These Highlights Do Not Include All The Information Needed To Use Chantix Safely And Effectively. See Full Prescribing Information For Chantix.
0fefd790-b526-4f2f-8c83-5f39f7306556
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Serious neuropsychiatric events including, but not limited to, depression, suicidal ideation, suicide attempt, and completed suicide have been reported in patients taking CHANTIX. Some reported cases may have been complicated by the symptoms of nicotine withdrawal in patients who stopped smoking. Depressed mood may be a symptom of nicotine withdrawal. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these symptoms have occurred in patients taking CHANTIX who continued to smoke. All patients being treated with CHANTIX should be observed for neuropsychiatric symptoms including changes in behavior, hostility, agitation, depressed mood, and suicide-related events, including ideation, behavior, and attempted suicide. These symptoms, as well as worsening of pre-existing psychiatric illness and completed suicide, have been reported in some patients attempting to quit smoking while taking CHANTIX in the postmarketing experience. When symptoms were reported, most were during CHANTIX treatment, but some were following discontinuation of CHANTIX therapy. These events have occurred in patients with and without pre-existing psychiatric disease. Patients with serious psychiatric illness such as schizophrenia, bipolar disorder, and major depressive disorder did not participate in the premarketing studies of CHANTIX. Advise patients and caregivers that the patient should stop taking CHANTIX and contact a healthcare provider immediately if agitation, hostility, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. In many postmarketing cases, resolution of symptoms after discontinuation of CHANTIX was reported, although in some cases the symptoms persisted; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve. The risks of CHANTIX should be weighed against the benefits of its use. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo. The health benefits of quitting smoking are immediate and substantial. [see Warnings and Precautions (5.1) and Adverse Reactions (6.2) ]
Indications and Usage
CHANTIX is indicated for use as an aid to smoking cessation treatment.
Dosage and Administration
• Begin CHANTIX dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin CHANTIX dosing and then quit smoking between days 8 and 35 of treatment. ( 2.1 ) • Starting week: 0.5 mg once daily on days 1–3 and 0.5 mg twice daily on days 4–7. ( 2.1 ) • Continuing weeks: 1 mg twice daily for a total of 12 weeks. ( 2.1 ) • An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. ( 2.1 ) • Renal impairment: Reduce the dose in patients with severe renal impairment (estimated creatinine clearance <30 mL/min). ( 2.2 ) • Consider dose reduction for patients who cannot tolerate adverse effects. ( 2.1 ) • Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. ( 2.1 ) • Provide patients with appropriate educational materials and counseling to support the quit attempt. ( 2.1 )
Warnings and Precautions
• Seizures: New or worsening seizures have been observed in patients taking CHANTIX. CHANTIX should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. ( 5.2 ) • Interaction with alcohol: Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether CHANTIX affects them. ( 5.3 ) • Accidental injury: Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how CHANTIX may affect them. ( 5.4 ) • Cardiovascular events: A meta-analysis of 15 clinical trials, including a trial in patients with stable cardiovascular disease, demonstrated that while cardiovascular events were infrequent overall, some were reported more frequently in patients treated with CHANTIX. These events occurred primarily in patients with known cardiovascular disease. In both the clinical trial and meta-analysis, all-cause and cardiovascular mortality was lower in patients treated with CHANTIX. Instruct patients to notify their health care providers of new or worsening cardiovascular symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke. ( 5.5 and 6.1 ) • Angioedema and hypersensitivity reactions: Such reactions, including angioedema, infrequently life threatening, have been reported. Instruct patients to discontinue CHANTIX and immediately seek medical care if symptoms occur. ( 5.6 and 6.2 ) • Serious skin reactions: Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue CHANTIX and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. ( 5.7 and 6.2) • Nausea: Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. ( 5.8)
Contraindications
CHANTIX is contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to CHANTIX.
Adverse Reactions
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling: • Neuropsychiatric symptoms and suicidality [see Boxed Warning and Warnings and Precautions (5.1) ] • Seizures [see Warnings and Precautions (5.2) ] • Interaction with Alcohol [see Warnings and Precautions (5.3) ] • Accidental injury [see Warnings and Precautions (5.4) ] • Cardiovascular Events [see Warnings and Precautions (5.5) ] • Angioedema and hypersensitivity reactions [see Warnings and Precautions (5.6) ] • Serious skin reactions [see Warnings and Precautions (5.7) ] In the placebo-controlled premarketing studies, the most common adverse events associated with CHANTIX (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting. The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for CHANTIX, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in CHANTIX-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo). Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
Drug Interactions
Based on varenicline characteristics and clinical experience to date, CHANTIX has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3) ].
Storage and Handling
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with " Pfizer " on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with " Pfizer " on one side and "CHX 1.0" on the other side. CHANTIX is supplied in the following package configurations: Description NDC Starting 4-week card: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53 Starting Month Box: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53
How Supplied
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with " Pfizer " on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with " Pfizer " on one side and "CHX 1.0" on the other side. CHANTIX is supplied in the following package configurations: Description NDC Starting 4-week card: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53 Starting Month Box: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53
Medication Information
Warnings and Precautions
• Seizures: New or worsening seizures have been observed in patients taking CHANTIX. CHANTIX should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. ( 5.2 ) • Interaction with alcohol: Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether CHANTIX affects them. ( 5.3 ) • Accidental injury: Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how CHANTIX may affect them. ( 5.4 ) • Cardiovascular events: A meta-analysis of 15 clinical trials, including a trial in patients with stable cardiovascular disease, demonstrated that while cardiovascular events were infrequent overall, some were reported more frequently in patients treated with CHANTIX. These events occurred primarily in patients with known cardiovascular disease. In both the clinical trial and meta-analysis, all-cause and cardiovascular mortality was lower in patients treated with CHANTIX. Instruct patients to notify their health care providers of new or worsening cardiovascular symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke. ( 5.5 and 6.1 ) • Angioedema and hypersensitivity reactions: Such reactions, including angioedema, infrequently life threatening, have been reported. Instruct patients to discontinue CHANTIX and immediately seek medical care if symptoms occur. ( 5.6 and 6.2 ) • Serious skin reactions: Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue CHANTIX and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. ( 5.7 and 6.2) • Nausea: Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. ( 5.8)
Indications and Usage
CHANTIX is indicated for use as an aid to smoking cessation treatment.
Dosage and Administration
• Begin CHANTIX dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin CHANTIX dosing and then quit smoking between days 8 and 35 of treatment. ( 2.1 ) • Starting week: 0.5 mg once daily on days 1–3 and 0.5 mg twice daily on days 4–7. ( 2.1 ) • Continuing weeks: 1 mg twice daily for a total of 12 weeks. ( 2.1 ) • An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. ( 2.1 ) • Renal impairment: Reduce the dose in patients with severe renal impairment (estimated creatinine clearance <30 mL/min). ( 2.2 ) • Consider dose reduction for patients who cannot tolerate adverse effects. ( 2.1 ) • Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. ( 2.1 ) • Provide patients with appropriate educational materials and counseling to support the quit attempt. ( 2.1 )
Contraindications
CHANTIX is contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to CHANTIX.
Adverse Reactions
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling: • Neuropsychiatric symptoms and suicidality [see Boxed Warning and Warnings and Precautions (5.1) ] • Seizures [see Warnings and Precautions (5.2) ] • Interaction with Alcohol [see Warnings and Precautions (5.3) ] • Accidental injury [see Warnings and Precautions (5.4) ] • Cardiovascular Events [see Warnings and Precautions (5.5) ] • Angioedema and hypersensitivity reactions [see Warnings and Precautions (5.6) ] • Serious skin reactions [see Warnings and Precautions (5.7) ] In the placebo-controlled premarketing studies, the most common adverse events associated with CHANTIX (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting. The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for CHANTIX, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in CHANTIX-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo). Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
Drug Interactions
Based on varenicline characteristics and clinical experience to date, CHANTIX has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3) ].
Storage and Handling
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with " Pfizer " on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with " Pfizer " on one side and "CHX 1.0" on the other side. CHANTIX is supplied in the following package configurations: Description NDC Starting 4-week card: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53 Starting Month Box: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53
How Supplied
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with " Pfizer " on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with " Pfizer " on one side and "CHX 1.0" on the other side. CHANTIX is supplied in the following package configurations: Description NDC Starting 4-week card: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53 Starting Month Box: 0.5 mg × 11 tablets and 1 mg × 42 tablets NDC 63187-618-53
Description
Serious neuropsychiatric events including, but not limited to, depression, suicidal ideation, suicide attempt, and completed suicide have been reported in patients taking CHANTIX. Some reported cases may have been complicated by the symptoms of nicotine withdrawal in patients who stopped smoking. Depressed mood may be a symptom of nicotine withdrawal. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these symptoms have occurred in patients taking CHANTIX who continued to smoke. All patients being treated with CHANTIX should be observed for neuropsychiatric symptoms including changes in behavior, hostility, agitation, depressed mood, and suicide-related events, including ideation, behavior, and attempted suicide. These symptoms, as well as worsening of pre-existing psychiatric illness and completed suicide, have been reported in some patients attempting to quit smoking while taking CHANTIX in the postmarketing experience. When symptoms were reported, most were during CHANTIX treatment, but some were following discontinuation of CHANTIX therapy. These events have occurred in patients with and without pre-existing psychiatric disease. Patients with serious psychiatric illness such as schizophrenia, bipolar disorder, and major depressive disorder did not participate in the premarketing studies of CHANTIX. Advise patients and caregivers that the patient should stop taking CHANTIX and contact a healthcare provider immediately if agitation, hostility, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. In many postmarketing cases, resolution of symptoms after discontinuation of CHANTIX was reported, although in some cases the symptoms persisted; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve. The risks of CHANTIX should be weighed against the benefits of its use. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo. The health benefits of quitting smoking are immediate and substantial. [see Warnings and Precautions (5.1) and Adverse Reactions (6.2) ]
Section 34077-8
Pregnancy Category C.
There are no adequate and well-controlled studies of CHANTIX use in pregnant women. In animal studies, CHANTIX caused decreased fetal weights, increased auditory startle response, and decreased fertility in offspring. CHANTIX should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
In reproductive and developmental toxicity studies, pregnant rats and rabbits received varenicline succinate during organogenesis at oral doses up to 15 and 30 mg/kg/day, respectively. These exposures were 36 (rats) and 50 (rabbits) times the human exposure (based on AUC) at the maximum recommended human dose (MRHD) of 1 mg twice daily. While no fetal structural abnormalities occurred in either species, reduced fetal weights occurred in rabbits at the highest dose (exposures 50 times the human exposure at the MRHD based on AUC). Fetal weight reduction did not occur at animal exposures 23 times the human exposure at the MRHD based on AUC.
In a pre- and postnatal development study, pregnant rats received up to 15 mg/kg/day of oral varenicline succinate from organogenesis through lactation. These resulted in exposures up to 36 times the human exposure (based on AUC) at the MRHD of 1 mg twice daily. Decreased fertility and increased auditory startle response occurred in offspring.
Section 42229-5
Patients with Impaired Renal Function: No dosage adjustment is necessary for patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30 mL/min), the recommended starting dose of CHANTIX is 0.5 mg once daily. The dose may then be titrated as needed to a maximum dose of 0.5 mg twice a day. For patients with end-stage renal disease undergoing hemodialysis, a maximum dose of 0.5 mg once daily may be administered if tolerated [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
Section 43683-2
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) (see USP Controlled Room Temperature).
5.8 Nausea
Nausea was the most common adverse reaction reported with CHANTIX treatment. Nausea was generally described as mild or moderate and often transient; however, for some patients, it was persistent over several months. The incidence of nausea was dose-dependent. Initial dose-titration was beneficial in reducing the occurrence of nausea. For patients treated to the maximum recommended dose of 1 mg twice daily following initial dosage titration, the incidence of nausea was 30% compared with 10% in patients taking a comparable placebo regimen. In patients taking CHANTIX 0.5 mg twice daily following initial titration, the incidence was 16% compared with 11% for placebo. Approximately 3% of patients treated with CHANTIX 1 mg twice daily in studies involving 12 weeks of treatment discontinued treatment prematurely because of nausea. For patients with intolerable nausea, a dose reduction should be considered.
5.2 Seizures
During clinical trials and the post-marketing experience, there have been reports of seizures in patients treated with CHANTIX. Some patients had no history of seizures, whereas others had a history of seizure disorder that was remote or well-controlled. In most cases, the seizure occurred within the first month of therapy. Weigh this potential risk against the potential benefits before prescribing CHANTIX in patients with a history of seizures or other factors that can lower the seizure threshold. Advise patients to discontinue CHANTIX and contact a healthcare provider immediately if they experience a seizure while on treatment [see Adverse Reactions (6.2) ].
10 Overdosage
In case of overdose, standard supportive measures should be instituted as required.
Varenicline has been shown to be dialyzed in patients with end stage renal disease [see Clinical Pharmacology (12.3)], however, there is no experience in dialysis following overdose.
11 Description
CHANTIX tablets contain varenicline (as the tartrate salt), which is a partial agonist selective for α4β2 nicotinic acetylcholine receptor subtypes.
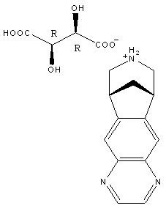
Varenicline, as the tartrate salt, is a powder which is a white to off-white to slightly yellow solid with the following chemical name: 7,8,9,10-tetrahydro-6,10-methano-6H-pyrazino[2,3- h][3]benzazepine, (2R,3R)-2,3-dihydroxybutanedioate (1:1). It is highly soluble in water. Varenicline tartrate has a molecular weight of 361.35 Daltons, and a molecular formula of C13H13N3 ∙ C4H6O6. The chemical structure is:
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with "Pfizer" on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with "Pfizer" on one side and "CHX 1.0" on the other side. Each 0.5 mg CHANTIX tablet contains 0.85 mg of varenicline tartrate equivalent to 0.5 mg of varenicline free base; each 1mg CHANTIX tablet contains 1.71 mg of varenicline tartrate equivalent to 1 mg of varenicline free base. The following inactive ingredients are included in the tablets: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry® White (for 0.5 mg), Opadry® Blue (for 1 mg), and Opadry® Clear.
