These Highlights Do Not Include All The Information Needed To Use Dextrose Injection (5%) Safely And Effectively. See Full Prescribing Information For Dextrose Injection (5%).
10e98cb0-31a5-4ad1-3eaa-aa4af387a42d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.1 , 2.2 , 2.3 ) 06/2025 Contraindications ( 4 ) 06/2025 Warnings and Precautions ( 5.1 , 5.2 , 5.3 , 5.4 , 5.5 , 5.6 , 5.7 ) 06/2025
Indications and Usage
Dextrose Injection (5%) is indicated as a source of water and calories in adult and pediatric patients and may also be used as a diluent for reconstitution of a powder or liquid drug product.
Dosage and Administration
• Only for intravenous infusion. ( 2.1 ) • Infusion rate depends on the age, weight, clinical, and metabolic conditions of the patient and concomitant therapy. See full prescribing information for more information on preparation, administration, and dosing considerations. ( 2.1 , 2.2 , 2.3 )
Warnings and Precautions
• Neonatal Hypoglycemia : Closely monitor blood glucose concentration to ensure adequate glycemic control. ( 5.1 ) • Hyperglycemia and Hyperosmolar Hyperglycemic State : Use with caution in patients with known subclinical or overt diabetes mellitus. ( 5.2 ) • Hypersensitivity Reactions : Monitor for signs and symptoms and discontinue infusion immediately if reaction occurs. ( 5.3 ) • Phlebitis and Thrombosis : Remove catheter as soon as possible if thrombophlebitis develops. ( 2.1 , 5.4 ) • Hyponatremia : Monitor serum sodium and chloride concentrations, fluid status, acid-base balance, and neurologic status. ( 5.5 ) • Electrolyte Imbalance and Fluid Overload : Monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during administration. ( 5.6 ) • Refeeding Syndrome : Monitory laboratory parameters. ( 5.7 )
Contraindications
Dextrose Injection (5%) is contraindicated in patients with: • Clinically significant hyperglycemia [see Warnings and Precautions (5.2) ] . • Known hypersensitivity to dextrose [see Warnings and Precautions (5.3) ] .
Adverse Reactions
The following clinically significant adverse reactions are also described elsewhere in the labeling: • Neonatal Hypoglycemia [see Warnings and Precautions (5.1) ] • Hyperglycemia and Hyperosmolar Hyperglycemic State [see Warnings and Precautions (5.2) ] • Hypersensitivity Reactions [see Warnings and Precautions (5.3) ] • Phlebitis and Thrombosis [see Warnings and Precautions (5.4) ] • Hyponatremia [see Warnings and Precautions (5.5) ] • Electrolyte Imbalance, Fluid Overload and Hypervolemia [see Warnings and Precautions (5.6) ] • Refeeding Syndrome [see Warnings and Precautions (5.7) ] The following adverse reactions associated with the use of Dextrose Injection (5%) were identified in clinical trials or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Administration site conditions: blister, erythema, extravasation, pain, phlebitis, vein damage, thrombosis Immune system disorders: anaphylaxis, angioedema, bronchospasm, chills, hypotension, pruritus, pyrexia, rash Cardiovascular disorders: cyanosis, volume overload
Drug Interactions
Storage and Handling
Dextrose Injection, USP (5%) is a clear, colorless, sterile solution of dextrose supplied in single-dose flexible plastic ADD-Vantage TM diluent containers. This dosage form is intended to be used as a diluent for the contents of an ADD-Vantage vial or a single-dose powdered drug vial with a 20 mm closure using the ADD-Vantage ADDAPTOR TM . Unit of Sale Strength Volume NDC 0409-7100-66 Case of 50 – 50 mL bags 5% (2.5 g/50 mL) (50 mg/mL) 50 mL bags NDC 0409-7100-67 Case of 50 – 100 mL bags 5% (5 g/100 mL) (50 mg/mL) 100 mL bags NDC 0409-7100-02 Case of 24 – 250 mL bags 5% (12.5 g/250 mL) (50 mg/mL) 250 mL bags Do not remove the container from the overwrap until intended for use. Use the product immediately after the introduction of additives. Storage and Handling Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. Do not freeze. To contact Pfizer’s Medical Information Department, please visit www.pfizermedinfo.com or call 1‑800-438‑1985. Distributed by Hospira, Inc., Lake Forest, IL 60045 USA LAB-1143-4.0
How Supplied
Dextrose Injection, USP (5%) is a clear, colorless, sterile solution of dextrose supplied in single-dose flexible plastic ADD-Vantage TM diluent containers. This dosage form is intended to be used as a diluent for the contents of an ADD-Vantage vial or a single-dose powdered drug vial with a 20 mm closure using the ADD-Vantage ADDAPTOR TM . Unit of Sale Strength Volume NDC 0409-7100-66 Case of 50 – 50 mL bags 5% (2.5 g/50 mL) (50 mg/mL) 50 mL bags NDC 0409-7100-67 Case of 50 – 100 mL bags 5% (5 g/100 mL) (50 mg/mL) 100 mL bags NDC 0409-7100-02 Case of 24 – 250 mL bags 5% (12.5 g/250 mL) (50 mg/mL) 250 mL bags Do not remove the container from the overwrap until intended for use. Use the product immediately after the introduction of additives. Storage and Handling Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. Do not freeze. To contact Pfizer’s Medical Information Department, please visit www.pfizermedinfo.com or call 1‑800-438‑1985. Distributed by Hospira, Inc., Lake Forest, IL 60045 USA LAB-1143-4.0
Medication Information
Warnings and Precautions
• Neonatal Hypoglycemia : Closely monitor blood glucose concentration to ensure adequate glycemic control. ( 5.1 ) • Hyperglycemia and Hyperosmolar Hyperglycemic State : Use with caution in patients with known subclinical or overt diabetes mellitus. ( 5.2 ) • Hypersensitivity Reactions : Monitor for signs and symptoms and discontinue infusion immediately if reaction occurs. ( 5.3 ) • Phlebitis and Thrombosis : Remove catheter as soon as possible if thrombophlebitis develops. ( 2.1 , 5.4 ) • Hyponatremia : Monitor serum sodium and chloride concentrations, fluid status, acid-base balance, and neurologic status. ( 5.5 ) • Electrolyte Imbalance and Fluid Overload : Monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during administration. ( 5.6 ) • Refeeding Syndrome : Monitory laboratory parameters. ( 5.7 )
Indications and Usage
Dextrose Injection (5%) is indicated as a source of water and calories in adult and pediatric patients and may also be used as a diluent for reconstitution of a powder or liquid drug product.
