These Highlights Do Not Include All The Information Needed To Use Derma-smoothe/fs Scalp Oil Safely And Effectively. See Full Prescribing Information For Derma-smoothe/fs Scalp Oil.
113e167a-a9fe-440e-9446-d6d87963ef46
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Derma-Smoothe/FS ® Scalp Oil is a corticosteroid indicated for the treatment of psoriasis of the scalp in adults. ( 1 )
Indications and Usage
Derma-Smoothe/FS ® Scalp Oil is a corticosteroid indicated for the treatment of psoriasis of the scalp in adults. ( 1 )
Dosage and Administration
DERMA-SMOOTHE/FS Scalp Oil is for topical use only. Not for oral, ophthalmic, or intravaginal use. Wet or dampen hair and scalp thoroughly. Apply a thin film of DERMA-SMOOTHE/FS Scalp Oil on the scalp, massage well and cover scalp with the supplied shower cap. Leave on overnight or for a minimum of 4 hours then wash hair with regular shampoo and rinse thoroughly. Use daily as needed. Discontinue DERMA-SMOOTHE/FS Scalp Oil when control of disease is achieved within 2 weeks, or contact the healthcare provider if no improvement is seen within 2 weeks. Do not use DERMA-SMOOTHE/FS Scalp Oil on the face unless directed by the healthcare provider. Do not apply to intertriginous areas due to the increased risk of local adverse reactions [see Adverse Reactions (6) ] . Do not apply to the diaper area; diapers or plastic pants may constitute occlusive use. [see Warnings and Precautions (5.1) ]
Warnings and Precautions
Endocrine System Adverse Reactions: o Topical corticosteroids can produce reversible HPA axis suppression, Cushing's syndrome, hyperglycemia, and glucosuria. ( 5.1 ) o Systemic absorption may require evaluation for hypothalamic-pituitary-adrenal (HPA) axis suppression. Potent corticosteroids use on large areas, prolonged use or occlusive use, altered skin barrier, liver failure, and young age may increase systemic absorption. Modify use should HPA axis suppression develop. ( 5.1 ) Local Adverse Reactions: Local adverse reactions may include atrophy, striae irritation, acneiform eruptions, hypopigmentation, and allergic contact dermatitis, and may be more likely with occlusive use or more potent corticosteroids. ( 5.2 , 6.1 ) Ophthalmic Adverse Reactions: May increase the risks of glaucoma and posterior subcapsular cataract. Avoid contact of DERMA-SMOOTHE/FS Scalp Oil with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation. ( 5.3 )
Contraindications
None.
Adverse Reactions
Local adverse reactions may occur with use of topical corticosteroids, including Derma-Smoothe/FS Scalp Oil, and may be more likely to occur with occlusive use, prolonged use or use of higher potency corticosteroids. Some local adverse reactions may be irreversible. Reactions may include atrophy, striae, telangiectasias, burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria [see Adverse Reactions (6.1) ] .
Storage and Handling
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% (NDC # 68791-102-04) is supplied in bottles containing 4 fluid ounces and with 2 shower caps.
How Supplied
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% (NDC # 68791-102-04) is supplied in bottles containing 4 fluid ounces and with 2 shower caps.
