |
| HDL-C | 22 | +9.1 | 24 | 0.0 | +9.1 |
| LDL-C | 89 | +44.5 | 108 | -4.8 | +49.3 |
Omega-3-acid ethyl esters 4 grams per day reduced median TG, VLDL-C, and non-HDL-C levels and increased median HDL-C from baseline relative to placebo. Treatment with omega-3-acid ethyl esters to reduce very high TG levels may result in elevations in LDL-C and non-HDL-C in some individuals. Patients should be monitored to ensure that the LDL-C level does not increase excessively.
The effect of omega-3-acid ethyl esters on the risk of pancreatitis has not been determined.
The effect of omega-3-acid ethyl esters on cardiovascular mortality and morbidity has not been determined.
5.1 Monitoring: Laboratory Tests
In patients with hepatic impairment, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels should be monitored periodically during therapy with omega-3-acid ethyl esters. In some patients, increases in ALT levels without a concurrent increase in AST levels were observed.
In some patients, omega-3-acid ethyl esters increases LDL-C levels. LDL-C levels should be monitored periodically during therapy with omega-3-acid ethyl esters.
Laboratory studies should be performed periodically to measure the patient's TG levels during therapy with omega-3-acid ethyl esters.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Omega-3-Acid Ethyl Esters Capsules USP are available as follows: 1 gram – slightly yellow to yellow liquid, oblong, clear, soft gel capsules, imprinted with "GLW" in white ink across one side.
Bottles of 120: NDC 51407-763-12
5.3 Recurrent Atrial Fibrillation (af) Or Flutter
In a double-blind, placebo-controlled trial of 663 subjects with symptomatic paroxysmal AF (n = 542) or persistent AF (n = 121), recurrent AF or flutter was observed in subjects randomized to omega-3-acid ethyl esters who received 8 grams per day for 7 days and 4 grams per day thereafter for 23 weeks at a higher rate relative to placebo. Subjects in this trial had median baseline triglycerides of 127 mg per dL, had no substantial structural heart disease, were taking no anti-arrhythmic therapy (rate control permitted), and were in normal sinus rhythm at baseline.
At 24 weeks, in the paroxysmal AF stratum, there were 129 (47%) first recurrent symptomatic AF or flutter events on placebo and 141 (53%) on omega-3-acid ethyl esters [primary endpoint, HR 1.19; 95% CI: 0.93, 1.35]. In the persistent AF stratum, there were 19 (35%) events on placebo and 34 (52%) events on omega-3-acid ethyl esters [HR 1.63; 95% CI: 0.91, 2.18]. For both strata combined, the HR was 1.25; 95% CI: 1.00, 1.40. Although the clinical significance of these results is uncertain, there is a possible association between omega-3-acid ethyl esters and more frequent recurrences of symptomatic atrial fibrillation or flutter in patients with paroxysmal or persistent atrial fibrillation, particularly within the first 2 or 3 months of initiating therapy.
Omega-3-acid ethyl esters are not indicated for the treatment of AF or flutter.
Principal Display Panel 120 Capsule Bottle Label
NDC 51407-763-12
Omega-3-Acid
Ethyl Esters
Capsules, USP
1 gram*
Swallow capsules whole.
Rx Only
120 Capsules
7.1 Anticoagulants Or Other Drugs Affecting Coagulation
Some trials with omega-3-acids demonstrated prolongation of bleeding time. The prolongation of bleeding time reported in these trials has not exceeded normal limits and did not produce clinically significant bleeding episodes. Clinical trials have not been done to thoroughly examine the effect of omega-3-acid ethyl esters and concomitant anticoagulants. Patients receiving treatment with omega-3-acid ethyl esters and an anticoagulant or other drug affecting coagulation (e.g., anti-platelet agents) should be monitored periodically.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a rat carcinogenicity study with oral gavage doses of 100, 600, and 2,000 mg per kg per day, males were treated with omega-3-acid ethyl esters for 101 weeks and females for 89 weeks without an increased incidence of tumors (up to 5 times human systemic exposures following an oral dose of 4 grams per day based on a body surface area comparison). Standard lifetime carcinogenicity bioassays were not conducted in mice.
Omega-3-acid ethyl esters were not mutagenic or clastogenic with or without metabolic activation in the bacterial mutagenesis (Ames) test with Salmonella typhimuriumand Escherichia colior in the chromosomal aberration assay in Chinese hamster V79 lung cells or human lymphocytes. Omega-3-acid ethyl esters were negative in the in vivomouse micronucleus assay.