Medication Guide
CHANTIX® (CHANT-iks)
(varenicline) Tablets
What is the most important information I should know about CHANTIX?
Some people have had serious side effects while using CHANTIX to help them quit smoking, including:
New or worse mental health problems, such as changes in behavior, hostility, agitation, depressed mood, and suicidal thoughts or actions. Some people had these symptoms when they began taking CHANTIX, and others developed them after several weeks of treatment, or after stopping CHANTIX.
Before taking CHANTIX, tell your doctor if you have ever had depression or other mental health problems. You should also tell your doctor about any symptoms you had during other times you tried to quit smoking, with or without CHANTIX.
Stop taking CHANTIX and call your doctor right away if you, your family, or caregiver notice agitation, hostility, depression or changes in your behavior or thinking that are not typical for you, or you develop any of the following symptoms:
-
•thoughts about suicide or dying, or attempts to commit suicide
-
•new or worse depression, anxiety, or panic attacks
-
•feeling very agitated or restless
-
•acting aggressive, being angry, or violent
-
•acting on dangerous impulses
-
•an extreme increase in activity and talking (mania)
-
•abnormal thoughts or sensations
-
•seeing or hearing things that are not there (hallucinations)
-
•feeling people are against you (paranoia)
-
•feeling confused
-
•other unusual changes in behavior or mood
When you try to quit smoking, with or without CHANTIX, you may have symptoms that may be due to nicotine withdrawal, including urge to smoke, depressed mood, trouble sleeping, irritability, frustration, anger, feeling anxious, difficulty concentrating, restlessness, decreased heart rate, and increased appetite or weight gain. Some people have even experienced suicidal thoughts when trying to quit smoking without medication. Sometimes quitting smoking can lead to worsening of mental health problems that you already have, such as depression.
-
See " What are the possible side effects of CHANTIX? " for more information about other side effects.
What is CHANTIX?
CHANTIX is a prescription medicine to help people stop smoking.
Quitting smoking can lower your chances of having lung disease, heart disease or getting certain types of cancer that are related to smoking.
It is not known if CHANTIX is safe and effective in children.
It is not known if CHANTIX is safe and effective when used with other stop smoking medicines.
Who should not take CHANTIX?
Do not take CHANTIX if you have had a serious allergic or skin reaction to CHANTIX. Symptoms may include:
-
•swelling of the face, mouth (tongue, lips, gums), throat or neck
-
•trouble breathing
-
•rash, with peeling skin
-
•blisters in your mouth
What should I tell my doctor before taking CHANTIX?
See "What is the most important information I should know about CHANTIX?"
Before you take CHANTIX, tell your doctor if you:
-
•use other treatments to quit smoking. Using CHANTIX with a nicotine patch may cause nausea, vomiting, headache, dizziness, upset stomach, and tiredness to happen more often than if you just use a nicotine patch alone.
-
•have kidney problems or get kidney dialysis. Your doctor may prescribe a lower dose of CHANTIX for you.
-
•have a history of seizures
-
•drink alcohol
-
•have heart or blood vessel problems
-
•have any other medical conditions
-
•are pregnant or plan to become pregnant. It is not known if CHANTIX will harm your unborn baby.
-
•are breastfeeding. It is not known if CHANTIX passes into breast milk. You and your doctor should decide if you will breastfeed or take CHANTIX. You should not do both.
Tell your doctor about all the medicines you take, including prescription and over the counter medicines, vitamins and herbal supplements. Your doctor may need to change the dose of some of your medicines when you stop smoking.
You should not use CHANTIX while using other medicines to quit smoking. Tell your doctor if you use other treatments to quit smoking.
Know the medicines you take. Keep a list of them with you to show your doctor and pharmacist when you get a new medicine.
How should I take CHANTIX?
-
•There are 2 ways that you can use CHANTIX to help you quit smoking. Talk to your doctor about the following 2 ways to use CHANTIX:
-
Choose a quit date when you will stop smoking. Start taking CHANTIX 1 week (7 days) before your quit date.
-
OR
-
Start taking CHANTIX before you choose a quit date. Pick a date to quit smoking that is between days 8 and 35 of treatment.
-
Starting CHANTIX before your quit date gives CHANTIX time to build up in your body. You can keep smoking during this time. Take CHANTIX exactly as prescribed by your doctor.
-
•CHANTIX comes as a white tablet (0.5 mg) and a blue tablet (1 mg). You start with the white tablet and then usually go to the blue tablet. See the chart below for dosing instructions for adults.
|
Day 1 to Day 3 |
|
|
Day 4 to Day 7 |
|
|
Day 8 to end of treatment |
|
-
•Make sure that you try to stop smoking on your quit date. If you slip-up and smoke, try again. Some people need to take CHANTIX for a few weeks for CHANTIX to work best.
-
•Most people will take CHANTIX for up to 12 weeks. If you have completely quit smoking by 12 weeks, your doctor may prescribe CHANTIX for another 12 weeks to help you stay cigarette-free.
-
•Take CHANTIX after eating and with a full glass (8 ounces) of water.
-
•This dosing schedule may not be right for everyone. Talk to your doctor if you are having side effects such as nausea, strange dreams, or sleep problems. Your doctor may want to reduce your dose.
-
•If you miss a dose of CHANTIX, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Just take your next dose at your regular time.
What should I avoid while taking CHANTIX?
-
•Use caution when driving or operating machinery until you know how CHANTIX affects you. CHANTIX may make you feel sleepy, dizzy, or have trouble concentrating, making it hard to drive or perform other activities safely.
-
•Decrease the amount of alcoholic beverages that you drink during treatment with CHANTIX until you know if CHANTIX affects your ability to tolerate alcohol. Some people have experienced the following when drinking alcohol during treatment with CHANTIX:
-
•increased drunkenness (intoxication)
-
•unusual or sometimes aggressive behavior
-
•no memory of things that have happened
-
What are the possible side effects of CHANTIX?
Serious side effects of CHANTIX may include:
-
•See "What is the most important information I should know about CHANTIX?"
-
•Seizures. Some people have had seizures during treatment with CHANTIX. In most cases, the seizures have happened during the first month of treatment with CHANTIX. If you have a seizure during treatment with CHANTIX, stop taking CHANTIX and contact your healthcare provider right away.
-
•New or worse heart or blood vessel (cardiovascular) problems, mostly in people, who already have cardiovascular problems. Tell your doctor if you have any changes in symptoms during treatment with CHANTIX.
Get emergency medical help right away if you have any of the following symptoms of a heart attack, including:
-
•chest discomfort (uncomfortable pressure, squeezing, fullness or pain) that lasts more than a few minutes, or that goes away and comes back
-
•pain or discomfort in one or both arms, back, neck, jaw or stomach
-
•shortness of breath, sweating, nausea, vomiting, or feeling lightheaded associated with chest discomfort
-
-
•Allergic reactions can happen with CHANTIX. Some of these allergic reactions can be life-threatening.
-
•Serious skin reactions, including rash, swelling, redness, and peeling of the skin. Some of these skin reactions can become life-threatening.
Stop taking CHANTIX and get medical help right away if you have any of the following symptoms:
-
•swelling of the face, mouth (tongue, lips, and gums), throat or neck
-
•trouble breathing
-
•rash with peeling skin
-
•blisters in your mouth
The most common side effects of CHANTIX include:
-
•nausea
-
•sleep problems (trouble sleeping or vivid, unusual, or strange dreams)
-
•constipation
-
•gas
-
•vomiting
Tell your doctor about side effects that bother you or that do not go away.
These are not all the side effects of CHANTIX. Ask your doctor or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store CHANTIX?
-
•Store CHANTIX at room temperature, between 68°F to 77°F (20°C to 25°C).
-
•Keep CHANTIX and all medicines out of the reach of children.
General information about CHANTIX
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CHANTIX for a condition for which it was not prescribed. Do not give your CHANTIX to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about CHANTIX that is written for healthcare professionals.
For more information about CHANTIX and tips on how to quit smoking, go to www.CHANTIX.com or call 1-877-242-6849.
If you are motivated to quit smoking and did not succeed during prior CHANTIX treatment for reasons other than side effects, or if you returned to smoking after treatment, speak with your doctor about whether another course of CHANTIX therapy may be right for you.
What are the ingredients in CHANTIX?
Active ingredient: varenicline tartrate
Inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry ® White (for 0.5 mg), Opadry ® Blue (for 1 mg), and Opadry® Clear.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
October 2014
LAB-0328-13.0
Relabeled by:
Proficient Rx LP
Thousand Oaks, CA 91320
8.4 Pediatric Use
Safety and effectiveness of CHANTIX in pediatric patients have not been established.
8.5 Geriatric Use
A combined single- and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once daily or twice daily to 16 healthy elderly male and female smokers (aged 65–75 yrs) for 7 consecutive days was similar to that of younger subjects. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.2) ].
No dosage adjustment is recommended for elderly patients.
14.2 Urge to Smoke
Based on responses to the Brief Questionnaire of Smoking Urges and the Minnesota Nicotine Withdrawal scale "urge to smoke" item, CHANTIX reduced urge to smoke compared to placebo.
14 Clinical Studies
The efficacy of CHANTIX in smoking cessation was demonstrated in six clinical trials in which a total of 3659 chronic cigarette smokers (≥10 cigarettes per day) were treated with CHANTIX. In all clinical studies, abstinence from smoking was determined by patient self-report and verified by measurement of exhaled carbon monoxide (CO≤10 ppm) at weekly visits. Among the CHANTIX-treated patients enrolled in these studies, the completion rate was 65%. Except for the dose-ranging study (Study 1) and the maintenance of abstinence study (Study 6), patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. Most patients enrolled in these trials were white (79–96%). All studies enrolled almost equal numbers of men and women. The average age of patients in these studies was 43 years. Patients on average had smoked about 21 cigarettes per day for an average of approximately 25 years. Patients set a date to stop smoking (target quit date) with dosing starting 1 week before this date.
Three additional studies were conducted in patients with cardiovascular disease, in patients with chronic obstructive pulmonary disease [see Clinical Studies (14.4)], and in patients instructed to select their quit date within days 8 and 35 of treatment [see Clinical Studies (14.5)].
In all studies, patients were provided with an educational booklet on smoking cessation and received up to 10 minutes of smoking cessation counseling at each weekly treatment visit according to Agency for Healthcare Research and Quality guidelines.
4 Contraindications
CHANTIX is contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to CHANTIX.
6 Adverse Reactions
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling:
-
•Neuropsychiatric symptoms and suicidality [see Boxed Warning and Warnings and Precautions (5.1)]
-
•Seizures [see Warnings and Precautions (5.2)]
-
•Interaction with Alcohol [see Warnings and Precautions (5.3)]
-
•Accidental injury [see Warnings and Precautions (5.4)]
-
•Cardiovascular Events [see Warnings and Precautions (5.5)]
-
•Angioedema and hypersensitivity reactions [see Warnings and Precautions (5.6)]
-
•Serious skin reactions [see Warnings and Precautions (5.7)]
In the placebo-controlled premarketing studies, the most common adverse events associated with CHANTIX (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting.
The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for CHANTIX, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in CHANTIX-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo).
Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
7 Drug Interactions
Based on varenicline characteristics and clinical experience to date, CHANTIX has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3)].
8.3 Nursing Mothers
It is not known whether CHANTIX is excreted in human milk. In animal studies varenicline was excreted in milk of lactating animals. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from CHANTIX, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.6 Renal Impairment
Varenicline is substantially eliminated by renal glomerular filtration along with active tubular secretion. Dose reduction is not required in patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30 mL/min), and for patients with end-stage renal disease undergoing hemodialysis, dosage adjustment is needed. [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
5.4 Accidental Injury
There have been postmarketing reports of traffic accidents, near-miss incidents in traffic, or other accidental injuries in patients taking CHANTIX. In some cases, the patients reported somnolence, dizziness, loss of consciousness or difficulty concentrating that resulted in impairment, or concern about potential impairment, in driving or operating machinery. Advise patients to use caution driving or operating machinery or engaging in other potentially hazardous activities until they know how CHANTIX may affect them.
1 Indications and Usage
CHANTIX is indicated for use as an aid to smoking cessation treatment.
14.7 Re Treatment Study
CHANTIX was evaluated in a double-blind, placebo-controlled trial of patients who had made a previous attempt to quit smoking with CHANTIX, and either did not succeed in quitting or relapsed after treatment. Subjects were randomized 1:1 to CHANTIX 1 mg twice daily (n=249) or placebo (n=245) for 12 weeks of treatment and followed for 40 weeks post-treatment. Patients included in this study had taken CHANTIX for a smoking-cessation attempt in the past (for a total treatment duration of a minimum of two weeks), at least three months prior to study entry, and had been smoking for at least four weeks.
Patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (45%) compared to patients treated with placebo (12%) and from weeks 9 through 52 (20%) compared to subjects treated with placebo (3%).
| Weeks 9 through 12 | Weeks 9 through 52 | |||
|---|---|---|---|---|
|
CHANTIX
1 mg BID |
Placebo |
CHANTIX
1 mg BID |
Placebo | |
| BID = twice daily | ||||
|
Retreatment Study |
45% |
12% |
20% |
3% |
12.1 Mechanism of Action
Varenicline binds with high affinity and selectivity at α4β2 neuronal nicotinic acetylcholine receptors. The efficacy of CHANTIX in smoking cessation is believed to be the result of varenicline's activity at α4β2 sub-type of the nicotinic receptor where its binding produces agonist activity, while simultaneously preventing nicotine binding to these receptors.
Electrophysiology studies in vitro and neurochemical studies in vivo have shown that varenicline binds to α4β2 neuronal nicotinic acetylcholine receptors and stimulates receptor-mediated activity, but at a significantly lower level than nicotine. Varenicline blocks the ability of nicotine to activate α4β2 receptors and thus to stimulate the central nervous mesolimbic dopamine system, believed to be the neuronal mechanism underlying reinforcement and reward experienced upon smoking. Varenicline is highly selective and binds more potently to α4β2 receptors than to other common nicotinic receptors (>500-fold α3β4, >3500-fold α7, >20,000-fold α1βγδ), or to non-nicotinic receptors and transporters (>2000-fold). Varenicline also binds with moderate affinity (Ki = 350 nM) to the 5-HT3 receptor.