Medication Information
Warnings and Precautions
Endocrine System Adverse Reactions: o Topical corticosteroids can produce reversible HPA axis suppression, Cushing's syndrome, hyperglycemia, and glucosuria. ( 5.1 ) o Systemic absorption may require evaluation for hypothalamic-pituitary-adrenal (HPA) axis suppression. Potent corticosteroids use on large areas, prolonged use or occlusive use, altered skin barrier, liver failure, and young age may increase systemic absorption. Modify use should HPA axis suppression develop. ( 5.1 ) Local Adverse Reactions: Local adverse reactions may include atrophy, striae irritation, acneiform eruptions, hypopigmentation, and allergic contact dermatitis, and may be more likely with occlusive use or more potent corticosteroids. ( 5.2 , 6.1 ) Ophthalmic Adverse Reactions: May increase the risks of glaucoma and posterior subcapsular cataract. Avoid contact of DERMA-SMOOTHE/FS Scalp Oil with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation. ( 5.3 )
Indications and Usage
Derma-Smoothe/FS ® Scalp Oil is a corticosteroid indicated for the treatment of psoriasis of the scalp in adults. ( 1 )
Dosage and Administration
DERMA-SMOOTHE/FS Scalp Oil is for topical use only. Not for oral, ophthalmic, or intravaginal use. Wet or dampen hair and scalp thoroughly. Apply a thin film of DERMA-SMOOTHE/FS Scalp Oil on the scalp, massage well and cover scalp with the supplied shower cap. Leave on overnight or for a minimum of 4 hours then wash hair with regular shampoo and rinse thoroughly. Use daily as needed. Discontinue DERMA-SMOOTHE/FS Scalp Oil when control of disease is achieved within 2 weeks, or contact the healthcare provider if no improvement is seen within 2 weeks. Do not use DERMA-SMOOTHE/FS Scalp Oil on the face unless directed by the healthcare provider. Do not apply to intertriginous areas due to the increased risk of local adverse reactions [see Adverse Reactions (6) ] . Do not apply to the diaper area; diapers or plastic pants may constitute occlusive use. [see Warnings and Precautions (5.1) ]
Contraindications
None.
Adverse Reactions
Local adverse reactions may occur with use of topical corticosteroids, including Derma-Smoothe/FS Scalp Oil, and may be more likely to occur with occlusive use, prolonged use or use of higher potency corticosteroids. Some local adverse reactions may be irreversible. Reactions may include atrophy, striae, telangiectasias, burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria [see Adverse Reactions (6.1) ] .
Storage and Handling
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% (NDC # 68791-102-04) is supplied in bottles containing 4 fluid ounces and with 2 shower caps.
How Supplied
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% (NDC # 68791-102-04) is supplied in bottles containing 4 fluid ounces and with 2 shower caps.
Description
Derma-Smoothe/FS ® Scalp Oil is a corticosteroid indicated for the treatment of psoriasis of the scalp in adults. ( 1 )
Section 42229-5
DERMA-SMOOTHE/FS® Scalp Oil is indicated for the treatment of psoriasis of the scalp in adults.
Section 44425-7
Storage: Keep tightly closed. Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature].
Section 59845-8
Administration Instructions
Advise patients that DERMA-SMOOTHE/FS Scalp Oil is for topical use only [see Dosage and Administration (2)].
Instruct patients not to apply DERMA-SMOOTHE/FS Scalp Oil to the diaper area as diapers or plastic pants may constitute occlusive use [see Dosage and Administration (2)].
Advise patients to avoid use of DERMA-SMOOTHE/FS Scalp Oil on the face, axillae, or groin unless directed by their healthcare provider [see Dosage and Administration (2)].
Advise patients to discontinue therapy when control of disease is achieved. Instruct patients to contact their healthcare provider if no improvement is seen within 2 weeks [see Dosage and Administration (2)].
Endocrine System Adverse Reactions
Instruct patients not to use other corticosteroid-containing products while using DERMA-SMOOTHE/FS Scalp Oil without first consulting their healthcare provider [see Warnings and Precautions (5.1)].
Ophthalmic Adverse Reactions
Advise patients to avoid contact with the eyes and in case of contact, wash eyes liberally with water. Instruct patients to tell their healthcare provider if they develop any visual symptoms [see Warnings and Precautions (5.3)].
Pregnancy and Lactation
Advise women to use DERMA-SMOOTHE/FS Scalp Oil on the smallest area of skin and for the shortest duration possible while pregnant or breastfeeding. Advise patients that are breastfeeding not to apply DERMA-SMOOTHE/FS Scalp Oil directly to the nipple and areola to avoid direct infant exposure [See Use in Specific Populations (8.1 and 8.2)].