Dosage and Administration
• Only for intravenous infusion. ( 2.1 ) • Infusion rate depends on the age, weight, clinical, and metabolic conditions of the patient and concomitant therapy. See full prescribing information for more information on preparation, administration, and dosing considerations. ( 2.1 , 2.2 , 2.3 )
Contraindications
Dextrose Injection (5%) is contraindicated in patients with: • Clinically significant hyperglycemia [see Warnings and Precautions (5.2) ] . • Known hypersensitivity to dextrose [see Warnings and Precautions (5.3) ] .
Adverse Reactions
The following clinically significant adverse reactions are also described elsewhere in the labeling: • Neonatal Hypoglycemia [see Warnings and Precautions (5.1) ] • Hyperglycemia and Hyperosmolar Hyperglycemic State [see Warnings and Precautions (5.2) ] • Hypersensitivity Reactions [see Warnings and Precautions (5.3) ] • Phlebitis and Thrombosis [see Warnings and Precautions (5.4) ] • Hyponatremia [see Warnings and Precautions (5.5) ] • Electrolyte Imbalance, Fluid Overload and Hypervolemia [see Warnings and Precautions (5.6) ] • Refeeding Syndrome [see Warnings and Precautions (5.7) ] The following adverse reactions associated with the use of Dextrose Injection (5%) were identified in clinical trials or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Administration site conditions: blister, erythema, extravasation, pain, phlebitis, vein damage, thrombosis Immune system disorders: anaphylaxis, angioedema, bronchospasm, chills, hypotension, pruritus, pyrexia, rash Cardiovascular disorders: cyanosis, volume overload
Drug Interactions
Storage and Handling
Dextrose Injection, USP (5%) is a clear, colorless, sterile solution of dextrose supplied in single-dose flexible plastic ADD-Vantage TM diluent containers. This dosage form is intended to be used as a diluent for the contents of an ADD-Vantage vial or a single-dose powdered drug vial with a 20 mm closure using the ADD-Vantage ADDAPTOR TM . Unit of Sale Strength Volume NDC 0409-7100-66 Case of 50 – 50 mL bags 5% (2.5 g/50 mL) (50 mg/mL) 50 mL bags NDC 0409-7100-67 Case of 50 – 100 mL bags 5% (5 g/100 mL) (50 mg/mL) 100 mL bags NDC 0409-7100-02 Case of 24 – 250 mL bags 5% (12.5 g/250 mL) (50 mg/mL) 250 mL bags Do not remove the container from the overwrap until intended for use. Use the product immediately after the introduction of additives. Storage and Handling Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. Do not freeze. To contact Pfizer’s Medical Information Department, please visit www.pfizermedinfo.com or call 1‑800-438‑1985. Distributed by Hospira, Inc., Lake Forest, IL 60045 USA LAB-1143-4.0
How Supplied
Dextrose Injection, USP (5%) is a clear, colorless, sterile solution of dextrose supplied in single-dose flexible plastic ADD-Vantage TM diluent containers. This dosage form is intended to be used as a diluent for the contents of an ADD-Vantage vial or a single-dose powdered drug vial with a 20 mm closure using the ADD-Vantage ADDAPTOR TM . Unit of Sale Strength Volume NDC 0409-7100-66 Case of 50 – 50 mL bags 5% (2.5 g/50 mL) (50 mg/mL) 50 mL bags NDC 0409-7100-67 Case of 50 – 100 mL bags 5% (5 g/100 mL) (50 mg/mL) 100 mL bags NDC 0409-7100-02 Case of 24 – 250 mL bags 5% (12.5 g/250 mL) (50 mg/mL) 250 mL bags Do not remove the container from the overwrap until intended for use. Use the product immediately after the introduction of additives. Storage and Handling Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. Do not freeze. To contact Pfizer’s Medical Information Department, please visit www.pfizermedinfo.com or call 1‑800-438‑1985. Distributed by Hospira, Inc., Lake Forest, IL 60045 USA LAB-1143-4.0
Description
Dosage and Administration ( 2.1 , 2.2 , 2.3 ) 06/2025 Contraindications ( 4 ) 06/2025 Warnings and Precautions ( 5.1 , 5.2 , 5.3 , 5.4 , 5.5 , 5.6 , 5.7 ) 06/2025
Section 43683-2
10 Overdosage
A medication error resulting in a high infusion rate of Dextrose Injection (5%) can cause hyperglycemia, hyperosmolality, and adverse effects on fluid and electrolyte balance [see Warnings and Precautions (5.2, 5.6)].
Severe hyperglycemia and severe dilutional hyponatremia, and their complications, can be fatal. In the event of overdosage (overhydration or solute overload) during Dextrose Injection (5%) treatment, discontinue the infusion. Institute corrective measures such as administration of exogenous insulin, and treat adverse effects on the CNS, respiratory, and cardiovascular systems [see Warnings and Precautions (5.2, 5.6)].
8.1 Pregnancy
Risk Summary
Dextrose Injection (5%) has been used for decades during labor and delivery. Although there are a few case reports that describe adverse effects of dextrose use in other stages of pregnancy, exposure during pregnancy in general is not expected to cause major birth defects, miscarriage, or adverse maternal or fetal outcomes. Animal reproduction studies have not been conducted with dextrose.
The background risk of major birth defects and miscarriage for the indicated populations are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
Risk Summary
Dextrose Injection (5%) has been used for decades and is not expected to cause harm to a breastfed infant. There are no data on the effects of Dextrose Injection (5%) on levels of glucose in human milk, on the breastfed infant, or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Dextrose Injection (5%) and any potential adverse effects on the breastfed infant from Dextrose Injection (5%) or from the underlying maternal condition.
11 Description
Dextrose, USP is chemically designated D-glucose, monohydrate (C6H12O6 • H2O), a hexose sugar freely soluble in water.
The molecular weight of dextrose (D-glucose) monohydrate is 198.17. It has the following structural formula:
Water for Injection, USP is chemically designated H2O.
Dextrose Injection, USP (5%) is a clear, sterile, non-pyrogenic solutions of Dextrose, USP in Water for Injection in a polyvinylchloride flexible plastic container for intravenous administration after admixture with a single-dose powdered or liquid (up to 10 mL) drug vial [see Dosage and Administration (2.1)].
Partial-fill containers, designed to facilitate admixture, are available in 50 mL, 100 mL, and 250 mL sizes. See Table 1 for the content and characteristics of this solution.
The solution contains no bacteriostatic, antimicrobial agent or added buffer and is supplied as single-dose containers. The pH is 4.3 (range is 3.2 to 6.5).
|
Strength |
Fill Volume |
Amount of Dextrose Hydrous per Container |
kcal Caloric value calculated on the basis of 3.4 kcal/g of dextrose, hydrous
per Container
|
mOsmol per Liter |
|
5% Dextrose |
50 mL |
2.5 grams |
8.5 |
253 |
|
100 mL |
5 grams |
17 |
253 |
|
|
250 mL |
12.5 grams |
42.5 |
253 |
Dextrose is derived from corn.
Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
5.5 Hyponatremia
Dextrose Injection (5%) may cause hyponatremia. Hyponatremia can lead to acute hyponatremic encephalopathy characterized by headache, nausea, seizures, lethargy, and vomiting. The risk of hospital-acquired hyponatremia is increased in younger pediatric patients, geriatric patients, patients treated with diuretics, and patients with cardiac or pulmonary failure or with the syndrome of inappropriate antidiuretic hormone (SIADH) (e.g., postoperative patients, patients concomitantly treated with arginine vasopressin analogs or certain antiepileptic, psychotropic, and cytotoxic drugs) [see Drug Interactions (7.1), Use in Specific Populations (8.4)].
Avoid Dextrose Injection (5%) in patients with or at risk for hyponatremia. If use cannot be avoided, closely monitor serum sodium concentrations, chloride concentrations, fluid status, acid-base balance, and neurologic status [see Warnings and Precautions (5.6)].
8.4 Pediatric Use
Dextrose Injection (5%) is indicated in pediatric patients as a source of water and calories, and may also be used as a diluent for reconstitution of a powder or liquid drug product.
Neonates, especially preterm neonates with low birth weight, are at increased risk of developing hypo- or hyperglycemia. Therefore, they need close monitoring during treatment with intravenous glucose infusions to ensure adequate glycemic control in order to avoid potential long-term adverse effects.
Dextrose Injection (5%) can cause imbalances in fluid and electrolytes in pediatric patients and requires close monitoring of volume status and plasma electrolyte concentrations, particularly in pediatric patients who may have impaired ability to regulate fluids and electrolytes. Pediatric patients are at increased risk for developing hyponatremic encephalopathy [see Warnings and Precautions (5.5, 5.6)].
In very low birth weight neonates, excessive or rapid administration of Dextrose Injection (5%) may increase the risk of intracerebral hemorrhage.
8.5 Geriatric Use
Dextrose Injection (5%) has not been studied in sufficient number of patients aged 65 and over to determine whether they respond differently from younger patients. Geriatric patients are at increased risk of developing hyponatremia and hyponatremic encephalopathy [see Warnings and Precautions (5.5)]. Other reported clinical experience has not identified differences in responses between the geriatric and younger adult patients. In general, the infusion rate for geriatric patients should start slow and be titrated up cautiously, reflecting their greater risk for electrolyte abnormalities and fluid overload.
Dextrose is known to be substantially excreted by the kidney, and the risk of adverse reactions to Dextrose Injection (5%) may be greater in patients with impaired renal function. Because geriatric patients are more likely to have impaired renal function, care should be taken in selection of the infusion rate, and patients should be closely monitored during Dextrose Injection (5%) treatment.
4 Contraindications
Dextrose Injection (5%) is contraindicated in patients with:
-
•Clinically significant hyperglycemia [see Warnings and Precautions (5.2)].
-
•Known hypersensitivity to dextrose [see Warnings and Precautions (5.3)].
6 Adverse Reactions
The following clinically significant adverse reactions are also described elsewhere in the labeling:
-
•Neonatal Hypoglycemia [see Warnings and Precautions (5.1)]
-
•Hyperglycemia and Hyperosmolar Hyperglycemic State [see Warnings and Precautions (5.2)]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
-
•Phlebitis and Thrombosis [see Warnings and Precautions (5.4)]
-
•Hyponatremia [see Warnings and Precautions (5.5)]
-
•Electrolyte Imbalance, Fluid Overload and Hypervolemia [see Warnings and Precautions (5.6)]
-
•Refeeding Syndrome [see Warnings and Precautions (5.7)]
The following adverse reactions associated with the use of Dextrose Injection (5%) were identified in clinical trials or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Administration site conditions: blister, erythema, extravasation, pain, phlebitis, vein damage, thrombosis
Immune system disorders: anaphylaxis, angioedema, bronchospasm, chills, hypotension, pruritus, pyrexia, rash
Cardiovascular disorders: cyanosis, volume overload
7 Drug Interactions
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of dextrose have not been fully characterized.
12.3 Pharmacokinetics
Dextrose is oxidized to carbon dioxide and water.
5.7 Refeeding Syndrome
Refeeding severely undernourished patients may result in refeeding syndrome, characterized by the intracellular shift of potassium, phosphorus, and magnesium as the patient becomes anabolic. Thiamine deficiency and fluid retention may also develop. To prevent these complications, monitor severely undernourished patients and slowly increase nutrient intake.
1 Indications and Usage
Dextrose Injection (5%) is indicated as a source of water and calories in adult and pediatric patients and may also be used as a diluent for reconstitution of a powder or liquid drug product.
12 Clinical Pharmacology
12.1 Mechanism of Action
Dextrose provides a source of carbohydrate calories and is used to supplement nutrition by providing glucose parenterally.
2.3 Dosage Considerations
The choice of dextrose concentration, rate, and volume depends on the age, weight, clinical, and metabolic conditions of the patient and concomitant therapy.
Dextrose Injection (5%) administration rate should be based on the patient’s tolerance of dextrose, especially for preterm neonates with low birth weight.
Increase the infusion rate gradually as needed; frequently monitor blood glucose concentrations to avoid hyperglycemia [see Warnings and Precautions (5.2), Use in Specific Populations (8.4)].
5.1 Neonatal Hypoglycemia
Neonates, especially preterm neonates with low birth weight, are at increased risk of developing hypoglycemia. Closely monitor blood glucose concentration during treatment with Dextrose Injection (5%) to ensure adequate glycemic control in order to avoid potential long-term adverse effects.
5 Warnings and Precautions
-
•Neonatal Hypoglycemia: Closely monitor blood glucose concentration to ensure adequate glycemic control. (5.1)
-
•Hyperglycemia and Hyperosmolar Hyperglycemic State: Use with caution in patients with known subclinical or overt diabetes mellitus. (5.2)
-
•Hypersensitivity Reactions: Monitor for signs and symptoms and discontinue infusion immediately if reaction occurs. (5.3)
-
•Phlebitis and Thrombosis: Remove catheter as soon as possible if thrombophlebitis develops. (2.1, 5.4)
-
•Hyponatremia: Monitor serum sodium and chloride concentrations, fluid status, acid-base balance, and neurologic status. (5.5)
-
•Electrolyte Imbalance and Fluid Overload: Monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during administration. (5.6)
-
•Refeeding Syndrome: Monitory laboratory parameters. (5.7)
2 Dosage and Administration
3 Dosage Forms and Strengths
Injection:
-
•5% (5 g/100 mL) (50 mg/mL) of dextrose hydrous in a clear, sterile, and nonpyrogenic solution in single-dose flexible plastic ADD-VantageTM diluent containers: 50 mL, 100 mL and 250 mL.
This dextrose product is intended to be used as a diluent for the contents of an ADD- Vantage vial or a single-dose powdered drug vial with 20 mm closure using the ADD-Vantage ADDAPTORTM.