9.1 Controlled Substance
Varenicline is not a controlled substance.
14.3 Long Term Abstinence
Studies 1 through 5 included 40 weeks of post-treatment follow-up. In each study, CHANTIX-treated patients were more likely to maintain abstinence throughout the follow-up period than were patients treated with placebo (Figure 2, Table 7).
Figure 2: Continuous Abstinence, Weeks 9 through 52
|
CHANTIX
0.5 mg BID |
CHANTIX
1 mg BID |
CHANTIX
Flexible |
Bupropion
SR |
Placebo | |
|---|---|---|---|---|---|
| BID = twice daily | |||||
|
Study 2 |
19% |
23% |
4% |
||
|
Study 3 |
22% |
8% |
|||
|
Study 4 |
21% |
16% |
8% |
||
|
Study 5 |
22% |
14% |
10% |
5.5 Cardiovascular Events
In a placebo-controlled clinical trial of CHANTIX administered to patients with stable cardiovascular disease, with approximately 350 patients per treatment arm, all-cause and cardiovascular mortality was lower in patients treated with CHANTIX, but certain nonfatal cardiovascular events occurred more frequently in patients treated with CHANTIX than in patients treated with placebo [see Clinical Trials Experience (6.1)]. Table 3 below shows the incidence of deaths and of selected nonfatal serious cardiovascular events occurring more frequently in the CHANTIX arm compared to the placebo arm. These events were adjudicated by an independent blinded committee. Nonfatal serious cardiovascular events not listed occurred at the same incidence or more commonly in the placebo arm. Patients with more than one cardiovascular event of the same type are counted only once per row. Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina.
| Mortality and Cardiovascular Events |
CHANTIX
(N=353) n (%) |
Placebo
(N=350) n (%) |
|---|---|---|
|
Mortality (Cardiovascular & All-cause up to 52 wks) |
||
|
Cardiovascular death |
1 (0.3) |
2 (0.6) |
|
All-cause mortality |
2 (0.6) |
5 (1.4) |
|
Nonfatal Cardiovascular Events (rate on CHANTIX > Placebo) |
||
|
Up to 30 days after treatment |
||
|
Nonfatal myocardial infarction |
4 (1.1) |
1 (0.3) |
|
Nonfatal Stroke |
2 (0.6) |
0 (0) |
|
Beyond 30 days after treatment & up to 52 weeks |
||
|
Nonfatal myocardial infarction |
3 (0.8) |
2 (0.6) |
|
Need for coronary revascularization |
7 (2.0) |
2 (0.6) |
|
Hospitalization for angina pectoris |
6 (1.7) |
4 (1.1) |
|
Transient ischemia attack |
1 (0.3) |
0 (0) |
|
New diagnosis of peripheral vascular disease (PVD) or admission for a PVD procedure |
5 (1.4) |
2 (0.6) |
A meta-analysis of 15 clinical trials of ≥ 12 weeks treatment duration, including 7002 patients (4190 CHANTIX, 2812 placebo), was conducted to systematically assess the cardiovascular safety of CHANTIX. The study in patients with stable cardiovascular disease described above was included in the meta-analysis. There were lower rates of all-cause mortality (CHANTIX 6 [0.14%]; placebo 7 [0.25%]) and cardiovascular mortality (CHANTIX 2 [0.05%]; placebo 2 [0.07%]) in the CHANTIX arms compared with the placebo arms in the meta-analysis.
The key cardiovascular safety analysis included occurrence and timing of a composite endpoint of Major Adverse Cardiovascular Events (MACE), defined as cardiovascular death, nonfatal MI, and nonfatal stroke. These events included in the endpoint were adjudicated by a blinded, independent committee. Overall, a small number of MACE occurred in the trials included in the meta-analysis, as described in Table 4. These events occurred primarily in patients with known cardiovascular disease.
|
CHANTIX
N=4190 |
Placebo
N=2812 |
|
|---|---|---|
|
MACE cases, n (%) |
13 (0.31%) |
6 (0.21%) |
|
Patient-years of exposure |
1316 |
839 |
|
Hazard Ratio (95% CI) |
||
|
1.95 (0.79, 4.82) |
||
|
Rate Difference per 1,000 patient-years (95% CI) |
||
|
6.30 (-2.40, 15.10) |
The meta-analysis showed that exposure to CHANTIX resulted in a hazard ratio for MACE of 1.95 (95% confidence interval from 0.79 to 4.82) for patients up to 30 days after treatment; this is equivalent to an estimated increase of 6.3 MACE events per 1,000 patient-years of exposure. The meta-analysis showed higher rates of CV endpoints in patients on CHANTIX relative to placebo across different time frames and pre-specified sensitivity analyses, including various study groupings and CV outcomes. Although these findings were not statistically significant they were consistent. Because the number of events was small overall, the power for finding a statistically significant difference in a signal of this magnitude is low.
CHANTIX was not studied in patients with unstable cardiovascular disease or cardiovascular events occurring within two months before screening. Patients should be advised to notify a health care provider of new or worsening symptoms of cardiovascular disease. The risks of CHANTIX should be weighed against the benefits of its use in smokers with cardiovascular disease. Smoking is an independent and major risk factor for cardiovascular disease. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo.
5 Warnings and Precautions
-
•Seizures: New or worsening seizures have been observed in patients taking CHANTIX. CHANTIX should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. (5.2)
-
•Interaction with alcohol: Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether CHANTIX affects them. (5.3)
-
•Accidental injury: Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how CHANTIX may affect them. (5.4)
-
•Cardiovascular events: A meta-analysis of 15 clinical trials, including a trial in patients with stable cardiovascular disease, demonstrated that while cardiovascular events were infrequent overall, some were reported more frequently in patients treated with CHANTIX. These events occurred primarily in patients with known cardiovascular disease. In both the clinical trial and meta-analysis, all-cause and cardiovascular mortality was lower in patients treated with CHANTIX. Instruct patients to notify their health care providers of new or worsening cardiovascular symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke. (5.5 and 6.1)
-
•Angioedema and hypersensitivity reactions: Such reactions, including angioedema, infrequently life threatening, have been reported. Instruct patients to discontinue CHANTIX and immediately seek medical care if symptoms occur. (5.6 and 6.2)
-
•Serious skin reactions: Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue CHANTIX and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. (5.7 and 6.2)
-
•Nausea: Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. (5.8)
5.7 Serious Skin Reactions
There have been postmarketing reports of rare but serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients using CHANTIX [see Adverse Reactions (6.2)]. As these skin reactions can be life-threatening, instruct patients to stop taking CHANTIX and contact a healthcare provider immediately at the first appearance of a skin rash with mucosal lesions or any other signs of hypersensitivity.
2 Dosage and Administration
-
•Begin CHANTIX dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin CHANTIX dosing and then quit smoking between days 8 and 35 of treatment. (2.1)
-
•Starting week: 0.5 mg once daily on days 1–3 and 0.5 mg twice daily on days 4–7. (2.1)
-
•Continuing weeks: 1 mg twice daily for a total of 12 weeks. (2.1)
-
•An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. (2.1)
-
•Renal impairment: Reduce the dose in patients with severe renal impairment (estimated creatinine clearance <30 mL/min). (2.2)
-
•Consider dose reduction for patients who cannot tolerate adverse effects. (2.1)
-
•Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. (2.1)
-
•Provide patients with appropriate educational materials and counseling to support the quit attempt. (2.1)
2.1 Usual Dosage for Adults
Smoking cessation therapies are more likely to succeed for patients who are motivated to stop smoking and who are provided additional advice and support. Provide patients with appropriate educational materials and counseling to support the quit attempt.
The patient should set a date to stop smoking. Begin CHANTIX dosing one week before this date. Alternatively, the patient can begin CHANTIX dosing and then quit smoking between days 8 and 35 of treatment.
CHANTIX should be taken after eating and with a full glass of water.
The recommended dose of CHANTIX is 1 mg twice daily following a 1-week titration as follows:
|
Days 1 – 3: |
0.5 mg once daily |
|
Days 4 – 7: |
0.5 mg twice daily |
|
Day 8 – end of treatment: |
1 mg twice daily |
Patients should be treated with CHANTIX for 12 weeks. For patients who have successfully stopped smoking at the end of 12 weeks, an additional course of 12 weeks' treatment with CHANTIX is recommended to further increase the likelihood of long-term abstinence.
Patients who are motivated to quit, and who did not succeed in stopping smoking during prior CHANTIX therapy for reasons other than intolerability due to adverse events or who relapsed after treatment, should be encouraged to make another attempt with CHANTIX once factors contributing to the failed attempt have been identified and addressed.
Consider a temporary or permanent dose reduction in patients who cannot tolerate the adverse effects of CHANTIX.
3 Dosage Forms and Strengths
Capsular, biconvex tablets: 0.5 mg (white to off-white, debossed with "Pfizer" on one side and "CHX 0.5" on the other side) and 1 mg (light blue, debossed with "Pfizer" on one side and "CHX 1.0" on the other side).
5.3 Interaction With Alcohol
There have been post-marketing reports of patients experiencing increased intoxicating effects of alcohol while taking CHANTIX. Some cases described unusual and sometimes aggressive behavior, and were often accompanied by amnesia for the events. Advise patients to reduce the amount of alcohol they consume while taking CHANTIX until they know whether CHANTIX affects their tolerance for alcohol [see Adverse Reactions (6.2) ] .
6.2 Postmarketing Experience
The following adverse events have been reported during post-approval use of CHANTIX. Because these events are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
There have been reports of depression, mania, psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide in patients attempting to quit smoking while taking CHANTIX [see Boxed Warning, Warnings and Precautions (5.1)]. Smoking cessation with or without treatment is associated with nicotine withdrawal symptoms and the exacerbation of underlying psychiatric illness. Not all patients had known pre-existing psychiatric illness and not all had discontinued smoking.
There have been post-marketing reports of new or worsening seizures in patients treated with CHANTIX [see Warnings and Precautions (5.2)].
There have been post-marketing reports of patients experiencing increased intoxicating effects of alcohol while taking CHANTIX. Some reported neuropsychiatric events, including unusual and sometimes aggressive behavior [see Warnings and Precautions (5.1) and (5.3)].
There have been reports of hypersensitivity reactions, including angioedema [see Warnings and Precautions (5.6)].
There have also been reports of serious skin reactions, including Stevens- Johnson Syndrome and erythema multiforme, in patients taking CHANTIX [see Warnings and Precautions (5.7)].
There have been reports of myocardial infarction (MI) and cerebrovascular accident (CVA) including ischemic and hemorrhagic events in patients taking CHANTIX. In the majority of the reported cases, patients had pre-existing cardiovascular disease and/or other risk factors. Although smoking is a risk factor for MI and CVA, based on temporal relationship between medication use and events, a contributory role of varenicline cannot be ruled out.
8 Use in Specific Populations
-
•Pregnancy: CHANTIX should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. (8.1)
-
•Nursing Mothers: Discontinue drug or nursing taking into consideration importance of drug to mother. (8.3)
-
•Pediatric Use: Safety and effectiveness not established. (8.4)
-
•Renal Impairment: Dosage adjustment is required for severe renal impairment. (2.2, 8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reactions rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During the premarketing development of CHANTIX, over 4500 subjects were exposed to CHANTIX, with over 450 treated for at least 24 weeks and approximately 100 for a year. Most study participants were treated for 12 weeks or less.
The most common adverse event associated with CHANTIX treatment is nausea, occurring in 30% of patients treated at the recommended dose, compared with 10% in patients taking a comparable placebo regimen [see Warnings and Precautions (5.6)].
Table 5 shows the adverse events for CHANTIX and placebo in the 12- week fixed dose premarketing studies with titration in the first week [Studies 2 (titrated arm only), 4, and 5]. Adverse events were categorized using the Medical Dictionary for Regulatory Activities (MedDRA, Version 7.1).
MedDRA High Level Group Terms (HLGT) reported in ≥ 5% of patients in the CHANTIX 1 mg twice daily dose group, and more commonly than in the placebo group, are listed, along with subordinate Preferred Terms (PT) reported in ≥ 1% of CHANTIX patients (and at least 0.5% more frequent than placebo). Closely related Preferred Terms such as 'Insomnia', 'Initial insomnia', 'Middle insomnia', 'Early morning awakening' were grouped, but individual patients reporting two or more grouped events are only counted once.
|
SYSTEM ORGAN CLASS
High Level Group Term |
CHANTIX
0.5 mg BID |
CHANTIX
1 mg BID |
Placebo |
|---|---|---|---|
| Preferred Term | N=129 | N=821 | N=805 |
|
GASTROINTESTINAL (GI) |
|||
|
GI Signs and Symptoms |
|||
|
Nausea |
16 |
30 |
10 |
|
Abdominal Pain Includes PTs Abdominal (pain, pain upper, pain lower, discomfort, tenderness, distension) and Stomach discomfort
|
5 |
7 |
5 |
|
Flatulence |
9 |
6 |
3 |
|
Dyspepsia |
5 |
5 |
3 |
|
Vomiting |
1 |
5 |
2 |
|
GI Motility/Defecation Conditions |
|||
|
Constipation |
5 |
8 |
3 |
|
Gastroesophageal reflux disease |
1 |
1 |
0 |
|
Salivary Gland Conditions |
|||
|
Dry mouth |
4 |
6 |
4 |
|
PSYCHIATRIC DISORDERS |
|||
|
Sleep Disorder/Disturbances |
|||
|
Insomnia Includes PTs Insomnia/Initial insomnia/Middle insomnia/Early morning awakening
|
19 |
18 |
13 |
|
Abnormal dreams |
9 |
13 |
5 |
|
Sleep disorder |
2 |
5 |
3 |
|
Nightmare |
2 |
1 |
0 |
|
NERVOUS SYSTEM |
|||
|
Headaches |
|||
|
Headache |
19 |
15 |
13 |
|
Neurological Disorders NEC |
|||
|
Dysgeusia |
8 |
5 |
4 |
|
Somnolence |
3 |
3 |
2 |
|
Lethargy |
2 |
1 |
0 |
|
GENERAL DISORDERS |
|||
|
General Disorders NEC |
|||
|
Fatigue/Malaise/Asthenia |
4 |
7 |
6 |
|
RESPIR/THORACIC/MEDIAST |
|||
|
Respiratory Disorders NEC |
|||
|
Rhinorrhea |
0 |
1 |
0 |
|
Dyspnea |
2 |
1 |
1 |
|
Upper Respiratory Tract Disorder |
7 |
5 |
4 |
|
SKIN/SUBCUTANEOUS TISSUE |
|||
|
Epidermal and Dermal Conditions |
|||
|
Rash |
1 |
3 |
2 |
|
Pruritis |
0 |
1 |
1 |
|
METABOLISM & NUTRITION |
|||
|
Appetite/General Nutrit. Disorders |
|||
|
Increased appetite |
4 |
3 |
2 |
|
Decreased appetite/Anorexia |
1 |
2 |
1 |
The overall pattern and frequency of adverse events during the longer-term premarketing trials was similar to those described in Table 5, though several of the most common events were reported by a greater proportion of patients with long-term use (e.g., nausea was reported in 40% of patients treated with CHANTIX 1 mg twice daily in a one-year study, compared to 8% of placebo-treated patients).
