These Highlights Do Not Include All The Information Needed To Use Alunbrig Safely And Effectively. See Full Prescribing Information For Alunbrig.
0fe9ff20-d402-41f3-bc1e-7002ea7007db
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ALUNBRIG is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) as detected by an FDA-approved test [see Dosage and Administration (2.1) ] .
Indications and Usage
ALUNBRIG is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) as detected by an FDA-approved test [see Dosage and Administration (2.1) ] .
Dosage and Administration
90 mg orally once daily for the first 7 days; then increase to 180 mg orally once daily. May be taken with or without food. ( 2.2 )
Warnings and Precautions
Interstitial Lung Disease (ILD)/Pneumonitis : Monitor for new or worsening respiratory symptoms, particularly during the first week of treatment. Withhold ALUNBRIG for new or worsening respiratory symptoms and promptly evaluate for ILD/pneumonitis. Upon recovery, either dose reduce or permanently discontinue ALUNBRIG. ( 2.3 , 5.1 ) Hypertension : Monitor blood pressure after 2 weeks and then at least monthly during treatment. For severe hypertension, withhold ALUNBRIG, then dose reduce or permanently discontinue. ( 2.3 , 5.2 ) Bradycardia : Monitor heart rate and blood pressure regularly during treatment. If symptomatic, withhold ALUNBRIG, then dose reduce or permanently discontinue. ( 2.3 , 5.3 ) Visual Disturbance : Advise patients to report visual symptoms. Withhold ALUNBRIG and obtain ophthalmologic evaluation, then dose reduce or permanently discontinue ALUNBRIG. ( 2.3 , 5.4 ) Creatine Phosphokinase (CPK) Elevation : Monitor CPK levels regularly during treatment. Based on the severity and with muscle pain or weakness, withhold ALUNBRIG, then resume or reduce dose. ( 2.3 , 5.5 ) Pancreatic Enzymes Elevation : Monitor lipase and amylase levels regularly during treatment. Based on the severity, withhold ALUNBRIG, then resume or reduce dose. ( 2.3 , 5.6 ) Hepatotoxicity : Monitor alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total bilirubin levels regularly during treatment. Based on severity, withhold dose, then resume at lower level. ( 2.3 , 5.7 ) Hyperglycemia : Assess fasting serum glucose prior to starting ALUNBRIG and regularly during treatment. If not adequately controlled with optimal medical management, withhold ALUNBRIG, then consider dose reduction or permanently discontinue, based on severity. ( 2.3 , 5.8 ) Photosensitivity : Advise patients to limit sun exposure. Based on severity withhold ALUNBRIG, then resume at the same dose, reduce the dose, or permanently discontinue. ( 2.3 , 5.9 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.10 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the prescribing information: Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.1) ] Hypertension [see Warnings and Precautions (5.2) ] Bradycardia [see Warnings and Precautions (5.3) ] Visual Disturbance [see Warnings and Precautions (5.4) ] Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5) ] Pancreatic Enzymes Elevation [see Warnings and Precautions (5.6) ] Hepatotoxicity [see Warnings and Precautions (5.7) ] Hyperglycemia [see Warnings and Precautions (5.8) ] Photosensitivity [see Warnings and Precautions (5.9) ]
Drug Interactions
CYP3A Inhibitors : Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inhibitors. If coadministration of a strong or moderate CYP3A inhibitor is unavoidable, reduce the dose of ALUNBRIG. ( 2.4 , 7.1 ) CYP3A Inducers : Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inducers. If coadministration of a moderate CYP3A inducer is unavoidable, increase the dose of ALUNBRIG. ( 2.5 , 7.1 )
Storage and Handling
180 mg tablets: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side; available in: Bottle of 23 tablets NDC 63020-180-23 Bottle of 30 tablets NDC 63020-180-30 90 mg tablets: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side; available in: Bottle of 7 tablets NDC 63020-090-07 Bottle of 30 tablets NDC 63020-090-30 30 mg tablets: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side; available in: Bottle of 30 tablets NDC 63020-113-30 90 mg / 7 count tablets (NDC 63020-090-07) and 180 mg / 23 count tablets (NDC 63020-180-23) are also available in a single carton as a one-month initiation pack: One carton containing one bottle of 90 mg tablets (7 count) and one bottle of 180 mg tablets (23 count) NDC 63020-198-30
How Supplied
180 mg tablets: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side; available in: Bottle of 23 tablets NDC 63020-180-23 Bottle of 30 tablets NDC 63020-180-30 90 mg tablets: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side; available in: Bottle of 7 tablets NDC 63020-090-07 Bottle of 30 tablets NDC 63020-090-30 30 mg tablets: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side; available in: Bottle of 30 tablets NDC 63020-113-30 90 mg / 7 count tablets (NDC 63020-090-07) and 180 mg / 23 count tablets (NDC 63020-180-23) are also available in a single carton as a one-month initiation pack: One carton containing one bottle of 90 mg tablets (7 count) and one bottle of 180 mg tablets (23 count) NDC 63020-198-30
Medication Information
Warnings and Precautions
Interstitial Lung Disease (ILD)/Pneumonitis : Monitor for new or worsening respiratory symptoms, particularly during the first week of treatment. Withhold ALUNBRIG for new or worsening respiratory symptoms and promptly evaluate for ILD/pneumonitis. Upon recovery, either dose reduce or permanently discontinue ALUNBRIG. ( 2.3 , 5.1 ) Hypertension : Monitor blood pressure after 2 weeks and then at least monthly during treatment. For severe hypertension, withhold ALUNBRIG, then dose reduce or permanently discontinue. ( 2.3 , 5.2 ) Bradycardia : Monitor heart rate and blood pressure regularly during treatment. If symptomatic, withhold ALUNBRIG, then dose reduce or permanently discontinue. ( 2.3 , 5.3 ) Visual Disturbance : Advise patients to report visual symptoms. Withhold ALUNBRIG and obtain ophthalmologic evaluation, then dose reduce or permanently discontinue ALUNBRIG. ( 2.3 , 5.4 ) Creatine Phosphokinase (CPK) Elevation : Monitor CPK levels regularly during treatment. Based on the severity and with muscle pain or weakness, withhold ALUNBRIG, then resume or reduce dose. ( 2.3 , 5.5 ) Pancreatic Enzymes Elevation : Monitor lipase and amylase levels regularly during treatment. Based on the severity, withhold ALUNBRIG, then resume or reduce dose. ( 2.3 , 5.6 ) Hepatotoxicity : Monitor alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total bilirubin levels regularly during treatment. Based on severity, withhold dose, then resume at lower level. ( 2.3 , 5.7 ) Hyperglycemia : Assess fasting serum glucose prior to starting ALUNBRIG and regularly during treatment. If not adequately controlled with optimal medical management, withhold ALUNBRIG, then consider dose reduction or permanently discontinue, based on severity. ( 2.3 , 5.8 ) Photosensitivity : Advise patients to limit sun exposure. Based on severity withhold ALUNBRIG, then resume at the same dose, reduce the dose, or permanently discontinue. ( 2.3 , 5.9 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.10 , 8.1 , 8.3 )
Indications and Usage
ALUNBRIG is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) as detected by an FDA-approved test [see Dosage and Administration (2.1) ] .
Dosage and Administration
90 mg orally once daily for the first 7 days; then increase to 180 mg orally once daily. May be taken with or without food. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the prescribing information: Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.1) ] Hypertension [see Warnings and Precautions (5.2) ] Bradycardia [see Warnings and Precautions (5.3) ] Visual Disturbance [see Warnings and Precautions (5.4) ] Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5) ] Pancreatic Enzymes Elevation [see Warnings and Precautions (5.6) ] Hepatotoxicity [see Warnings and Precautions (5.7) ] Hyperglycemia [see Warnings and Precautions (5.8) ] Photosensitivity [see Warnings and Precautions (5.9) ]
Drug Interactions
CYP3A Inhibitors : Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inhibitors. If coadministration of a strong or moderate CYP3A inhibitor is unavoidable, reduce the dose of ALUNBRIG. ( 2.4 , 7.1 ) CYP3A Inducers : Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inducers. If coadministration of a moderate CYP3A inducer is unavoidable, increase the dose of ALUNBRIG. ( 2.5 , 7.1 )
Storage and Handling
180 mg tablets: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side; available in: Bottle of 23 tablets NDC 63020-180-23 Bottle of 30 tablets NDC 63020-180-30 90 mg tablets: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side; available in: Bottle of 7 tablets NDC 63020-090-07 Bottle of 30 tablets NDC 63020-090-30 30 mg tablets: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side; available in: Bottle of 30 tablets NDC 63020-113-30 90 mg / 7 count tablets (NDC 63020-090-07) and 180 mg / 23 count tablets (NDC 63020-180-23) are also available in a single carton as a one-month initiation pack: One carton containing one bottle of 90 mg tablets (7 count) and one bottle of 180 mg tablets (23 count) NDC 63020-198-30
How Supplied
180 mg tablets: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side; available in: Bottle of 23 tablets NDC 63020-180-23 Bottle of 30 tablets NDC 63020-180-30 90 mg tablets: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side; available in: Bottle of 7 tablets NDC 63020-090-07 Bottle of 30 tablets NDC 63020-090-30 30 mg tablets: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side; available in: Bottle of 30 tablets NDC 63020-113-30 90 mg / 7 count tablets (NDC 63020-090-07) and 180 mg / 23 count tablets (NDC 63020-180-23) are also available in a single carton as a one-month initiation pack: One carton containing one bottle of 90 mg tablets (7 count) and one bottle of 180 mg tablets (23 count) NDC 63020-198-30
Description
ALUNBRIG is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) as detected by an FDA-approved test [see Dosage and Administration (2.1) ] .
Section 42229-5
Advanced ALK-positive NSCLC Without Prior ALK-targeted Therapy
In ALTA 1L, the safety of ALUNBRIG was evaluated in 136 patients with advanced ALK-positive NSCLC who had not previously received an ALK-targeted therapy [see Clinical Studies (14)]. The median duration of treatment with ALUNBRIG when administered as 90 mg orally once daily for the first 7 days; then increased to 180 mg orally once daily, was 24.3 months. A total of 106 (78%) patients were exposed to ALUNBRIG for greater than or equal to 6 months including 92 (68%) patients exposed for greater than or equal to 1 year. The median relative dose intensity was 97% for ALUNBRIG.
The study population (N = 275) characteristics were: median age 59 years (range: 27 to 89), age less than 65 years (68%), female (55%), White (59%), Asian (39%), Stage IV disease (93%), NSCLC adenocarcinoma histology (96%), never smoker (58%), ECOG Performance Status (PS) 0 or 1 (95%), and CNS metastases at baseline (30%) [see Clinical Studies (14)].
Serious adverse reactions occurred in 33% of patients receiving ALUNBRIG. The most common serious adverse reactions were pneumonia (4.4%), ILD/pneumonitis (3.7%), pyrexia (2.9%), dyspnea (2.2%), pulmonary embolism (2.2%), and asthenia (2.2%). Fatal adverse reactions occurred in 2.9% of patients and included pneumonia (1.5%), cerebrovascular accident (0.7%), and multiple organ dysfunction syndrome (0.7%).