In a rat fertility study with oral gavage doses of 100, 600, and 2,000 mg per kg per day, males were treated for 10 weeks prior to mating and females were treated for 2 weeks prior to and throughout mating, gestation, and lactation. No adverse effect on fertility was observed at 2,000 mg per kg per day (5 times human systemic exposure following an oral dose of 4 grams per day based on a body surface area comparison).
Structured Label Content
Section 42229-5 (42229-5)
Usage Considerations:Patients should be placed on an appropriate lipid-lowering diet before receiving omega-3-acid ethyl esters and should continue this diet during treatment with omega-3-acid ethyl esters.
Laboratory studies should be done to ascertain that the lipid levels are consistently abnormal before instituting therapy with omega-3-acid ethyl esters. Every attempt should be made to control serum lipids with appropriate diet, exercise, weight loss in obese patients, and control of any medical problems such as diabetes mellitus and hypothyroidism that are contributing to the lipid abnormalities. Medications known to exacerbate hypertriglyceridemia (such as beta blockers, thiazides, estrogens) should be discontinued or changed if possible prior to consideration of triglyceride-lowering drug therapy.
Section 44425-7 (44425-7)
Store at 20°-25°C (68°-77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Do not freeze. Protect from light. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
11 Description (11 DESCRIPTION)
Omega-3-acid ethyl esters, USP, lipid-regulating agents, are supplied as a liquid-filled gel capsule for oral administration. Each 1-gram capsule of omega-3-acid ethyl esters, USP contains at least 900 mg of the ethyl esters of omega-3 fatty acids sourced from fish oils. These are predominantly a combination of ethyl esters of eicosapentaenoic acid (EP — approximately 465 mg) and docosahexaenoic acid (DHA — approximately 375 mg).
The empirical formula of EPA ethyl ester is C 22H 34O 2, and the molecular weight of EPA ethyl ester is 330.51. The structural formula of EPA ethyl ester is:
The empirical formula of DHA ethyl ester is C 24H 36O 2, and the molecular weight of DHA ethyl ester is 356.55. The structural formula of DHA ethyl ester is:
Omega-3-Acid Ethyl Esters Capsules USP also contain the following inactive ingredients: 4 mg alpha-tocopherol, gelatin, glycerin, hypromellose, propylene glycol, and titanium dioxide.
5.2 Fish Allergy
Omega-3-acid ethyl esters contain ethyl esters of omega-3 fatty acids (EPA and DHA) obtained from the oil of several fish sources. It is not known whether patients with allergies to fish and/or shellfish, are at increased risk of an allergic reaction to omega-3-acid ethyl esters. Omega-3-acid ethyl esters should be used with caution in patients with known hypersensitivity to fish and/or shellfish.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
A limited number of subjects older than 65 years were enrolled in the clinical trials of omega-3-acid ethyl esters. Safety and efficacy findings in subjects older than 60 years did not appear to differ from those of subjects younger than 60 years.
4 Contraindications (4 CONTRAINDICATIONS)
Omega-3-acid ethyl esters are contraindicated in patients with known hypersensitivity (e.g., anaphylactic reaction) to omega-3-acid ethyl esters or any of its components.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (incidence >3% and greater than placebo) were eructation, dyspepsia, and taste perversion. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact KD Pharma USA at 1-877-241-7978 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions (7 DRUG INTERACTIONS)
Omega-3-acids may prolong bleeding time. Patients taking omega-3-acid ethyl esters and an anticoagulant or other drug affecting coagulation (e.g., anti-platelet agents) should be monitored periodically. ( 7.1)
8.3 Nursing Mothers
Studies with omega-3-acid ethyl esters have demonstrated excretion in human milk. The effect of this excretion on the infant of a nursing mother is unknown; caution should be exercised when omega-3-acid ethyl esters are administered to a nursing mother. An animal study in lactating rats given oral gavage 14C-ethyl EPA demonstrated that drug levels were 6 to 14 times higher in milk than in plasma.
Patient Information (PATIENT INFORMATION)
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP
(oh may' ga as' id eth' il es' ters).
What are OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP are a prescription medicine used along with a low-fat and low-cholesterol diet to lower very high triglyceride (fat) levels in adults.
It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP change your risk of having inflammation of your pancreas (pancreatitis).
It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP prevent you from having a heart attack or stroke.
It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP are safe and effective in children.