5.4 Phlebitis and Thrombosis
The infusion of hypertonic solutions into a peripheral vein may result in vein irritation, vein damage, and/or thrombosis [see Dosage and Administration (2.1)]. If thrombophlebitis develops, remove the catheter as soon as possible.
8 Use in Specific Populations
Pediatric Use: Increased risk of hypoglycemia/hyperglycemia and imbalances in fluid/electrolytes; monitor serum glucose concentrations, volume status, and electrolytes. (8.4)
5.3 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis, have been reported with Dextrose Injection (5%) administration [see Adverse Reactions (6)]. Stop administration of Dextrose Injection (5%) immediately if signs or symptoms of a hypersensitivity reaction develop. Initiate appropriate treatment as clinically indicated.
16 How Supplied/storage and Handling
Dextrose Injection, USP (5%) is a clear, colorless, sterile solution of dextrose supplied in single-dose flexible plastic ADD-VantageTM diluent containers. This dosage form is intended to be used as a diluent for the contents of an ADD-Vantage vial or a single-dose powdered drug vial with a 20 mm closure using the ADD-Vantage ADDAPTORTM.
|
Unit of Sale |
Strength |
Volume |
|
NDC 0409-7100-66
|
5% (2.5 g/50 mL) (50 mg/mL) |
50 mL bags |
|
NDC 0409-7100-67
|
5% (5 g/100 mL) (50 mg/mL) |
100 mL bags |
|
NDC 0409-7100-02
|
5% (12.5 g/250 mL) (50 mg/mL) |
250 mL bags |
Do not remove the container from the overwrap until intended for use.
Use the product immediately after the introduction of additives.
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
Do not freeze.
To contact Pfizer’s Medical Information Department, please visit www.pfizermedinfo.com or call 1‑800-438‑1985.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
LAB-1143-4.0
2.2 Important Preparation Information
Visually inspect the Dextrose Injection (5%) for particulate matter and discoloration. Do not administer Dextrose Injection (5%) if the solution is cloudy, there are precipitates, and the container is damaged.
To reduce the risk of air embolism, adhere to the following preparation instructions for Dextrose Injection (5%):
-
•Use a non-vented infusion set or close the vent on a vented set.
-
•Use a dedicated line without any connections (do not connect flexible containers in series).
-
•Do not pressurize the flexible container to increase flow rates.
-
•If using a pumping device to administer Dextrose Injection (5%), turn off the pump before the container is empty.
To Open:
-
•Peel overwrap at corner and remove solution container.
-
oUse unit within 30 days of opening overwrap, as long as the use date does not exceed the printed expiration date.
-
-
•Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
To Assemble Vial and Flexible Diluent Container:
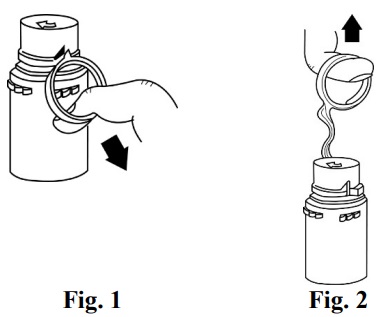
NOTE: Once the breakaway cap has been removed, do not access the vial with the syringe. |
|
|
|
NOTE: Once vial is seated, do not attempt to remove. (SEE FIGURE 4.)
|
|
|
To Reconstitute the Drug:
If the rubber stopper is not removed from the vial, and the medication is not released on the first attempt, the inner cap may be manipulated back into the rubber stopper without removing the drug vial from the diluent container. Repeat steps 3 through 5. |
Preparation for Administration:
-
1.Confirm the activation and admixture of vial contents.
-
2.Check for leaks by squeezing the container firmly. If leaks are found, discard the unit because sterility may be compromised.
-
3.Close the flow control clamp of the administration set.
-
4.Remove the cover from the outlet port at the bottom of the container.
-
5.Insert the piercing pin of the administration set into the port with a twisting motion until the pin is firmly seated. Note: See full directions on the administration set carton.
-
6.Lift the free end of the hanger loop on the bottom of the vial, breaking the two tie strings. Bend the loop outward to lock it in the upright position, then suspend the container from the hanger.
-
7.Squeeze and release the drip chamber to establish the proper fluid level in the chamber.
-
8.Open the flow control clamp and clear air from set. Close clamp.
-
9.Attach the set to the venipuncture device. If the device is not indwelling, prime and make venipuncture.
-
10.Regulate the rate of administration with the flow control clamp. Do not use the flexible container in series connections.
2.1 Important Administration Instructions
Dextrose Injection (5%) is intended for intravenous use.
-
•Use a peripheral vein to administer if the final dextrose concentration is 5% or less and the osmolarity is less than 900 mOsm/L.
-
•To avoid venous irritation, consider using a central vein to administer hypertonic solutions with osmolarity of 900 mOsm/L or greater [see Warnings and Precautions (5.4)].
-
•Do not administer Dextrose Injection (5%) simultaneously with blood products through the same administration set because of the possibility of pseudoagglutination or hemolysis.
-
•Use of a final filter is recommended during administration of parenteral solutions, where possible.
-
•Discard the unused portion.
-
•Do not use the Dextrose Injection (5%) ADD-VantageTM diluent containers with chemotherapy agents.
Principal Display Panel 50 Ml Bag Label
PULL INNER PLUG/STOPPER
AND MIX DRUG BEFORE USE
ADD-Vantage™ Unit
NDC 0409-7100-68
5% DEXTROSE
Injection, USP
50
mL
EACH 100 mL CONTAINS DEXTROSE, HYDROUS 5 g.
253 mOsmol/LITER (calc.). pH 4.3 (3.2 TO 6.5)
DEXTROSE SOLUTIONS WITHOUT SALTS SHOULD
NOT BE USED IN BLOOD TRANSFUSIONS BECAUSE
OF POSSIBLE ROULEAU FORMATION. FOR USE ONLY
WITH ADD-Vantage™ SYSTEM COMPONENTS. SINGLE-
DOSE CONTAINER. FOR IV USE. USUAL DOSAGE: SEE
PACKAGE INSERT. STERILE, NONPYROGENIC. USE ONLY
IF SOLUTION IS CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
DISTRIBUTED BY
HOSPIRA, INC.,
LAKE FOREST, IL 60045 USA
Rx ONLY
IM-4480
14475201
3
v
CONTAINS DEHP
Principal Display Panel 100 Ml Bag Label
PULL INNER PLUG/STOPPER
AND MIX DRUG BEFORE USE
ADD-Vantage™ Unit
NDC 0409-7100-69
5% DEXTROSE
Injection, USP
100
mL
EACH 100 mL CONTAINS DEXTROSE, HYDROUS 5 g.