In ALTA 1L, 13% of patients receiving ALUNBRIG permanently discontinued ALUNBRIG for adverse reactions. The most frequent adverse reactions that led to discontinuation were ILD/pneumonitis (3.7%) and pneumonia (2.2%).
In ALTA 1L, 38% of patients required a dose reduction due to adverse reactions. The most common adverse reaction that led to dose reduction was increased creatine phosphokinase (15%), increased lipase (6.6%), increased amylase (4.4%), increased aspartate aminotransferase (2.2%), ILD/pneumonitis (2.2%) and hypertension (2.2%).
Table 3 and Table 4 summarize the common adverse reactions and laboratory abnormalities observed in ALTA 1L.
| Adverse Reactions | ALUNBRIG N = 136 |
Crizotinib N = 137 |
||
|---|---|---|---|---|
| All Grades (%) |
Grades 3-4 (%) |
All Grades (%) |
Grades 3-4 (%) |
|
| Gastrointestinal Disorders | ||||
| Diarrhea | 53 | 2.2 | 57 | 2.9 |
| Nausea | 30 | 2.2 | 58 | 2.9 |
| Abdominal pain Includes abdominal discomfort, abdominal distension, abdominal pain, abdominal pain lower, abdominal pain upper, and epigastric discomfort
|
24 | 0.7 | 33 | 3.6 |
| Vomiting | 21 | 0.7 | 44 | 2.2 |
| Constipation | 18 | 0 | 42 | 0 |
| Stomatitis Includes aphthous ulcer, mouth ulceration, oral mucosal blistering and stomatitis
|
13 | 0.7 | 8.8 | 0 |
| Dyspepsia | 8 | 0 | 16 | 0.7 |
| Gastroesophageal reflux disease | 0.7 | 0 | 11 | 0 |
| Skin and Subcutaneous Tissue Disorders | ||||
| Rash Includes dermatitis, dermatitis acneiform, dermatitis bullous, dermatitis contact, drug eruption, erythema, palmar-plantar erythrodysesthesia syndrome, rash, rash erythematous, rash macular, rash maculo-papular, rash papular, rash pruritic, rash pustular, toxic skin eruption, urticaria
|
40 | 2.9 | 17 | 0 |
| Pruritus Included pruritus, allergic pruritus, and generalized pruritus
|
20 | 0.7 | 5.8 | 0.7 |
| Respiratory, Thoracic and Mediastinal Disorders | ||||
| Cough | 35 | 0 | 20 | 0 |
| Dyspnea Include dyspnea and exertional dyspnea
|
25 | 2.9 | 22 Includes Grade 5 events
|
3.6 |
| ILD/Pneumonitis | 5.1 | 2.9 | 2.2 | 0.7 |
| Pulmonary embolism | 2.2 | 2.2 | 5.8 | 2.9 |
| Vascular Disorders | ||||
| Hypertension Includes hypertension and systolic hypertension
|
32 | 13 | 8 | 2.9 |
| General Disorders and Administration Site Conditions | ||||
| Fatigue Includes asthenia and fatigue
|
32 | 1.5 | 40 | 2.2 |
| Edema Includes angioedema, eye swelling, eyelid edema, face edema, generalized edema, lip swelling, peripheral edema, periorbital edema, peripheral swelling, skin swelling, swelling and swelling face
|
18 | 0.7 | 48 | 0.7 |
| Pyrexia | 15 | 0.7 | 15 | 0 |
| Musculoskeletal and Connective Tissue Disorders | ||||
| Myalgia Includes muscle spasms, muscle twitching, musculoskeletal discomfort, musculoskeletal pain, and myalgia
|
28 | 0 | 23 | 0 |
| Back pain | 21 | 0.7 | 17 | 1.5 |
| Arthralgia | 14 | 0 | 12 | 0 |
| Pain in extremity | 5.1 | 0 | 15 | 0.7 |
| Nervous System Disorders | ||||
| Headache Includes headache and migraine
|
22 | 2.2 | 17 | 0 |
| Dizziness | 15 | 0.7 | 20 | 0.7 |
| Peripheral neuropathy Includes burning sensation, dysesthesia, hyperesthesia, hypoesthesia, neuralgia, peripheral neuropathy, paraesthesia, peripheral sensory neuropathy and polyneuropathy
|
11 | 0.7 | 18 | 0 |
| Dysgeusia | 2.9 | 0 | 14 | 0 |
| Investigations | ||||
| Increased Blood cholesterol Includes blood cholesterol increased, hypercholesterolaemia
|
13 | 0 | 0.7 | 0 |
| Cardiac Disorders | ||||
| Bradycardia Includes bradycardia, heart rate decreased, sinus bradycardia
|
12 | 0.7 | 23 | 0 |
| Infections and Infestations | ||||
| Pneumonia Includes lower respiratory tract infection, lung infection, pneumonia, aspiration pneumonia, and cryptococcal pneumonia
|
15 | 5.1 | 6.6 | 2.9 |
| Upper respiratory tract infection Includes upper respiratory tract infection and viral upper respiratory tract infection
|
12 | 0 | 10 | 0 |
| Nasopharyngitis | 8 | 0 | 11 | 0 |
| Urinary tract infection | 5.9 | 0.7 | 8.8 | 2.2 |
| Metabolism and Nutrition Disorders | ||||
| Decreased Appetite | 8.8 | 0.7 | 19 | 2.9 |
| Eye Disorders | ||||
| Visual Disturbance Includes cataract, glaucoma, hypermetropia, night blindness, papilloedema, photophobia, photopsia, blurred vision, reduced visual acuity, visual field defect, visual impairment, and vitreous floaters
|
7.4 | 0 | 53 | 0.7 |
| Laboratory Abnormality | ALUNBRIG N = 136 Denominator for each laboratory parameter may vary and is defined as the number of patients who had both, baseline and post-baseline test
|
Crizotinib N = 137 |
||
|---|---|---|---|---|
| All Grades (%) |
Grades 3-4 (%) |
All Grades (%) |
Grades 3-4 (%) |
|
| Chemistry | ||||
| Increased creatine phosphokinase | 81 | 24 | 68 | 4.8 |
| Increased aspartate aminotransferase | 72 | 4.5 | 70 | 5.2 |
| Increased lipase | 59 | 17 | 36 | 9.8 |
| Hyperglycemia Elevated blood insulin was also observed in both arms
|
56 | 7.5 | 37 | 3.7 |
| Increased alanine aminotransferase | 52 | 5.2 | 77 | 13 |
| Increased amylase | 52 | 6.8 | 25 | 3 |
| Decreased phosphorous | 41 | 3.7 | 39 | 6 |
| Increased alkaline phosphatase | 36 | 3 | 49 | 1.5 |
| Increased creatinine | 25 | 0 | 33 | 0 |
| Potassium increased | 24 | 1.5 | 31 | 3.7 |
| Increased calcium | 22 | 0 | 1.5 | 0 |
| Decreased magnesium | 21 | 0 | 6.9 | 0 |
| Decreased albumin | 15 | 0.8 | 52 | 3.7 |
| Decreased calcium | 15 | 0 | 67 | 1.5 |
| Hematology | ||||
| Hemoglobin decreased | 41 | 2.3 | 36 | 1.5 |
| Lymphocyte count decreased | 42 | 9.3 | 30 | 5.4 |
| Neutrophil count decreased | 12 | 0 | 34 | 6.8 |
Clinically relevant adverse reactions in patients who received ALUNBRIG included photosensitivity (3.7%).
Section 42230-3
| PATIENT INFORMATION ALUNBRIG® (uh-lun-brig) (brigatinib) tablets |
|||||
|---|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: October 2024 | ||||
|
What is the most important information I should know about ALUNBRIG? ALUNBRIG can cause serious side effects, including:
|
|||||
|
|
||||
|
|||||
|
|
||||
|
|||||
|
|
||||
|
|||||
|
|
||||
| See "What are the possible side effects of ALUNBRIG?" for information about side effects. | |||||
|
What is ALUNBRIG?
ALUNBRIG is a prescription medicine used to treat adults with non-small cell lung cancer (NSCLC):
|
|||||
Before you take ALUNBRIG, tell your healthcare provider about all of your medical conditions, including if you:
|
|||||
How should I take ALUNBRIG?
|
|||||
What should I avoid while taking ALUNBRIG?
|
|||||
|
What are the possible side effects of ALUNBRIG? ALUNBRIG may cause serious side effects, including: The most common side effects of ALUNBRIG include: |
|||||
|
|
|
|||
| ALUNBRIG may cause fertility problems in males. This may affect your ability to father a child. Talk to your healthcare provider if you have concerns about fertility. These are not all of the possible side effects of ALUNBRIG. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||
How should I store ALUNBRIG?
|
|||||
|
General information about the safe and effective use of ALUNBRIG.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ALUNBRIG for a condition for which it was not prescribed. Do not give ALUNBRIG to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about ALUNBRIG that is written for health professionals. |
|||||
|
What are the ingredients in ALUNBRIG? Active ingredient: brigatinib Inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (Type A), magnesium stearate, and hydrophobic colloidal silica. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol, and titanium dioxide. Distributed by: Takeda Pharmaceuticals America, Inc., Cambridge, MA 02142 ALUNBRIG and are registered trademarks of ARIAD Pharmaceuticals, Inc.©2024 ARIAD Pharmaceuticals, Inc. All rights reserved. For more information, go to www.alunbrig.com or call 1-844-217-6468. ABG346 R6 |
Section 44425-7
Store at controlled room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) (see USP).
11 Description
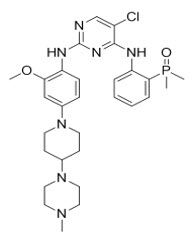
Brigatinib is a kinase inhibitor. The chemical name for brigatinib is 5-chloro-N4-[2-(dimethylphosphoryl)phenyl]-N2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl}pyrimidine-2,4-diamine. The molecular formula is C29H39ClN7O2P which corresponds to a formula weight of 584.10 g/mol. Brigatinib has no chiral centers. The chemical structure is shown below:
Brigatinib is an off-white to beige/tan solid. The pKas were determined to be: 1.73 ± 0.02 (base), 3.65 ± 0.01 (base), 4.72 ± 0.01 (base), and 8.04 ± 0.01 (base).
ALUNBRIG is supplied for oral use as film-coated tablets containing 180 mg, 90 mg or 30 mg of brigatinib and the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (Type A), magnesium stearate, and hydrophobic colloidal silica. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol, and titanium dioxide.
5.3 Bradycardia
In ALTA 1L, heart rates less than 50 beats per minute (bpm) occurred in 8.1% of patients receiving ALUNBRIG. Grade 3 bradycardia occurred in 1 patient (0.7%).
In ALTA, heart rates less than 50 beats per minute (bpm) occurred in 5.7% of patients in the 90 mg group and 7.6% of patients in the 90→180 mg group. Grade 2 bradycardia occurred in 1 (0.9%) patient in the 90 mg group.