11 Description
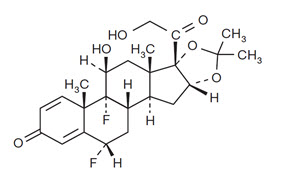
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% contains fluocinolone acetonide [(6α, 11β, 16α)-6,9-difluoro-11,21-dihydroxy-16,17[(1–methylethylidene) bis(oxy)]-pregna-1,4-diene-3,20-dione, cyclic 16,17 acetal with acetone], a synthetic corticosteroid for topical dermatologic use. Chemically, fluocinolone acetonide is C24 H30 F2 O6. It has the following structural formula:
Fluocinolone acetonide has a molecular weight of 452.50. It is a white crystalline powder that is odorless, stable in light, and melts at 270°C with decomposition; soluble in alcohol, acetone and methanol; slightly soluble in chloroform; insoluble in water.
Each gram of DERMA-SMOOTHE/FS Scalp Oil contains approximately 0.11 mg of fluocinolone acetonide in a blend of oils, which contains isopropyl alcohol, isopropyl myristate, light mineral oil, oleth-2, refined peanut oil and fragrances.
Each packaged product contains 2 shower caps. The shower cap is made of low density polyethylene material with rubber elastic.
DERMA-SMOOTHE/FS Scalp Oil is formulated with 48% refined peanut oil. The bulk refined peanut oil used in DERMA-SMOOTHE/FS Scalp Oil is heated at 246°C (475°F) for at least 15 minutes. The refined peanut oil used in DERMA-SMOOTHE/FS Scalp Oil is routinely tested for peanut proteins through amino acid analysis; the quantity of amino acids is below 0.5 parts per million (ppm).
14 Clinical Studies
In a vehicle-controlled study for the treatment of psoriasis of the scalp in adults, after 21 days of treatment, 60% of patients on active treatment and 21% of patients on the drug vehicle had excellent to cleared clinical response.
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of the labeling:
• Endocrine System Adverse Reactions [see Warnings and Precautions (5.1)]
• Local Adverse Reactions [see Warnings and Precautions (5.2)]
• Ophthalmic Adverse Reactions [see Warnings and Precautions (5.3)]
12.2 Pharmacodynamics
Vasoconstrictor Assay
DERMA-SMOOTHE/FS Scalp Oil is in the low to medium range of potency as compared with other topical corticosteroids in vasoconstrictor studies. However, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
HPA axis suppression following administration of DERMA-SMOOTHE/FS Scalp Oil was not assessed.
12.3 Pharmacokinetics
Topical corticosteroids can be absorbed from intact healthy skin. The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the product formulation and the integrity of the epidermal barrier. Occlusion, inflammation and/or other disease processes in the skin may increase percutaneous absorption. The use of pharmacodynamic endpoints for assessing the systemic exposure of topical corticosteroids may be necessary due to the fact that circulating levels are often below the level of detection. Once absorbed through the skin, topical corticosteroids are metabolized, primarily in the liver, and are then excreted by the kidneys. Some corticosteroids and their metabolites are also excreted in the bile.
1 Indications and Usage
Derma-Smoothe/FS® Scalp Oil is a corticosteroid indicated for the treatment of psoriasis of the scalp in adults. (1)
Principal Display Panel
PRINCIPAL DISPLAY PANEL - 118.28 mL Carton Label
NDC 68791-102-04
Rx only
Derma-Smoothe/FS®
Fluocinolone Acetonide
0.01% Topical Oil
(SCALP OIL)
FOR TOPICAL USE ONLY
NOT FOR ORAL, OPHTHALMIC,
OR INTRAVAGINAL USE
SHAKE WELL BEFORE USE
Net Contents 118.28 mL
(4 Fl. oz.)
ROYAL
PHARMACEUTICALS®
PRINCIPAL DISPLAY PANEL - 20 mL Carton Physcian Sample Label
NOT FOR SALE
PHYSICIAN SAMPLE
NDC 68791-102-01
Rx only
Derma-Smoothe/FS®
Fluocinolone Acetonide 0.01% Topical Oil
(SCALP OIL)
FOR TOPICAL USE ONLY
NOT FOR ORAL OPHTHALMIC,
OR INTRAVAGINAL USE
SHAKE WELL BEFORE USE
NET Contents: 20 mL
Royal Pharmaceuticals®
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action in psoriasis of the scalp is unknown.