Following is a list of treatment-emergent adverse events reported by patients treated with CHANTIX during all premarketing clinical trials and updated based on pooled data from 18 placebo-controlled pre- and post-marketing studies, including approximately 5,000 patients treated with varenicline. Adverse events were categorized using MedDRA, Version 16.0. The listing does not include those events already listed in the previous tables or elsewhere in labeling, those events for which a drug cause was remote, those events which were so general as to be uninformative, and those events reported only once which did not have a substantial probability of being acutely life-threatening.
Blood and Lymphatic System Disorders. Infrequent: anemia, lymphadenopathy. Rare: leukocytosis, splenomegaly, thrombocytopenia.
Cardiac Disorders. Infrequent: angina pectoris, myocardial infarction, palpitations, tachycardia. Rare: acute coronary syndrome, arrhythmia, atrial fibrillation, bradycardia, cardiac flutter, cor pulmonale, coronary artery disease, ventricular extrasystoles.
Ear and Labyrinth Disorders. Infrequent: tinnitus, vertigo. Rare: deafness, Meniere's disease.
Endocrine Disorders. Infrequent: thyroid gland disorders.
Eye Disorders. Infrequent: conjunctivitis, eye irritation, eye pain, vision blurred, visual impairment. Rare: blindness transient, cataract subcapsular, dry eye, night blindness, ocular vascular disorder, photophobia, vitreous floaters.
Gastrointestinal Disorders. Frequent: diarrhea, toothache. Infrequent: dysphagia, eructation, gastritis, gastrointestinal hemorrhage, mouth ulceration. Rare: enterocolitis, esophagitis, gastric ulcer, intestinal obstruction, pancreatitis acute.
General Disorders and Administration Site Conditions. Frequent: chest pain. Infrequent: chest discomfort, chills, edema, influenza-like illness, pyrexia.
Hepatobiliary Disorders. Rare: gall bladder disorder.
Investigations. Frequent: liver function test abnormal, weight increased. Infrequent: electrocardiogram abnormal. Rare: muscle enzyme increased, urine analysis abnormal.
Metabolism and Nutrition Disorders. Infrequent: diabetes mellitus, hypoglycemia. Rare: hyperlipidemia, hypokalemia.
Musculoskeletal and Connective Tissue Disorders. Frequent: arthralgia, back pain, myalgia. Infrequent: arthritis, muscle cramp, musculoskeletal pain. Rare: myositis, osteoporosis.
Nervous System Disorders. Frequent: disturbance in attention, dizziness. Infrequent: amnesia, convulsion, migraine, parosmia, syncope, tremor. Rare: balance disorder, cerebrovascular accident, dysarthria, mental impairment, multiple sclerosis, VIIth nerve paralysis, nystagmus, psychomotor hyperactivity, psychomotor skills impaired, restless legs syndrome, sensory disturbance, transient ischemic attack, visual field defect.
Psychiatric Disorders. Infrequent: dissociation, libido decreased, mood swings, thinking abnormal. Rare: bradyphrenia, disorientation, euphoric mood.
Renal and Urinary Disorders. Infrequent: nocturia, pollakiuria, urine abnormality. Rare: nephrolithiasis, polyuria, renal failure acute, urethral syndrome, urinary retention.
Reproductive System and Breast Disorders. Frequent: menstrual disorder. Infrequent: erectile dysfunction. Rare: sexual dysfunction.
Respiratory, Thoracic and Mediastinal Disorders. Frequent: respiratory disorders. Infrequent: asthma, epistaxis, rhinitis allergic, upper respiratory tract inflammation. Rare: pleurisy, pulmonary embolism.
Skin and Subcutaneous Tissue Disorders. Infrequent: acne, dry skin, eczema, erythema, hyperhidrosis, urticaria. Rare: photosensitivity reaction, psoriasis.
Vascular Disorders. Infrequent: hot flush. Rare: thrombosis.
CHANTIX has also been studied in postmarketing trials including (1) a trial conducted in patients with chronic obstructive pulmonary disease (COPD), (2) a trial conducted in generally healthy patients (similar to those in the premarketing studies) in which they were allowed to select a quit date between days 8 and 35 of treatment ("alternative quit date instruction trial"), (3) a trial conducted in patients who did not succeed in stopping smoking during prior CHANTIX therapy, or who relapsed after treatment ("re-treatment trial"), (4) a trial conducted in patients with stable cardiovascular disease, (5) a trial conducted in patients with stable schizophrenia or schizoaffective disorder and (6) a trial conducted in patients with major depressive disorder.
Adverse events in the trial of patients with COPD, in the alternative quit date instruction trial, were quantitatively and qualitatively similar to those observed in premarketing studies. In the re-treatment trial, the profile of common adverse events was similar to that previously reported, but, in addition, varenicline-treated patients also commonly reported diarrhea (6% vs 4% in placebo-treated patients), depressed mood disorders and disturbances (6% vs 1%), and other mood disorders and disturbances (5% vs 2%).
In the trial of patients with stable cardiovascular disease, more types and a greater number of cardiovascular events were reported compared to premarketing studies. Treatment-emergent (on-treatment or 30 days after treatment) cardiovascular events reported with a frequency ≥ 1% in either treatment group in this study were angina pectoris (3.7% and 2.0% for varenicline and placebo, respectively), chest pain (2.5% vs. 2.3%), peripheral edema (2.0% vs. 1.1%), hypertension (1.4% vs. 2.6%), and palpitations (0.6 % vs. 1.1%). Deaths and serious cardiovascular events occurring over the 52 weeks of the study (treatment emergent and non-treatment emergent) were adjudicated by a blinded, independent committee. The following treatment-emergent adjudicated events occurred with a frequency ≥1% in either treatment group: nonfatal MI (1.1% vs. 0.3% for varenicline and placebo, respectively), and hospitalization for angina pectoris (0.6% vs. 1.1%). During non-treatment follow up to 52 weeks, the adjudicated events included need for coronary revascularization (2.0% vs. 0.6%), hospitalization for angina pectoris (1.7% vs. 1.1%), and new diagnosis of peripheral vascular disease (PVD) or admission for a PVD procedure (1.4% vs. 0.6%). Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina. Cardiovascular death occurred in 0.3% of patients in the varenicline arm and 0.6% of patients in the placebo arm over the course of the 52-week study.
In the trial of patients with stable schizophrenia or schizoaffective disorder, 128 smokers on antipsychotic medication were randomized 2:1 to varenicline (1 mg twice daily) or placebo for 12 weeks with 12-week non-drug follow-up. The most common adverse events in patients taking varenicline were nausea (24% vs. 14.0% on placebo), headache (11% vs. 19% on placebo) and vomiting (11% vs. 9% on placebo). Among reported neuropsychiatric adverse events, insomnia was the only event that occurred in either treatment group in ≥5% of subjects at a rate higher in the varenicline group than in placebo (10% vs. 5%). These common and neuropsychiatric adverse events occurred on treatment or within 30 days after the last dose of study drug. There was no consistent worsening of schizophrenia in either treatment group as measured by the Positive and Negative Syndrome Scale. There were no overall changes in extra-pyramidal signs, as measured by the Simpson-Angus Rating Scale. The Columbia-Suicide Severity Rating Scale was administered at baseline and at clinic visits during the treatment and non-treatment follow-up phases. Over half of the patients had a lifetime history of suicidal behavior and/or ideation (62% on varenicline vs. 51% on placebo), but at baseline, no patients in the varenicline group reported suicidal behavior and/or ideation vs. one patient in the placebo group (2%). Suicidal behavior and/or ideation were reported in 11% of the varenicline-treated and 9% of the placebo-treated patients during the treatment phase. During the post-treatment phase, suicidal behavior and/or ideation were reported in 11% of patients in the varenicline group and 5% of patients in the placebo group. Many of the patients reporting suicidal behavior and ideation in the follow-up phase had not reported such experiences in the treatment phase. However, no new suicidal ideation or behavior emerged in either treatment group shortly (within one week) after treatment discontinuation (a phenomenon noted in post-marketing reporting). There were no completed suicides. There was one suicide attempt in a varenicline-treated patient. The limited data available from this single smoking cessation study are not sufficient to allow conclusions to be drawn.
In the trial of patients with major depressive disorder, the most common adverse events (≥ 10%) in subjects taking varenicline were nausea (27% vs. 10% on placebo), headache (17 vs 11%), abnormal dreams (11% vs 8%), insomnia (11% vs 5%) and irritability (11% vs. 8%). Additionally, the following psychiatric AEs were reported in ≥ 2% of patients in either treatment group (varenicline or placebo, respectively): anxiety (7% vs. 9%), agitation (7% vs. 4%), depressed mood disorders and disturbances (11% vs. 9%), tension (4% vs. 3%), hostility (2% vs. 0.4%) and restlessness (2% vs. 2%). Patients treated with varenicline were more likely than patients treated with placebo to report one of various events related to hostility and aggression (3% vs 1%). Psychiatric scales showed no differences between the varenicline and placebo groups and no overall worsening of depression during the study in either treatment group. The percentage of subjects with suicidal ideation and/or behavior was similar between the varenicline and placebo groups during treatment (6% and 8%, respectively) and the non-treatment follow-up (6% and 6%, respectively). There was one event of intentional self-injury/possible suicide attempt during treatment (Day 73) in a subject in the placebo group. Suicide could not be ruled out in one subject who died by an overdose of illicit drugs 76 days after last dose of study drug in the varenicline group.
17 Patient Counseling Information
16 How Supplied/storage and Handling
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with "Pfizer" on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with "Pfizer" on one side and "CHX 1.0" on the other side. CHANTIX is supplied in the following package configurations:
| Description | NDC | |
|---|---|---|
|
Starting 4-week card: 0.5 mg × 11 tablets and 1 mg × 42 tablets |
NDC 63187-618-53 |
|
|
Starting Month Box: 0.5 mg × 11 tablets and 1 mg × 42 tablets |
NDC 63187-618-53 |
Warning: Serious Neuropsychiatric Events
Serious neuropsychiatric events including, but not limited to, depression, suicidal ideation, suicide attempt, and completed suicide have been reported in patients taking CHANTIX. Some reported cases may have been complicated by the symptoms of nicotine withdrawal in patients who stopped smoking. Depressed mood may be a symptom of nicotine withdrawal. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these symptoms have occurred in patients taking CHANTIX who continued to smoke.
All patients being treated with CHANTIX should be observed for neuropsychiatric symptoms including changes in behavior, hostility, agitation, depressed mood, and suicide-related events, including ideation, behavior, and attempted suicide. These symptoms, as well as worsening of pre-existing psychiatric illness and completed suicide, have been reported in some patients attempting to quit smoking while taking CHANTIX in the postmarketing experience. When symptoms were reported, most were during CHANTIX treatment, but some were following discontinuation of CHANTIX therapy.
These events have occurred in patients with and without pre-existing psychiatric disease. Patients with serious psychiatric illness such as schizophrenia, bipolar disorder, and major depressive disorder did not participate in the premarketing studies of CHANTIX.
Advise patients and caregivers that the patient should stop taking CHANTIX and contact a healthcare provider immediately if agitation, hostility, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. In many postmarketing cases, resolution of symptoms after discontinuation of CHANTIX was reported, although in some cases the symptoms persisted; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.
The risks of CHANTIX should be weighed against the benefits of its use. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo. The health benefits of quitting smoking are immediate and substantial. [see Warnings and Precautions (5.1) and Adverse Reactions (6.2)]
14.5 Subjects With Major Depressive Disorder
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 18 to 75 years with major depressive disorder without psychotic features (DSM-IV TR). If on medication, subjects were to be on a stable antidepressant regimen for at least two months. If not on medication, subjects were to have experienced a major depressive episode in the past 2 years, which was successfully treated. Subjects were randomized to CHANTIX 1 mg twice daily (n=256) or placebo (n=269) for a treatment of 12 weeks and then followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (36%) compared to subjects treated with placebo (16%) and from week 9 through 52 (20%) compared to subjects treated with placebo (10%).
| Weeks 9 through 12 | Weeks 9 through 52 | |||
|---|---|---|---|---|
|
CHANTIX
1 mg BID |
Placebo |
CHANTIX
1 mg BID |
Placebo | |
| BID = twice daily | ||||
|
MDD Study |
36% |
16% |
20% |
10% |
5.1 Neuropsychiatric Symptoms and Suicidality
Serious neuropsychiatric symptoms have been reported in patients being treated with CHANTIX [see Boxed Warning and Adverse Reactions (6.2)]. These postmarketing reports have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Some reported cases may have been complicated by the symptoms of nicotine withdrawal in patients who stopped smoking. Depressed mood may be a symptom of nicotine withdrawal. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these symptoms have occurred in patients taking CHANTIX who continued to smoke. When symptoms were reported, most were during CHANTIX treatment, but some were following discontinuation of CHANTIX therapy.