Monitor heart rate and blood pressure during treatment with ALUNBRIG. Monitor patients more frequently if concomitant use of drug known to cause bradycardia cannot be avoided [see Warnings and Precautions (5.2)].
For symptomatic bradycardia, withhold ALUNBRIG and review concomitant medications for those known to cause bradycardia. If a concomitant medication known to cause bradycardia is identified and discontinued or dose adjusted, resume ALUNBRIG at the same dose following resolution of symptomatic bradycardia; otherwise, reduce the dose of ALUNBRIG following resolution of symptomatic bradycardia. Discontinue ALUNBRIG for life-threatening bradycardia if no contributing concomitant medication is identified [see Dosage and Administration (2.3)].
5.2 Hypertension
In ALTA 1L, hypertension was reported in 32% of patients receiving ALUNBRIG; Grade 3 hypertension occurred in 13% of patients.
In ALTA, hypertension was reported in 11% of patients in the 90 mg group who received ALUNBRIG and 21% of patients in the 90→180 mg group. Grade 3 hypertension occurred in 5.9% of patients overall.
Control blood pressure prior to treatment with ALUNBRIG. Monitor blood pressure after 2 weeks and at least monthly thereafter during treatment with ALUNBRIG. Withhold ALUNBRIG for Grade 3 hypertension despite optimal antihypertensive therapy. Upon resolution or improvement to Grade 1, resume ALUNBRIG at the same dose. Consider permanent discontinuation of treatment with ALUNBRIG for Grade 4 hypertension or recurrence of Grade 3 hypertension [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
Use caution when administering ALUNBRIG in combination with antihypertensive agents that cause bradycardia [see Warnings and Precautions (5.3)].
5.8 Hyperglycemia
In ALTA 1L, 56% of patients who received ALUNBRIG experienced new or worsening hyperglycemia. Grade 3 hyperglycemia, based on laboratory assessment of serum fasting glucose levels, occurred in 7.5% of patients.
In ALTA, 43% of patients who received ALUNBRIG experienced new or worsening hyperglycemia. Grade 3 hyperglycemia, based on laboratory assessment of serum fasting glucose levels, occurred in 3.7% of patients. Two of 20 (10%) patients with diabetes or glucose intolerance at baseline required initiation of insulin while receiving ALUNBRIG.
Assess fasting serum glucose prior to initiation of ALUNBRIG and monitor periodically thereafter. Initiate or optimize antihyperglycemic medications as needed. If adequate hyperglycemic control cannot be achieved with optimal medical management, withhold ALUNBRIG until adequate hyperglycemic control is achieved and consider reducing the dose of ALUNBRIG as described in Table 1 or permanently discontinuing ALUNBRIG [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
8.4 Pediatric Use
The safety and effectiveness of ALUNBRIG in pediatric patients have not been established.
8.5 Geriatric Use
Of the 359 patients enrolled in the ALTA 1L ALUNBRIG arm and in ALTA, 26.7% were 65 and older and 7.5% were 75 and older. No overall differences in safety or effectiveness were observed between patients ≥65 years and younger patients.
5.7 Hepatotoxicity
In ALTA 1L, aspartate aminotransferase (AST) elevations occurred in 72% of patients and Grade 3 or 4 AST elevations occurred in 4.5% of patients. Alanine aminotransferase (ALT) elevations occurred in 52% of patients and Grade 3 or 4 ALT elevations occurred in 5.2% of patients. One patient (0.7%) had a serious adverse reaction of hepatocellular injury.
In ALTA, AST elevations occurred in 38% of patients in the 90 mg group and 65% of patients in the 90→180 mg group. ALT elevations occurred in 34% of patients in the 90 mg group and 40% of patients in the 90→180 mg group. Grade 3 or 4 AST elevations occurred in 0.9% of patients in the 90 mg group and did not occur in any patients in the 90→180 mg group. Grade 3 or 4 ALT elevations did not occur in any patients in the 90 mg group and in 2.7% of patients in the 90→180 mg group.
Monitor AST, ALT and total bilirubin during treatment with ALUNBRIG, especially during first 3 months. Withhold ALUNBRIG for Grade 3 or 4 hepatic enzyme elevation with bilirubin less than or equal to 2 × ULN. Upon resolution or recovery to Grade 1 or less (less than or equal to 3 × ULN) or to baseline, resume ALUNBRIG at a next lower dose as described in Table 2. Permanently discontinue ALUNBRIG for Grade 2 to 4 hepatic enzyme elevation with concurrent total bilirubin elevation greater than 2 times the ULN in the absence of cholestasis or hemolysis [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the prescribing information:
- Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.1)]
- Hypertension [see Warnings and Precautions (5.2)]
- Bradycardia [see Warnings and Precautions (5.3)]
- Visual Disturbance [see Warnings and Precautions (5.4)]
- Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5)]
- Pancreatic Enzymes Elevation [see Warnings and Precautions (5.6)]
- Hepatotoxicity [see Warnings and Precautions (5.7)]
- Hyperglycemia [see Warnings and Precautions (5.8)]
- Photosensitivity [see Warnings and Precautions (5.9)]
7 Drug Interactions
- CYP3A Inhibitors: Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inhibitors. If coadministration of a strong or moderate CYP3A inhibitor is unavoidable, reduce the dose of ALUNBRIG. (2.4, 7.1)
- CYP3A Inducers: Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inducers. If coadministration of a moderate CYP3A inducer is unavoidable, increase the dose of ALUNBRIG. (2.5, 7.1)
5.9 Photosensitivity
In ALTA 1L, photosensitivity occurred in 3.7% of patients who received ALUNBRIG, with 0.7% Grade 3 to 4.
In ALTA, 0.9% of patients who received ALUNBRIG in the 90 mg group experienced photosensitivity and 0.9% of patients in the 90 mg→180 mg group. Grade 3 to 4 photosensitivity was not reported in patients in the 90 mg group or in the 90→180 mg group.
Advise patients to limit sun exposure while taking brigatinib, and for at least 5 days after discontinuation of treatment. Advise patients, when outdoors to wear a hat and protective clothing, and use a broad-spectrum Ultraviolet A (UVA)/ Ultraviolet B (UVB) sunscreen and lip balm (SPF ≥30) to help protect against sunburn. Based on the severity withhold ALUNBRIG, then resume at the same dose, or reduce the dose, or permanently discontinue as described in Table 2 [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
8.7 Renal Impairment
No dose adjustment is recommended for patients with mild or moderate renal impairment [creatinine clearance (CLcr) 30 to 89 mL/min by Cockcroft-Gault]. Reduce the dose of ALUNBRIG for patients with severe renal impairment (CLcr 15 to 29 mL/min) [see Dosage and Administration (2.7), Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Brigatinib exposure-response relationships and the time course of the pharmacodynamic response are unknown.
12.3 Pharmacokinetics
The geometric mean (CV%) steady-state maximum concentration (Cmax) of brigatinib at ALUNBRIG doses of 90 mg and 180 mg once daily was 552 (49%) ng/mL and 1452 (60%) ng/mL, respectively, and the corresponding area under the concentration-time curve (AUC0-Tau) was 8165 (45%) ng∙h/mL and 20276 (62%) ng∙h/mL. After a single dose and multiple dosing of ALUNBRIG, systemic exposure of brigatinib was dose proportional over the dose range of 60 mg (0.3 times the recommended dose of 180 mg) to 240 mg (1.3 times the recommended dose of 180 mg) once daily. The mean accumulation ratio after repeat dosing was 1.9 to 2.4.
2.1 Patient Selection
Select patients for the treatment of metastatic NSCLC with ALUNBRIG based on the presence of ALK positivity in tumor specimens [see Clinical Studies (14)].
Information on FDA-approved tests for the detection of ALK rearrangements in NSCLC is available at http://www.fda.gov/CompanionDiagnostics.
2.2 Recommended Dosage
The recommended dosage for ALUNBRIG is:
- 90 mg orally once daily for the first 7 days; then increase the dose to 180 mg orally once daily.
Administer ALUNBRIG until disease progression or unacceptable toxicity.
If ALUNBRIG is interrupted for 14 days or longer for reasons other than adverse reactions, resume treatment at 90 mg once daily for 7 days before increasing to the previously tolerated dose.
ALUNBRIG may be taken with or without food. Instruct patients to swallow tablets whole. Do not crush or chew tablets.
If a dose of ALUNBRIG is missed or vomiting occurs after taking a dose, do not administer an additional dose and take the next dose of ALUNBRIG at the scheduled time.
5.4 Visual Disturbance
In ALTA 1L, Grade 1 or 2 adverse reactions leading to visual disturbance including blurred vision, photophobia, photopsia, and reduced visual acuity were reported in 7.4% of patients receiving ALUNBRIG.
In ALTA, adverse reactions leading to visual disturbance including blurred vision, diplopia, and reduced visual acuity, were reported in 7.3% of patients receiving ALUNBRIG in the 90 mg group and 10% of patients in the 90→180 mg group. Grade 3 macular edema and cataract occurred in 1 patient each in the 90→180 mg group.
Advise patients to report any visual symptoms. Withhold ALUNBRIG and obtain an ophthalmologic evaluation in patients with new or worsening visual symptoms of Grade 2 or greater severity. Upon recovery of Grade 2 or Grade 3 visual disturbances to Grade 1 severity or baseline, resume ALUNBRIG at a reduced dose. Permanently discontinue treatment with ALUNBRIG for Grade 4 visual disturbances [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
8.6 Hepatic Impairment
No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A) or moderate hepatic impairment (Child-Pugh B). Reduce the dose of ALUNBRIG for patients with severe hepatic impairment (Child-Pugh C) [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
1 Indications and Usage
ALUNBRIG is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) as detected by an FDA-approved test [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
Brigatinib is a tyrosine kinase inhibitor (TKI) with in vitro activity at clinically achievable concentrations against multiple kinases including ALK, ROS1, insulin-like growth factor-1 receptor (IGF-1R), and FLT-3 as well as EGFR deletion and point mutations. Brigatinib inhibited autophosphorylation of ALK and ALK-mediated phosphorylation of the downstream signaling proteins STAT3, AKT, ERK1/2, and S6 in in vitro and in vivo assays. Brigatinib also inhibited the in vitro proliferation of cell lines expressing EML4-ALK and NPM-ALK fusion proteins and demonstrated dose-dependent inhibition of EML4-ALK-positive NSCLC xenograft growth in mice.
At clinically achievable concentrations (≤500 nM), brigatinib inhibited the in vitro viability of cells expressing EML4-ALK and 17 mutant forms associated with resistance to ALK inhibitors including crizotinib, as well as EGFR-Del (E746-A750), ROS1-L2026M, FLT3-F691L, and FLT3-D835Y. Brigatinib exhibited in vivo antitumor activity against 4 mutant forms of EML4-ALK, including G1202R and L1196M mutants identified in NSCLC tumors in patients who have progressed on crizotinib. Brigatinib also reduced tumor burden and prolonged survival in mice implanted intracranially with an ALK-driven tumor cell line.