5 Warnings and Precautions
-
Endocrine System Adverse Reactions:
o Topical corticosteroids can produce reversible HPA axis suppression, Cushing's syndrome, hyperglycemia, and glucosuria. (5.1)
o Systemic absorption may require evaluation for hypothalamic-pituitary-adrenal (HPA) axis suppression. Potent corticosteroids use on large areas, prolonged use or occlusive use, altered skin barrier, liver failure, and young age may increase systemic absorption. Modify use should HPA axis suppression develop. (5.1) - Local Adverse Reactions: Local adverse reactions may include atrophy, striae irritation, acneiform eruptions, hypopigmentation, and allergic contact dermatitis, and may be more likely with occlusive use or more potent corticosteroids. (5.2, 6.1)
- Ophthalmic Adverse Reactions: May increase the risks of glaucoma and posterior subcapsular cataract. Avoid contact of DERMA-SMOOTHE/FS Scalp Oil with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation. (5.3)
2 Dosage and Administration
DERMA-SMOOTHE/FS Scalp Oil is for topical use only. Not for oral, ophthalmic, or intravaginal use.
Wet or dampen hair and scalp thoroughly. Apply a thin film of DERMA-SMOOTHE/FS Scalp Oil on the scalp, massage well and cover scalp with the supplied shower cap. Leave on overnight or for a minimum of 4 hours then wash hair with regular shampoo and rinse thoroughly. Use daily as needed.
Discontinue DERMA-SMOOTHE/FS Scalp Oil when control of disease is achieved within 2 weeks, or contact the healthcare provider if no improvement is seen within 2 weeks.
Do not use DERMA-SMOOTHE/FS Scalp Oil on the face unless directed by the healthcare provider. Do not apply to intertriginous areas due to the increased risk of local adverse reactions [see Adverse Reactions (6)].
Do not apply to the diaper area; diapers or plastic pants may constitute occlusive use. [see Warnings and Precautions (5.1)]
3 Dosage Forms and Strengths
DERMA-SMOOTHE/FS Scalp Oil is a topical oil containing 0.01% fluocinolone acetonide, supplied in bottles containing 4 fluid ounces and with 2 shower caps.
5.2 local Adverse Reactions
Local adverse reactions may occur with use of topical corticosteroids, including Derma-Smoothe/FS Scalp Oil, and may be more likely to occur with occlusive use, prolonged use or use of higher potency corticosteroids. Some local adverse reactions may be irreversible. Reactions may include atrophy, striae, telangiectasias, burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria [see Adverse Reactions (6.1)].
6.2 postmarketing Experience
The following adverse reactions have been identified during post-approval use of products containing topical corticosteroids. Because postmarketing adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Endocrine Disorders: HPA axis suppression and Cushing’s syndrome
- Eye Disorders: glaucoma and cataracts
- Nervous System Disorders: intracranial hypertension including bulging fontanelles, headaches, and bilateral papilledema
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying condition, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
An open-label safety study was conducted in 29 pediatric subjects 3 months to 2 years old to assess the HPA axis by ACTH stimulation testing following use of the formulation of DERMA-SMOOTHE/FS Scalp Oil twice daily for 4 weeks. DERMA-SMOOTHE/FS Scalp Oil is not approved for use in pediatric patients for the treatment of psoriasis of the scalp. The most common adverse reactions were reported in the study:
| Adverse Reaction | n (%) |
|---|---|
| Cough | 6 (20) |
| Rhinorrhea | 4 (13) |
| Pyrexia | 3 (10) |
| Nasopharyngitis | 2 (7) |
| Hypopigmentation | 2 (7) |
| Abscess | 1 (3) |
| Atopic Dermatitis | 1 (3) |
| Eczema | 1 (3) |
| Hyperpigmentation | 1 (3) |
| Molluscum | 1 (3) |
| Rash | 1 (3) |
| Diarrhea | 1 (3) |
| Otitis Media | 1 (3) |
| URI | 1 (3) |
| Vomiting | 1 (3) |
5.4 allergic Contact Dermatitis
Use of topical corticosteroids can cause allergic contact dermatitis. Allergic contact dermatitis to any component of topical corticosteroids is usually diagnosed by a failure to heal rather than a clinical exacerbation. Clinical diagnosis of allergic contact dermatitis can be confirmed by patch testing.
