Fresh'n Soft®
10d21a95-b74f-4a10-984b-c6e350281e6d
34390-5
HUMAN OTC DRUG LABEL
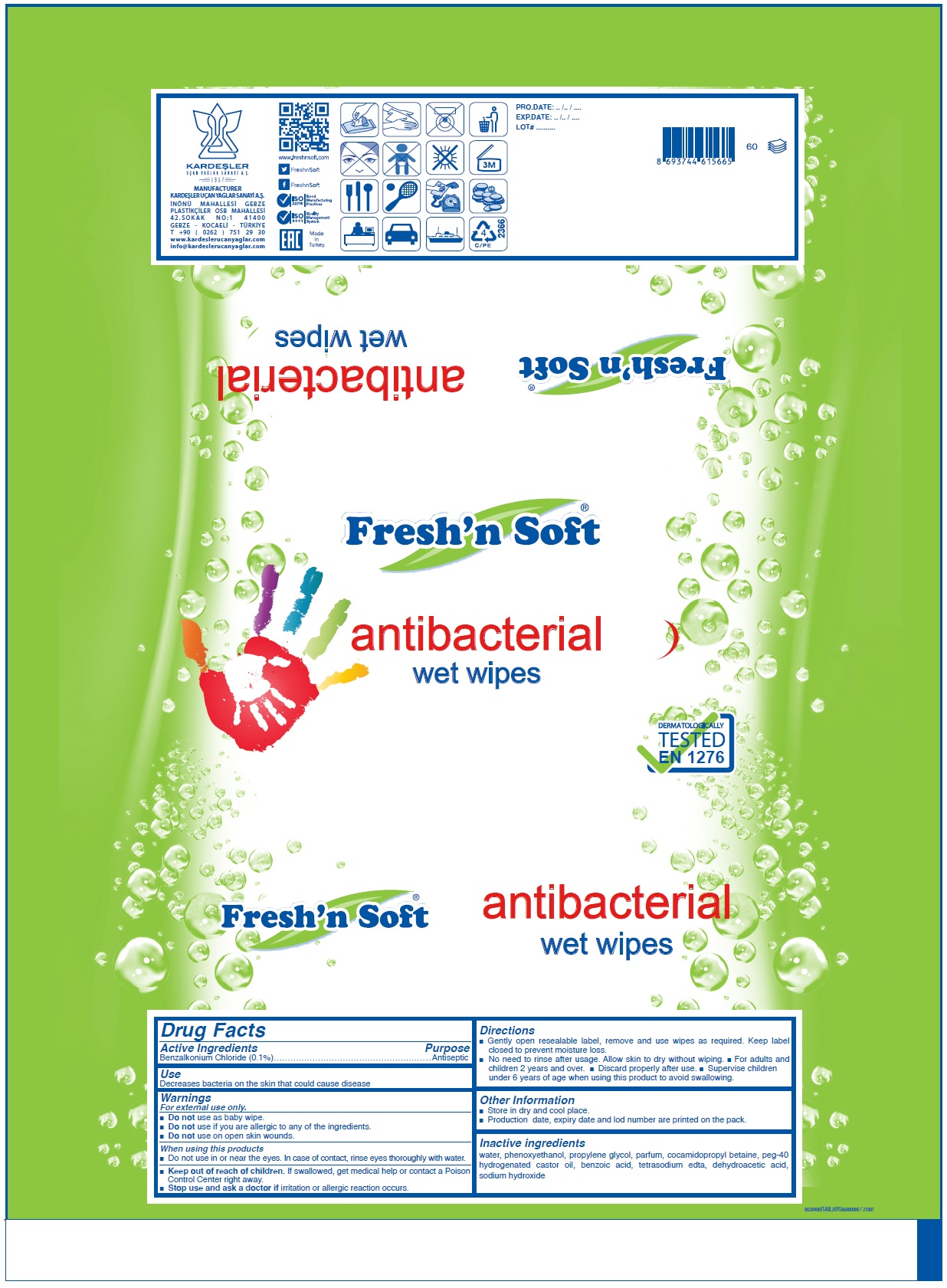
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzalkonium Chloride (0.1%)
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Benzalkonium Chloride (0.1%)
Use
Decreases bacteria on the skin that could cause disease
Section 42229-5
DERMATOLOGICALLY TESTED
EN 1276
KARDESLER
UCAN YAGLAR SANAYI A.S.