5 Warnings and Precautions
- Interstitial Lung Disease (ILD)/Pneumonitis: Monitor for new or worsening respiratory symptoms, particularly during the first week of treatment. Withhold ALUNBRIG for new or worsening respiratory symptoms and promptly evaluate for ILD/pneumonitis. Upon recovery, either dose reduce or permanently discontinue ALUNBRIG. (2.3, 5.1)
- Hypertension: Monitor blood pressure after 2 weeks and then at least monthly during treatment. For severe hypertension, withhold ALUNBRIG, then dose reduce or permanently discontinue. (2.3, 5.2)
- Bradycardia: Monitor heart rate and blood pressure regularly during treatment. If symptomatic, withhold ALUNBRIG, then dose reduce or permanently discontinue. (2.3, 5.3)
- Visual Disturbance: Advise patients to report visual symptoms. Withhold ALUNBRIG and obtain ophthalmologic evaluation, then dose reduce or permanently discontinue ALUNBRIG. (2.3, 5.4)
- Creatine Phosphokinase (CPK) Elevation: Monitor CPK levels regularly during treatment. Based on the severity and with muscle pain or weakness, withhold ALUNBRIG, then resume or reduce dose. (2.3, 5.5)
- Pancreatic Enzymes Elevation: Monitor lipase and amylase levels regularly during treatment. Based on the severity, withhold ALUNBRIG, then resume or reduce dose. (2.3, 5.6)
- Hepatotoxicity: Monitor alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total bilirubin levels regularly during treatment. Based on severity, withhold dose, then resume at lower level. (2.3, 5.7)
- Hyperglycemia: Assess fasting serum glucose prior to starting ALUNBRIG and regularly during treatment. If not adequately controlled with optimal medical management, withhold ALUNBRIG, then consider dose reduction or permanently discontinue, based on severity. (2.3, 5.8)
- Photosensitivity: Advise patients to limit sun exposure. Based on severity withhold ALUNBRIG, then resume at the same dose, reduce the dose, or permanently discontinue. (2.3, 5.9)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.10, 8.1, 8.3)
5.10 Embryo Fetal Toxicity
Based on its mechanism of action and findings in animals, ALUNBRIG can cause fetal harm when administered to pregnant women. There are no clinical data on the use of ALUNBRIG in pregnant women. Administration of brigatinib to pregnant rats during the period of organogenesis resulted in dose-related skeletal anomalies at doses as low as 12.5 mg/kg/day (approximately 0.7 times the human exposure by AUC at 180 mg once daily), as well as increased post-implantation loss, malformations, and decreased fetal body weight at doses of 25 mg/kg/day (approximately 1.26 times the human exposure at 180 mg once daily) or higher.
Advise women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ALUNBRIG and for at least 4 months following the final dose. Advise males with female partners of reproductive potential to use effective contraception during treatment and for at least 3 months after the last dose of ALUNBRIG [see Use in Specific Populations (8.1, 8.3), Clinical Pharmacology (12.1)].
2 Dosage and Administration
90 mg orally once daily for the first 7 days; then increase to 180 mg orally once daily. May be taken with or without food. (2.2)
3 Dosage Forms and Strengths
- 180 mg: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side
- 90 mg: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side
- 30 mg: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.6 Pancreatic Enzymes Elevation
In ALTA 1L, amylase elevation occurred in 52% of patients and Grade 3 or 4 amylase elevation occurred in 6.8% of patients. Lipase elevations occurred in 59% of patients and Grade 3 or 4 lipase elevation occurred in 17% of patients.
In ALTA, amylase elevation occurred in 27% of patients in the 90 mg group and 39% of patients in the 90→180 mg group. Lipase elevations occurred in 21% of patients in the 90 mg group and 45% of patients in the 90→180 mg group. Grade 3 or 4 amylase elevation occurred in 3.7% of patients in the 90 mg group and 2.7% of patients in the 90→180 mg group. Grade 3 or 4 lipase elevation occurred in 4.6% of patients in the 90 mg group and 5.5% of patients in the 90→180 mg group.
Monitor lipase and amylase during treatment with ALUNBRIG. Withhold ALUNBRIG for Grade 3 or 4 pancreatic enzyme elevation. Upon resolution or recovery to Grade 1 or baseline, resume ALUNBRIG at the same dose or at a reduced dose as described in Table 2 [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
180 mg tablets: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side; available in:
| Bottle of 23 tablets | NDC 63020-180-23 |
| Bottle of 30 tablets | NDC 63020-180-30 |
90 mg tablets: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side; available in:
| Bottle of 7 tablets | NDC 63020-090-07 |
| Bottle of 30 tablets | NDC 63020-090-30 |
30 mg tablets: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side; available in:
| Bottle of 30 tablets | NDC 63020-113-30 |
90 mg / 7 count tablets (NDC 63020-090-07) and 180 mg / 23 count tablets (NDC 63020-180-23) are also available in a single carton as a one-month initiation pack:
| One carton containing one bottle of 90 mg tablets (7 count) and one bottle of 180 mg tablets (23 count) | NDC 63020-198-30 |
Principal Display Panel Kit Carton
NDC 63020-198-30
INITIATION PACK
ALUNBRIG®
(brigatinib) tablets
Rx Only | Oral Use
Usual Dose: One 90 mg tablet orally once daily for the first 7 days;
if tolerated, increase to one 180 mg tablet orally once daily.
See accompanying full Prescribing Information for complete details.
90 mg
7 Tablets
Take 1 tablet on
Day 1 - Day 7
180 mg
23 Tablets
Take 1 tablet on
Day 8 - Day 30
5.5 Creatine Phosphokinase (cpk) Elevation
In ALTA 1L, creatine phosphokinase (CPK) elevation occurred in 81% of patients who received ALUNBRIG. The incidence of Grade 3 or 4 CPK elevation was 24%. Dose reduction for CPK elevation occurred in 15% of patients.
In ALTA, CPK elevation occurred in 27% of patients receiving ALUNBRIG in the 90 mg group and 48% of patients in the 90 mg→180 mg group. The incidence of Grade 3 to 4 CPK elevation was 2.8% in the 90 mg group and 12% in the 90→180 mg group.
Dose reduction for CPK elevation occurred in 1.8% of patients in the 90 mg group and 4.5% in the 90→180 mg group.
Advise patients to report any unexplained muscle pain, tenderness, or weakness. Monitor CPK levels during ALUNBRIG treatment. Withhold ALUNBRIG for Grade 3 or 4 CPK elevation with Grade 2 or higher muscle pain or weakness. Upon resolution or recovery to Grade 1 CPK elevation or baseline, resume ALUNBRIG at the same dose or at a reduced dose as described in Table 2 [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
2.3 Dosage Modifications for Adverse Reactions
ALUNBRIG dosage reductions for adverse reactions are summarized in Table 1.
| Dosage Reduction | |||
|---|---|---|---|
| Dosage | First | Second | Third |
| 90 mg once daily | 60 mg once daily | permanently discontinue | N/A Not applicable
|
| 180 mg once daily | 120 mg once daily | 90 mg once daily | 60 mg once daily |
Once reduced for adverse reactions, do not subsequently increase the dosage of ALUNBRIG. Permanently discontinue ALUNBRIG if patients are unable to tolerate the 60 mg once daily dose.
Recommendations for dosage modifications of ALUNBRIG for the management of adverse reactions are provided in Table 2.
| Adverse Reaction | Severity Graded per National Cancer Institute Common Terminology Criteria for Adverse Events. Version 4.0 (NCI CTCAE v4).
|
Dosage Modifications |
|---|---|---|
| bpm = beats per minute; DBP = diastolic blood pressure; HR = heart rate; SBP = systolic blood pressure; ULN = upper limit of normal | ||
| Interstitial Lung Disease (ILD) /Pneumonitis [see Warnings and Precautions (5.1)] | Grade 1 |
|
| Grade 2 |
|
|
| Grade 3 or 4 | Permanently discontinue ALUNBRIG for ILD/pneumonitis. | |
| Hypertension [see Warnings and Precautions (5.2)] | Grade 3 hypertension (SBP greater than or equal to 160 mmHg or DBP greater than or equal to 100 mmHg, medical intervention indicated, more than one antihypertensive drug, or more intensive therapy than previously used indicated) |
|
| Grade 4 hypertension (life-threatening consequences, urgent intervention indicated) |
|
|
| Bradycardia (HR less than 60 bpm) [see Warnings and Precautions (5.3)] | Symptomatic bradycardia |
|
| Bradycardia with life-threatening consequences, urgent intervention indicated |
|
|
| Visual Disturbance [see Warnings and Precautions (5.4)] | Grade 2 or 3 visual disturbance | Withhold ALUNBRIG until recovery to Grade 1 or baseline, then resume at the next lower dose (Table 1). |
| Grade 4 visual disturbance | Permanently discontinue ALUNBRIG. | |
| Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5)] | Grade 3 or 4 CPK elevation (greater than 5× ULN) with Grade 2 or higher muscle pain or weakness |
|
| Lipase/Amylase Elevation [see Warnings and Precautions (5.6)] | Grade 3 lipase or amylase elevation (greater than 2× ULN) |
|
| Grade 4 lipase or amylase elevation (greater than 5× ULN) | Withhold ALUNBRIG until recovery to Grade 1 or less (less than or equal to 1.5× ULN) or to baseline, then resume ALUNBRIG at next lower dose (Table 1). | |
| Hepatotoxicity (Elevation of alanine aminotransferase (ALT) or aspartate aminotransferase (AST)) [see Warnings and Precautions (5.7)] | Grade 3 or 4 elevation (greater than 5 × ULN) of either ALT or AST with bilirubin less than or equal to 2 × ULN | Withhold ALUNBRIG until recovery to Grade 1 or less (less than or equal to 3× ULN) or to baseline, then resume ALUNBRIG at next lower dose (Table 1). |
| Grade 2 to 4 elevation (greater than 3 × ULN) of ALT or AST with concurrent total bilirubin elevation greater than 2 × ULN in the absence of cholestasis or hemolysis | Permanently discontinue ALUNBRIG. | |
| Hyperglycemia [see Warnings and Precautions (5.8)] | Grade 3 (greater than 250 mg/dL or 13.9 mmol/L) or 4 | If adequate hyperglycemic control cannot be achieved with optimal medical management, withhold ALUNBRIG until adequate hyperglycemic control is achieved and resume at the next lower dose (Table 1) or permanently discontinue ALUNBRIG. |
| Other | Grade 3 |
|
| Grade 4 |
|
5.1 Interstitial Lung Disease (ild)/pneumonitis
Severe, life-threatening, and fatal pulmonary adverse reactions consistent with interstitial lung disease (ILD)/pneumonitis have occurred with ALUNBRIG.
In Trial ALTA 1L, ILD/pneumonitis occurred in 5.1% of patients receiving ALUNBRIG. ILD/pneumonitis occurred within 8 days of initiation of ALUNBRIG in 2.9% of patients, with Grade 3 to 4 reactions occurring in 2.2% of patients.