These events have occurred in patients with and without pre-existing psychiatric disease; some patients have experienced worsening of their psychiatric illnesses. All patients being treated with CHANTIX should be observed for neuropsychiatric symptoms or worsening of pre-existing psychiatric illness. Patients with serious psychiatric illness such as schizophrenia, bipolar disorder, and major depressive disorder did not participate in the premarketing studies of CHANTIX. Limited safety data are available from post-marketing smoking cessation studies in two patient groups: 1) patients with major depressive disorder, and 2) patients with stable schizophrenia or schizoaffective disorder [see Adverse Reactions (6.1) , Clinical Studies (14.5) ] .
Some reported neuropsychiatric events, including unusual and sometimes aggressive behavior directed to oneself or others, may have been worsened by concomitant use of alcohol [see Interaction with Alcohol (5.3) , Adverse Reactions (6.2) ] .
Advise patients and caregivers that the patient should stop taking CHANTIX and contact a healthcare provider immediately if agitation, depressed mood, changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. In many postmarketing cases, resolution of symptoms after discontinuation of CHANTIX was reported, although in some cases the symptoms persisted, therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.
The risks of CHANTIX should be weighed against the benefits of its use. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo. The health benefits of quitting smoking are immediate and substantial.
-
Since the initial signal of neuropsychiatric symptoms and suicidality emerged, additional analyses and studies have been conducted to further evaluate this association.
5.6 Angioedema and Hypersensitivity Reactions
There have been postmarketing reports of hypersensitivity reactions including angioedema in patients treated with CHANTIX [see Adverse Reactions (6.2), and Patient Counseling Information (17)]. Clinical signs included swelling of the face, mouth (tongue, lips, and gums), extremities, and neck (throat and larynx). There were infrequent reports of life-threatening angioedema requiring emergent medical attention due to respiratory compromise. Instruct patients to discontinue CHANTIX and immediately seek medical care if they experience these symptoms.
7.1 Use With Other Drugs for Smoking Cessation
Safety and efficacy of CHANTIX in combination with other smoking cessation therapies have not been studied.
7.2 Effect of Smoking Cessation On Other Drugs
Physiological changes resulting from smoking cessation, with or without treatment with CHANTIX, may alter the pharmacokinetics or pharmacodynamics of certain drugs (e.g., theophylline, warfarin, insulin) for which dosage adjustment may be necessary.
14.6 Alternative Instructions for Setting A Quit Date
CHANTIX was evaluated in a double-blind, placebo-controlled trial where patients were instructed to select a target quit date between Day 8 and Day 35 of treatment. Subjects were randomized 3:1 to CHANTIX 1 mg twice daily (n=486) or placebo (n=165) for 12 weeks of treatment and followed for another 12 weeks post-treatment. Patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (54%) compared to patients treated with placebo (19%) and from weeks 9 through 24 (35%) compared to subjects treated with placebo (13%).
Principal Display Panel 0.5 / 1 Mg Starting Pack Carton
Contains:
1 Starting Week (0.5 mg* x 11 tablets)
3 Continuing Weeks (1 mg† x 42 tablets)
NDC 63187-618-53
Rx only
CHANTIX
®
(varenicline) TABLETS
STARTING MONTH BOX
(Your first 4 weeks)
Use the Starting Week (green) blister card
first when beginning to take Chantix.
ALWAYS DISPENSE WITH ENCLOSED MEDICATION GUIDE
14.4 Subjects With Cardiovascular and Chronic Obstructive Pulmonary Disease
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 35 to 75 years with stable, documented cardiovascular disease (diagnoses other than, or in addition to, hypertension) that had been diagnosed for more than 2 months. Subjects were randomized to CHANTIX 1 mg twice daily (n=353) or placebo (n=350) for a treatment of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (47%) compared to subjects treated with placebo (14%) and from week 9 through 52 (20%) compared to subjects treated with placebo (7%).
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged ≥ 35 years with mild-to-moderate COPD with post-bronchodilator FEV1/FVC <70% and FEV1 ≥ 50% of predicted normal value. Subjects were randomized to CHANTIX 1 mg twice daily (n=223) or placebo (n=237) for a treatment of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (41%) compared to subjects treated with placebo (9%) and from week 9 through 52 (19%) compared to subjects treated with placebo (6%).
| Weeks 9 through 12 | Weeks 9 through 52 | |||
|---|---|---|---|---|
|
CHANTIX
1 mg BID |
Placebo |
CHANTIX
1 mg BID |
Placebo | |
| BID = twice daily | ||||
|
CVD Study |
47% |
14% |
20% |
7% |
|
COPD Study |
41% |
9% |
19% |
6% |
Structured Label Content
Section 34077-8 (34077-8)
Pregnancy Category C.
There are no adequate and well-controlled studies of CHANTIX use in pregnant women. In animal studies, CHANTIX caused decreased fetal weights, increased auditory startle response, and decreased fertility in offspring. CHANTIX should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
In reproductive and developmental toxicity studies, pregnant rats and rabbits received varenicline succinate during organogenesis at oral doses up to 15 and 30 mg/kg/day, respectively. These exposures were 36 (rats) and 50 (rabbits) times the human exposure (based on AUC) at the maximum recommended human dose (MRHD) of 1 mg twice daily. While no fetal structural abnormalities occurred in either species, reduced fetal weights occurred in rabbits at the highest dose (exposures 50 times the human exposure at the MRHD based on AUC). Fetal weight reduction did not occur at animal exposures 23 times the human exposure at the MRHD based on AUC.
In a pre- and postnatal development study, pregnant rats received up to 15 mg/kg/day of oral varenicline succinate from organogenesis through lactation. These resulted in exposures up to 36 times the human exposure (based on AUC) at the MRHD of 1 mg twice daily. Decreased fertility and increased auditory startle response occurred in offspring.
Section 42229-5 (42229-5)
Patients with Impaired Renal Function: No dosage adjustment is necessary for patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30 mL/min), the recommended starting dose of CHANTIX is 0.5 mg once daily. The dose may then be titrated as needed to a maximum dose of 0.5 mg twice a day. For patients with end-stage renal disease undergoing hemodialysis, a maximum dose of 0.5 mg once daily may be administered if tolerated [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) (see USP Controlled Room Temperature).
5.8 Nausea
Nausea was the most common adverse reaction reported with CHANTIX treatment. Nausea was generally described as mild or moderate and often transient; however, for some patients, it was persistent over several months. The incidence of nausea was dose-dependent. Initial dose-titration was beneficial in reducing the occurrence of nausea. For patients treated to the maximum recommended dose of 1 mg twice daily following initial dosage titration, the incidence of nausea was 30% compared with 10% in patients taking a comparable placebo regimen. In patients taking CHANTIX 0.5 mg twice daily following initial titration, the incidence was 16% compared with 11% for placebo. Approximately 3% of patients treated with CHANTIX 1 mg twice daily in studies involving 12 weeks of treatment discontinued treatment prematurely because of nausea. For patients with intolerable nausea, a dose reduction should be considered.
5.2 Seizures
During clinical trials and the post-marketing experience, there have been reports of seizures in patients treated with CHANTIX. Some patients had no history of seizures, whereas others had a history of seizure disorder that was remote or well-controlled. In most cases, the seizure occurred within the first month of therapy. Weigh this potential risk against the potential benefits before prescribing CHANTIX in patients with a history of seizures or other factors that can lower the seizure threshold. Advise patients to discontinue CHANTIX and contact a healthcare provider immediately if they experience a seizure while on treatment [see Adverse Reactions (6.2) ].
10 Overdosage (10 OVERDOSAGE)
In case of overdose, standard supportive measures should be instituted as required.
Varenicline has been shown to be dialyzed in patients with end stage renal disease [see Clinical Pharmacology (12.3)], however, there is no experience in dialysis following overdose.
11 Description (11 DESCRIPTION)
CHANTIX tablets contain varenicline (as the tartrate salt), which is a partial agonist selective for α4β2 nicotinic acetylcholine receptor subtypes.
Varenicline, as the tartrate salt, is a powder which is a white to off-white to slightly yellow solid with the following chemical name: 7,8,9,10-tetrahydro-6,10-methano-6H-pyrazino[2,3- h][3]benzazepine, (2R,3R)-2,3-dihydroxybutanedioate (1:1). It is highly soluble in water. Varenicline tartrate has a molecular weight of 361.35 Daltons, and a molecular formula of C13H13N3 ∙ C4H6O6. The chemical structure is:
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with "Pfizer" on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with "Pfizer" on one side and "CHX 1.0" on the other side. Each 0.5 mg CHANTIX tablet contains 0.85 mg of varenicline tartrate equivalent to 0.5 mg of varenicline free base; each 1mg CHANTIX tablet contains 1.71 mg of varenicline tartrate equivalent to 1 mg of varenicline free base. The following inactive ingredients are included in the tablets: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry® White (for 0.5 mg), Opadry® Blue (for 1 mg), and Opadry® Clear.
Medication Guide (MEDICATION GUIDE)
CHANTIX® (CHANT-iks)
(varenicline) Tablets
What is the most important information I should know about CHANTIX?
Some people have had serious side effects while using CHANTIX to help them quit smoking, including:
New or worse mental health problems, such as changes in behavior, hostility, agitation, depressed mood, and suicidal thoughts or actions. Some people had these symptoms when they began taking CHANTIX, and others developed them after several weeks of treatment, or after stopping CHANTIX.
Before taking CHANTIX, tell your doctor if you have ever had depression or other mental health problems. You should also tell your doctor about any symptoms you had during other times you tried to quit smoking, with or without CHANTIX.
Stop taking CHANTIX and call your doctor right away if you, your family, or caregiver notice agitation, hostility, depression or changes in your behavior or thinking that are not typical for you, or you develop any of the following symptoms:
-
•thoughts about suicide or dying, or attempts to commit suicide
-
•new or worse depression, anxiety, or panic attacks
-
•feeling very agitated or restless
-
•acting aggressive, being angry, or violent
-
•acting on dangerous impulses
-
•an extreme increase in activity and talking (mania)
-
•abnormal thoughts or sensations
-
•seeing or hearing things that are not there (hallucinations)
-
•feeling people are against you (paranoia)
-
•feeling confused
-
•other unusual changes in behavior or mood
When you try to quit smoking, with or without CHANTIX, you may have symptoms that may be due to nicotine withdrawal, including urge to smoke, depressed mood, trouble sleeping, irritability, frustration, anger, feeling anxious, difficulty concentrating, restlessness, decreased heart rate, and increased appetite or weight gain. Some people have even experienced suicidal thoughts when trying to quit smoking without medication. Sometimes quitting smoking can lead to worsening of mental health problems that you already have, such as depression.
-
See " What are the possible side effects of CHANTIX? " for more information about other side effects.
What is CHANTIX?
CHANTIX is a prescription medicine to help people stop smoking.
Quitting smoking can lower your chances of having lung disease, heart disease or getting certain types of cancer that are related to smoking.
It is not known if CHANTIX is safe and effective in children.
It is not known if CHANTIX is safe and effective when used with other stop smoking medicines.
Who should not take CHANTIX?
Do not take CHANTIX if you have had a serious allergic or skin reaction to CHANTIX. Symptoms may include:
-
•swelling of the face, mouth (tongue, lips, gums), throat or neck
-
•trouble breathing
-
•rash, with peeling skin
-
•blisters in your mouth
What should I tell my doctor before taking CHANTIX?
See "What is the most important information I should know about CHANTIX?"
Before you take CHANTIX, tell your doctor if you:
-
•use other treatments to quit smoking. Using CHANTIX with a nicotine patch may cause nausea, vomiting, headache, dizziness, upset stomach, and tiredness to happen more often than if you just use a nicotine patch alone.
-
•have kidney problems or get kidney dialysis. Your doctor may prescribe a lower dose of CHANTIX for you.
-
•have a history of seizures
-
•drink alcohol
-
•have heart or blood vessel problems
-
•have any other medical conditions
-
•are pregnant or plan to become pregnant. It is not known if CHANTIX will harm your unborn baby.
-
•are breastfeeding. It is not known if CHANTIX passes into breast milk. You and your doctor should decide if you will breastfeed or take CHANTIX. You should not do both.
Tell your doctor about all the medicines you take, including prescription and over the counter medicines, vitamins and herbal supplements. Your doctor may need to change the dose of some of your medicines when you stop smoking.
You should not use CHANTIX while using other medicines to quit smoking. Tell your doctor if you use other treatments to quit smoking.
Know the medicines you take. Keep a list of them with you to show your doctor and pharmacist when you get a new medicine.
How should I take CHANTIX?
-
•There are 2 ways that you can use CHANTIX to help you quit smoking. Talk to your doctor about the following 2 ways to use CHANTIX:
-
Choose a quit date when you will stop smoking. Start taking CHANTIX 1 week (7 days) before your quit date.
-
OR
-
Start taking CHANTIX before you choose a quit date. Pick a date to quit smoking that is between days 8 and 35 of treatment.
-
Starting CHANTIX before your quit date gives CHANTIX time to build up in your body. You can keep smoking during this time. Take CHANTIX exactly as prescribed by your doctor.
-
•CHANTIX comes as a white tablet (0.5 mg) and a blue tablet (1 mg). You start with the white tablet and then usually go to the blue tablet. See the chart below for dosing instructions for adults.
|
Day 1 to Day 3 |
|
|
Day 4 to Day 7 |
|
|
Day 8 to end of treatment |
|
-
•Make sure that you try to stop smoking on your quit date. If you slip-up and smoke, try again. Some people need to take CHANTIX for a few weeks for CHANTIX to work best.
-
•Most people will take CHANTIX for up to 12 weeks. If you have completely quit smoking by 12 weeks, your doctor may prescribe CHANTIX for another 12 weeks to help you stay cigarette-free.
-
•Take CHANTIX after eating and with a full glass (8 ounces) of water.
-
•This dosing schedule may not be right for everyone. Talk to your doctor if you are having side effects such as nausea, strange dreams, or sleep problems. Your doctor may want to reduce your dose.
