These Highlights Do Not Include All The Information Needed To Use Desloratadine Tablets Safely And Effectively. See Full Prescribing Information For Desloratadine Tablets.
0fe374d0-c14b-4102-b568-eb79165fc7ef
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Desloratadine Tablets are a histamine-1 (H1) receptor antagonist indicated for: • Seasonal Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.1 ) • Perennial Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.2 ) • Chronic Idiopathic Urticaria: symptomatic relief of pruritus, reduction in the number of hives, and size of hives in patients 12 years of age and older. ( 1.3 )
Indications and Usage
Desloratadine Tablets are a histamine-1 (H1) receptor antagonist indicated for: • Seasonal Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.1 ) • Perennial Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.2 ) • Chronic Idiopathic Urticaria: symptomatic relief of pruritus, reduction in the number of hives, and size of hives in patients 12 years of age and older. ( 1.3 )
Dosage and Administration
Desloratadine Tablets may be taken without regard to meals.
Warnings and Precautions
• Hypersensitivity reactions including rash, pruritus, urticaria, edema, dyspnea, and anaphylaxis have been reported. In such cases, stop Desloratadine Tablets at once and consider alternative treatments. ( 5.1 )
Contraindications
Desloratadine Tablets are contraindicated in patients who are hypersensitive to this medication or to any of its ingredients or to loratadine [ see Warnings and Precautions (5.1) and Adverse Reactions (6.2) ].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label: • Hypersensitivity reactions. [ See Warnings and Precautions (5.1) .]
Storage and Handling
Desloratadine Tablets: Debossed "5", light blue, round tablets that are packaged in high-density polyethylene plastic bottles of 100 (NDC 69543-107-10) and 500 (NDC 69543-107-50).
How Supplied
Desloratadine Tablets: Debossed "5", light blue, round tablets that are packaged in high-density polyethylene plastic bottles of 100 (NDC 69543-107-10) and 500 (NDC 69543-107-50).
Patient Information
Desloratadine Tablets Read the Patient Information that comes with Desloratadine Tablets before you start taking it and each time you get a refill. There may be new information. This leaflet is a summary of the information for patients. Your doctor or pharmacist can give you additional information. This leaflet does not take the place of talking to your doctor about your medical condition or treatment. What are Desloratadine Tablets? Desloratadine Tablets are a prescription medicine that contains the medicine desloratadine (an antihistamine). Desloratadine Tablets are used to help control the symptoms of: • seasonal allergic rhinitis (sneezing, stuffy nose, runny nose and itching of the nose) in people 12 years of age and older. • perennial allergic rhinitis (sneezing, stuffy nose, runny nose and itching of the nose) in people 12 years of age and older. • chronic idiopathic urticaria (long-term itching) and to reduce the number and size of hives in people 12 years of age and older. Desloratadine Tablets, 5 mg is not for children younger than 12 years of age. Who should not take Desloratadine Tablets? Do not take Desloratadine Tablets if you: • are allergic to desloratadine or any of the ingredients in Desloratadine Tablets. See the end of this leaflet for a complete list of ingredients. • are allergic to loratadine (Alavert, Claritin). Talk to your doctor before taking this medicine if you have any questions about whether or not to take this medicine. What should I tell my doctor before taking Desloratadine Tablets? Before you take Desloratadine Tablets, tell your doctor if you: • have liver or kidney problems. • have any other medical conditions. • are pregnant or plan to become pregnant. It is not known if Desloratadine will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant. • are breast-feeding or plan to breast-feed. Desloratadine can pass into your breast milk. Talk to your doctor about the best way to feed your baby if you take Desloratadine. Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. Desloratadine may affect the way other medicines work, and other medicines may affect how Desloratadine works. Especially tell your doctor if you take: • ketoconazole (Nizoral) • erythromycin (Ery-tab, Eryc, PCE) • azithromycin (Zithromax, Zmax) • antihistamines • fluoxetine (Prozac) • cimetidine (Tagamet) Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine. How should I take Desloratadine Tablets? • Take Desloratadine Tablets exactly as your doctor tells you to take it. • Do not change your dose of Desloratadine Tablets or take more often than prescribed. • Desloratadine Tablets can be taken with or without food. • If you take too many Desloratadine Tablets, call your doctor or get medical attention right away. What are the possible side effects of Desloratadine Tablets? Desloratadine Tablets may cause serious side effects, including: • Allergic reactions. Stop taking Desloratadine Tablets and call your doctor right away or get emergency help if you have any of these symptoms: o rash o itching o hives o swelling of your lips, tongue, face, and throat o shortness of breath or trouble breathing The most common side effects of Desloratadine Tablets in adults and children 12 years of age and older with allergic rhinitis include: • sore throat • dry mouth • muscle pain • tiredness • sleepiness • menstrual pain Increased sleepiness or tiredness can happen if you take more Desloratadine Tablets than your doctor prescribed to you. Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Desloratadine Tablets. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to Virtus Pharmaceuticals, LLC at 1-888-848-3593 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. How should I store Desloratadine Tablets? • Store Desloratadine Tablets between 59°F to 86°F (15°C to 30°C). • Desloratadine Tablets are sensitive to heat. Do not store above 86°F (30°C). • Protect Desloratadine Tablets from moisture. Keep Desloratadine Tablets , and all medicines out of the reach of children. General information about Desloratadine Tablets Medicines are sometimes prescribed for purposes other than those listed in a patient information leaflet. Do not use Desloratadine Tablets for a condition for which it was not prescribed. Do not give Desloratadine Tablets to other people, even if they have the same condition you have. It may harm them. This Patient Information leaflet summarizes the most important information about Desloratadine Tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Desloratadine Tablets that is written for health professionals. What are the ingredients in Desloratadine Tablets? Active ingredient: Desloratadine Inactive ingredients in Desloratadine Tablets: microcrystalline cellulose NF, pregelatinized starch NF, croscarmellose sodium NF, talc USP, zinc stearate, USP and FD&C Blue #2 HT 11-14%. Manufactured for: Virtus Pharmaceuticals, LLC Langhorne, PA 19047 1-888-848-3593 MADE IN USA Rev. 10/2020 R-2010
Medication Information
Warnings and Precautions
• Hypersensitivity reactions including rash, pruritus, urticaria, edema, dyspnea, and anaphylaxis have been reported. In such cases, stop Desloratadine Tablets at once and consider alternative treatments. ( 5.1 )
Indications and Usage
Desloratadine Tablets are a histamine-1 (H1) receptor antagonist indicated for: • Seasonal Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.1 ) • Perennial Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.2 ) • Chronic Idiopathic Urticaria: symptomatic relief of pruritus, reduction in the number of hives, and size of hives in patients 12 years of age and older. ( 1.3 )
Dosage and Administration
Desloratadine Tablets may be taken without regard to meals.