In Trial ALTA, ILD/pneumonitis occurred in 3.7% of patients in the 90 mg group (90 mg once daily) and 9.1% of patients in the 90→180 mg group (180 mg once daily with 7-day lead-in at 90 mg once daily). Adverse reactions consistent with possible ILD/pneumonitis occurred within 9 days of initiation of ALUNBRIG (median onset was 2 days) in 6.4% of patients, with Grade 3 to 4 reactions occurring in 2.7%.
Monitor for new or worsening respiratory symptoms (e.g., dyspnea, cough, etc.), particularly during the first week of initiating ALUNBRIG. Withhold ALUNBRIG in any patient with new or worsening respiratory symptoms, and promptly evaluate for ILD/pneumonitis or other causes of respiratory symptoms (e.g., pulmonary embolism, tumor progression, and infectious pneumonia). For Grade 1 or 2 ILD/pneumonitis, either resume ALUNBRIG with dose reduction according to Table 1 after recovery to baseline or permanently discontinue ALUNBRIG. Permanently discontinue ALUNBRIG for Grade 3 or 4 ILD/pneumonitis or recurrence of Grade 1 or 2 ILD/pneumonitis [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
2.5 Dosage Modifications for Moderate Cyp3a Inducers
Avoid coadministration of moderate CYP3A inducers during treatment with ALUNBRIG [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If coadministration of a moderate CYP3A inducer cannot be avoided, increase the ALUNBRIG once daily dose in 30 mg increments after 7 days of treatment with the current ALUNBRIG dose as tolerated, up to a maximum of twice the ALUNBRIG dose that was tolerated prior to initiating the moderate CYP3A inducer.
After discontinuation of a moderate CYP3A inducer, resume the ALUNBRIG dose that was tolerated prior to initiating the moderate CYP3A inducer.
Principal Display Panel 30 Mg Tablet Bottle Carton
NDC 63020-113-30
ALUNBRIG®
(brigatinib) tablets
30 mg
Rx Only
30 Tablets
Principal Display Panel 90 Mg Tablet Bottle Carton
NDC 63020-090-30
ALUNBRIG®
(brigatinib) tablets
90 mg
Rx Only
30 Tablets
Principal Display Panel 180 Mg Tablet Bottle Carton
NDC 63020-180-30
ALUNBRIG®
(brigatinib) tablets
180 mg
Rx Only
30 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with brigatinib.
Treatment with brigatinib resulted in chromosomal damage in an in vivo mammalian erythrocyte micronucleus in the rat, but was not mutagenic in the Ames or in vitro mammalian chromosome aberration tests.
Dedicated animal fertility studies were not conducted with brigatinib. Testicular toxicity was observed in repeat-dose animal studies at doses resulting in exposure as low as 0.2 times the exposure in patients at the 180 mg dose. In rats, findings included lower weight of testes, seminal vesicles and prostate gland, and testicular tubular degeneration; these effects were not reversible during the 2 month recovery period. In monkeys, findings included reduced size of testes along with microscopic evidence of hypospermatogenesis; these effects were reversible during the recovery period.
2.4 Dosage Modifications for Strong Or Moderate Cyp3a Inhibitors
Avoid coadministration of strong or moderate CYP3A inhibitors during treatment with ALUNBRIG [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If coadministration of a strong CYP3A inhibitor cannot be avoided, reduce the ALUNBRIG once daily dose by approximately 50% (i.e., from 180 mg to 90 mg, or from 90 mg to 60 mg). If coadministration of a moderate CYP3A inhibitor cannot be avoided, reduce the ALUNBRIG once daily dose by approximately 40% (i.e., from 180 mg to 120 mg, 120 mg to 90 mg, or from 90 mg to 60 mg). After discontinuation of a strong or moderate CYP3A inhibitor, resume the ALUNBRIG dose that was tolerated prior to initiating the CYP3A inhibitor.
2.7 Dosage Modifications for Patients With Severe Renal Impairment
Reduce the ALUNBRIG once daily dose by approximately 50% (i.e., from 180 mg to 90 mg, or from 90 mg to 60 mg) for patients with severe renal impairment [creatinine clearance (CLcr) 15 to 29 mL/min by Cockcroft-Gault] [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2.6 Dosage Modifications for Patients With Severe Hepatic Impairment
Reduce the ALUNBRIG once daily dose by approximately 40% (i.e., from 180 mg to 120 mg, 120 mg to 90 mg, or from 90 mg to 60 mg) for patients with severe hepatic impairment (Child-Pugh C) [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Advanced ALK-positive NSCLC Without Prior ALK-targeted Therapy
In ALTA 1L, the safety of ALUNBRIG was evaluated in 136 patients with advanced ALK-positive NSCLC who had not previously received an ALK-targeted therapy [see Clinical Studies (14)]. The median duration of treatment with ALUNBRIG when administered as 90 mg orally once daily for the first 7 days; then increased to 180 mg orally once daily, was 24.3 months. A total of 106 (78%) patients were exposed to ALUNBRIG for greater than or equal to 6 months including 92 (68%) patients exposed for greater than or equal to 1 year. The median relative dose intensity was 97% for ALUNBRIG.
The study population (N = 275) characteristics were: median age 59 years (range: 27 to 89), age less than 65 years (68%), female (55%), White (59%), Asian (39%), Stage IV disease (93%), NSCLC adenocarcinoma histology (96%), never smoker (58%), ECOG Performance Status (PS) 0 or 1 (95%), and CNS metastases at baseline (30%) [see Clinical Studies (14)].
Serious adverse reactions occurred in 33% of patients receiving ALUNBRIG. The most common serious adverse reactions were pneumonia (4.4%), ILD/pneumonitis (3.7%), pyrexia (2.9%), dyspnea (2.2%), pulmonary embolism (2.2%), and asthenia (2.2%). Fatal adverse reactions occurred in 2.9% of patients and included pneumonia (1.5%), cerebrovascular accident (0.7%), and multiple organ dysfunction syndrome (0.7%).
In ALTA 1L, 13% of patients receiving ALUNBRIG permanently discontinued ALUNBRIG for adverse reactions. The most frequent adverse reactions that led to discontinuation were ILD/pneumonitis (3.7%) and pneumonia (2.2%).
In ALTA 1L, 38% of patients required a dose reduction due to adverse reactions. The most common adverse reaction that led to dose reduction was increased creatine phosphokinase (15%), increased lipase (6.6%), increased amylase (4.4%), increased aspartate aminotransferase (2.2%), ILD/pneumonitis (2.2%) and hypertension (2.2%).
Table 3 and Table 4 summarize the common adverse reactions and laboratory abnormalities observed in ALTA 1L.
| Adverse Reactions | ALUNBRIG N = 136 |
Crizotinib N = 137 |
||
|---|---|---|---|---|
| All Grades (%) |
Grades 3-4 (%) |
All Grades (%) |
Grades 3-4 (%) |
|
| Gastrointestinal Disorders | ||||
| Diarrhea | 53 | 2.2 | 57 | 2.9 |
| Nausea | 30 | 2.2 | 58 | 2.9 |
| Abdominal pain Includes abdominal discomfort, abdominal distension, abdominal pain, abdominal pain lower, abdominal pain upper, and epigastric discomfort
|
24 | 0.7 | 33 | 3.6 |
| Vomiting | 21 | 0.7 | 44 | 2.2 |
| Constipation | 18 | 0 | 42 | 0 |
| Stomatitis Includes aphthous ulcer, mouth ulceration, oral mucosal blistering and stomatitis
|
13 | 0.7 | 8.8 | 0 |
| Dyspepsia | 8 | 0 | 16 | 0.7 |
| Gastroesophageal reflux disease | 0.7 | 0 | 11 | 0 |
| Skin and Subcutaneous Tissue Disorders | ||||
| Rash Includes dermatitis, dermatitis acneiform, dermatitis bullous, dermatitis contact, drug eruption, erythema, palmar-plantar erythrodysesthesia syndrome, rash, rash erythematous, rash macular, rash maculo-papular, rash papular, rash pruritic, rash pustular, toxic skin eruption, urticaria
|
40 | 2.9 | 17 | 0 |
| Pruritus Included pruritus, allergic pruritus, and generalized pruritus
|
20 | 0.7 | 5.8 | 0.7 |
| Respiratory, Thoracic and Mediastinal Disorders | ||||
| Cough | 35 | 0 | 20 | 0 |
| Dyspnea Include dyspnea and exertional dyspnea
|
25 | 2.9 | 22 Includes Grade 5 events
|
3.6 |
| ILD/Pneumonitis | 5.1 | 2.9 | 2.2 | 0.7 |
| Pulmonary embolism | 2.2 | 2.2 | 5.8 | 2.9 |
| Vascular Disorders | ||||
| Hypertension Includes hypertension and systolic hypertension
|
32 | 13 | 8 | 2.9 |
| General Disorders and Administration Site Conditions | ||||
| Fatigue Includes asthenia and fatigue
|
32 | 1.5 | 40 | 2.2 |
| Edema Includes angioedema, eye swelling, eyelid edema, face edema, generalized edema, lip swelling, peripheral edema, periorbital edema, peripheral swelling, skin swelling, swelling and swelling face
|
18 | 0.7 | 48 | 0.7 |
| Pyrexia | 15 | 0.7 | 15 | 0 |
| Musculoskeletal and Connective Tissue Disorders | ||||
| Myalgia Includes muscle spasms, muscle twitching, musculoskeletal discomfort, musculoskeletal pain, and myalgia
|
28 | 0 | 23 | 0 |
| Back pain | 21 | 0.7 | 17 | 1.5 |
| Arthralgia | 14 | 0 | 12 | 0 |
| Pain in extremity | 5.1 | 0 | 15 | 0.7 |
| Nervous System Disorders | ||||
| Headache Includes headache and migraine
|
22 | 2.2 | 17 | 0 |
| Dizziness | 15 | 0.7 | 20 | 0.7 |
| Peripheral neuropathy Includes burning sensation, dysesthesia, hyperesthesia, hypoesthesia, neuralgia, peripheral neuropathy, paraesthesia, peripheral sensory neuropathy and polyneuropathy
|
11 | 0.7 | 18 | 0 |
| Dysgeusia | 2.9 | 0 | 14 | 0 |
| Investigations | ||||
| Increased Blood cholesterol Includes blood cholesterol increased, hypercholesterolaemia
|
13 | 0 | 0.7 | 0 |
| Cardiac Disorders | ||||
| Bradycardia Includes bradycardia, heart rate decreased, sinus bradycardia
|
12 | 0.7 | 23 | 0 |
| Infections and Infestations | ||||
| Pneumonia Includes lower respiratory tract infection, lung infection, pneumonia, aspiration pneumonia, and cryptococcal pneumonia
|
15 | 5.1 | 6.6 | 2.9 |
| Upper respiratory tract infection Includes upper respiratory tract infection and viral upper respiratory tract infection
|
12 | 0 | 10 | 0 |
| Nasopharyngitis | 8 | 0 | 11 | 0 |
| Urinary tract infection | 5.9 | 0.7 | 8.8 | 2.2 |
| Metabolism and Nutrition Disorders | ||||
| Decreased Appetite | 8.8 | 0.7 | 19 | 2.9 |
| Eye Disorders | ||||
| Visual Disturbance Includes cataract, glaucoma, hypermetropia, night blindness, papilloedema, photophobia, photopsia, blurred vision, reduced visual acuity, visual field defect, visual impairment, and vitreous floaters
|
7.4 | 0 | 53 | 0.7 |
| Laboratory Abnormality | ALUNBRIG N = 136 Denominator for each laboratory parameter may vary and is defined as the number of patients who had both, baseline and post-baseline test
|
Crizotinib N = 137 |
||
|---|---|---|---|---|
| All Grades (%) |
Grades 3-4 (%) |
All Grades (%) |
Grades 3-4 (%) |
|
| Chemistry | ||||
| Increased creatine phosphokinase | 81 | 24 | 68 | 4.8 |
| Increased aspartate aminotransferase | 72 | 4.5 | 70 | 5.2 |
| Increased lipase | 59 | 17 | 36 | 9.8 |
| Hyperglycemia Elevated blood insulin was also observed in both arms
|
56 | 7.5 | 37 | 3.7 |
| Increased alanine aminotransferase | 52 | 5.2 | 77 | 13 |
| Increased amylase | 52 | 6.8 | 25 | 3 |
| Decreased phosphorous | 41 | 3.7 | 39 | 6 |
| Increased alkaline phosphatase | 36 | 3 | 49 | 1.5 |
| Increased creatinine | 25 | 0 | 33 | 0 |
| Potassium increased | 24 | 1.5 | 31 | 3.7 |
| Increased calcium | 22 | 0 | 1.5 | 0 |
| Decreased magnesium | 21 | 0 | 6.9 | 0 |
| Decreased albumin | 15 | 0.8 | 52 | 3.7 |
| Decreased calcium | 15 | 0 | 67 | 1.5 |
| Hematology | ||||
| Hemoglobin decreased | 41 | 2.3 | 36 | 1.5 |
| Lymphocyte count decreased | 42 | 9.3 | 30 | 5.4 |
| Neutrophil count decreased | 12 | 0 | 34 | 6.8 |
Clinically relevant adverse reactions in patients who received ALUNBRIG included photosensitivity (3.7%).