253 mOsmol/LITER (calc.). pH 4.3 (3.2 TO 6.5)
DEXTROSE SOLUTIONS WITHOUT SALTS SHOULD
NOT BE USED IN BLOOD TRANSFUSIONS BECAUSE OF
POSSIBLE ROULEAU FORMATION. FOR USE ONLY WITH
ADD-Vantage™ SYSTEM COMPONENTS. SINGLE-DOSE
CONTAINER. FOR IV USE. USUAL DOSAGE: SEE PACKAGE
INSERT. STERILE, NONPYROGENIC. USE ONLY IF SOLUTION
IS CLEAR AND CONTAINER IS UNDAMAGED. MUST NOT
BE USED IN SERIES CONNECTIONS.
DISTRIBUTED BY
HOSPIRA, INC.,
LAKE FOREST, IL 60045 USA
Rx ONLY
IM-4481
14475601
3
v
CONTAINS DEHP
Principal Display Panel 250 Ml Bag Label
LOT
EXP.
PULL INNER PLUG/STOPPER
AND MIX DRUG BEFORE USE
ADD-Vantage™ Unit
NDC 0409-7100-04
5% DEXTROSE
Injection, USP
250
mL
EACH 100 mL CONTAINS
DEXTROSE, HYDROUS 5 g.
253 mOsmol/LITER (calc).
pH 4.3 (3.2 TO 6.5)
DEXTROSE SOLUTIONS
WITHOUT SALTS SHOULD
NOT BE USED IN BLOOD
TRANSFUSIONS BECAUSE
OF POSSIBLE ROULEAU
FORMATION. FOR USE ONLY
WITH ADD-Vantage™ SYSTEM
COMPONENTS. SINGLE-
DOSE CONTAINER. FOR
INTRAVENOUS USE. USUAL
DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY
IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES
CONNECTIONS.
Rx ONLY
DISTRIBUTED BY
HOSPIRA, INC.,
LAKE FOREST, IL 60045 USA
Hospira
3
v
CONTAINS DEHP
IM-4482
14475801
5.6 Electrolyte Imbalance and Fluid Overload
Electrolyte deficits, particularly serum potassium and phosphate, may occur during prolonged use of Dextrose Injection (5%).
Depending on the administered volume and the infusion rate, Dextrose Injection (5%) can cause fluid overload, including pulmonary edema.
Avoid Dextrose Injection (5%) in patients at risk for fluid and/or solute overload. If use cannot be avoided in these patients, monitor fluid balance, electrolyte concentrations, and acid-base balance, especially during prolonged use. Additional monitoring is recommended for patients with water and electrolyte disturbances that could be aggravated by increased glucose, insulin administration, and/or free water load.
Principal Display Panel Overwrap – Wr 0581
TO OPEN – PEEL AT NOTCH
Five/ADD-Vantage™ Units
For use only with ADD-Vantage™ system components.
The overwrap is a moisture barrier. Use units within 30 days of opening overwrap, as long as the use
date does not exceed the printed expiration date. After removing the overwrap, check for minute
leaks by squeezing container firmly. If leaks are found, discard unit as sterility may be
impaired. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]
Rx only
14475301
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
WR-0581
Principal Display Panel Overwrap – Wr 0583
One/ADD-Vantage™ Unit
TO OPEN–PEEL AT CORNER
For use only with ADD-Vantage™ system components.
The overwrap is a moisture barrier. Use unit within 30 days of opening
overwrap, as long as the use date does not exceed the printed
expiration date. Store at 20 to 25°C (68 to 77°F). [See USP Controlled
Room Temperature.] Protect from freezing. See insert.
After removing the overwrap, check for minute leaks by
squeezing container firmly. If leaks are found, discard
unit as sterility may be impaired.
Rx only
14475901
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
WR-0583
5.2 Hyperglycemia and Hyperosmolar Hyperglycemic State
The use of Dextrose Injection (5%) in patients with impaired glucose tolerance may worsen hyperglycemia. Administration of dextrose at a rate exceeding the patient’s utilization rate may lead to hyperglycemia, coma, and death.
Hyperglycemia is associated with an increase in serum osmolality, resulting in osmotic diuresis, dehydration, and electrolyte losses [see Warnings and Precautions (5.6), Use in Specific Populations (8.4)]. Patients with underlying CNS disease and renal impairment who receive dextrose infusions may be at greater risk of developing hyperosmolar hyperglycemic state.
Monitor blood glucose levels and treat hyperglycemia to maintain levels within normal limits while administering Dextrose Injection (5%). Insulin may be administered or adjusted to maintain optimal blood glucose levels during Dextrose Injection (5%) administration.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies with dextrose to evaluate the drug’s carcinogenic potential, mutagenic potential, or effects on fertility have not been performed.
7.1 Drugs With Effects On Glycemic Control and Electrolyte Balance
Dextrose Injection (5%) can affect glycemic control, vasopressin, and fluid and/or electrolyte balance [see Warnings and Precautions (5.1, 5.2, 5.5, 5.6)]. Monitor patients’ blood glucose concentrations, fluid balance, serum electrolyte concentrations, and acid-base balance.
Concomitant administration of Dextrose Injection (5%) with drugs associated with hyponatremia may increase the risk of developing hyponatremia. Drugs associated with hyponatremia include diuretics and those that cause SIADH (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), arginine vasopressin analogs, certain antiepileptic, psychotropic, and cytotoxic drugs). Avoid use of Dextrose Injection (5%) in patients receiving drugs associated with hyponatremia. If use cannot be avoided, closely monitor serum sodium concentrations during concomitant use [see Warnings and Precautions (5.5)].
Structured Label Content
Section 43683-2 (43683-2)
10 Overdosage (10 OVERDOSAGE)
A medication error resulting in a high infusion rate of Dextrose Injection (5%) can cause hyperglycemia, hyperosmolality, and adverse effects on fluid and electrolyte balance [see Warnings and Precautions (5.2, 5.6)].
Severe hyperglycemia and severe dilutional hyponatremia, and their complications, can be fatal. In the event of overdosage (overhydration or solute overload) during Dextrose Injection (5%) treatment, discontinue the infusion. Institute corrective measures such as administration of exogenous insulin, and treat adverse effects on the CNS, respiratory, and cardiovascular systems [see Warnings and Precautions (5.2, 5.6)].
8.1 Pregnancy
Risk Summary
Dextrose Injection (5%) has been used for decades during labor and delivery. Although there are a few case reports that describe adverse effects of dextrose use in other stages of pregnancy, exposure during pregnancy in general is not expected to cause major birth defects, miscarriage, or adverse maternal or fetal outcomes. Animal reproduction studies have not been conducted with dextrose.
The background risk of major birth defects and miscarriage for the indicated populations are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
Risk Summary
Dextrose Injection (5%) has been used for decades and is not expected to cause harm to a breastfed infant. There are no data on the effects of Dextrose Injection (5%) on levels of glucose in human milk, on the breastfed infant, or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Dextrose Injection (5%) and any potential adverse effects on the breastfed infant from Dextrose Injection (5%) or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Dextrose, USP is chemically designated D-glucose, monohydrate (C6H12O6 • H2O), a hexose sugar freely soluble in water.