5.5 concomitant Skin Infections
Use of topical corticosteroids may delay healing or worsen concomitant skin infections. Treat concomitant skin infections with an appropriate antimicrobial agent. If the infection persists unchanged, discontinue DERMA-SMOOTHE/FS Scalp Oil until the infection has been adequately treated.
5.3 ophthalmic Adverse Reactions
Use of topical corticosteroids may increase the risks of glaucoma and posterior subcapsular cataract. Glaucoma and cataracts have been reported in postmarketing experience with the use of topical corticosteroid products. Avoid contact of DERMA-SMOOTHE/FS Scalp Oil with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
16 How Supplied / Storage and Handling
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% (NDC # 68791-102-04) is supplied in bottles containing 4 fluid ounces and with 2 shower caps.
5.1 endocrine System Adverse Reactions
Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency. Cushing's syndrome, hyperglycemia, and glucosuria can result from systemic absorption of topical corticosteroids.
HPA axis suppression and Cushing's syndrome have been reported in patients receiving topical corticosteroids.
Conditions which increase systemic absorption include the use of more potent corticosteroids, use over large surface areas, use over prolonged periods, use of occlusive dressings, altered skin barrier, liver failure, and young age. Use of more than one corticosteroid-containing product at the same time may increase total systemic corticosteroid exposure. Because of the potential for systemic absorption, use of topical corticosteroids may require that patients be periodically evaluated for HPA axis suppression. The ACTH stimulation test may be helpful in evaluating patients for HPA axis suppression.
If HPA axis suppression is documented, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent corticosteroid. Manifestations of adrenal insufficiency may require supplemental systemic corticosteroids. Recovery of HPA axis function is generally prompt upon discontinuation of topical corticosteroids.
5.6 Use in Peanut Sensitive Individuals
Use caution in prescribing DERMA-SMOOTHE/FS Scalp Oil for peanut-sensitive individuals [see Description (11)].
Should signs of hypersensitivity present (wheal and flare reactions, pruritus, or other manifestations), or should disease exacerbations occur, discontinue DERMA-SMOOTHE/FS Scalp Oil immediately and institute appropriate therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity, genotoxicity, or fertility studies were conducted with DERMA-SMOOTHE/FS Scalp Oil. However, some corticosteroids are genotoxic in various genotoxicity tests (i.e., the in vitro human peripheral blood lymphocyte chromosome aberration assay with metabolic activation, the in vivo mouse bone marrow micronucleus assay, the Chinese hamster micronucleus test, and the in vitro mouse lymphoma gene mutation assay).
Structured Label Content
Section 42229-5 (42229-5)
DERMA-SMOOTHE/FS® Scalp Oil is indicated for the treatment of psoriasis of the scalp in adults.
Section 44425-7 (44425-7)
Storage: Keep tightly closed. Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature].
Section 59845-8 (59845-8)
Administration Instructions
Advise patients that DERMA-SMOOTHE/FS Scalp Oil is for topical use only [see Dosage and Administration (2)].
Instruct patients not to apply DERMA-SMOOTHE/FS Scalp Oil to the diaper area as diapers or plastic pants may constitute occlusive use [see Dosage and Administration (2)].
Advise patients to avoid use of DERMA-SMOOTHE/FS Scalp Oil on the face, axillae, or groin unless directed by their healthcare provider [see Dosage and Administration (2)].
Advise patients to discontinue therapy when control of disease is achieved. Instruct patients to contact their healthcare provider if no improvement is seen within 2 weeks [see Dosage and Administration (2)].
Endocrine System Adverse Reactions
Instruct patients not to use other corticosteroid-containing products while using DERMA-SMOOTHE/FS Scalp Oil without first consulting their healthcare provider [see Warnings and Precautions (5.1)].