Contraindications
Desloratadine Tablets are contraindicated in patients who are hypersensitive to this medication or to any of its ingredients or to loratadine [ see Warnings and Precautions (5.1) and Adverse Reactions (6.2) ].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label: • Hypersensitivity reactions. [ See Warnings and Precautions (5.1) .]
Storage and Handling
Desloratadine Tablets: Debossed "5", light blue, round tablets that are packaged in high-density polyethylene plastic bottles of 100 (NDC 69543-107-10) and 500 (NDC 69543-107-50).
How Supplied
Desloratadine Tablets: Debossed "5", light blue, round tablets that are packaged in high-density polyethylene plastic bottles of 100 (NDC 69543-107-10) and 500 (NDC 69543-107-50).
Patient Information
Desloratadine Tablets
Read the Patient Information that comes with Desloratadine Tablets before you start taking it and each time you get a refill. There may be new information. This leaflet is a summary of the information for patients. Your doctor or pharmacist can give you additional information. This leaflet does not take the place of talking to your doctor about your medical condition or treatment.
What are Desloratadine Tablets?
Desloratadine Tablets are a prescription medicine that contains the medicine desloratadine (an antihistamine).
Desloratadine Tablets are used to help control the symptoms of:
-
•seasonal allergic rhinitis (sneezing, stuffy nose, runny nose and itching of the nose) in people 12 years of age and older.
-
•perennial allergic rhinitis (sneezing, stuffy nose, runny nose and itching of the nose) in people 12 years of age and older.
-
•chronic idiopathic urticaria (long-term itching) and to reduce the number and size of hives in people 12 years of age and older.
Desloratadine Tablets, 5 mg is not for children younger than 12 years of age.
Who should not take Desloratadine Tablets?
Do not take Desloratadine Tablets if you:
-
•are allergic to desloratadine or any of the ingredients in Desloratadine Tablets. See the end of this leaflet for a complete list of ingredients.
-
•are allergic to loratadine (Alavert, Claritin).
Talk to your doctor before taking this medicine if you have any questions about whether or not to take this medicine.
What should I tell my doctor before taking Desloratadine Tablets?
Before you take Desloratadine Tablets, tell your doctor if you:
-
•have liver or kidney problems.
-
•have any other medical conditions.
-
•are pregnant or plan to become pregnant. It is not known if Desloratadine will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
-
•are breast-feeding or plan to breast-feed. Desloratadine can pass into your breast milk. Talk to your doctor about the best way to feed your baby if you take Desloratadine.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. Desloratadine may affect the way other medicines work, and other medicines may affect how Desloratadine works. Especially tell your doctor if you take:
-
•ketoconazole (Nizoral)
-
•erythromycin (Ery-tab, Eryc, PCE)
-
•azithromycin (Zithromax, Zmax)
-
•antihistamines
-
•fluoxetine (Prozac)
-
•cimetidine (Tagamet)
Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine.
How should I take Desloratadine Tablets?
-
•Take Desloratadine Tablets exactly as your doctor tells you to take it.
-
•Do not change your dose of Desloratadine Tablets or take more often than prescribed.
-
•Desloratadine Tablets can be taken with or without food.
-
•If you take too many Desloratadine Tablets, call your doctor or get medical attention right away.
What are the possible side effects of Desloratadine Tablets?
Desloratadine Tablets may cause serious side effects, including:
-
•Allergic reactions. Stop taking Desloratadine Tablets and call your doctor right away or get emergency help if you have any of these symptoms:
-
orash
-
oitching
-
ohives
-
oswelling of your lips, tongue, face, and throat
-
oshortness of breath or trouble breathing
The most common side effects of Desloratadine Tablets in adults and children 12 years of age and older with allergic rhinitis include:
-
•sore throat
-
•dry mouth
-
•muscle pain
-
•tiredness
-
•sleepiness
-
•menstrual pain
Increased sleepiness or tiredness can happen if you take more Desloratadine Tablets than your doctor prescribed to you.
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of Desloratadine Tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to Virtus Pharmaceuticals, LLC at 1-888-848-3593 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How should I store Desloratadine Tablets?
-
•Store Desloratadine Tablets between 59°F to 86°F (15°C to 30°C).
-
•Desloratadine Tablets are sensitive to heat. Do not store above 86°F (30°C).
-
•Protect Desloratadine Tablets from moisture.
Keep Desloratadine Tablets, and all medicines out of the reach of children.
General information about Desloratadine Tablets
Medicines are sometimes prescribed for purposes other than those listed in a patient information leaflet. Do not use Desloratadine Tablets for a condition for which it was not prescribed. Do not give Desloratadine Tablets to other people, even if they have the same condition you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Desloratadine Tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Desloratadine Tablets that is written for health professionals.
What are the ingredients in Desloratadine Tablets?
Active ingredient: Desloratadine
Inactive ingredients in Desloratadine Tablets:
microcrystalline cellulose NF, pregelatinized starch NF, croscarmellose sodium NF, talc USP, zinc stearate, USP and FD&C Blue #2 HT 11-14%.
Manufactured for:
Virtus Pharmaceuticals, LLC
Langhorne, PA 19047
1-888-848-3593
MADE IN USA
Rev. 10/2020
R-2010
Description
Desloratadine Tablets are a histamine-1 (H1) receptor antagonist indicated for: • Seasonal Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.1 ) • Perennial Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. ( 1.2 ) • Chronic Idiopathic Urticaria: symptomatic relief of pruritus, reduction in the number of hives, and size of hives in patients 12 years of age and older. ( 1.3 )
Section 42229-5
Adults and Adolescents
Section 44425-7
Storage
-
•Desloratadine Tablets: Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature]. Heat sensitive. Avoid exposure at or above 30°C (86°F).