-
•If you miss a dose of CHANTIX, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Just take your next dose at your regular time.
What should I avoid while taking CHANTIX?
-
•Use caution when driving or operating machinery until you know how CHANTIX affects you. CHANTIX may make you feel sleepy, dizzy, or have trouble concentrating, making it hard to drive or perform other activities safely.
-
•Decrease the amount of alcoholic beverages that you drink during treatment with CHANTIX until you know if CHANTIX affects your ability to tolerate alcohol. Some people have experienced the following when drinking alcohol during treatment with CHANTIX:
-
•increased drunkenness (intoxication)
-
•unusual or sometimes aggressive behavior
-
•no memory of things that have happened
-
What are the possible side effects of CHANTIX?
Serious side effects of CHANTIX may include:
-
•See "What is the most important information I should know about CHANTIX?"
-
•Seizures. Some people have had seizures during treatment with CHANTIX. In most cases, the seizures have happened during the first month of treatment with CHANTIX. If you have a seizure during treatment with CHANTIX, stop taking CHANTIX and contact your healthcare provider right away.
-
•New or worse heart or blood vessel (cardiovascular) problems, mostly in people, who already have cardiovascular problems. Tell your doctor if you have any changes in symptoms during treatment with CHANTIX.
Get emergency medical help right away if you have any of the following symptoms of a heart attack, including:
-
•chest discomfort (uncomfortable pressure, squeezing, fullness or pain) that lasts more than a few minutes, or that goes away and comes back
-
•pain or discomfort in one or both arms, back, neck, jaw or stomach
-
•shortness of breath, sweating, nausea, vomiting, or feeling lightheaded associated with chest discomfort
-
-
•Allergic reactions can happen with CHANTIX. Some of these allergic reactions can be life-threatening.
-
•Serious skin reactions, including rash, swelling, redness, and peeling of the skin. Some of these skin reactions can become life-threatening.
Stop taking CHANTIX and get medical help right away if you have any of the following symptoms:
-
•swelling of the face, mouth (tongue, lips, and gums), throat or neck
-
•trouble breathing
-
•rash with peeling skin
-
•blisters in your mouth
The most common side effects of CHANTIX include:
-
•nausea
-
•sleep problems (trouble sleeping or vivid, unusual, or strange dreams)
-
•constipation
-
•gas
-
•vomiting
Tell your doctor about side effects that bother you or that do not go away.
These are not all the side effects of CHANTIX. Ask your doctor or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store CHANTIX?
-
•Store CHANTIX at room temperature, between 68°F to 77°F (20°C to 25°C).
-
•Keep CHANTIX and all medicines out of the reach of children.
General information about CHANTIX
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CHANTIX for a condition for which it was not prescribed. Do not give your CHANTIX to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about CHANTIX that is written for healthcare professionals.
For more information about CHANTIX and tips on how to quit smoking, go to www.CHANTIX.com or call 1-877-242-6849.
If you are motivated to quit smoking and did not succeed during prior CHANTIX treatment for reasons other than side effects, or if you returned to smoking after treatment, speak with your doctor about whether another course of CHANTIX therapy may be right for you.
What are the ingredients in CHANTIX?
Active ingredient: varenicline tartrate
Inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry ® White (for 0.5 mg), Opadry ® Blue (for 1 mg), and Opadry® Clear.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
October 2014
LAB-0328-13.0
Relabeled by:
Proficient Rx LP
Thousand Oaks, CA 91320
8.4 Pediatric Use
Safety and effectiveness of CHANTIX in pediatric patients have not been established.
8.5 Geriatric Use
A combined single- and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once daily or twice daily to 16 healthy elderly male and female smokers (aged 65–75 yrs) for 7 consecutive days was similar to that of younger subjects. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.2) ].
No dosage adjustment is recommended for elderly patients.
14.2 Urge to Smoke
Based on responses to the Brief Questionnaire of Smoking Urges and the Minnesota Nicotine Withdrawal scale "urge to smoke" item, CHANTIX reduced urge to smoke compared to placebo.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of CHANTIX in smoking cessation was demonstrated in six clinical trials in which a total of 3659 chronic cigarette smokers (≥10 cigarettes per day) were treated with CHANTIX. In all clinical studies, abstinence from smoking was determined by patient self-report and verified by measurement of exhaled carbon monoxide (CO≤10 ppm) at weekly visits. Among the CHANTIX-treated patients enrolled in these studies, the completion rate was 65%. Except for the dose-ranging study (Study 1) and the maintenance of abstinence study (Study 6), patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. Most patients enrolled in these trials were white (79–96%). All studies enrolled almost equal numbers of men and women. The average age of patients in these studies was 43 years. Patients on average had smoked about 21 cigarettes per day for an average of approximately 25 years. Patients set a date to stop smoking (target quit date) with dosing starting 1 week before this date.
Three additional studies were conducted in patients with cardiovascular disease, in patients with chronic obstructive pulmonary disease [see Clinical Studies (14.4)], and in patients instructed to select their quit date within days 8 and 35 of treatment [see Clinical Studies (14.5)].
In all studies, patients were provided with an educational booklet on smoking cessation and received up to 10 minutes of smoking cessation counseling at each weekly treatment visit according to Agency for Healthcare Research and Quality guidelines.
4 Contraindications (4 CONTRAINDICATIONS)
CHANTIX is contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to CHANTIX.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling:
-
•Neuropsychiatric symptoms and suicidality [see Boxed Warning and Warnings and Precautions (5.1)]
-
•Seizures [see Warnings and Precautions (5.2)]
-
•Interaction with Alcohol [see Warnings and Precautions (5.3)]
-
•Accidental injury [see Warnings and Precautions (5.4)]
-
•Cardiovascular Events [see Warnings and Precautions (5.5)]
-
•Angioedema and hypersensitivity reactions [see Warnings and Precautions (5.6)]
-
•Serious skin reactions [see Warnings and Precautions (5.7)]
In the placebo-controlled premarketing studies, the most common adverse events associated with CHANTIX (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting.
The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for CHANTIX, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in CHANTIX-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo).
Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
7 Drug Interactions (7 DRUG INTERACTIONS)
Based on varenicline characteristics and clinical experience to date, CHANTIX has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3)].
8.3 Nursing Mothers
It is not known whether CHANTIX is excreted in human milk. In animal studies varenicline was excreted in milk of lactating animals. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from CHANTIX, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.6 Renal Impairment
Varenicline is substantially eliminated by renal glomerular filtration along with active tubular secretion. Dose reduction is not required in patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30 mL/min), and for patients with end-stage renal disease undergoing hemodialysis, dosage adjustment is needed. [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
5.4 Accidental Injury
There have been postmarketing reports of traffic accidents, near-miss incidents in traffic, or other accidental injuries in patients taking CHANTIX. In some cases, the patients reported somnolence, dizziness, loss of consciousness or difficulty concentrating that resulted in impairment, or concern about potential impairment, in driving or operating machinery. Advise patients to use caution driving or operating machinery or engaging in other potentially hazardous activities until they know how CHANTIX may affect them.
1 Indications and Usage (1 INDICATIONS AND USAGE)
CHANTIX is indicated for use as an aid to smoking cessation treatment.
14.7 Re Treatment Study (14.7 Re-Treatment Study)
CHANTIX was evaluated in a double-blind, placebo-controlled trial of patients who had made a previous attempt to quit smoking with CHANTIX, and either did not succeed in quitting or relapsed after treatment. Subjects were randomized 1:1 to CHANTIX 1 mg twice daily (n=249) or placebo (n=245) for 12 weeks of treatment and followed for 40 weeks post-treatment. Patients included in this study had taken CHANTIX for a smoking-cessation attempt in the past (for a total treatment duration of a minimum of two weeks), at least three months prior to study entry, and had been smoking for at least four weeks.
Patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (45%) compared to patients treated with placebo (12%) and from weeks 9 through 52 (20%) compared to subjects treated with placebo (3%).
| Weeks 9 through 12 | Weeks 9 through 52 | |||
|---|---|---|---|---|
|
CHANTIX
1 mg BID |
Placebo |
CHANTIX
1 mg BID |
Placebo | |
| BID = twice daily | ||||
|
Retreatment Study |
45% |
12% |
20% |
3% |
12.1 Mechanism of Action
Varenicline binds with high affinity and selectivity at α4β2 neuronal nicotinic acetylcholine receptors. The efficacy of CHANTIX in smoking cessation is believed to be the result of varenicline's activity at α4β2 sub-type of the nicotinic receptor where its binding produces agonist activity, while simultaneously preventing nicotine binding to these receptors.
Electrophysiology studies in vitro and neurochemical studies in vivo have shown that varenicline binds to α4β2 neuronal nicotinic acetylcholine receptors and stimulates receptor-mediated activity, but at a significantly lower level than nicotine. Varenicline blocks the ability of nicotine to activate α4β2 receptors and thus to stimulate the central nervous mesolimbic dopamine system, believed to be the neuronal mechanism underlying reinforcement and reward experienced upon smoking. Varenicline is highly selective and binds more potently to α4β2 receptors than to other common nicotinic receptors (>500-fold α3β4, >3500-fold α7, >20,000-fold α1βγδ), or to non-nicotinic receptors and transporters (>2000-fold). Varenicline also binds with moderate affinity (Ki = 350 nM) to the 5-HT3 receptor.
9.1 Controlled Substance
Varenicline is not a controlled substance.
14.3 Long Term Abstinence (14.3 Long-Term Abstinence)
Studies 1 through 5 included 40 weeks of post-treatment follow-up. In each study, CHANTIX-treated patients were more likely to maintain abstinence throughout the follow-up period than were patients treated with placebo (Figure 2, Table 7).
Figure 2: Continuous Abstinence, Weeks 9 through 52
|
CHANTIX
0.5 mg BID |
CHANTIX
1 mg BID |
CHANTIX
Flexible |
Bupropion
SR |
Placebo | |
|---|---|---|---|---|---|
| BID = twice daily | |||||
|
Study 2 |
19% |
23% |
4% |
||
|
Study 3 |
22% |
8% |
|||
|
Study 4 |
21% |
16% |
8% |
||
|
Study 5 |
22% |
14% |
10% |
5.5 Cardiovascular Events
In a placebo-controlled clinical trial of CHANTIX administered to patients with stable cardiovascular disease, with approximately 350 patients per treatment arm, all-cause and cardiovascular mortality was lower in patients treated with CHANTIX, but certain nonfatal cardiovascular events occurred more frequently in patients treated with CHANTIX than in patients treated with placebo [see Clinical Trials Experience (6.1)]. Table 3 below shows the incidence of deaths and of selected nonfatal serious cardiovascular events occurring more frequently in the CHANTIX arm compared to the placebo arm. These events were adjudicated by an independent blinded committee. Nonfatal serious cardiovascular events not listed occurred at the same incidence or more commonly in the placebo arm. Patients with more than one cardiovascular event of the same type are counted only once per row. Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina.
| Mortality and Cardiovascular Events |
CHANTIX
(N=353) n (%) |
Placebo
(N=350) n (%) |
|---|---|---|
|
Mortality (Cardiovascular & All-cause up to 52 wks) |
||
|
Cardiovascular death |
1 (0.3) |
2 (0.6) |
|
All-cause mortality |
2 (0.6) |
5 (1.4) |
|
Nonfatal Cardiovascular Events (rate on CHANTIX > Placebo) |
||
|
Up to 30 days after treatment |
||
|
Nonfatal myocardial infarction |
4 (1.1) |
1 (0.3) |
|
Nonfatal Stroke |
2 (0.6) |
0 (0) |
|
Beyond 30 days after treatment & up to 52 weeks |
||
|
Nonfatal myocardial infarction |
3 (0.8) |
2 (0.6) |
|
Need for coronary revascularization |
7 (2.0) |
2 (0.6) |
|
Hospitalization for angina pectoris |
6 (1.7) |
4 (1.1) |
|
Transient ischemia attack |
1 (0.3) |
0 (0) |
|
New diagnosis of peripheral vascular disease (PVD) or admission for a PVD procedure |
5 (1.4) |
2 (0.6) |
A meta-analysis of 15 clinical trials of ≥ 12 weeks treatment duration, including 7002 patients (4190 CHANTIX, 2812 placebo), was conducted to systematically assess the cardiovascular safety of CHANTIX. The study in patients with stable cardiovascular disease described above was included in the meta-analysis. There were lower rates of all-cause mortality (CHANTIX 6 [0.14%]; placebo 7 [0.25%]) and cardiovascular mortality (CHANTIX 2 [0.05%]; placebo 2 [0.07%]) in the CHANTIX arms compared with the placebo arms in the meta-analysis.
The key cardiovascular safety analysis included occurrence and timing of a composite endpoint of Major Adverse Cardiovascular Events (MACE), defined as cardiovascular death, nonfatal MI, and nonfatal stroke. These events included in the endpoint were adjudicated by a blinded, independent committee. Overall, a small number of MACE occurred in the trials included in the meta-analysis, as described in Table 4. These events occurred primarily in patients with known cardiovascular disease.
|
CHANTIX
N=4190 |
Placebo
N=2812 |
|
|---|---|---|
|
MACE cases, n (%) |
13 (0.31%) |
6 (0.21%) |
|
Patient-years of exposure |
1316 |
839 |
|
Hazard Ratio (95% CI) |
||
|
1.95 (0.79, 4.82) |
||
|
Rate Difference per 1,000 patient-years (95% CI) |
||
|
6.30 (-2.40, 15.10) |
The meta-analysis showed that exposure to CHANTIX resulted in a hazard ratio for MACE of 1.95 (95% confidence interval from 0.79 to 4.82) for patients up to 30 days after treatment; this is equivalent to an estimated increase of 6.3 MACE events per 1,000 patient-years of exposure. The meta-analysis showed higher rates of CV endpoints in patients on CHANTIX relative to placebo across different time frames and pre-specified sensitivity analyses, including various study groupings and CV outcomes. Although these findings were not statistically significant they were consistent. Because the number of events was small overall, the power for finding a statistically significant difference in a signal of this magnitude is low.