Who should not take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
Do not take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP if you are allergic to omega-3-acid ethyl esters or any of the ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. See the end of this leaflet for a complete list of ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
Before taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, tell your healthcare provider about all of your medical conditions, including if you:
- have diabetes.
- have a low thyroid problem (hypothyroidism).
- have a liver problem.
- have a pancreas problem.
- have a certain heart rhythm problem called atrial fibrillation or flutter.
- are allergic to fish or shellfish. It is not known if people who are allergic to fish or shellfish are also allergic to OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
- are pregnant or plan to become pregnant. It is not known if OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP will harm your unborn baby.
- are breastfeeding or plan to breastfeed. OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP can pass into your breast milk. Talk to your healthcare provider about the best way to feed your baby if you take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements.
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP can interact with certain other medicines that you are taking. Using OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP with medicines that affect blood clotting (anticoagulants or blood thinners) may cause serious side effects.
How should I take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
- Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP exactly as your healthcare provider tells you to take them.
- You should not take more than 4 capsules of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP each day. Either take all 4 capsules at one time or 2 capsules two times a day.
- Do not change your dose or stop OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP without talking to your healthcare provider.
- Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP with food.
- Take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP whole. Do not break, open, crush, dissolve, or chew OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP before swallowing. If you cannot swallow OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP whole, tell your healthcare provider. You may need a different medicine.
- If you miss a dose of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, take the missed dose as soon as you remember. If you miss one day of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, do not double your dose the next time you take it.
- Your healthcare provider may start you on a cholesterol-lowering diet before giving you OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP. Stay on this diet while taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
- Your healthcare provider should do blood tests to check your triglyceride, bad cholesterol (LDL-C) and liver function (ALT and AST) levels while you take OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
What are the possible side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause serious side effects, including:
- changes in certain blood tests.OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause an increase in the results of blood tests used to check your liver function and your bad cholesterol levels.
- increased risk of a heart rhythm problem in people who have a heart rhythm problem.OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP may cause an increase in the frequency of a heart rhythm problem (atrial fibrillation or flutter), especially if the first few months of taking OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP, if you already have a heart rhythm problem.
The most common side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP include:
- burping
- upset stomach
- a change in your sense of taste
These are not all the possible side effects of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
- Store OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not freeze OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP.
- Safely throw away medicine that is out of date or no longer needed.
Keep OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP and all medicines out of the reach of children.
General information about the safe and effective use of OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP for a condition for which it was not prescribed. Do not give OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP to other people, even if they have the same symptoms you have. It may harm them.
You can ask your healthcare provider or pharmacist for information about OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP that is written for health professionals.
What are the ingredients in OMEGA-3-ACID ETHYL ESTERS CAPSULES, USP?
Active Ingredient: omega-3-acid ethyl esters, mostly EPA and DHA
Inactive Ingredients: 4 mg alpha-tocopherol, gelatin, glycerin, hypromellose, propylene glycol, and titanium dioxide.
Manufactured By:
Patheon Softgels Inc.
High Point, NC 27265
USA
Manufactured For:
KD Pharma USA Inc.
12021 SW 144th Street
Miami, Florida 33186
USA
This patient labeling has been approved by the U.S. Food and Drug Administration.
Revised: November 2022
Revision: 1
Marketed by:
GSMS, Inc.
Camarillo, CA 93012 USA
1 Indications and Usage (1 INDICATIONS AND USAGE)
Omega-3-acid ethyl esters capsules are indicated as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (greater than or equal to 500 mg per dL) hypertriglyceridemia (HTG).
12.1 Mechanism of Action
The mechanism of action of omega-3-acid ethyl esters is not completely understood. Potential mechanisms of action include inhibition of acyl-CoA:1,2-diacylglycerol acyltransferase, increased mitochondrial and peroxisomal β-oxidation in the liver, decreased lipogenesis in the liver, and increased plasma lipoprotein lipase activity. Omega-3-acid ethyl esters may reduce the synthesis of triglycerides in the liver because EPA and DHA are poor substrates for the enzymes responsible for TG synthesis, and EPA and DHA inhibit esterification of other fatty acids.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- In patients with hepatic impairment, monitor ALT and AST levels periodically during therapy. ( 5.1)
- Omega-3-acid ethyl esters may increase levels of LDL. Monitor LDL levels periodically during therapy. ( 5.1)
- Use with caution in patients with known hypersensitivity to fish and/or shellfish. ( 5.2)
- There is a possible association between omega-3-acid ethyl esters and more frequent recurrences of symptomatic atrial fibrillation or flutter in patients with paroxysmal or persistent atrial fibrillation, particularly within the first months of initiating therapy. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Assess triglyceride levels carefully before initiating therapy. Identify other causes (e.g., diabetes mellitus, hypothyroidism, medications) of high triglyceride levels and manage as appropriate [see Indications and Usage (1)].