Ophthalmic Adverse Reactions
Advise patients to avoid contact with the eyes and in case of contact, wash eyes liberally with water. Instruct patients to tell their healthcare provider if they develop any visual symptoms [see Warnings and Precautions (5.3)].
Pregnancy and Lactation
Advise women to use DERMA-SMOOTHE/FS Scalp Oil on the smallest area of skin and for the shortest duration possible while pregnant or breastfeeding. Advise patients that are breastfeeding not to apply DERMA-SMOOTHE/FS Scalp Oil directly to the nipple and areola to avoid direct infant exposure [See Use in Specific Populations (8.1 and 8.2)].
11 Description (11 DESCRIPTION)
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% contains fluocinolone acetonide [(6α, 11β, 16α)-6,9-difluoro-11,21-dihydroxy-16,17[(1–methylethylidene) bis(oxy)]-pregna-1,4-diene-3,20-dione, cyclic 16,17 acetal with acetone], a synthetic corticosteroid for topical dermatologic use. Chemically, fluocinolone acetonide is C24 H30 F2 O6. It has the following structural formula:
Fluocinolone acetonide has a molecular weight of 452.50. It is a white crystalline powder that is odorless, stable in light, and melts at 270°C with decomposition; soluble in alcohol, acetone and methanol; slightly soluble in chloroform; insoluble in water.
Each gram of DERMA-SMOOTHE/FS Scalp Oil contains approximately 0.11 mg of fluocinolone acetonide in a blend of oils, which contains isopropyl alcohol, isopropyl myristate, light mineral oil, oleth-2, refined peanut oil and fragrances.
Each packaged product contains 2 shower caps. The shower cap is made of low density polyethylene material with rubber elastic.
DERMA-SMOOTHE/FS Scalp Oil is formulated with 48% refined peanut oil. The bulk refined peanut oil used in DERMA-SMOOTHE/FS Scalp Oil is heated at 246°C (475°F) for at least 15 minutes. The refined peanut oil used in DERMA-SMOOTHE/FS Scalp Oil is routinely tested for peanut proteins through amino acid analysis; the quantity of amino acids is below 0.5 parts per million (ppm).
14 Clinical Studies (14 CLINICAL STUDIES)
In a vehicle-controlled study for the treatment of psoriasis of the scalp in adults, after 21 days of treatment, 60% of patients on active treatment and 21% of patients on the drug vehicle had excellent to cleared clinical response.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in more detail in other sections of the labeling:
• Endocrine System Adverse Reactions [see Warnings and Precautions (5.1)]
• Local Adverse Reactions [see Warnings and Precautions (5.2)]
• Ophthalmic Adverse Reactions [see Warnings and Precautions (5.3)]
12.2 Pharmacodynamics
Vasoconstrictor Assay
DERMA-SMOOTHE/FS Scalp Oil is in the low to medium range of potency as compared with other topical corticosteroids in vasoconstrictor studies. However, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
HPA axis suppression following administration of DERMA-SMOOTHE/FS Scalp Oil was not assessed.
12.3 Pharmacokinetics
Topical corticosteroids can be absorbed from intact healthy skin. The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the product formulation and the integrity of the epidermal barrier. Occlusion, inflammation and/or other disease processes in the skin may increase percutaneous absorption. The use of pharmacodynamic endpoints for assessing the systemic exposure of topical corticosteroids may be necessary due to the fact that circulating levels are often below the level of detection. Once absorbed through the skin, topical corticosteroids are metabolized, primarily in the liver, and are then excreted by the kidneys. Some corticosteroids and their metabolites are also excreted in the bile.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Derma-Smoothe/FS® Scalp Oil is a corticosteroid indicated for the treatment of psoriasis of the scalp in adults. (1)
Principal Display Panel
PRINCIPAL DISPLAY PANEL - 118.28 mL Carton Label
NDC 68791-102-04
Rx only
Derma-Smoothe/FS®
Fluocinolone Acetonide
0.01% Topical Oil
(SCALP OIL)
FOR TOPICAL USE ONLY
NOT FOR ORAL, OPHTHALMIC,
OR INTRAVAGINAL USE
SHAKE WELL BEFORE USE
Net Contents 118.28 mL
(4 Fl. oz.)