CHANTIX was not studied in patients with unstable cardiovascular disease or cardiovascular events occurring within two months before screening. Patients should be advised to notify a health care provider of new or worsening symptoms of cardiovascular disease. The risks of CHANTIX should be weighed against the benefits of its use in smokers with cardiovascular disease. Smoking is an independent and major risk factor for cardiovascular disease. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Seizures: New or worsening seizures have been observed in patients taking CHANTIX. CHANTIX should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. (5.2)
-
•Interaction with alcohol: Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether CHANTIX affects them. (5.3)
-
•Accidental injury: Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how CHANTIX may affect them. (5.4)
-
•Cardiovascular events: A meta-analysis of 15 clinical trials, including a trial in patients with stable cardiovascular disease, demonstrated that while cardiovascular events were infrequent overall, some were reported more frequently in patients treated with CHANTIX. These events occurred primarily in patients with known cardiovascular disease. In both the clinical trial and meta-analysis, all-cause and cardiovascular mortality was lower in patients treated with CHANTIX. Instruct patients to notify their health care providers of new or worsening cardiovascular symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke. (5.5 and 6.1)
-
•Angioedema and hypersensitivity reactions: Such reactions, including angioedema, infrequently life threatening, have been reported. Instruct patients to discontinue CHANTIX and immediately seek medical care if symptoms occur. (5.6 and 6.2)
-
•Serious skin reactions: Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue CHANTIX and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. (5.7 and 6.2)
-
•Nausea: Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. (5.8)
5.7 Serious Skin Reactions
There have been postmarketing reports of rare but serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients using CHANTIX [see Adverse Reactions (6.2)]. As these skin reactions can be life-threatening, instruct patients to stop taking CHANTIX and contact a healthcare provider immediately at the first appearance of a skin rash with mucosal lesions or any other signs of hypersensitivity.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Begin CHANTIX dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin CHANTIX dosing and then quit smoking between days 8 and 35 of treatment. (2.1)
-
•Starting week: 0.5 mg once daily on days 1–3 and 0.5 mg twice daily on days 4–7. (2.1)
-
•Continuing weeks: 1 mg twice daily for a total of 12 weeks. (2.1)
-
•An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. (2.1)
-
•Renal impairment: Reduce the dose in patients with severe renal impairment (estimated creatinine clearance <30 mL/min). (2.2)
-
•Consider dose reduction for patients who cannot tolerate adverse effects. (2.1)
-
•Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. (2.1)
-
•Provide patients with appropriate educational materials and counseling to support the quit attempt. (2.1)
2.1 Usual Dosage for Adults
Smoking cessation therapies are more likely to succeed for patients who are motivated to stop smoking and who are provided additional advice and support. Provide patients with appropriate educational materials and counseling to support the quit attempt.
The patient should set a date to stop smoking. Begin CHANTIX dosing one week before this date. Alternatively, the patient can begin CHANTIX dosing and then quit smoking between days 8 and 35 of treatment.
CHANTIX should be taken after eating and with a full glass of water.
The recommended dose of CHANTIX is 1 mg twice daily following a 1-week titration as follows:
|
Days 1 – 3: |
0.5 mg once daily |
|
Days 4 – 7: |
0.5 mg twice daily |
|
Day 8 – end of treatment: |
1 mg twice daily |
Patients should be treated with CHANTIX for 12 weeks. For patients who have successfully stopped smoking at the end of 12 weeks, an additional course of 12 weeks' treatment with CHANTIX is recommended to further increase the likelihood of long-term abstinence.
Patients who are motivated to quit, and who did not succeed in stopping smoking during prior CHANTIX therapy for reasons other than intolerability due to adverse events or who relapsed after treatment, should be encouraged to make another attempt with CHANTIX once factors contributing to the failed attempt have been identified and addressed.
Consider a temporary or permanent dose reduction in patients who cannot tolerate the adverse effects of CHANTIX.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsular, biconvex tablets: 0.5 mg (white to off-white, debossed with "Pfizer" on one side and "CHX 0.5" on the other side) and 1 mg (light blue, debossed with "Pfizer" on one side and "CHX 1.0" on the other side).
5.3 Interaction With Alcohol (5.3 Interaction with Alcohol)
There have been post-marketing reports of patients experiencing increased intoxicating effects of alcohol while taking CHANTIX. Some cases described unusual and sometimes aggressive behavior, and were often accompanied by amnesia for the events. Advise patients to reduce the amount of alcohol they consume while taking CHANTIX until they know whether CHANTIX affects their tolerance for alcohol [see Adverse Reactions (6.2) ] .
6.2 Postmarketing Experience
The following adverse events have been reported during post-approval use of CHANTIX. Because these events are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
There have been reports of depression, mania, psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide in patients attempting to quit smoking while taking CHANTIX [see Boxed Warning, Warnings and Precautions (5.1)]. Smoking cessation with or without treatment is associated with nicotine withdrawal symptoms and the exacerbation of underlying psychiatric illness. Not all patients had known pre-existing psychiatric illness and not all had discontinued smoking.
There have been post-marketing reports of new or worsening seizures in patients treated with CHANTIX [see Warnings and Precautions (5.2)].
There have been post-marketing reports of patients experiencing increased intoxicating effects of alcohol while taking CHANTIX. Some reported neuropsychiatric events, including unusual and sometimes aggressive behavior [see Warnings and Precautions (5.1) and (5.3)].
There have been reports of hypersensitivity reactions, including angioedema [see Warnings and Precautions (5.6)].
There have also been reports of serious skin reactions, including Stevens- Johnson Syndrome and erythema multiforme, in patients taking CHANTIX [see Warnings and Precautions (5.7)].
There have been reports of myocardial infarction (MI) and cerebrovascular accident (CVA) including ischemic and hemorrhagic events in patients taking CHANTIX. In the majority of the reported cases, patients had pre-existing cardiovascular disease and/or other risk factors. Although smoking is a risk factor for MI and CVA, based on temporal relationship between medication use and events, a contributory role of varenicline cannot be ruled out.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•Pregnancy: CHANTIX should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. (8.1)
-
•Nursing Mothers: Discontinue drug or nursing taking into consideration importance of drug to mother. (8.3)
-
•Pediatric Use: Safety and effectiveness not established. (8.4)
-
•Renal Impairment: Dosage adjustment is required for severe renal impairment. (2.2, 8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reactions rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During the premarketing development of CHANTIX, over 4500 subjects were exposed to CHANTIX, with over 450 treated for at least 24 weeks and approximately 100 for a year. Most study participants were treated for 12 weeks or less.
The most common adverse event associated with CHANTIX treatment is nausea, occurring in 30% of patients treated at the recommended dose, compared with 10% in patients taking a comparable placebo regimen [see Warnings and Precautions (5.6)].
Table 5 shows the adverse events for CHANTIX and placebo in the 12- week fixed dose premarketing studies with titration in the first week [Studies 2 (titrated arm only), 4, and 5]. Adverse events were categorized using the Medical Dictionary for Regulatory Activities (MedDRA, Version 7.1).
MedDRA High Level Group Terms (HLGT) reported in ≥ 5% of patients in the CHANTIX 1 mg twice daily dose group, and more commonly than in the placebo group, are listed, along with subordinate Preferred Terms (PT) reported in ≥ 1% of CHANTIX patients (and at least 0.5% more frequent than placebo). Closely related Preferred Terms such as 'Insomnia', 'Initial insomnia', 'Middle insomnia', 'Early morning awakening' were grouped, but individual patients reporting two or more grouped events are only counted once.
|
SYSTEM ORGAN CLASS
High Level Group Term |
CHANTIX
0.5 mg BID |
CHANTIX
1 mg BID |
Placebo |
|---|---|---|---|
| Preferred Term | N=129 | N=821 | N=805 |
|
GASTROINTESTINAL (GI) |
|||
|
GI Signs and Symptoms |
|||
|
Nausea |
16 |
30 |
10 |
|
Abdominal Pain Includes PTs Abdominal (pain, pain upper, pain lower, discomfort, tenderness, distension) and Stomach discomfort
|
5 |
7 |
5 |
|
Flatulence |
9 |
6 |
3 |
|
Dyspepsia |
5 |
5 |
3 |
|
Vomiting |
1 |
5 |
2 |
|
GI Motility/Defecation Conditions |
|||
|
Constipation |
5 |
8 |
3 |
|
Gastroesophageal reflux disease |
1 |
1 |
0 |
|
Salivary Gland Conditions |
|||
|
Dry mouth |
4 |
6 |
4 |
|
PSYCHIATRIC DISORDERS |
|||
|
Sleep Disorder/Disturbances |
|||
|
Insomnia Includes PTs Insomnia/Initial insomnia/Middle insomnia/Early morning awakening
|
19 |
18 |
13 |
|
Abnormal dreams |
9 |
13 |
5 |
|
Sleep disorder |
2 |
5 |
3 |
|
Nightmare |
2 |
1 |
0 |
|
NERVOUS SYSTEM |
|||
|
Headaches |
|||
|
Headache |
19 |
15 |
13 |
|
Neurological Disorders NEC |
|||
|
Dysgeusia |
8 |
5 |
4 |
|
Somnolence |
3 |
3 |
2 |
|
Lethargy |
2 |
1 |
0 |
|
GENERAL DISORDERS |
|||
|
General Disorders NEC |
|||
|
Fatigue/Malaise/Asthenia |
4 |
7 |
6 |
|
RESPIR/THORACIC/MEDIAST |
|||
|
Respiratory Disorders NEC |
|||
|
Rhinorrhea |
0 |
1 |
0 |
|
Dyspnea |
2 |
1 |
1 |
|
Upper Respiratory Tract Disorder |
7 |
5 |
4 |
|
SKIN/SUBCUTANEOUS TISSUE |
|||
|
Epidermal and Dermal Conditions |
|||
|
Rash |
1 |
3 |
2 |
|
Pruritis |
0 |
1 |
1 |
|
METABOLISM & NUTRITION |
|||
|
Appetite/General Nutrit. Disorders |
|||
|
Increased appetite |
4 |
3 |
2 |
|
Decreased appetite/Anorexia |
1 |
2 |
1 |
The overall pattern and frequency of adverse events during the longer-term premarketing trials was similar to those described in Table 5, though several of the most common events were reported by a greater proportion of patients with long-term use (e.g., nausea was reported in 40% of patients treated with CHANTIX 1 mg twice daily in a one-year study, compared to 8% of placebo-treated patients).
Following is a list of treatment-emergent adverse events reported by patients treated with CHANTIX during all premarketing clinical trials and updated based on pooled data from 18 placebo-controlled pre- and post-marketing studies, including approximately 5,000 patients treated with varenicline. Adverse events were categorized using MedDRA, Version 16.0. The listing does not include those events already listed in the previous tables or elsewhere in labeling, those events for which a drug cause was remote, those events which were so general as to be uninformative, and those events reported only once which did not have a substantial probability of being acutely life-threatening.
Blood and Lymphatic System Disorders. Infrequent: anemia, lymphadenopathy. Rare: leukocytosis, splenomegaly, thrombocytopenia.
Cardiac Disorders. Infrequent: angina pectoris, myocardial infarction, palpitations, tachycardia. Rare: acute coronary syndrome, arrhythmia, atrial fibrillation, bradycardia, cardiac flutter, cor pulmonale, coronary artery disease, ventricular extrasystoles.
Ear and Labyrinth Disorders. Infrequent: tinnitus, vertigo. Rare: deafness, Meniere's disease.
Endocrine Disorders. Infrequent: thyroid gland disorders.
Eye Disorders. Infrequent: conjunctivitis, eye irritation, eye pain, vision blurred, visual impairment. Rare: blindness transient, cataract subcapsular, dry eye, night blindness, ocular vascular disorder, photophobia, vitreous floaters.
Gastrointestinal Disorders. Frequent: diarrhea, toothache. Infrequent: dysphagia, eructation, gastritis, gastrointestinal hemorrhage, mouth ulceration. Rare: enterocolitis, esophagitis, gastric ulcer, intestinal obstruction, pancreatitis acute.
General Disorders and Administration Site Conditions. Frequent: chest pain. Infrequent: chest discomfort, chills, edema, influenza-like illness, pyrexia.
Hepatobiliary Disorders. Rare: gall bladder disorder.
Investigations. Frequent: liver function test abnormal, weight increased. Infrequent: electrocardiogram abnormal. Rare: muscle enzyme increased, urine analysis abnormal.
Metabolism and Nutrition Disorders. Infrequent: diabetes mellitus, hypoglycemia. Rare: hyperlipidemia, hypokalemia.
Musculoskeletal and Connective Tissue Disorders. Frequent: arthralgia, back pain, myalgia. Infrequent: arthritis, muscle cramp, musculoskeletal pain. Rare: myositis, osteoporosis.
Nervous System Disorders. Frequent: disturbance in attention, dizziness. Infrequent: amnesia, convulsion, migraine, parosmia, syncope, tremor. Rare: balance disorder, cerebrovascular accident, dysarthria, mental impairment, multiple sclerosis, VIIth nerve paralysis, nystagmus, psychomotor hyperactivity, psychomotor skills impaired, restless legs syndrome, sensory disturbance, transient ischemic attack, visual field defect.
Psychiatric Disorders. Infrequent: dissociation, libido decreased, mood swings, thinking abnormal. Rare: bradyphrenia, disorientation, euphoric mood.
Renal and Urinary Disorders. Infrequent: nocturia, pollakiuria, urine abnormality. Rare: nephrolithiasis, polyuria, renal failure acute, urethral syndrome, urinary retention.
Reproductive System and Breast Disorders. Frequent: menstrual disorder. Infrequent: erectile dysfunction. Rare: sexual dysfunction.
Respiratory, Thoracic and Mediastinal Disorders. Frequent: respiratory disorders. Infrequent: asthma, epistaxis, rhinitis allergic, upper respiratory tract inflammation. Rare: pleurisy, pulmonary embolism.
Skin and Subcutaneous Tissue Disorders. Infrequent: acne, dry skin, eczema, erythema, hyperhidrosis, urticaria. Rare: photosensitivity reaction, psoriasis.