- Patients should be placed on an appropriate lipid-lowering diet before receiving omega-3-acid ethyl esters, and should continue this diet during treatment with omega-3-acid ethyl esters. In clinical studies, omega-3-acid ethyl esters was administered with meals.
The daily dose of omega-3-acid ethyl esters is 4 grams per day. The daily dose may be taken as a single 4-gram dose (4 capsules) or as two 2-gram doses (2 capsules given twice daily).
Patients should be advised to swallow omega-3-acid ethyl esters whole. Do not break open, crush, dissolve, or chew omega-3-acid ethyl esters.
9 Drug Abuse and Dependence (9 DRUG ABUSE AND DEPENDENCE)
Omega-3-acid ethyl esters do not have any known drug abuse or withdrawal effects.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Omega-3-acid ethyl esters capsules USP are supplied as 1-gram oblong, clear, soft gel capsules filled with slightly yellow to yellow liquid and imprinted with "GLW" in white ink across one side.
6.2 Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the events described below have been identified during post-approval use of omega-3-acid ethyl esters. Because these events are reported voluntarily from a population of unknown size, it is not possible to reliably estimate their frequency or to always establish a causal relationship to drug exposure.
The following events have been reported: anaphylactic reaction, hemorrhagic diathesis, urticaria.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Use during pregnancy only if the potential benefit justifies the potential risk to the fetus. ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse reactions reported in at least 3% of subjects treated with Omega-3-acid ethyl esters and at with omega-3-acid ethyl esters based on pooled data across 23 clinical trials are listed in Table 1.
| Adverse Reaction
Trials included subjects with HTG and severe HTG.
|
Omega-3-acid ethyl esters (n = 655) | Placebo (n = 370) | ||
|---|---|---|---|---|
| n | % | n | % | |
| Eructation | 29 | 4 | 5 | 1 |
| Dyspepsia | 22 | 3 | 6 | 2 |
| Taste perversion | 27 | 4 | 1 | <1 |
Additional adverse reactions from clinical trials are listed below:
14.1 Severe Hypertriglyceridemia
The effects of omega-3-acid ethyl esters 4 grams per day were assessed in 2 randomized, placebo-controlled, doubleblind, parallel-group trials of 84 adult subjects (42 on omega-3-acid ethyl esters, 42 on placebo) with very high triglyceride levels. Subjects whose baseline triglyceride levels were between 500 and 2,000 mg per dL were enrolled in these 2 trials of 6 and 16 weeks' duration. The median triglyceride and LDL-C levels in these subjects were 792 mg per dL and 100 mg per dL, respectively. Median high-density lipoprotein cholesterol (HDL-C) level was 23.0 mg per dL.
The changes in the major lipoprotein lipid parameters for the groups receiving omega-3-acid ethyl esters or placebo are shown in Table 2.
| Parameter | Omega-3-acid ethyl esters
n=42 |
Placebo
n=42 |
Difference | ||
|---|---|---|---|---|---|
| BL | % Change | BL | % Change | ||
| BL = Baseline (mg per dL); % Change = Median Percent Change from Baseline; Difference = Omega-3-acid ethyl esters Median % Change – Placebo Median % Change; VLDL-C = Very-low-density lipoprotein (VLDL) cholesterol. | |||||
| TG | 816 | -44.9 | 788 | +6.7 | -51.6 |
| Non-HDL-C | 271 | -13.8 | 292 | -3.6 | -10.2 |
| TC | 296 | -9.7 | 314 | -1.7 | -8.0 |
| VLDL-C | 175 | -41.7 | 175 | -0.9 | -40.8 |
| HDL-C | 22 | +9.1 | 24 | 0.0 | +9.1 |
| LDL-C | 89 | +44.5 | 108 | -4.8 | +49.3 |
Omega-3-acid ethyl esters 4 grams per day reduced median TG, VLDL-C, and non-HDL-C levels and increased median HDL-C from baseline relative to placebo. Treatment with omega-3-acid ethyl esters to reduce very high TG levels may result in elevations in LDL-C and non-HDL-C in some individuals. Patients should be monitored to ensure that the LDL-C level does not increase excessively.
The effect of omega-3-acid ethyl esters on the risk of pancreatitis has not been determined.