Section 42230-3 (42230-3)
| PATIENT INFORMATION ALUNBRIG® (uh-lun-brig) (brigatinib) tablets |
|||||
|---|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: October 2024 | ||||
|
What is the most important information I should know about ALUNBRIG? ALUNBRIG can cause serious side effects, including:
|
|||||
|
|
||||
|
|||||
|
|
||||
|
|||||
|
|
||||
|
|||||
|
|
||||
| See "What are the possible side effects of ALUNBRIG?" for information about side effects. | |||||
|
What is ALUNBRIG?
ALUNBRIG is a prescription medicine used to treat adults with non-small cell lung cancer (NSCLC):
|
|||||
Before you take ALUNBRIG, tell your healthcare provider about all of your medical conditions, including if you:
|
|||||
How should I take ALUNBRIG?
|
|||||
What should I avoid while taking ALUNBRIG?
|
|||||
|
What are the possible side effects of ALUNBRIG? ALUNBRIG may cause serious side effects, including: The most common side effects of ALUNBRIG include: |
|||||
|
|
|
|||
| ALUNBRIG may cause fertility problems in males. This may affect your ability to father a child. Talk to your healthcare provider if you have concerns about fertility. These are not all of the possible side effects of ALUNBRIG. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||
How should I store ALUNBRIG?
|
|||||
|
General information about the safe and effective use of ALUNBRIG.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ALUNBRIG for a condition for which it was not prescribed. Do not give ALUNBRIG to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about ALUNBRIG that is written for health professionals. |
|||||
|
What are the ingredients in ALUNBRIG? Active ingredient: brigatinib Inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (Type A), magnesium stearate, and hydrophobic colloidal silica. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol, and titanium dioxide. Distributed by: Takeda Pharmaceuticals America, Inc., Cambridge, MA 02142 ALUNBRIG and are registered trademarks of ARIAD Pharmaceuticals, Inc.©2024 ARIAD Pharmaceuticals, Inc. All rights reserved. For more information, go to www.alunbrig.com or call 1-844-217-6468. ABG346 R6 |
Section 44425-7 (44425-7)
Store at controlled room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) (see USP).
11 Description (11 DESCRIPTION)
Brigatinib is a kinase inhibitor. The chemical name for brigatinib is 5-chloro-N4-[2-(dimethylphosphoryl)phenyl]-N2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl}pyrimidine-2,4-diamine. The molecular formula is C29H39ClN7O2P which corresponds to a formula weight of 584.10 g/mol. Brigatinib has no chiral centers. The chemical structure is shown below:
Brigatinib is an off-white to beige/tan solid. The pKas were determined to be: 1.73 ± 0.02 (base), 3.65 ± 0.01 (base), 4.72 ± 0.01 (base), and 8.04 ± 0.01 (base).
ALUNBRIG is supplied for oral use as film-coated tablets containing 180 mg, 90 mg or 30 mg of brigatinib and the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (Type A), magnesium stearate, and hydrophobic colloidal silica. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol, and titanium dioxide.
5.3 Bradycardia
In ALTA 1L, heart rates less than 50 beats per minute (bpm) occurred in 8.1% of patients receiving ALUNBRIG. Grade 3 bradycardia occurred in 1 patient (0.7%).
In ALTA, heart rates less than 50 beats per minute (bpm) occurred in 5.7% of patients in the 90 mg group and 7.6% of patients in the 90→180 mg group. Grade 2 bradycardia occurred in 1 (0.9%) patient in the 90 mg group.
Monitor heart rate and blood pressure during treatment with ALUNBRIG. Monitor patients more frequently if concomitant use of drug known to cause bradycardia cannot be avoided [see Warnings and Precautions (5.2)].
For symptomatic bradycardia, withhold ALUNBRIG and review concomitant medications for those known to cause bradycardia. If a concomitant medication known to cause bradycardia is identified and discontinued or dose adjusted, resume ALUNBRIG at the same dose following resolution of symptomatic bradycardia; otherwise, reduce the dose of ALUNBRIG following resolution of symptomatic bradycardia. Discontinue ALUNBRIG for life-threatening bradycardia if no contributing concomitant medication is identified [see Dosage and Administration (2.3)].
5.2 Hypertension
In ALTA 1L, hypertension was reported in 32% of patients receiving ALUNBRIG; Grade 3 hypertension occurred in 13% of patients.
In ALTA, hypertension was reported in 11% of patients in the 90 mg group who received ALUNBRIG and 21% of patients in the 90→180 mg group. Grade 3 hypertension occurred in 5.9% of patients overall.
Control blood pressure prior to treatment with ALUNBRIG. Monitor blood pressure after 2 weeks and at least monthly thereafter during treatment with ALUNBRIG. Withhold ALUNBRIG for Grade 3 hypertension despite optimal antihypertensive therapy. Upon resolution or improvement to Grade 1, resume ALUNBRIG at the same dose. Consider permanent discontinuation of treatment with ALUNBRIG for Grade 4 hypertension or recurrence of Grade 3 hypertension [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
Use caution when administering ALUNBRIG in combination with antihypertensive agents that cause bradycardia [see Warnings and Precautions (5.3)].
5.8 Hyperglycemia
In ALTA 1L, 56% of patients who received ALUNBRIG experienced new or worsening hyperglycemia. Grade 3 hyperglycemia, based on laboratory assessment of serum fasting glucose levels, occurred in 7.5% of patients.
In ALTA, 43% of patients who received ALUNBRIG experienced new or worsening hyperglycemia. Grade 3 hyperglycemia, based on laboratory assessment of serum fasting glucose levels, occurred in 3.7% of patients. Two of 20 (10%) patients with diabetes or glucose intolerance at baseline required initiation of insulin while receiving ALUNBRIG.
Assess fasting serum glucose prior to initiation of ALUNBRIG and monitor periodically thereafter. Initiate or optimize antihyperglycemic medications as needed. If adequate hyperglycemic control cannot be achieved with optimal medical management, withhold ALUNBRIG until adequate hyperglycemic control is achieved and consider reducing the dose of ALUNBRIG as described in Table 1 or permanently discontinuing ALUNBRIG [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
8.4 Pediatric Use
The safety and effectiveness of ALUNBRIG in pediatric patients have not been established.
8.5 Geriatric Use
Of the 359 patients enrolled in the ALTA 1L ALUNBRIG arm and in ALTA, 26.7% were 65 and older and 7.5% were 75 and older. No overall differences in safety or effectiveness were observed between patients ≥65 years and younger patients.
5.7 Hepatotoxicity
In ALTA 1L, aspartate aminotransferase (AST) elevations occurred in 72% of patients and Grade 3 or 4 AST elevations occurred in 4.5% of patients. Alanine aminotransferase (ALT) elevations occurred in 52% of patients and Grade 3 or 4 ALT elevations occurred in 5.2% of patients. One patient (0.7%) had a serious adverse reaction of hepatocellular injury.
In ALTA, AST elevations occurred in 38% of patients in the 90 mg group and 65% of patients in the 90→180 mg group. ALT elevations occurred in 34% of patients in the 90 mg group and 40% of patients in the 90→180 mg group. Grade 3 or 4 AST elevations occurred in 0.9% of patients in the 90 mg group and did not occur in any patients in the 90→180 mg group. Grade 3 or 4 ALT elevations did not occur in any patients in the 90 mg group and in 2.7% of patients in the 90→180 mg group.
Monitor AST, ALT and total bilirubin during treatment with ALUNBRIG, especially during first 3 months. Withhold ALUNBRIG for Grade 3 or 4 hepatic enzyme elevation with bilirubin less than or equal to 2 × ULN. Upon resolution or recovery to Grade 1 or less (less than or equal to 3 × ULN) or to baseline, resume ALUNBRIG at a next lower dose as described in Table 2. Permanently discontinue ALUNBRIG for Grade 2 to 4 hepatic enzyme elevation with concurrent total bilirubin elevation greater than 2 times the ULN in the absence of cholestasis or hemolysis [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the prescribing information:
- Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.1)]
- Hypertension [see Warnings and Precautions (5.2)]
- Bradycardia [see Warnings and Precautions (5.3)]
- Visual Disturbance [see Warnings and Precautions (5.4)]
- Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5)]
- Pancreatic Enzymes Elevation [see Warnings and Precautions (5.6)]
- Hepatotoxicity [see Warnings and Precautions (5.7)]
- Hyperglycemia [see Warnings and Precautions (5.8)]
- Photosensitivity [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- CYP3A Inhibitors: Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inhibitors. If coadministration of a strong or moderate CYP3A inhibitor is unavoidable, reduce the dose of ALUNBRIG. (2.4, 7.1)
- CYP3A Inducers: Avoid coadministration of ALUNBRIG with strong or moderate CYP3A inducers. If coadministration of a moderate CYP3A inducer is unavoidable, increase the dose of ALUNBRIG. (2.5, 7.1)
5.9 Photosensitivity
In ALTA 1L, photosensitivity occurred in 3.7% of patients who received ALUNBRIG, with 0.7% Grade 3 to 4.