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Section 51945-4
PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label
Rx only
NDC: 69543-107-10
Desloratadine
TABLETS
5 mg
100 Tablets
VIRTUS®
PHARMACEUTICALS
10 Overdosage
In the event of overdose, consider standard measures to remove any unabsorbed drug. Symptomatic and supportive treatment is recommended. Desloratadine and 3-hydroxydesloratadine are not eliminated by hemodialysis.
Information regarding acute overdosage is limited to experience from post-marketing adverse event reports and from clinical trials conducted during the development of the Desloratadine product. In a dose-ranging trial, at doses of 10 mg and 20 mg/day somnolence was reported.
In another study, no clinically relevant adverse events were reported in normal male and female volunteers who were given single daily doses of Desloratadine 45 mg for 10 days [see Clinical Pharmacology (12.2) ].
8.1 Pregnancy
Risk Summary
The limited available data with Desloratadine in pregnant women are not sufficient to inform a drug-associated risk for major birth defects and miscarriage. There are no adequate and well-controlled studies in pregnant women. Desloratadine given during organogenesis to pregnant rats was not teratogenic at the summed area under the concentration-time curve (AUC)-based exposures of desloratadine and its metabolite approximately 320 times that at the recommended human daily oral dose (RHD) of 5 mg/day. Desloratadine given during organogenesis to pregnant rabbits was not teratogenic at the AUC-based exposures of desloratadine approximately 230 times that at the RHD. Desloratadine given to pregnant rats during organogenesis through lactation resulted in reduced body weight and slow righting reflex of F1 pups at the summed AUC-based exposures of desloratadine and its metabolite approximately 70 times or greater than that at the RHD [see Data ].
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Desloratadine was given orally during organogenesis to pregnant rats at doses of 6, 24 and 48 mg/kg/day (approximately 50, 200 and 320 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). No fetal malformations were present. Reduced fetal weights and skeletal variations noted at doses of 24 and 48 mg/kg/day were likely secondary to the maternal toxicities of reduced body weight gain and food consumption observed at the same doses. Desloratadine was also given orally during organogenesis to pregnant rabbits at doses of 15, 30 and 60 mg/kg/day (approximately 30, 70 and 230 times the AUC-based exposure of desloratadine at the RHD). No adverse effects to the fetus were noted. Reduced maternal body weight gain was noted in rabbits at 60 mg/kg/day. In a peri- and post-natal development study, desloratadine was given to rats orally during the perinatal (Gestation Day 6) through lactation periods (Postpartum Day 21) at doses of 3, 9 and 18 mg/kg/day. Reduced body weight and slow righting reflex were reported in F1 pups at doses of 9 mg/kg/day or greater (approximately 70 times or greater than the summed AUC-based exposure of desloratadine and its metabolite at the RHD). Desloratadine had no effect on F1 pup development at 3 mg/kg/day (approximately 10 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). Maternal toxicities including reduced body weight gain and food consumption were noted at 18 mg/kg/day for F0 dams. F1 offspring were subsequently mated and there was no developmental toxicity for F2 pups observed.
8.2 Lactation
Risk Summary
Desloratadine passes into breast milk. There are not sufficient data on the effects of desloratadine on the breastfed infant or the effects of desloratadine on milk production. The decision should be made whether to discontinue nursing or to discontinue desloratadine, taking into account the developmental and health benefits of breastfeeding, the nursing mother's clinical need, and any potential adverse effects on the breastfed infant from desloratadine or from the underlying maternal condition.
11 Description
Desloratadine Tablets are light blue, round, tablets containing 5 mg desloratadine, an antihistamine, to be administered orally. Desloratadine Tablets also contain the following excipients: microcrystalline cellulose NF, pregelatinized starch NF, croscarmellose sodium NF, talc USP, zinc stearate, USP and FD&C Blue #2 HT 11-14%.
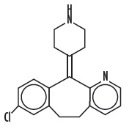
Desloratadine is a white to off-white powder that is slightly soluble in water, but very soluble in ethanol and propylene glycol. It has an empirical formula: C19H19ClN2 and a molecular weight of 310.8. The chemical name is 8-chloro-6,11-dihydro-11-(4-piperdinylidene)-5H-benzo[5,6]cyclohepta[1,2-b]pyridine and has the following structure:
7.2 Fluoxetine
In controlled clinical studies co-administration of desloratadine with fluoxetine, a selective serotonin reuptake inhibitor (SSRI), resulted in increased plasma concentrations of desloratadine and 3 hydroxydesloratadine, but there were no clinically relevant changes in the safety profile of desloratadine. [See Clinical Pharmacology (12.3) .]
7.3 Cimetidine
In controlled clinical studies co-administration of desloratadine with cimetidine, a histamine H2-receptor antagonist, resulted in increased plasma concentrations of desloratadine and 3 hydroxydesloratadine, but there were no clinically relevant changes in the safety profile of desloratadine. [See Clinical Pharmacology (12.3).]
8.4 Pediatric Use
The recommended dose of Desloratadine Oral Solution in the pediatric population is based on cross-study comparison of the plasma concentration of Desloratadine in adults and pediatric subjects. The safety of Desloratadine Oral Solution has been established in 246 pediatric subjects aged 6 months to 11 years in three placebo-controlled clinical studies. Since the course of seasonal and perennial allergic rhinitis and chronic idiopathic urticaria and the effects of Desloratadine are sufficiently similar in the pediatric and adult populations, it allows extrapolation from the adult efficacy data to pediatric patients. The effectiveness of Desloratadine Oral Solution in these age groups is supported by evidence from adequate and well-controlled studies of Desloratadine Tablets in adults. The safety and effectiveness of Desloratadine Tablets or Desloratadine Oral Solution have not been demonstrated in pediatric patients less than 6 months of age. [See Clinical Pharmacology (12.3).]
8.5 Geriatric Use
Clinical studies of desloratadine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. [See Clinical Pharmacology (12.3).]