ROYAL
PHARMACEUTICALS®
PRINCIPAL DISPLAY PANEL - 20 mL Carton Physcian Sample Label
NOT FOR SALE
PHYSICIAN SAMPLE
NDC 68791-102-01
Rx only
Derma-Smoothe/FS®
Fluocinolone Acetonide 0.01% Topical Oil
(SCALP OIL)
FOR TOPICAL USE ONLY
NOT FOR ORAL OPHTHALMIC,
OR INTRAVAGINAL USE
SHAKE WELL BEFORE USE
NET Contents: 20 mL
Royal Pharmaceuticals®
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action in psoriasis of the scalp is unknown.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
Endocrine System Adverse Reactions:
o Topical corticosteroids can produce reversible HPA axis suppression, Cushing's syndrome, hyperglycemia, and glucosuria. (5.1)
o Systemic absorption may require evaluation for hypothalamic-pituitary-adrenal (HPA) axis suppression. Potent corticosteroids use on large areas, prolonged use or occlusive use, altered skin barrier, liver failure, and young age may increase systemic absorption. Modify use should HPA axis suppression develop. (5.1) - Local Adverse Reactions: Local adverse reactions may include atrophy, striae irritation, acneiform eruptions, hypopigmentation, and allergic contact dermatitis, and may be more likely with occlusive use or more potent corticosteroids. (5.2, 6.1)
- Ophthalmic Adverse Reactions: May increase the risks of glaucoma and posterior subcapsular cataract. Avoid contact of DERMA-SMOOTHE/FS Scalp Oil with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
DERMA-SMOOTHE/FS Scalp Oil is for topical use only. Not for oral, ophthalmic, or intravaginal use.
Wet or dampen hair and scalp thoroughly. Apply a thin film of DERMA-SMOOTHE/FS Scalp Oil on the scalp, massage well and cover scalp with the supplied shower cap. Leave on overnight or for a minimum of 4 hours then wash hair with regular shampoo and rinse thoroughly. Use daily as needed.
Discontinue DERMA-SMOOTHE/FS Scalp Oil when control of disease is achieved within 2 weeks, or contact the healthcare provider if no improvement is seen within 2 weeks.
Do not use DERMA-SMOOTHE/FS Scalp Oil on the face unless directed by the healthcare provider. Do not apply to intertriginous areas due to the increased risk of local adverse reactions [see Adverse Reactions (6)].
Do not apply to the diaper area; diapers or plastic pants may constitute occlusive use. [see Warnings and Precautions (5.1)]
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
DERMA-SMOOTHE/FS Scalp Oil is a topical oil containing 0.01% fluocinolone acetonide, supplied in bottles containing 4 fluid ounces and with 2 shower caps.
5.2 local Adverse Reactions (5.2 Local Adverse Reactions)
Local adverse reactions may occur with use of topical corticosteroids, including Derma-Smoothe/FS Scalp Oil, and may be more likely to occur with occlusive use, prolonged use or use of higher potency corticosteroids. Some local adverse reactions may be irreversible. Reactions may include atrophy, striae, telangiectasias, burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria [see Adverse Reactions (6.1)].