In ALTA, 0.9% of patients who received ALUNBRIG in the 90 mg group experienced photosensitivity and 0.9% of patients in the 90 mg→180 mg group. Grade 3 to 4 photosensitivity was not reported in patients in the 90 mg group or in the 90→180 mg group.
Advise patients to limit sun exposure while taking brigatinib, and for at least 5 days after discontinuation of treatment. Advise patients, when outdoors to wear a hat and protective clothing, and use a broad-spectrum Ultraviolet A (UVA)/ Ultraviolet B (UVB) sunscreen and lip balm (SPF ≥30) to help protect against sunburn. Based on the severity withhold ALUNBRIG, then resume at the same dose, or reduce the dose, or permanently discontinue as described in Table 2 [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
8.7 Renal Impairment
No dose adjustment is recommended for patients with mild or moderate renal impairment [creatinine clearance (CLcr) 30 to 89 mL/min by Cockcroft-Gault]. Reduce the dose of ALUNBRIG for patients with severe renal impairment (CLcr 15 to 29 mL/min) [see Dosage and Administration (2.7), Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Brigatinib exposure-response relationships and the time course of the pharmacodynamic response are unknown.
12.3 Pharmacokinetics
The geometric mean (CV%) steady-state maximum concentration (Cmax) of brigatinib at ALUNBRIG doses of 90 mg and 180 mg once daily was 552 (49%) ng/mL and 1452 (60%) ng/mL, respectively, and the corresponding area under the concentration-time curve (AUC0-Tau) was 8165 (45%) ng∙h/mL and 20276 (62%) ng∙h/mL. After a single dose and multiple dosing of ALUNBRIG, systemic exposure of brigatinib was dose proportional over the dose range of 60 mg (0.3 times the recommended dose of 180 mg) to 240 mg (1.3 times the recommended dose of 180 mg) once daily. The mean accumulation ratio after repeat dosing was 1.9 to 2.4.
2.1 Patient Selection
Select patients for the treatment of metastatic NSCLC with ALUNBRIG based on the presence of ALK positivity in tumor specimens [see Clinical Studies (14)].
Information on FDA-approved tests for the detection of ALK rearrangements in NSCLC is available at http://www.fda.gov/CompanionDiagnostics.
2.2 Recommended Dosage
The recommended dosage for ALUNBRIG is:
- 90 mg orally once daily for the first 7 days; then increase the dose to 180 mg orally once daily.
Administer ALUNBRIG until disease progression or unacceptable toxicity.
If ALUNBRIG is interrupted for 14 days or longer for reasons other than adverse reactions, resume treatment at 90 mg once daily for 7 days before increasing to the previously tolerated dose.
ALUNBRIG may be taken with or without food. Instruct patients to swallow tablets whole. Do not crush or chew tablets.
If a dose of ALUNBRIG is missed or vomiting occurs after taking a dose, do not administer an additional dose and take the next dose of ALUNBRIG at the scheduled time.
5.4 Visual Disturbance
In ALTA 1L, Grade 1 or 2 adverse reactions leading to visual disturbance including blurred vision, photophobia, photopsia, and reduced visual acuity were reported in 7.4% of patients receiving ALUNBRIG.
In ALTA, adverse reactions leading to visual disturbance including blurred vision, diplopia, and reduced visual acuity, were reported in 7.3% of patients receiving ALUNBRIG in the 90 mg group and 10% of patients in the 90→180 mg group. Grade 3 macular edema and cataract occurred in 1 patient each in the 90→180 mg group.
Advise patients to report any visual symptoms. Withhold ALUNBRIG and obtain an ophthalmologic evaluation in patients with new or worsening visual symptoms of Grade 2 or greater severity. Upon recovery of Grade 2 or Grade 3 visual disturbances to Grade 1 severity or baseline, resume ALUNBRIG at a reduced dose. Permanently discontinue treatment with ALUNBRIG for Grade 4 visual disturbances [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
8.6 Hepatic Impairment
No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A) or moderate hepatic impairment (Child-Pugh B). Reduce the dose of ALUNBRIG for patients with severe hepatic impairment (Child-Pugh C) [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ALUNBRIG is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) as detected by an FDA-approved test [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
Brigatinib is a tyrosine kinase inhibitor (TKI) with in vitro activity at clinically achievable concentrations against multiple kinases including ALK, ROS1, insulin-like growth factor-1 receptor (IGF-1R), and FLT-3 as well as EGFR deletion and point mutations. Brigatinib inhibited autophosphorylation of ALK and ALK-mediated phosphorylation of the downstream signaling proteins STAT3, AKT, ERK1/2, and S6 in in vitro and in vivo assays. Brigatinib also inhibited the in vitro proliferation of cell lines expressing EML4-ALK and NPM-ALK fusion proteins and demonstrated dose-dependent inhibition of EML4-ALK-positive NSCLC xenograft growth in mice.
At clinically achievable concentrations (≤500 nM), brigatinib inhibited the in vitro viability of cells expressing EML4-ALK and 17 mutant forms associated with resistance to ALK inhibitors including crizotinib, as well as EGFR-Del (E746-A750), ROS1-L2026M, FLT3-F691L, and FLT3-D835Y. Brigatinib exhibited in vivo antitumor activity against 4 mutant forms of EML4-ALK, including G1202R and L1196M mutants identified in NSCLC tumors in patients who have progressed on crizotinib. Brigatinib also reduced tumor burden and prolonged survival in mice implanted intracranially with an ALK-driven tumor cell line.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Interstitial Lung Disease (ILD)/Pneumonitis: Monitor for new or worsening respiratory symptoms, particularly during the first week of treatment. Withhold ALUNBRIG for new or worsening respiratory symptoms and promptly evaluate for ILD/pneumonitis. Upon recovery, either dose reduce or permanently discontinue ALUNBRIG. (2.3, 5.1)
- Hypertension: Monitor blood pressure after 2 weeks and then at least monthly during treatment. For severe hypertension, withhold ALUNBRIG, then dose reduce or permanently discontinue. (2.3, 5.2)
- Bradycardia: Monitor heart rate and blood pressure regularly during treatment. If symptomatic, withhold ALUNBRIG, then dose reduce or permanently discontinue. (2.3, 5.3)
- Visual Disturbance: Advise patients to report visual symptoms. Withhold ALUNBRIG and obtain ophthalmologic evaluation, then dose reduce or permanently discontinue ALUNBRIG. (2.3, 5.4)
- Creatine Phosphokinase (CPK) Elevation: Monitor CPK levels regularly during treatment. Based on the severity and with muscle pain or weakness, withhold ALUNBRIG, then resume or reduce dose. (2.3, 5.5)
- Pancreatic Enzymes Elevation: Monitor lipase and amylase levels regularly during treatment. Based on the severity, withhold ALUNBRIG, then resume or reduce dose. (2.3, 5.6)
- Hepatotoxicity: Monitor alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total bilirubin levels regularly during treatment. Based on severity, withhold dose, then resume at lower level. (2.3, 5.7)
- Hyperglycemia: Assess fasting serum glucose prior to starting ALUNBRIG and regularly during treatment. If not adequately controlled with optimal medical management, withhold ALUNBRIG, then consider dose reduction or permanently discontinue, based on severity. (2.3, 5.8)
- Photosensitivity: Advise patients to limit sun exposure. Based on severity withhold ALUNBRIG, then resume at the same dose, reduce the dose, or permanently discontinue. (2.3, 5.9)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.10, 8.1, 8.3)
5.10 Embryo Fetal Toxicity (5.10 Embryo-Fetal Toxicity)
Based on its mechanism of action and findings in animals, ALUNBRIG can cause fetal harm when administered to pregnant women. There are no clinical data on the use of ALUNBRIG in pregnant women. Administration of brigatinib to pregnant rats during the period of organogenesis resulted in dose-related skeletal anomalies at doses as low as 12.5 mg/kg/day (approximately 0.7 times the human exposure by AUC at 180 mg once daily), as well as increased post-implantation loss, malformations, and decreased fetal body weight at doses of 25 mg/kg/day (approximately 1.26 times the human exposure at 180 mg once daily) or higher.
Advise women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ALUNBRIG and for at least 4 months following the final dose. Advise males with female partners of reproductive potential to use effective contraception during treatment and for at least 3 months after the last dose of ALUNBRIG [see Use in Specific Populations (8.1, 8.3), Clinical Pharmacology (12.1)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
90 mg orally once daily for the first 7 days; then increase to 180 mg orally once daily. May be taken with or without food. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 180 mg: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side
- 90 mg: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side
- 30 mg: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.6 Pancreatic Enzymes Elevation
In ALTA 1L, amylase elevation occurred in 52% of patients and Grade 3 or 4 amylase elevation occurred in 6.8% of patients. Lipase elevations occurred in 59% of patients and Grade 3 or 4 lipase elevation occurred in 17% of patients.
In ALTA, amylase elevation occurred in 27% of patients in the 90 mg group and 39% of patients in the 90→180 mg group. Lipase elevations occurred in 21% of patients in the 90 mg group and 45% of patients in the 90→180 mg group. Grade 3 or 4 amylase elevation occurred in 3.7% of patients in the 90 mg group and 2.7% of patients in the 90→180 mg group. Grade 3 or 4 lipase elevation occurred in 4.6% of patients in the 90 mg group and 5.5% of patients in the 90→180 mg group.
Monitor lipase and amylase during treatment with ALUNBRIG. Withhold ALUNBRIG for Grade 3 or 4 pancreatic enzyme elevation. Upon resolution or recovery to Grade 1 or baseline, resume ALUNBRIG at the same dose or at a reduced dose as described in Table 2 [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
180 mg tablets: oval, white to off-white film-coated tablets with "U13" debossed on one side and plain on the other side; available in:
| Bottle of 23 tablets | NDC 63020-180-23 |
| Bottle of 30 tablets | NDC 63020-180-30 |
90 mg tablets: oval, white to off-white film-coated tablets with "U7" debossed on one side and plain on the other side; available in:
| Bottle of 7 tablets | NDC 63020-090-07 |
| Bottle of 30 tablets | NDC 63020-090-30 |
30 mg tablets: round, white to off-white film-coated tablets with "U3" debossed on one side and plain on the other side; available in:
| Bottle of 30 tablets | NDC 63020-113-30 |
90 mg / 7 count tablets (NDC 63020-090-07) and 180 mg / 23 count tablets (NDC 63020-180-23) are also available in a single carton as a one-month initiation pack:
| One carton containing one bottle of 90 mg tablets (7 count) and one bottle of 180 mg tablets (23 count) | NDC 63020-198-30 |
Principal Display Panel Kit Carton (PRINCIPAL DISPLAY PANEL - Kit Carton)
NDC 63020-198-30
INITIATION PACK
ALUNBRIG®
(brigatinib) tablets
Rx Only | Oral Use
Usual Dose: One 90 mg tablet orally once daily for the first 7 days;
if tolerated, increase to one 180 mg tablet orally once daily.