The molecular weight of dextrose (D-glucose) monohydrate is 198.17. It has the following structural formula:
Water for Injection, USP is chemically designated H2O.
Dextrose Injection, USP (5%) is a clear, sterile, non-pyrogenic solutions of Dextrose, USP in Water for Injection in a polyvinylchloride flexible plastic container for intravenous administration after admixture with a single-dose powdered or liquid (up to 10 mL) drug vial [see Dosage and Administration (2.1)].
Partial-fill containers, designed to facilitate admixture, are available in 50 mL, 100 mL, and 250 mL sizes. See Table 1 for the content and characteristics of this solution.
The solution contains no bacteriostatic, antimicrobial agent or added buffer and is supplied as single-dose containers. The pH is 4.3 (range is 3.2 to 6.5).
|
Strength |
Fill Volume |
Amount of Dextrose Hydrous per Container |
kcal Caloric value calculated on the basis of 3.4 kcal/g of dextrose, hydrous
per Container
|
mOsmol per Liter |
|
5% Dextrose |
50 mL |
2.5 grams |
8.5 |
253 |
|
100 mL |
5 grams |
17 |
253 |
|
|
250 mL |
12.5 grams |
42.5 |
253 |
Dextrose is derived from corn.
Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
5.5 Hyponatremia
Dextrose Injection (5%) may cause hyponatremia. Hyponatremia can lead to acute hyponatremic encephalopathy characterized by headache, nausea, seizures, lethargy, and vomiting. The risk of hospital-acquired hyponatremia is increased in younger pediatric patients, geriatric patients, patients treated with diuretics, and patients with cardiac or pulmonary failure or with the syndrome of inappropriate antidiuretic hormone (SIADH) (e.g., postoperative patients, patients concomitantly treated with arginine vasopressin analogs or certain antiepileptic, psychotropic, and cytotoxic drugs) [see Drug Interactions (7.1), Use in Specific Populations (8.4)].
Avoid Dextrose Injection (5%) in patients with or at risk for hyponatremia. If use cannot be avoided, closely monitor serum sodium concentrations, chloride concentrations, fluid status, acid-base balance, and neurologic status [see Warnings and Precautions (5.6)].
8.4 Pediatric Use
Dextrose Injection (5%) is indicated in pediatric patients as a source of water and calories, and may also be used as a diluent for reconstitution of a powder or liquid drug product.
Neonates, especially preterm neonates with low birth weight, are at increased risk of developing hypo- or hyperglycemia. Therefore, they need close monitoring during treatment with intravenous glucose infusions to ensure adequate glycemic control in order to avoid potential long-term adverse effects.
Dextrose Injection (5%) can cause imbalances in fluid and electrolytes in pediatric patients and requires close monitoring of volume status and plasma electrolyte concentrations, particularly in pediatric patients who may have impaired ability to regulate fluids and electrolytes. Pediatric patients are at increased risk for developing hyponatremic encephalopathy [see Warnings and Precautions (5.5, 5.6)].
In very low birth weight neonates, excessive or rapid administration of Dextrose Injection (5%) may increase the risk of intracerebral hemorrhage.
8.5 Geriatric Use
Dextrose Injection (5%) has not been studied in sufficient number of patients aged 65 and over to determine whether they respond differently from younger patients. Geriatric patients are at increased risk of developing hyponatremia and hyponatremic encephalopathy [see Warnings and Precautions (5.5)]. Other reported clinical experience has not identified differences in responses between the geriatric and younger adult patients. In general, the infusion rate for geriatric patients should start slow and be titrated up cautiously, reflecting their greater risk for electrolyte abnormalities and fluid overload.
Dextrose is known to be substantially excreted by the kidney, and the risk of adverse reactions to Dextrose Injection (5%) may be greater in patients with impaired renal function. Because geriatric patients are more likely to have impaired renal function, care should be taken in selection of the infusion rate, and patients should be closely monitored during Dextrose Injection (5%) treatment.
4 Contraindications (4 CONTRAINDICATIONS)
Dextrose Injection (5%) is contraindicated in patients with:
-
•Clinically significant hyperglycemia [see Warnings and Precautions (5.2)].
-
•Known hypersensitivity to dextrose [see Warnings and Precautions (5.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are also described elsewhere in the labeling:
-
•Neonatal Hypoglycemia [see Warnings and Precautions (5.1)]
-
•Hyperglycemia and Hyperosmolar Hyperglycemic State [see Warnings and Precautions (5.2)]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
-
•Phlebitis and Thrombosis [see Warnings and Precautions (5.4)]
-
•Hyponatremia [see Warnings and Precautions (5.5)]
-
•Electrolyte Imbalance, Fluid Overload and Hypervolemia [see Warnings and Precautions (5.6)]
-
•Refeeding Syndrome [see Warnings and Precautions (5.7)]
The following adverse reactions associated with the use of Dextrose Injection (5%) were identified in clinical trials or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Administration site conditions: blister, erythema, extravasation, pain, phlebitis, vein damage, thrombosis
Immune system disorders: anaphylaxis, angioedema, bronchospasm, chills, hypotension, pruritus, pyrexia, rash
Cardiovascular disorders: cyanosis, volume overload
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of dextrose have not been fully characterized.
12.3 Pharmacokinetics
Dextrose is oxidized to carbon dioxide and water.
5.7 Refeeding Syndrome
Refeeding severely undernourished patients may result in refeeding syndrome, characterized by the intracellular shift of potassium, phosphorus, and magnesium as the patient becomes anabolic. Thiamine deficiency and fluid retention may also develop. To prevent these complications, monitor severely undernourished patients and slowly increase nutrient intake.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Dextrose Injection (5%) is indicated as a source of water and calories in adult and pediatric patients and may also be used as a diluent for reconstitution of a powder or liquid drug product.
12 Clinical Pharmacology (12 CLINICAL PHARMACOLOGY)
12.1 Mechanism of Action
Dextrose provides a source of carbohydrate calories and is used to supplement nutrition by providing glucose parenterally.
2.3 Dosage Considerations
The choice of dextrose concentration, rate, and volume depends on the age, weight, clinical, and metabolic conditions of the patient and concomitant therapy.
Dextrose Injection (5%) administration rate should be based on the patient’s tolerance of dextrose, especially for preterm neonates with low birth weight.
Increase the infusion rate gradually as needed; frequently monitor blood glucose concentrations to avoid hyperglycemia [see Warnings and Precautions (5.2), Use in Specific Populations (8.4)].