4 Contraindications
Desloratadine Tablets are contraindicated in patients who are hypersensitive to this medication or to any of its ingredients or to loratadine [see Warnings and Precautions (5.1) and Adverse Reactions (6.2) ].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label:
-
•Hypersensitivity reactions. [See Warnings and Precautions (5.1) .]
8.6 Renal Impairment
Dosage adjustment for patients with renal impairment is recommended [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
8.7 Hepatic Impairment
Dosage adjustment for patients with hepatic impairment is recommended [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
1 Indications and Usage
Desloratadine Tablets are a histamine-1 (H1) receptor antagonist indicated for:
-
•Seasonal Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. (1.1)
-
•Perennial Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. (1.2)
-
•Chronic Idiopathic Urticaria: symptomatic relief of pruritus, reduction in the number of hives, and size of hives in patients 12 years of age and older. (1.3)
12.1 Mechanism of Action
Desloratadine is a long-acting tricyclic histamine antagonist with selective H1-receptor histamine antagonist activity. Receptor binding data indicates that at a concentration of 2-3 ng/mL (7 nanomolar), desloratadine shows significant interaction with the human histamine H1-receptor. Desloratadine inhibited histamine release from human mast cells in vitro. Results of a radiolabeled tissue distribution study in rats and a radioligand H1-receptor binding study in guinea pigs showed that desloratadine did not readily cross the blood brain barrier. The clinical significance of this finding is unknown.
5 Warnings and Precautions
-
•Hypersensitivity reactions including rash, pruritus, urticaria, edema, dyspnea, and anaphylaxis have been reported. In such cases, stop Desloratadine Tablets at once and consider alternative treatments. (5.1)
2 Dosage and Administration
Desloratadine Tablets may be taken without regard to meals.
9 Drug Abuse and Dependence
There is no information to indicate that abuse or dependency occurs with Desloratadine Tablets.
3 Dosage Forms and Strengths
Desloratadine Tablets are light blue round tablets debossed with "5" containing 5 mg desloratadine.
17.1 Information for Patients
-
•Patients should be instructed to use Desloratadine Tablets as directed.
-
•As there are no food effects on bioavailability, patients can be instructed that Desloratadine Tablets may be taken without regard to meals.
-
•Patients should be advised not to increase the dose or dosing frequency as studies have not demonstrated increased effectiveness at higher doses and somnolence may occur.
6.2 Post Marketing Experience
Because adverse events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following spontaneous adverse events have been reported during the marketing of desloratadine:
Cardiac disorders: tachycardia, palpitations
Respiratory, thoracic and mediastinal disorders: dyspnea
Skin and subcutaneous tissue disorders: rash, pruritus
Nervous system disorders: psychomotor hyperactivity, movement disorders (including dystonia, tics, and extrapyramidal symptoms), seizures (reported in patients with and without a known seizure disorder)
Immune system disorders: hypersensitivity reactions (such as urticaria, edema and anaphylaxis)
Investigations: elevated liver enzymes including bilirubin
Hepatobiliary disorders: hepatitis
Metabolism and nutrition disorders: increased appetite
8 Use in Specific Populations
1.1 Seasonal Allergic Rhinitis
Desloratadine Tablets are indicated for the relief of the nasal and non-nasal symptoms of seasonal allergic rhinitis in patients 12 years of age and older.
5.1 Hypersensitivity Reactions
Hypersensitivity reactions including rash, pruritus, urticaria, edema, dyspnea, and anaphylaxis have been reported after administration of desloratadine. If such a reaction occurs, therapy with Desloratadine Tablets should be stopped and alternative treatment should be considered. [See Adverse Reactions (6.2) .]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
1.2 Perennial Allergic Rhinitis
Desloratadine Tablets are indicated for the relief of the nasal and non-nasal symptoms of perennial allergic rhinitis in patients 12 years of age and older.
14.1 Seasonal Allergic Rhinitis
The clinical efficacy and safety of Desloratadine Tablets were evaluated in over 2300 patients 12 to 75 years of age with seasonal allergic rhinitis. A total of 1838 patients received 2.5 to 20 mg/day of Desloratadine in 4 double-blind, randomized, placebo-controlled clinical trials of 2 to 4 weeks' duration conducted in the United States. The results of these studies demonstrated the efficacy and safety of Desloratadine 5 mg in the treatment of adult and adolescent patients with seasonal allergic rhinitis. In a dose-ranging trial, Desloratadine 2.5 to 20 mg/day was studied. Doses of 5, 7.5, 10, and 20 mg/day were superior to placebo; and no additional benefit was seen at doses above 5.0 mg. In the same study, an increase in the incidence of somnolence was observed at doses of 10 mg/day and 20 mg/day (5.2% and 7.6%, respectively), compared to placebo (2.3%).
In two 4-week studies of 924 patients (aged 15 to 75 years) with seasonal allergic rhinitis and concomitant asthma, Desloratadine Tablets 5 mg once daily improved rhinitis symptoms, with no decrease in pulmonary function. This supports the safety of administering Desloratadine Tablets to adult patients with seasonal allergic rhinitis with mild to moderate asthma.
Desloratadine Tablets 5 mg once daily significantly reduced the Total Symptom Score (the sum of individual scores of nasal and non-nasal symptoms) in patients with seasonal allergic rhinitis. See Table 3.
|
Treatment Group
(n) |
Mean Baseline
At baseline, a total nasal symptom score (sum of 4 individual symptoms) of at least 6 and a total non-nasal symptom score (sum of 4 individual symptoms) of at least 5 (each symptom scored 0 to 3 where 0=no symptom and 3=severe symptoms) was required for trial eligibility. TSS ranges from 0=no symptoms to 24=maximal symptoms.
(SEM) |
Change from Baseline
Mean reduction in TSS averaged over the 2-week treatment period.
(SEM) |
Placebo Comparison
(P-value) |
|---|---|---|---|
| SEM = Standard Error of the Mean | |||
|
Desloratadine
|
14.2 (0.3) |
-4.3 (0.3) |
P<0.01 |
|
Placebo (173) |
13.7 (0.3) |
-2.5 (0.3) |
There were no significant differences in the effectiveness of Desloratadine Tablets 5 mg across subgroups of patients defined by gender, age, or race.