Vascular Disorders. Infrequent: hot flush. Rare: thrombosis.
CHANTIX has also been studied in postmarketing trials including (1) a trial conducted in patients with chronic obstructive pulmonary disease (COPD), (2) a trial conducted in generally healthy patients (similar to those in the premarketing studies) in which they were allowed to select a quit date between days 8 and 35 of treatment ("alternative quit date instruction trial"), (3) a trial conducted in patients who did not succeed in stopping smoking during prior CHANTIX therapy, or who relapsed after treatment ("re-treatment trial"), (4) a trial conducted in patients with stable cardiovascular disease, (5) a trial conducted in patients with stable schizophrenia or schizoaffective disorder and (6) a trial conducted in patients with major depressive disorder.
Adverse events in the trial of patients with COPD, in the alternative quit date instruction trial, were quantitatively and qualitatively similar to those observed in premarketing studies. In the re-treatment trial, the profile of common adverse events was similar to that previously reported, but, in addition, varenicline-treated patients also commonly reported diarrhea (6% vs 4% in placebo-treated patients), depressed mood disorders and disturbances (6% vs 1%), and other mood disorders and disturbances (5% vs 2%).
In the trial of patients with stable cardiovascular disease, more types and a greater number of cardiovascular events were reported compared to premarketing studies. Treatment-emergent (on-treatment or 30 days after treatment) cardiovascular events reported with a frequency ≥ 1% in either treatment group in this study were angina pectoris (3.7% and 2.0% for varenicline and placebo, respectively), chest pain (2.5% vs. 2.3%), peripheral edema (2.0% vs. 1.1%), hypertension (1.4% vs. 2.6%), and palpitations (0.6 % vs. 1.1%). Deaths and serious cardiovascular events occurring over the 52 weeks of the study (treatment emergent and non-treatment emergent) were adjudicated by a blinded, independent committee. The following treatment-emergent adjudicated events occurred with a frequency ≥1% in either treatment group: nonfatal MI (1.1% vs. 0.3% for varenicline and placebo, respectively), and hospitalization for angina pectoris (0.6% vs. 1.1%). During non-treatment follow up to 52 weeks, the adjudicated events included need for coronary revascularization (2.0% vs. 0.6%), hospitalization for angina pectoris (1.7% vs. 1.1%), and new diagnosis of peripheral vascular disease (PVD) or admission for a PVD procedure (1.4% vs. 0.6%). Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina. Cardiovascular death occurred in 0.3% of patients in the varenicline arm and 0.6% of patients in the placebo arm over the course of the 52-week study.
In the trial of patients with stable schizophrenia or schizoaffective disorder, 128 smokers on antipsychotic medication were randomized 2:1 to varenicline (1 mg twice daily) or placebo for 12 weeks with 12-week non-drug follow-up. The most common adverse events in patients taking varenicline were nausea (24% vs. 14.0% on placebo), headache (11% vs. 19% on placebo) and vomiting (11% vs. 9% on placebo). Among reported neuropsychiatric adverse events, insomnia was the only event that occurred in either treatment group in ≥5% of subjects at a rate higher in the varenicline group than in placebo (10% vs. 5%). These common and neuropsychiatric adverse events occurred on treatment or within 30 days after the last dose of study drug. There was no consistent worsening of schizophrenia in either treatment group as measured by the Positive and Negative Syndrome Scale. There were no overall changes in extra-pyramidal signs, as measured by the Simpson-Angus Rating Scale. The Columbia-Suicide Severity Rating Scale was administered at baseline and at clinic visits during the treatment and non-treatment follow-up phases. Over half of the patients had a lifetime history of suicidal behavior and/or ideation (62% on varenicline vs. 51% on placebo), but at baseline, no patients in the varenicline group reported suicidal behavior and/or ideation vs. one patient in the placebo group (2%). Suicidal behavior and/or ideation were reported in 11% of the varenicline-treated and 9% of the placebo-treated patients during the treatment phase. During the post-treatment phase, suicidal behavior and/or ideation were reported in 11% of patients in the varenicline group and 5% of patients in the placebo group. Many of the patients reporting suicidal behavior and ideation in the follow-up phase had not reported such experiences in the treatment phase. However, no new suicidal ideation or behavior emerged in either treatment group shortly (within one week) after treatment discontinuation (a phenomenon noted in post-marketing reporting). There were no completed suicides. There was one suicide attempt in a varenicline-treated patient. The limited data available from this single smoking cessation study are not sufficient to allow conclusions to be drawn.
In the trial of patients with major depressive disorder, the most common adverse events (≥ 10%) in subjects taking varenicline were nausea (27% vs. 10% on placebo), headache (17 vs 11%), abnormal dreams (11% vs 8%), insomnia (11% vs 5%) and irritability (11% vs. 8%). Additionally, the following psychiatric AEs were reported in ≥ 2% of patients in either treatment group (varenicline or placebo, respectively): anxiety (7% vs. 9%), agitation (7% vs. 4%), depressed mood disorders and disturbances (11% vs. 9%), tension (4% vs. 3%), hostility (2% vs. 0.4%) and restlessness (2% vs. 2%). Patients treated with varenicline were more likely than patients treated with placebo to report one of various events related to hostility and aggression (3% vs 1%). Psychiatric scales showed no differences between the varenicline and placebo groups and no overall worsening of depression during the study in either treatment group. The percentage of subjects with suicidal ideation and/or behavior was similar between the varenicline and placebo groups during treatment (6% and 8%, respectively) and the non-treatment follow-up (6% and 6%, respectively). There was one event of intentional self-injury/possible suicide attempt during treatment (Day 73) in a subject in the placebo group. Suicide could not be ruled out in one subject who died by an overdose of illicit drugs 76 days after last dose of study drug in the varenicline group.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with "Pfizer" on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with "Pfizer" on one side and "CHX 1.0" on the other side. CHANTIX is supplied in the following package configurations:
| Description | NDC | |
|---|---|---|
|
Starting 4-week card: 0.5 mg × 11 tablets and 1 mg × 42 tablets |
NDC 63187-618-53 |
|
|
Starting Month Box: 0.5 mg × 11 tablets and 1 mg × 42 tablets |
NDC 63187-618-53 |
Warning: Serious Neuropsychiatric Events (WARNING: SERIOUS NEUROPSYCHIATRIC EVENTS)
Serious neuropsychiatric events including, but not limited to, depression, suicidal ideation, suicide attempt, and completed suicide have been reported in patients taking CHANTIX. Some reported cases may have been complicated by the symptoms of nicotine withdrawal in patients who stopped smoking. Depressed mood may be a symptom of nicotine withdrawal. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these symptoms have occurred in patients taking CHANTIX who continued to smoke.
All patients being treated with CHANTIX should be observed for neuropsychiatric symptoms including changes in behavior, hostility, agitation, depressed mood, and suicide-related events, including ideation, behavior, and attempted suicide. These symptoms, as well as worsening of pre-existing psychiatric illness and completed suicide, have been reported in some patients attempting to quit smoking while taking CHANTIX in the postmarketing experience. When symptoms were reported, most were during CHANTIX treatment, but some were following discontinuation of CHANTIX therapy.
These events have occurred in patients with and without pre-existing psychiatric disease. Patients with serious psychiatric illness such as schizophrenia, bipolar disorder, and major depressive disorder did not participate in the premarketing studies of CHANTIX.
Advise patients and caregivers that the patient should stop taking CHANTIX and contact a healthcare provider immediately if agitation, hostility, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. In many postmarketing cases, resolution of symptoms after discontinuation of CHANTIX was reported, although in some cases the symptoms persisted; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.
The risks of CHANTIX should be weighed against the benefits of its use. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo. The health benefits of quitting smoking are immediate and substantial. [see Warnings and Precautions (5.1) and Adverse Reactions (6.2)]
14.5 Subjects With Major Depressive Disorder (14.5 Subjects with Major Depressive Disorder)
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 18 to 75 years with major depressive disorder without psychotic features (DSM-IV TR). If on medication, subjects were to be on a stable antidepressant regimen for at least two months. If not on medication, subjects were to have experienced a major depressive episode in the past 2 years, which was successfully treated. Subjects were randomized to CHANTIX 1 mg twice daily (n=256) or placebo (n=269) for a treatment of 12 weeks and then followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (36%) compared to subjects treated with placebo (16%) and from week 9 through 52 (20%) compared to subjects treated with placebo (10%).
| Weeks 9 through 12 | Weeks 9 through 52 | |||
|---|---|---|---|---|
|
CHANTIX
1 mg BID |
Placebo |
CHANTIX
1 mg BID |
Placebo | |
| BID = twice daily | ||||
|
MDD Study |
36% |
16% |
20% |
10% |
5.1 Neuropsychiatric Symptoms and Suicidality
Serious neuropsychiatric symptoms have been reported in patients being treated with CHANTIX [see Boxed Warning and Adverse Reactions (6.2)]. These postmarketing reports have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Some reported cases may have been complicated by the symptoms of nicotine withdrawal in patients who stopped smoking. Depressed mood may be a symptom of nicotine withdrawal. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these symptoms have occurred in patients taking CHANTIX who continued to smoke. When symptoms were reported, most were during CHANTIX treatment, but some were following discontinuation of CHANTIX therapy.
These events have occurred in patients with and without pre-existing psychiatric disease; some patients have experienced worsening of their psychiatric illnesses. All patients being treated with CHANTIX should be observed for neuropsychiatric symptoms or worsening of pre-existing psychiatric illness. Patients with serious psychiatric illness such as schizophrenia, bipolar disorder, and major depressive disorder did not participate in the premarketing studies of CHANTIX. Limited safety data are available from post-marketing smoking cessation studies in two patient groups: 1) patients with major depressive disorder, and 2) patients with stable schizophrenia or schizoaffective disorder [see Adverse Reactions (6.1) , Clinical Studies (14.5) ] .
Some reported neuropsychiatric events, including unusual and sometimes aggressive behavior directed to oneself or others, may have been worsened by concomitant use of alcohol [see Interaction with Alcohol (5.3) , Adverse Reactions (6.2) ] .
Advise patients and caregivers that the patient should stop taking CHANTIX and contact a healthcare provider immediately if agitation, depressed mood, changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. In many postmarketing cases, resolution of symptoms after discontinuation of CHANTIX was reported, although in some cases the symptoms persisted, therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.
The risks of CHANTIX should be weighed against the benefits of its use. CHANTIX has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo. The health benefits of quitting smoking are immediate and substantial.
-
Since the initial signal of neuropsychiatric symptoms and suicidality emerged, additional analyses and studies have been conducted to further evaluate this association.
5.6 Angioedema and Hypersensitivity Reactions
There have been postmarketing reports of hypersensitivity reactions including angioedema in patients treated with CHANTIX [see Adverse Reactions (6.2), and Patient Counseling Information (17)]. Clinical signs included swelling of the face, mouth (tongue, lips, and gums), extremities, and neck (throat and larynx). There were infrequent reports of life-threatening angioedema requiring emergent medical attention due to respiratory compromise. Instruct patients to discontinue CHANTIX and immediately seek medical care if they experience these symptoms.
7.1 Use With Other Drugs for Smoking Cessation
Safety and efficacy of CHANTIX in combination with other smoking cessation therapies have not been studied.
7.2 Effect of Smoking Cessation On Other Drugs (7.2 Effect of Smoking Cessation on Other Drugs)
Physiological changes resulting from smoking cessation, with or without treatment with CHANTIX, may alter the pharmacokinetics or pharmacodynamics of certain drugs (e.g., theophylline, warfarin, insulin) for which dosage adjustment may be necessary.
14.6 Alternative Instructions for Setting A Quit Date (14.6 Alternative Instructions for Setting a Quit Date)
CHANTIX was evaluated in a double-blind, placebo-controlled trial where patients were instructed to select a target quit date between Day 8 and Day 35 of treatment. Subjects were randomized 3:1 to CHANTIX 1 mg twice daily (n=486) or placebo (n=165) for 12 weeks of treatment and followed for another 12 weeks post-treatment. Patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (54%) compared to patients treated with placebo (19%) and from weeks 9 through 24 (35%) compared to subjects treated with placebo (13%).
Principal Display Panel 0.5 / 1 Mg Starting Pack Carton (PRINCIPAL DISPLAY PANEL - 0.5 / 1 mg Starting Pack Carton)
Contains:
1 Starting Week (0.5 mg* x 11 tablets)
3 Continuing Weeks (1 mg† x 42 tablets)
NDC 63187-618-53
Rx only
CHANTIX
®
(varenicline) TABLETS
STARTING MONTH BOX
(Your first 4 weeks)
Use the Starting Week (green) blister card
first when beginning to take Chantix.
ALWAYS DISPENSE WITH ENCLOSED MEDICATION GUIDE
14.4 Subjects With Cardiovascular and Chronic Obstructive Pulmonary Disease (14.4 Subjects with Cardiovascular and Chronic Obstructive Pulmonary Disease)
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 35 to 75 years with stable, documented cardiovascular disease (diagnoses other than, or in addition to, hypertension) that had been diagnosed for more than 2 months. Subjects were randomized to CHANTIX 1 mg twice daily (n=353) or placebo (n=350) for a treatment of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (47%) compared to subjects treated with placebo (14%) and from week 9 through 52 (20%) compared to subjects treated with placebo (7%).
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged ≥ 35 years with mild-to-moderate COPD with post-bronchodilator FEV1/FVC <70% and FEV1 ≥ 50% of predicted normal value. Subjects were randomized to CHANTIX 1 mg twice daily (n=223) or placebo (n=237) for a treatment of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (41%) compared to subjects treated with placebo (9%) and from week 9 through 52 (19%) compared to subjects treated with placebo (6%).
| Weeks 9 through 12 | Weeks 9 through 52 | |||
|---|---|---|---|---|
|
CHANTIX
1 mg BID |
Placebo |
CHANTIX
1 mg BID |
Placebo | |
| BID = twice daily | ||||
|
CVD Study |
47% |
14% |
20% |
7% |
|
COPD Study |
41% |
9% |
19% |
6% |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:35:18.835550 · Updated: 2026-03-14T21:43:05.271638