See accompanying full Prescribing Information for complete details.
90 mg
7 Tablets
Take 1 tablet on
Day 1 - Day 7
180 mg
23 Tablets
Take 1 tablet on
Day 8 - Day 30
5.5 Creatine Phosphokinase (cpk) Elevation (5.5 Creatine Phosphokinase (CPK) Elevation)
In ALTA 1L, creatine phosphokinase (CPK) elevation occurred in 81% of patients who received ALUNBRIG. The incidence of Grade 3 or 4 CPK elevation was 24%. Dose reduction for CPK elevation occurred in 15% of patients.
In ALTA, CPK elevation occurred in 27% of patients receiving ALUNBRIG in the 90 mg group and 48% of patients in the 90 mg→180 mg group. The incidence of Grade 3 to 4 CPK elevation was 2.8% in the 90 mg group and 12% in the 90→180 mg group.
Dose reduction for CPK elevation occurred in 1.8% of patients in the 90 mg group and 4.5% in the 90→180 mg group.
Advise patients to report any unexplained muscle pain, tenderness, or weakness. Monitor CPK levels during ALUNBRIG treatment. Withhold ALUNBRIG for Grade 3 or 4 CPK elevation with Grade 2 or higher muscle pain or weakness. Upon resolution or recovery to Grade 1 CPK elevation or baseline, resume ALUNBRIG at the same dose or at a reduced dose as described in Table 2 [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
2.3 Dosage Modifications for Adverse Reactions
ALUNBRIG dosage reductions for adverse reactions are summarized in Table 1.
| Dosage Reduction | |||
|---|---|---|---|
| Dosage | First | Second | Third |
| 90 mg once daily | 60 mg once daily | permanently discontinue | N/A Not applicable
|
| 180 mg once daily | 120 mg once daily | 90 mg once daily | 60 mg once daily |
Once reduced for adverse reactions, do not subsequently increase the dosage of ALUNBRIG. Permanently discontinue ALUNBRIG if patients are unable to tolerate the 60 mg once daily dose.
Recommendations for dosage modifications of ALUNBRIG for the management of adverse reactions are provided in Table 2.
| Adverse Reaction | Severity Graded per National Cancer Institute Common Terminology Criteria for Adverse Events. Version 4.0 (NCI CTCAE v4).
|
Dosage Modifications |
|---|---|---|
| bpm = beats per minute; DBP = diastolic blood pressure; HR = heart rate; SBP = systolic blood pressure; ULN = upper limit of normal | ||
| Interstitial Lung Disease (ILD) /Pneumonitis [see Warnings and Precautions (5.1)] | Grade 1 |
|
| Grade 2 |
|
|
| Grade 3 or 4 | Permanently discontinue ALUNBRIG for ILD/pneumonitis. | |
| Hypertension [see Warnings and Precautions (5.2)] | Grade 3 hypertension (SBP greater than or equal to 160 mmHg or DBP greater than or equal to 100 mmHg, medical intervention indicated, more than one antihypertensive drug, or more intensive therapy than previously used indicated) |
|
| Grade 4 hypertension (life-threatening consequences, urgent intervention indicated) |
|
|
| Bradycardia (HR less than 60 bpm) [see Warnings and Precautions (5.3)] | Symptomatic bradycardia |
|
| Bradycardia with life-threatening consequences, urgent intervention indicated |
|
|
| Visual Disturbance [see Warnings and Precautions (5.4)] | Grade 2 or 3 visual disturbance | Withhold ALUNBRIG until recovery to Grade 1 or baseline, then resume at the next lower dose (Table 1). |
| Grade 4 visual disturbance | Permanently discontinue ALUNBRIG. | |
| Creatine Phosphokinase (CPK) Elevation [see Warnings and Precautions (5.5)] | Grade 3 or 4 CPK elevation (greater than 5× ULN) with Grade 2 or higher muscle pain or weakness |
|
| Lipase/Amylase Elevation [see Warnings and Precautions (5.6)] | Grade 3 lipase or amylase elevation (greater than 2× ULN) |
|
| Grade 4 lipase or amylase elevation (greater than 5× ULN) | Withhold ALUNBRIG until recovery to Grade 1 or less (less than or equal to 1.5× ULN) or to baseline, then resume ALUNBRIG at next lower dose (Table 1). | |
| Hepatotoxicity (Elevation of alanine aminotransferase (ALT) or aspartate aminotransferase (AST)) [see Warnings and Precautions (5.7)] | Grade 3 or 4 elevation (greater than 5 × ULN) of either ALT or AST with bilirubin less than or equal to 2 × ULN | Withhold ALUNBRIG until recovery to Grade 1 or less (less than or equal to 3× ULN) or to baseline, then resume ALUNBRIG at next lower dose (Table 1). |
| Grade 2 to 4 elevation (greater than 3 × ULN) of ALT or AST with concurrent total bilirubin elevation greater than 2 × ULN in the absence of cholestasis or hemolysis | Permanently discontinue ALUNBRIG. | |
| Hyperglycemia [see Warnings and Precautions (5.8)] | Grade 3 (greater than 250 mg/dL or 13.9 mmol/L) or 4 | If adequate hyperglycemic control cannot be achieved with optimal medical management, withhold ALUNBRIG until adequate hyperglycemic control is achieved and resume at the next lower dose (Table 1) or permanently discontinue ALUNBRIG. |
| Other | Grade 3 |
|
| Grade 4 |
|
5.1 Interstitial Lung Disease (ild)/pneumonitis (5.1 Interstitial Lung Disease (ILD)/Pneumonitis)
Severe, life-threatening, and fatal pulmonary adverse reactions consistent with interstitial lung disease (ILD)/pneumonitis have occurred with ALUNBRIG.
In Trial ALTA 1L, ILD/pneumonitis occurred in 5.1% of patients receiving ALUNBRIG. ILD/pneumonitis occurred within 8 days of initiation of ALUNBRIG in 2.9% of patients, with Grade 3 to 4 reactions occurring in 2.2% of patients.
In Trial ALTA, ILD/pneumonitis occurred in 3.7% of patients in the 90 mg group (90 mg once daily) and 9.1% of patients in the 90→180 mg group (180 mg once daily with 7-day lead-in at 90 mg once daily). Adverse reactions consistent with possible ILD/pneumonitis occurred within 9 days of initiation of ALUNBRIG (median onset was 2 days) in 6.4% of patients, with Grade 3 to 4 reactions occurring in 2.7%.
Monitor for new or worsening respiratory symptoms (e.g., dyspnea, cough, etc.), particularly during the first week of initiating ALUNBRIG. Withhold ALUNBRIG in any patient with new or worsening respiratory symptoms, and promptly evaluate for ILD/pneumonitis or other causes of respiratory symptoms (e.g., pulmonary embolism, tumor progression, and infectious pneumonia). For Grade 1 or 2 ILD/pneumonitis, either resume ALUNBRIG with dose reduction according to Table 1 after recovery to baseline or permanently discontinue ALUNBRIG. Permanently discontinue ALUNBRIG for Grade 3 or 4 ILD/pneumonitis or recurrence of Grade 1 or 2 ILD/pneumonitis [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
2.5 Dosage Modifications for Moderate Cyp3a Inducers (2.5 Dosage Modifications for Moderate CYP3A Inducers)
Avoid coadministration of moderate CYP3A inducers during treatment with ALUNBRIG [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If coadministration of a moderate CYP3A inducer cannot be avoided, increase the ALUNBRIG once daily dose in 30 mg increments after 7 days of treatment with the current ALUNBRIG dose as tolerated, up to a maximum of twice the ALUNBRIG dose that was tolerated prior to initiating the moderate CYP3A inducer.
After discontinuation of a moderate CYP3A inducer, resume the ALUNBRIG dose that was tolerated prior to initiating the moderate CYP3A inducer.
Principal Display Panel 30 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 30 mg Tablet Bottle Carton)
NDC 63020-113-30
ALUNBRIG®
(brigatinib) tablets
30 mg
Rx Only
30 Tablets
Principal Display Panel 90 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 90 mg Tablet Bottle Carton)
NDC 63020-090-30
ALUNBRIG®
(brigatinib) tablets
90 mg
Rx Only
30 Tablets
Principal Display Panel 180 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 180 mg Tablet Bottle Carton)
NDC 63020-180-30
ALUNBRIG®
(brigatinib) tablets
180 mg
Rx Only
30 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with brigatinib.
Treatment with brigatinib resulted in chromosomal damage in an in vivo mammalian erythrocyte micronucleus in the rat, but was not mutagenic in the Ames or in vitro mammalian chromosome aberration tests.
Dedicated animal fertility studies were not conducted with brigatinib. Testicular toxicity was observed in repeat-dose animal studies at doses resulting in exposure as low as 0.2 times the exposure in patients at the 180 mg dose. In rats, findings included lower weight of testes, seminal vesicles and prostate gland, and testicular tubular degeneration; these effects were not reversible during the 2 month recovery period. In monkeys, findings included reduced size of testes along with microscopic evidence of hypospermatogenesis; these effects were reversible during the recovery period.
2.4 Dosage Modifications for Strong Or Moderate Cyp3a Inhibitors (2.4 Dosage Modifications for Strong or Moderate CYP3A Inhibitors)
Avoid coadministration of strong or moderate CYP3A inhibitors during treatment with ALUNBRIG [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If coadministration of a strong CYP3A inhibitor cannot be avoided, reduce the ALUNBRIG once daily dose by approximately 50% (i.e., from 180 mg to 90 mg, or from 90 mg to 60 mg). If coadministration of a moderate CYP3A inhibitor cannot be avoided, reduce the ALUNBRIG once daily dose by approximately 40% (i.e., from 180 mg to 120 mg, 120 mg to 90 mg, or from 90 mg to 60 mg). After discontinuation of a strong or moderate CYP3A inhibitor, resume the ALUNBRIG dose that was tolerated prior to initiating the CYP3A inhibitor.
2.7 Dosage Modifications for Patients With Severe Renal Impairment (2.7 Dosage Modifications for Patients with Severe Renal Impairment)
Reduce the ALUNBRIG once daily dose by approximately 50% (i.e., from 180 mg to 90 mg, or from 90 mg to 60 mg) for patients with severe renal impairment [creatinine clearance (CLcr) 15 to 29 mL/min by Cockcroft-Gault] [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
2.6 Dosage Modifications for Patients With Severe Hepatic Impairment (2.6 Dosage Modifications for Patients with Severe Hepatic Impairment)
Reduce the ALUNBRIG once daily dose by approximately 40% (i.e., from 180 mg to 120 mg, 120 mg to 90 mg, or from 90 mg to 60 mg) for patients with severe hepatic impairment (Child-Pugh C) [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:25.377549 · Updated: 2026-03-14T22:43:43.392452