1.3 Chronic Idiopathic Urticaria
Desloratadine Tablets are indicated for the symptomatic relief of pruritus, reduction in the number of hives, and size of hives, in patients with chronic idiopathic urticaria 12 years of age and older.
14.2 Perennial Allergic Rhinitis
The clinical efficacy and safety of Desloratadine Tablets 5 mg were evaluated in over 1300 patients 12 to 80 years of age with perennial allergic rhinitis. A total of 685 patients received 5 mg/day of Desloratadine in two double-blind, randomized, placebo-controlled clinical trials of 4 weeks' duration conducted in the United States and internationally. In one of these studies Desloratadine Tablets 5 mg once daily was shown to significantly reduce the Total Symptom Score in patients with perennial allergic rhinitis (Table 4).
|
Treatment Group
(n) |
Mean Baseline
At baseline, average of total symptom score (sum of 5 individual nasal symptoms and 3 non-nasal symptoms, each symptom scored 0 to 3 where 0=no symptom and 3=severe symptoms) of at least 10 was required for trial eligibility. TSS ranges from 0=no symptoms to 24=maximal symptoms.
(SEM) |
Change from Baseline
Mean reduction in TSS averaged over the 4 week treatment period.
(SEM) |
Placebo Comparison
(P-value) |
|---|---|---|---|
| SEM = Standard Error of the Mean | |||
|
Desloratadine
|
12.37 (0.18) |
-4.06 (0.21) |
P=0.01 |
|
Placebo (337) |
12.30 (0.18) |
-3.27 (0.21) |
14.3 Chronic Idiopathic Urticaria
The efficacy and safety of Desloratadine Tablets 5 mg once daily was studied in 416 chronic idiopathic urticaria patients 12 to 84 years of age, of whom 211 received Desloratadine. In two double-blind, placebo-controlled, randomized clinical trials of six weeks duration, at the pre-specified one-week primary time point evaluation, Desloratadine Tablets significantly reduced the severity of pruritus when compared to placebo (Table 5). Secondary endpoints were also evaluated, and during the first week of therapy Desloratadine Tablets 5 mg reduced the secondary endpoints, "Number of Hives" and the "Size of the Largest Hive," when compared to placebo.
|
Treatment Group
(n) |
Mean Baseline
(SEM) |
Change from Baseline
Mean reduction in pruritus averaged over the first week of treatment.
(SEM) |
Placebo Comparison
(P-value) |
|---|---|---|---|
| Pruritus scored 0 to 3 where 0=no symptom to 3=maximal symptom | |||
| SEM = Standard Error of the Mean | |||
|
Desloratadine
|
2.19 (0.04) |
-1.05 (0.07) |
P<0.01 |
|
Placebo (110) |
2.21 (0.04) |
-0.52 (0.07) |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Desloratadine Tablets: Debossed "5", light blue, round tablets that are packaged in high-density polyethylene plastic bottles of 100 (NDC 69543-107-10) and 500 (NDC 69543-107-50).
7.1 Inhibitors of Cytochrome P450 3a4
In controlled clinical studies co-administration of desloratadine with ketoconazole, erythromycin, or azithromycin resulted in increased plasma concentrations of desloratadine and 3 hydroxydesloratadine, but there were no clinically relevant changes in the safety profile of desloratadine. [See Clinical Pharmacology (12.3) .]
2.5 Adults With Hepatic Or Renal Impairment
In adult patients with liver or renal impairment, a starting dose of one 5 mg tablet every other day is recommended based on pharmacokinetic data. Dosing recommendation for children with liver or renal impairment cannot be made due to lack of data [see Clinical Pharmacology (12.3) ].
8.3 Females and Males of Reproductive Potential
Infertility
There are no data available on human infertility associated with desloratadine.
There were no clinically relevant effects of desloratadine on female fertility in rats. A male specific decrease in fertility occurred at an oral desloratadine dose of 12 mg/kg or greater in rats (approximately 65 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). Male fertility was unaffected at a desloratadine dose of 3 mg/kg (approximately 10 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). [See Nonclinical Toxicology (13.1) .]
2.1 Adults and Adolescents 12 Years of Age and Over
The recommended dose of Desloratadine Tablets is one 5-mg tablet once daily.
Structured Label Content
Section 42229-5 (42229-5)
Adults and Adolescents
Section 44425-7 (44425-7)
Storage
-
•Desloratadine Tablets: Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature]. Heat sensitive. Avoid exposure at or above 30°C (86°F).
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label
Rx only
NDC: 69543-107-10
Desloratadine
TABLETS
5 mg
100 Tablets
VIRTUS®
PHARMACEUTICALS
10 Overdosage (10 OVERDOSAGE)
In the event of overdose, consider standard measures to remove any unabsorbed drug. Symptomatic and supportive treatment is recommended. Desloratadine and 3-hydroxydesloratadine are not eliminated by hemodialysis.
Information regarding acute overdosage is limited to experience from post-marketing adverse event reports and from clinical trials conducted during the development of the Desloratadine product. In a dose-ranging trial, at doses of 10 mg and 20 mg/day somnolence was reported.
In another study, no clinically relevant adverse events were reported in normal male and female volunteers who were given single daily doses of Desloratadine 45 mg for 10 days [see Clinical Pharmacology (12.2) ].
8.1 Pregnancy
Risk Summary
The limited available data with Desloratadine in pregnant women are not sufficient to inform a drug-associated risk for major birth defects and miscarriage. There are no adequate and well-controlled studies in pregnant women. Desloratadine given during organogenesis to pregnant rats was not teratogenic at the summed area under the concentration-time curve (AUC)-based exposures of desloratadine and its metabolite approximately 320 times that at the recommended human daily oral dose (RHD) of 5 mg/day. Desloratadine given during organogenesis to pregnant rabbits was not teratogenic at the AUC-based exposures of desloratadine approximately 230 times that at the RHD. Desloratadine given to pregnant rats during organogenesis through lactation resulted in reduced body weight and slow righting reflex of F1 pups at the summed AUC-based exposures of desloratadine and its metabolite approximately 70 times or greater than that at the RHD [see Data ].