5.1 Neonatal Hypoglycemia
Neonates, especially preterm neonates with low birth weight, are at increased risk of developing hypoglycemia. Closely monitor blood glucose concentration during treatment with Dextrose Injection (5%) to ensure adequate glycemic control in order to avoid potential long-term adverse effects.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Neonatal Hypoglycemia: Closely monitor blood glucose concentration to ensure adequate glycemic control. (5.1)
-
•Hyperglycemia and Hyperosmolar Hyperglycemic State: Use with caution in patients with known subclinical or overt diabetes mellitus. (5.2)
-
•Hypersensitivity Reactions: Monitor for signs and symptoms and discontinue infusion immediately if reaction occurs. (5.3)
-
•Phlebitis and Thrombosis: Remove catheter as soon as possible if thrombophlebitis develops. (2.1, 5.4)
-
•Hyponatremia: Monitor serum sodium and chloride concentrations, fluid status, acid-base balance, and neurologic status. (5.5)
-
•Electrolyte Imbalance and Fluid Overload: Monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during administration. (5.6)
-
•Refeeding Syndrome: Monitory laboratory parameters. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection:
-
•5% (5 g/100 mL) (50 mg/mL) of dextrose hydrous in a clear, sterile, and nonpyrogenic solution in single-dose flexible plastic ADD-VantageTM diluent containers: 50 mL, 100 mL and 250 mL.
This dextrose product is intended to be used as a diluent for the contents of an ADD- Vantage vial or a single-dose powdered drug vial with 20 mm closure using the ADD-Vantage ADDAPTORTM.
5.4 Phlebitis and Thrombosis
The infusion of hypertonic solutions into a peripheral vein may result in vein irritation, vein damage, and/or thrombosis [see Dosage and Administration (2.1)]. If thrombophlebitis develops, remove the catheter as soon as possible.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pediatric Use: Increased risk of hypoglycemia/hyperglycemia and imbalances in fluid/electrolytes; monitor serum glucose concentrations, volume status, and electrolytes. (8.4)
5.3 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis, have been reported with Dextrose Injection (5%) administration [see Adverse Reactions (6)]. Stop administration of Dextrose Injection (5%) immediately if signs or symptoms of a hypersensitivity reaction develop. Initiate appropriate treatment as clinically indicated.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Dextrose Injection, USP (5%) is a clear, colorless, sterile solution of dextrose supplied in single-dose flexible plastic ADD-VantageTM diluent containers. This dosage form is intended to be used as a diluent for the contents of an ADD-Vantage vial or a single-dose powdered drug vial with a 20 mm closure using the ADD-Vantage ADDAPTORTM.
|
Unit of Sale |
Strength |
Volume |
|
NDC 0409-7100-66
|
5% (2.5 g/50 mL) (50 mg/mL) |
50 mL bags |
|
NDC 0409-7100-67
|
5% (5 g/100 mL) (50 mg/mL) |
100 mL bags |
|
NDC 0409-7100-02
|
5% (12.5 g/250 mL) (50 mg/mL) |
250 mL bags |
Do not remove the container from the overwrap until intended for use.
Use the product immediately after the introduction of additives.
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
Do not freeze.
To contact Pfizer’s Medical Information Department, please visit www.pfizermedinfo.com or call 1‑800-438‑1985.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
LAB-1143-4.0
2.2 Important Preparation Information
Visually inspect the Dextrose Injection (5%) for particulate matter and discoloration. Do not administer Dextrose Injection (5%) if the solution is cloudy, there are precipitates, and the container is damaged.
To reduce the risk of air embolism, adhere to the following preparation instructions for Dextrose Injection (5%):
-
•Use a non-vented infusion set or close the vent on a vented set.
-
•Use a dedicated line without any connections (do not connect flexible containers in series).
-
•Do not pressurize the flexible container to increase flow rates.
-
•If using a pumping device to administer Dextrose Injection (5%), turn off the pump before the container is empty.
To Open:
-
•Peel overwrap at corner and remove solution container.
-
oUse unit within 30 days of opening overwrap, as long as the use date does not exceed the printed expiration date.
-
-
•Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
To Assemble Vial and Flexible Diluent Container:
NOTE: Once the breakaway cap has been removed, do not access the vial with the syringe. |
|
|
|
NOTE: Once vial is seated, do not attempt to remove. (SEE FIGURE 4.)
|
|
|
To Reconstitute the Drug:
If the rubber stopper is not removed from the vial, and the medication is not released on the first attempt, the inner cap may be manipulated back into the rubber stopper without removing the drug vial from the diluent container. Repeat steps 3 through 5. |
Preparation for Administration:
-
1.Confirm the activation and admixture of vial contents.
-
2.Check for leaks by squeezing the container firmly. If leaks are found, discard the unit because sterility may be compromised.
-
3.Close the flow control clamp of the administration set.
-
4.Remove the cover from the outlet port at the bottom of the container.
-
5.Insert the piercing pin of the administration set into the port with a twisting motion until the pin is firmly seated. Note: See full directions on the administration set carton.
-
6.Lift the free end of the hanger loop on the bottom of the vial, breaking the two tie strings. Bend the loop outward to lock it in the upright position, then suspend the container from the hanger.
-
7.Squeeze and release the drip chamber to establish the proper fluid level in the chamber.
-
8.Open the flow control clamp and clear air from set. Close clamp.
-
9.Attach the set to the venipuncture device. If the device is not indwelling, prime and make venipuncture.
-
10.Regulate the rate of administration with the flow control clamp. Do not use the flexible container in series connections.
2.1 Important Administration Instructions
Dextrose Injection (5%) is intended for intravenous use.
-
•Use a peripheral vein to administer if the final dextrose concentration is 5% or less and the osmolarity is less than 900 mOsm/L.
-
•To avoid venous irritation, consider using a central vein to administer hypertonic solutions with osmolarity of 900 mOsm/L or greater [see Warnings and Precautions (5.4)].
-
•Do not administer Dextrose Injection (5%) simultaneously with blood products through the same administration set because of the possibility of pseudoagglutination or hemolysis.
-
•Use of a final filter is recommended during administration of parenteral solutions, where possible.
-
•Discard the unused portion.
-
•Do not use the Dextrose Injection (5%) ADD-VantageTM diluent containers with chemotherapy agents.
Principal Display Panel 50 Ml Bag Label (PRINCIPAL DISPLAY PANEL - 50 mL Bag Label)
PULL INNER PLUG/STOPPER
AND MIX DRUG BEFORE USE
ADD-Vantage™ Unit
NDC 0409-7100-68
5% DEXTROSE
Injection, USP
50
mL
EACH 100 mL CONTAINS DEXTROSE, HYDROUS 5 g.