1957
MANUFACTURER
KARDESLER UÇAN YAGLAR SANAYI A.S.
INÖNÜ MAHALLESI GEBZE
PLASTIKÇILER OSB MAHALLESI
42.SOKAK NO:1 41400
GEBZE - KOCAELI - TÜRKIYE
T +90 ( 0262 ) 751 29 30
www.kardeslerucanyaglar.com
[email protected]
www.freshnsoft.com
FreshnSoft
FreshnSoft
ISO 22716 Good Manufacturing Practices
ISO 9001 Quality Management System
EAC Made in Turkey.
Section 50565-1
• Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only.
• Do not use as baby wipe.
• Do not use if you are allergic to any of the ingredients.
• Do not use on open skin wounds.
When using this products
• Do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
• Stop use and ask a doctor if irritation or allergic reaction occurs.
Packaging
Directions
• Gently open resealable label, remove and use wipes as required. Keep label closed to prevent moisture loss.
• No need to rinse after usage. Allow skin to dry without wiping. • For adults and children 2 years and over. • Discard properly after use. • Supervise children under 6 years of age when using this product to avoid swallowing.
Drug Facts
Other Information
• Store in dry and cool place.
• Production date, expiry date and lot number are printed on the pack.
Active Ingredients
Benzalkonium Chloride (0.1%)
Inactive Ingredients
water, phenoxyethanol, propylene glycol, parfum, cocamidopropyl betaine, peg-40 hydrogenated castor oil, benzoic acid, tetrasodium edta, dehydroacetic acid, sodium hydroxide
Structured Label Content
Use
Decreases bacteria on the skin that could cause disease
Section 42229-5 (42229-5)
DERMATOLOGICALLY TESTED
EN 1276
KARDESLER
UCAN YAGLAR SANAYI A.S.
1957
MANUFACTURER
KARDESLER UÇAN YAGLAR SANAYI A.S.
INÖNÜ MAHALLESI GEBZE
PLASTIKÇILER OSB MAHALLESI
42.SOKAK NO:1 41400
GEBZE - KOCAELI - TÜRKIYE
T +90 ( 0262 ) 751 29 30
www.kardeslerucanyaglar.com
[email protected]
www.freshnsoft.com
FreshnSoft
FreshnSoft
ISO 22716 Good Manufacturing Practices
ISO 9001 Quality Management System
EAC Made in Turkey.
Section 50565-1 (50565-1)
• Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Antiseptic
Warnings
For external use only.
• Do not use as baby wipe.
• Do not use if you are allergic to any of the ingredients.
• Do not use on open skin wounds.
When using this products
• Do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
• Stop use and ask a doctor if irritation or allergic reaction occurs.
Packaging
Directions
• Gently open resealable label, remove and use wipes as required. Keep label closed to prevent moisture loss.
• No need to rinse after usage. Allow skin to dry without wiping. • For adults and children 2 years and over. • Discard properly after use. • Supervise children under 6 years of age when using this product to avoid swallowing.
Drug Facts
Other Information
• Store in dry and cool place.
• Production date, expiry date and lot number are printed on the pack.
Active Ingredients
Benzalkonium Chloride (0.1%)
Inactive Ingredients (Inactive ingredients)
water, phenoxyethanol, propylene glycol, parfum, cocamidopropyl betaine, peg-40 hydrogenated castor oil, benzoic acid, tetrasodium edta, dehydroacetic acid, sodium hydroxide
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:13.212824 · Updated: 2026-03-14T22:59:07.764303