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Desloratadine was given orally during organogenesis to pregnant rats at doses of 6, 24 and 48 mg/kg/day (approximately 50, 200 and 320 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). No fetal malformations were present. Reduced fetal weights and skeletal variations noted at doses of 24 and 48 mg/kg/day were likely secondary to the maternal toxicities of reduced body weight gain and food consumption observed at the same doses. Desloratadine was also given orally during organogenesis to pregnant rabbits at doses of 15, 30 and 60 mg/kg/day (approximately 30, 70 and 230 times the AUC-based exposure of desloratadine at the RHD). No adverse effects to the fetus were noted. Reduced maternal body weight gain was noted in rabbits at 60 mg/kg/day. In a peri- and post-natal development study, desloratadine was given to rats orally during the perinatal (Gestation Day 6) through lactation periods (Postpartum Day 21) at doses of 3, 9 and 18 mg/kg/day. Reduced body weight and slow righting reflex were reported in F1 pups at doses of 9 mg/kg/day or greater (approximately 70 times or greater than the summed AUC-based exposure of desloratadine and its metabolite at the RHD). Desloratadine had no effect on F1 pup development at 3 mg/kg/day (approximately 10 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). Maternal toxicities including reduced body weight gain and food consumption were noted at 18 mg/kg/day for F0 dams. F1 offspring were subsequently mated and there was no developmental toxicity for F2 pups observed.
8.2 Lactation
Risk Summary
Desloratadine passes into breast milk. There are not sufficient data on the effects of desloratadine on the breastfed infant or the effects of desloratadine on milk production. The decision should be made whether to discontinue nursing or to discontinue desloratadine, taking into account the developmental and health benefits of breastfeeding, the nursing mother's clinical need, and any potential adverse effects on the breastfed infant from desloratadine or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Desloratadine Tablets are light blue, round, tablets containing 5 mg desloratadine, an antihistamine, to be administered orally. Desloratadine Tablets also contain the following excipients: microcrystalline cellulose NF, pregelatinized starch NF, croscarmellose sodium NF, talc USP, zinc stearate, USP and FD&C Blue #2 HT 11-14%.
Desloratadine is a white to off-white powder that is slightly soluble in water, but very soluble in ethanol and propylene glycol. It has an empirical formula: C19H19ClN2 and a molecular weight of 310.8. The chemical name is 8-chloro-6,11-dihydro-11-(4-piperdinylidene)-5H-benzo[5,6]cyclohepta[1,2-b]pyridine and has the following structure:
7.2 Fluoxetine
In controlled clinical studies co-administration of desloratadine with fluoxetine, a selective serotonin reuptake inhibitor (SSRI), resulted in increased plasma concentrations of desloratadine and 3 hydroxydesloratadine, but there were no clinically relevant changes in the safety profile of desloratadine. [See Clinical Pharmacology (12.3) .]
7.3 Cimetidine
In controlled clinical studies co-administration of desloratadine with cimetidine, a histamine H2-receptor antagonist, resulted in increased plasma concentrations of desloratadine and 3 hydroxydesloratadine, but there were no clinically relevant changes in the safety profile of desloratadine. [See Clinical Pharmacology (12.3).]
8.4 Pediatric Use
The recommended dose of Desloratadine Oral Solution in the pediatric population is based on cross-study comparison of the plasma concentration of Desloratadine in adults and pediatric subjects. The safety of Desloratadine Oral Solution has been established in 246 pediatric subjects aged 6 months to 11 years in three placebo-controlled clinical studies. Since the course of seasonal and perennial allergic rhinitis and chronic idiopathic urticaria and the effects of Desloratadine are sufficiently similar in the pediatric and adult populations, it allows extrapolation from the adult efficacy data to pediatric patients. The effectiveness of Desloratadine Oral Solution in these age groups is supported by evidence from adequate and well-controlled studies of Desloratadine Tablets in adults. The safety and effectiveness of Desloratadine Tablets or Desloratadine Oral Solution have not been demonstrated in pediatric patients less than 6 months of age. [See Clinical Pharmacology (12.3).]
8.5 Geriatric Use
Clinical studies of desloratadine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. [See Clinical Pharmacology (12.3).]
4 Contraindications (4 CONTRAINDICATIONS)
Desloratadine Tablets are contraindicated in patients who are hypersensitive to this medication or to any of its ingredients or to loratadine [see Warnings and Precautions (5.1) and Adverse Reactions (6.2) ].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the label:
-
•Hypersensitivity reactions. [See Warnings and Precautions (5.1) .]
Patient Information (PATIENT INFORMATION)
Desloratadine Tablets
Read the Patient Information that comes with Desloratadine Tablets before you start taking it and each time you get a refill. There may be new information. This leaflet is a summary of the information for patients. Your doctor or pharmacist can give you additional information. This leaflet does not take the place of talking to your doctor about your medical condition or treatment.
What are Desloratadine Tablets?
Desloratadine Tablets are a prescription medicine that contains the medicine desloratadine (an antihistamine).
Desloratadine Tablets are used to help control the symptoms of:
-
•seasonal allergic rhinitis (sneezing, stuffy nose, runny nose and itching of the nose) in people 12 years of age and older.
-
•perennial allergic rhinitis (sneezing, stuffy nose, runny nose and itching of the nose) in people 12 years of age and older.
-
•chronic idiopathic urticaria (long-term itching) and to reduce the number and size of hives in people 12 years of age and older.
Desloratadine Tablets, 5 mg is not for children younger than 12 years of age.
Who should not take Desloratadine Tablets?
Do not take Desloratadine Tablets if you:
-
•are allergic to desloratadine or any of the ingredients in Desloratadine Tablets. See the end of this leaflet for a complete list of ingredients.
-
•are allergic to loratadine (Alavert, Claritin).
Talk to your doctor before taking this medicine if you have any questions about whether or not to take this medicine.
What should I tell my doctor before taking Desloratadine Tablets?
Before you take Desloratadine Tablets, tell your doctor if you:
-
•have liver or kidney problems.
-
•have any other medical conditions.