253 mOsmol/LITER (calc.). pH 4.3 (3.2 TO 6.5)
DEXTROSE SOLUTIONS WITHOUT SALTS SHOULD
NOT BE USED IN BLOOD TRANSFUSIONS BECAUSE
OF POSSIBLE ROULEAU FORMATION. FOR USE ONLY
WITH ADD-Vantage™ SYSTEM COMPONENTS. SINGLE-
DOSE CONTAINER. FOR IV USE. USUAL DOSAGE: SEE
PACKAGE INSERT. STERILE, NONPYROGENIC. USE ONLY
IF SOLUTION IS CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
DISTRIBUTED BY
HOSPIRA, INC.,
LAKE FOREST, IL 60045 USA
Rx ONLY
IM-4480
14475201
3
v
CONTAINS DEHP
Principal Display Panel 100 Ml Bag Label (PRINCIPAL DISPLAY PANEL - 100 mL Bag Label)
PULL INNER PLUG/STOPPER
AND MIX DRUG BEFORE USE
ADD-Vantage™ Unit
NDC 0409-7100-69
5% DEXTROSE
Injection, USP
100
mL
EACH 100 mL CONTAINS DEXTROSE, HYDROUS 5 g.
253 mOsmol/LITER (calc.). pH 4.3 (3.2 TO 6.5)
DEXTROSE SOLUTIONS WITHOUT SALTS SHOULD
NOT BE USED IN BLOOD TRANSFUSIONS BECAUSE OF
POSSIBLE ROULEAU FORMATION. FOR USE ONLY WITH
ADD-Vantage™ SYSTEM COMPONENTS. SINGLE-DOSE
CONTAINER. FOR IV USE. USUAL DOSAGE: SEE PACKAGE
INSERT. STERILE, NONPYROGENIC. USE ONLY IF SOLUTION
IS CLEAR AND CONTAINER IS UNDAMAGED. MUST NOT
BE USED IN SERIES CONNECTIONS.
DISTRIBUTED BY
HOSPIRA, INC.,
LAKE FOREST, IL 60045 USA
Rx ONLY
IM-4481
14475601
3
v
CONTAINS DEHP
Principal Display Panel 250 Ml Bag Label (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label)
LOT
EXP.
PULL INNER PLUG/STOPPER
AND MIX DRUG BEFORE USE
ADD-Vantage™ Unit
NDC 0409-7100-04
5% DEXTROSE
Injection, USP
250
mL
EACH 100 mL CONTAINS
DEXTROSE, HYDROUS 5 g.
253 mOsmol/LITER (calc).
pH 4.3 (3.2 TO 6.5)
DEXTROSE SOLUTIONS
WITHOUT SALTS SHOULD
NOT BE USED IN BLOOD
TRANSFUSIONS BECAUSE
OF POSSIBLE ROULEAU
FORMATION. FOR USE ONLY
WITH ADD-Vantage™ SYSTEM
COMPONENTS. SINGLE-
DOSE CONTAINER. FOR
INTRAVENOUS USE. USUAL
DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY
IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES
CONNECTIONS.
Rx ONLY
DISTRIBUTED BY
HOSPIRA, INC.,
LAKE FOREST, IL 60045 USA
Hospira
3
v
CONTAINS DEHP
IM-4482
14475801
5.6 Electrolyte Imbalance and Fluid Overload
Electrolyte deficits, particularly serum potassium and phosphate, may occur during prolonged use of Dextrose Injection (5%).
Depending on the administered volume and the infusion rate, Dextrose Injection (5%) can cause fluid overload, including pulmonary edema.
Avoid Dextrose Injection (5%) in patients at risk for fluid and/or solute overload. If use cannot be avoided in these patients, monitor fluid balance, electrolyte concentrations, and acid-base balance, especially during prolonged use. Additional monitoring is recommended for patients with water and electrolyte disturbances that could be aggravated by increased glucose, insulin administration, and/or free water load.
Principal Display Panel Overwrap – Wr 0581 (PRINCIPAL DISPLAY PANEL - OVERWRAP – WR-0581)
TO OPEN – PEEL AT NOTCH
Five/ADD-Vantage™ Units
For use only with ADD-Vantage™ system components.
The overwrap is a moisture barrier. Use units within 30 days of opening overwrap, as long as the use
date does not exceed the printed expiration date. After removing the overwrap, check for minute
leaks by squeezing container firmly. If leaks are found, discard unit as sterility may be
impaired. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]
Rx only
14475301
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
WR-0581
Principal Display Panel Overwrap – Wr 0583 (PRINCIPAL DISPLAY PANEL - OVERWRAP – WR-0583)
One/ADD-Vantage™ Unit
TO OPEN–PEEL AT CORNER
For use only with ADD-Vantage™ system components.
The overwrap is a moisture barrier. Use unit within 30 days of opening
overwrap, as long as the use date does not exceed the printed
expiration date. Store at 20 to 25°C (68 to 77°F). [See USP Controlled
Room Temperature.] Protect from freezing. See insert.
After removing the overwrap, check for minute leaks by
squeezing container firmly. If leaks are found, discard
unit as sterility may be impaired.
Rx only
14475901
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
WR-0583
5.2 Hyperglycemia and Hyperosmolar Hyperglycemic State
The use of Dextrose Injection (5%) in patients with impaired glucose tolerance may worsen hyperglycemia. Administration of dextrose at a rate exceeding the patient’s utilization rate may lead to hyperglycemia, coma, and death.
Hyperglycemia is associated with an increase in serum osmolality, resulting in osmotic diuresis, dehydration, and electrolyte losses [see Warnings and Precautions (5.6), Use in Specific Populations (8.4)]. Patients with underlying CNS disease and renal impairment who receive dextrose infusions may be at greater risk of developing hyperosmolar hyperglycemic state.
Monitor blood glucose levels and treat hyperglycemia to maintain levels within normal limits while administering Dextrose Injection (5%). Insulin may be administered or adjusted to maintain optimal blood glucose levels during Dextrose Injection (5%) administration.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies with dextrose to evaluate the drug’s carcinogenic potential, mutagenic potential, or effects on fertility have not been performed.
7.1 Drugs With Effects On Glycemic Control and Electrolyte Balance (7.1 Drugs with Effects on Glycemic Control and Electrolyte Balance)
Dextrose Injection (5%) can affect glycemic control, vasopressin, and fluid and/or electrolyte balance [see Warnings and Precautions (5.1, 5.2, 5.5, 5.6)]. Monitor patients’ blood glucose concentrations, fluid balance, serum electrolyte concentrations, and acid-base balance.
Concomitant administration of Dextrose Injection (5%) with drugs associated with hyponatremia may increase the risk of developing hyponatremia. Drugs associated with hyponatremia include diuretics and those that cause SIADH (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), arginine vasopressin analogs, certain antiepileptic, psychotropic, and cytotoxic drugs). Avoid use of Dextrose Injection (5%) in patients receiving drugs associated with hyponatremia. If use cannot be avoided, closely monitor serum sodium concentrations during concomitant use [see Warnings and Precautions (5.5)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:48.496972 · Updated: 2026-03-14T22:28:06.461439