The effect of omega-3-acid ethyl esters on cardiovascular mortality and morbidity has not been determined.
5.1 Monitoring: Laboratory Tests
In patients with hepatic impairment, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels should be monitored periodically during therapy with omega-3-acid ethyl esters. In some patients, increases in ALT levels without a concurrent increase in AST levels were observed.
In some patients, omega-3-acid ethyl esters increases LDL-C levels. LDL-C levels should be monitored periodically during therapy with omega-3-acid ethyl esters.
Laboratory studies should be performed periodically to measure the patient's TG levels during therapy with omega-3-acid ethyl esters.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Omega-3-Acid Ethyl Esters Capsules USP are available as follows: 1 gram – slightly yellow to yellow liquid, oblong, clear, soft gel capsules, imprinted with "GLW" in white ink across one side.
Bottles of 120: NDC 51407-763-12
5.3 Recurrent Atrial Fibrillation (af) Or Flutter (5.3 Recurrent Atrial Fibrillation (AF) or Flutter)
In a double-blind, placebo-controlled trial of 663 subjects with symptomatic paroxysmal AF (n = 542) or persistent AF (n = 121), recurrent AF or flutter was observed in subjects randomized to omega-3-acid ethyl esters who received 8 grams per day for 7 days and 4 grams per day thereafter for 23 weeks at a higher rate relative to placebo. Subjects in this trial had median baseline triglycerides of 127 mg per dL, had no substantial structural heart disease, were taking no anti-arrhythmic therapy (rate control permitted), and were in normal sinus rhythm at baseline.
At 24 weeks, in the paroxysmal AF stratum, there were 129 (47%) first recurrent symptomatic AF or flutter events on placebo and 141 (53%) on omega-3-acid ethyl esters [primary endpoint, HR 1.19; 95% CI: 0.93, 1.35]. In the persistent AF stratum, there were 19 (35%) events on placebo and 34 (52%) events on omega-3-acid ethyl esters [HR 1.63; 95% CI: 0.91, 2.18]. For both strata combined, the HR was 1.25; 95% CI: 1.00, 1.40. Although the clinical significance of these results is uncertain, there is a possible association between omega-3-acid ethyl esters and more frequent recurrences of symptomatic atrial fibrillation or flutter in patients with paroxysmal or persistent atrial fibrillation, particularly within the first 2 or 3 months of initiating therapy.
Omega-3-acid ethyl esters are not indicated for the treatment of AF or flutter.
Principal Display Panel 120 Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 120 Capsule Bottle Label)
NDC 51407-763-12
Omega-3-Acid
Ethyl Esters
Capsules, USP
1 gram*
Swallow capsules whole.
Rx Only
120 Capsules
7.1 Anticoagulants Or Other Drugs Affecting Coagulation (7.1 Anticoagulants or Other Drugs Affecting Coagulation)
Some trials with omega-3-acids demonstrated prolongation of bleeding time. The prolongation of bleeding time reported in these trials has not exceeded normal limits and did not produce clinically significant bleeding episodes. Clinical trials have not been done to thoroughly examine the effect of omega-3-acid ethyl esters and concomitant anticoagulants. Patients receiving treatment with omega-3-acid ethyl esters and an anticoagulant or other drug affecting coagulation (e.g., anti-platelet agents) should be monitored periodically.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a rat carcinogenicity study with oral gavage doses of 100, 600, and 2,000 mg per kg per day, males were treated with omega-3-acid ethyl esters for 101 weeks and females for 89 weeks without an increased incidence of tumors (up to 5 times human systemic exposures following an oral dose of 4 grams per day based on a body surface area comparison). Standard lifetime carcinogenicity bioassays were not conducted in mice.
Omega-3-acid ethyl esters were not mutagenic or clastogenic with or without metabolic activation in the bacterial mutagenesis (Ames) test with Salmonella typhimuriumand Escherichia colior in the chromosomal aberration assay in Chinese hamster V79 lung cells or human lymphocytes. Omega-3-acid ethyl esters were negative in the in vivomouse micronucleus assay.
In a rat fertility study with oral gavage doses of 100, 600, and 2,000 mg per kg per day, males were treated for 10 weeks prior to mating and females were treated for 2 weeks prior to and throughout mating, gestation, and lactation. No adverse effect on fertility was observed at 2,000 mg per kg per day (5 times human systemic exposure following an oral dose of 4 grams per day based on a body surface area comparison).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:37.055195 · Updated: 2026-03-14T22:25:11.616622