-
•are pregnant or plan to become pregnant. It is not known if Desloratadine will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
-
•are breast-feeding or plan to breast-feed. Desloratadine can pass into your breast milk. Talk to your doctor about the best way to feed your baby if you take Desloratadine.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. Desloratadine may affect the way other medicines work, and other medicines may affect how Desloratadine works. Especially tell your doctor if you take:
-
•ketoconazole (Nizoral)
-
•erythromycin (Ery-tab, Eryc, PCE)
-
•azithromycin (Zithromax, Zmax)
-
•antihistamines
-
•fluoxetine (Prozac)
-
•cimetidine (Tagamet)
Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine.
How should I take Desloratadine Tablets?
-
•Take Desloratadine Tablets exactly as your doctor tells you to take it.
-
•Do not change your dose of Desloratadine Tablets or take more often than prescribed.
-
•Desloratadine Tablets can be taken with or without food.
-
•If you take too many Desloratadine Tablets, call your doctor or get medical attention right away.
What are the possible side effects of Desloratadine Tablets?
Desloratadine Tablets may cause serious side effects, including:
-
•Allergic reactions. Stop taking Desloratadine Tablets and call your doctor right away or get emergency help if you have any of these symptoms:
-
orash
-
oitching
-
ohives
-
oswelling of your lips, tongue, face, and throat
-
oshortness of breath or trouble breathing
The most common side effects of Desloratadine Tablets in adults and children 12 years of age and older with allergic rhinitis include:
-
•sore throat
-
•dry mouth
-
•muscle pain
-
•tiredness
-
•sleepiness
-
•menstrual pain
Increased sleepiness or tiredness can happen if you take more Desloratadine Tablets than your doctor prescribed to you.
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of Desloratadine Tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to Virtus Pharmaceuticals, LLC at 1-888-848-3593 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How should I store Desloratadine Tablets?
-
•Store Desloratadine Tablets between 59°F to 86°F (15°C to 30°C).
-
•Desloratadine Tablets are sensitive to heat. Do not store above 86°F (30°C).
-
•Protect Desloratadine Tablets from moisture.
Keep Desloratadine Tablets, and all medicines out of the reach of children.
General information about Desloratadine Tablets
Medicines are sometimes prescribed for purposes other than those listed in a patient information leaflet. Do not use Desloratadine Tablets for a condition for which it was not prescribed. Do not give Desloratadine Tablets to other people, even if they have the same condition you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Desloratadine Tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Desloratadine Tablets that is written for health professionals.
What are the ingredients in Desloratadine Tablets?
Active ingredient: Desloratadine
Inactive ingredients in Desloratadine Tablets:
microcrystalline cellulose NF, pregelatinized starch NF, croscarmellose sodium NF, talc USP, zinc stearate, USP and FD&C Blue #2 HT 11-14%.
Manufactured for:
Virtus Pharmaceuticals, LLC
Langhorne, PA 19047
1-888-848-3593
MADE IN USA
Rev. 10/2020
R-2010
8.6 Renal Impairment
Dosage adjustment for patients with renal impairment is recommended [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
8.7 Hepatic Impairment
Dosage adjustment for patients with hepatic impairment is recommended [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Desloratadine Tablets are a histamine-1 (H1) receptor antagonist indicated for:
-
•Seasonal Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. (1.1)
-
•Perennial Allergic Rhinitis: relief of nasal and non-nasal symptoms in patients 12 years of age and older. (1.2)
-
•Chronic Idiopathic Urticaria: symptomatic relief of pruritus, reduction in the number of hives, and size of hives in patients 12 years of age and older. (1.3)
12.1 Mechanism of Action
Desloratadine is a long-acting tricyclic histamine antagonist with selective H1-receptor histamine antagonist activity. Receptor binding data indicates that at a concentration of 2-3 ng/mL (7 nanomolar), desloratadine shows significant interaction with the human histamine H1-receptor. Desloratadine inhibited histamine release from human mast cells in vitro. Results of a radiolabeled tissue distribution study in rats and a radioligand H1-receptor binding study in guinea pigs showed that desloratadine did not readily cross the blood brain barrier. The clinical significance of this finding is unknown.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Hypersensitivity reactions including rash, pruritus, urticaria, edema, dyspnea, and anaphylaxis have been reported. In such cases, stop Desloratadine Tablets at once and consider alternative treatments. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Desloratadine Tablets may be taken without regard to meals.
9 Drug Abuse and Dependence (9 DRUG ABUSE AND DEPENDENCE)
There is no information to indicate that abuse or dependency occurs with Desloratadine Tablets.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Desloratadine Tablets are light blue round tablets debossed with "5" containing 5 mg desloratadine.
17.1 Information for Patients
-
•Patients should be instructed to use Desloratadine Tablets as directed.
-
•As there are no food effects on bioavailability, patients can be instructed that Desloratadine Tablets may be taken without regard to meals.
-
•Patients should be advised not to increase the dose or dosing frequency as studies have not demonstrated increased effectiveness at higher doses and somnolence may occur.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
Because adverse events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following spontaneous adverse events have been reported during the marketing of desloratadine:
Cardiac disorders: tachycardia, palpitations
Respiratory, thoracic and mediastinal disorders: dyspnea
Skin and subcutaneous tissue disorders: rash, pruritus
Nervous system disorders: psychomotor hyperactivity, movement disorders (including dystonia, tics, and extrapyramidal symptoms), seizures (reported in patients with and without a known seizure disorder)
Immune system disorders: hypersensitivity reactions (such as urticaria, edema and anaphylaxis)
Investigations: elevated liver enzymes including bilirubin
Hepatobiliary disorders: hepatitis
Metabolism and nutrition disorders: increased appetite
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
1.1 Seasonal Allergic Rhinitis
Desloratadine Tablets are indicated for the relief of the nasal and non-nasal symptoms of seasonal allergic rhinitis in patients 12 years of age and older.
5.1 Hypersensitivity Reactions
Hypersensitivity reactions including rash, pruritus, urticaria, edema, dyspnea, and anaphylaxis have been reported after administration of desloratadine. If such a reaction occurs, therapy with Desloratadine Tablets should be stopped and alternative treatment should be considered. [See Adverse Reactions (6.2) .]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
1.2 Perennial Allergic Rhinitis
Desloratadine Tablets are indicated for the relief of the nasal and non-nasal symptoms of perennial allergic rhinitis in patients 12 years of age and older.