6.2 postmarketing Experience (6.2 Postmarketing Experience)
The following adverse reactions have been identified during post-approval use of products containing topical corticosteroids. Because postmarketing adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Endocrine Disorders: HPA axis suppression and Cushing’s syndrome
- Eye Disorders: glaucoma and cataracts
- Nervous System Disorders: intracranial hypertension including bulging fontanelles, headaches, and bilateral papilledema
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying condition, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
An open-label safety study was conducted in 29 pediatric subjects 3 months to 2 years old to assess the HPA axis by ACTH stimulation testing following use of the formulation of DERMA-SMOOTHE/FS Scalp Oil twice daily for 4 weeks. DERMA-SMOOTHE/FS Scalp Oil is not approved for use in pediatric patients for the treatment of psoriasis of the scalp. The most common adverse reactions were reported in the study:
| Adverse Reaction | n (%) |
|---|---|
| Cough | 6 (20) |
| Rhinorrhea | 4 (13) |
| Pyrexia | 3 (10) |
| Nasopharyngitis | 2 (7) |
| Hypopigmentation | 2 (7) |
| Abscess | 1 (3) |
| Atopic Dermatitis | 1 (3) |
| Eczema | 1 (3) |
| Hyperpigmentation | 1 (3) |
| Molluscum | 1 (3) |
| Rash | 1 (3) |
| Diarrhea | 1 (3) |
| Otitis Media | 1 (3) |
| URI | 1 (3) |
| Vomiting | 1 (3) |
5.4 allergic Contact Dermatitis (5.4 Allergic Contact Dermatitis)
Use of topical corticosteroids can cause allergic contact dermatitis. Allergic contact dermatitis to any component of topical corticosteroids is usually diagnosed by a failure to heal rather than a clinical exacerbation. Clinical diagnosis of allergic contact dermatitis can be confirmed by patch testing.
5.5 concomitant Skin Infections (5.5 Concomitant Skin Infections)
Use of topical corticosteroids may delay healing or worsen concomitant skin infections. Treat concomitant skin infections with an appropriate antimicrobial agent. If the infection persists unchanged, discontinue DERMA-SMOOTHE/FS Scalp Oil until the infection has been adequately treated.
5.3 ophthalmic Adverse Reactions (5.3 Ophthalmic Adverse Reactions)
Use of topical corticosteroids may increase the risks of glaucoma and posterior subcapsular cataract. Glaucoma and cataracts have been reported in postmarketing experience with the use of topical corticosteroid products. Avoid contact of DERMA-SMOOTHE/FS Scalp Oil with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
16 How Supplied / Storage and Handling (16 HOW SUPPLIED / STORAGE AND HANDLING)
DERMA-SMOOTHE/FS Scalp Oil (fluocinolone acetonide) topical oil, 0.01% (NDC # 68791-102-04) is supplied in bottles containing 4 fluid ounces and with 2 shower caps.
5.1 endocrine System Adverse Reactions (5.1 Endocrine System Adverse Reactions)
Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency. Cushing's syndrome, hyperglycemia, and glucosuria can result from systemic absorption of topical corticosteroids.
HPA axis suppression and Cushing's syndrome have been reported in patients receiving topical corticosteroids.
Conditions which increase systemic absorption include the use of more potent corticosteroids, use over large surface areas, use over prolonged periods, use of occlusive dressings, altered skin barrier, liver failure, and young age. Use of more than one corticosteroid-containing product at the same time may increase total systemic corticosteroid exposure. Because of the potential for systemic absorption, use of topical corticosteroids may require that patients be periodically evaluated for HPA axis suppression. The ACTH stimulation test may be helpful in evaluating patients for HPA axis suppression.
If HPA axis suppression is documented, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent corticosteroid. Manifestations of adrenal insufficiency may require supplemental systemic corticosteroids. Recovery of HPA axis function is generally prompt upon discontinuation of topical corticosteroids.
5.6 Use in Peanut Sensitive Individuals (5.6 Use in Peanut-Sensitive Individuals)
Use caution in prescribing DERMA-SMOOTHE/FS Scalp Oil for peanut-sensitive individuals [see Description (11)].
Should signs of hypersensitivity present (wheal and flare reactions, pruritus, or other manifestations), or should disease exacerbations occur, discontinue DERMA-SMOOTHE/FS Scalp Oil immediately and institute appropriate therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility (13.1 Carcinogenesis, mutagenesis, impairment of fertility)
No carcinogenicity, genotoxicity, or fertility studies were conducted with DERMA-SMOOTHE/FS Scalp Oil. However, some corticosteroids are genotoxic in various genotoxicity tests (i.e., the in vitro human peripheral blood lymphocyte chromosome aberration assay with metabolic activation, the in vivo mouse bone marrow micronucleus assay, the Chinese hamster micronucleus test, and the in vitro mouse lymphoma gene mutation assay).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:57.953441 · Updated: 2026-03-14T22:28:36.113572