14.1 Seasonal Allergic Rhinitis
The clinical efficacy and safety of Desloratadine Tablets were evaluated in over 2300 patients 12 to 75 years of age with seasonal allergic rhinitis. A total of 1838 patients received 2.5 to 20 mg/day of Desloratadine in 4 double-blind, randomized, placebo-controlled clinical trials of 2 to 4 weeks' duration conducted in the United States. The results of these studies demonstrated the efficacy and safety of Desloratadine 5 mg in the treatment of adult and adolescent patients with seasonal allergic rhinitis. In a dose-ranging trial, Desloratadine 2.5 to 20 mg/day was studied. Doses of 5, 7.5, 10, and 20 mg/day were superior to placebo; and no additional benefit was seen at doses above 5.0 mg. In the same study, an increase in the incidence of somnolence was observed at doses of 10 mg/day and 20 mg/day (5.2% and 7.6%, respectively), compared to placebo (2.3%).
In two 4-week studies of 924 patients (aged 15 to 75 years) with seasonal allergic rhinitis and concomitant asthma, Desloratadine Tablets 5 mg once daily improved rhinitis symptoms, with no decrease in pulmonary function. This supports the safety of administering Desloratadine Tablets to adult patients with seasonal allergic rhinitis with mild to moderate asthma.
Desloratadine Tablets 5 mg once daily significantly reduced the Total Symptom Score (the sum of individual scores of nasal and non-nasal symptoms) in patients with seasonal allergic rhinitis. See Table 3.
|
Treatment Group
(n) |
Mean Baseline
At baseline, a total nasal symptom score (sum of 4 individual symptoms) of at least 6 and a total non-nasal symptom score (sum of 4 individual symptoms) of at least 5 (each symptom scored 0 to 3 where 0=no symptom and 3=severe symptoms) was required for trial eligibility. TSS ranges from 0=no symptoms to 24=maximal symptoms.
(SEM) |
Change from Baseline
Mean reduction in TSS averaged over the 2-week treatment period.
(SEM) |
Placebo Comparison
(P-value) |
|---|---|---|---|
| SEM = Standard Error of the Mean | |||
|
Desloratadine
|
14.2 (0.3) |
-4.3 (0.3) |
P<0.01 |
|
Placebo (173) |
13.7 (0.3) |
-2.5 (0.3) |
There were no significant differences in the effectiveness of Desloratadine Tablets 5 mg across subgroups of patients defined by gender, age, or race.
1.3 Chronic Idiopathic Urticaria
Desloratadine Tablets are indicated for the symptomatic relief of pruritus, reduction in the number of hives, and size of hives, in patients with chronic idiopathic urticaria 12 years of age and older.
14.2 Perennial Allergic Rhinitis
The clinical efficacy and safety of Desloratadine Tablets 5 mg were evaluated in over 1300 patients 12 to 80 years of age with perennial allergic rhinitis. A total of 685 patients received 5 mg/day of Desloratadine in two double-blind, randomized, placebo-controlled clinical trials of 4 weeks' duration conducted in the United States and internationally. In one of these studies Desloratadine Tablets 5 mg once daily was shown to significantly reduce the Total Symptom Score in patients with perennial allergic rhinitis (Table 4).
|
Treatment Group
(n) |
Mean Baseline
At baseline, average of total symptom score (sum of 5 individual nasal symptoms and 3 non-nasal symptoms, each symptom scored 0 to 3 where 0=no symptom and 3=severe symptoms) of at least 10 was required for trial eligibility. TSS ranges from 0=no symptoms to 24=maximal symptoms.
(SEM) |
Change from Baseline
Mean reduction in TSS averaged over the 4 week treatment period.
(SEM) |
Placebo Comparison
(P-value) |
|---|---|---|---|
| SEM = Standard Error of the Mean | |||
|
Desloratadine
|
12.37 (0.18) |
-4.06 (0.21) |
P=0.01 |
|
Placebo (337) |
12.30 (0.18) |
-3.27 (0.21) |
14.3 Chronic Idiopathic Urticaria
The efficacy and safety of Desloratadine Tablets 5 mg once daily was studied in 416 chronic idiopathic urticaria patients 12 to 84 years of age, of whom 211 received Desloratadine. In two double-blind, placebo-controlled, randomized clinical trials of six weeks duration, at the pre-specified one-week primary time point evaluation, Desloratadine Tablets significantly reduced the severity of pruritus when compared to placebo (Table 5). Secondary endpoints were also evaluated, and during the first week of therapy Desloratadine Tablets 5 mg reduced the secondary endpoints, "Number of Hives" and the "Size of the Largest Hive," when compared to placebo.
|
Treatment Group
(n) |
Mean Baseline
(SEM) |
Change from Baseline
Mean reduction in pruritus averaged over the first week of treatment.
(SEM) |
Placebo Comparison
(P-value) |
|---|---|---|---|
| Pruritus scored 0 to 3 where 0=no symptom to 3=maximal symptom | |||
| SEM = Standard Error of the Mean | |||
|
Desloratadine
|
2.19 (0.04) |
-1.05 (0.07) |
P<0.01 |
|
Placebo (110) |
2.21 (0.04) |
-0.52 (0.07) |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Desloratadine Tablets: Debossed "5", light blue, round tablets that are packaged in high-density polyethylene plastic bottles of 100 (NDC 69543-107-10) and 500 (NDC 69543-107-50).
7.1 Inhibitors of Cytochrome P450 3a4 (7.1 Inhibitors of Cytochrome P450 3A4)
In controlled clinical studies co-administration of desloratadine with ketoconazole, erythromycin, or azithromycin resulted in increased plasma concentrations of desloratadine and 3 hydroxydesloratadine, but there were no clinically relevant changes in the safety profile of desloratadine. [See Clinical Pharmacology (12.3) .]
2.5 Adults With Hepatic Or Renal Impairment (2.5 Adults with Hepatic or Renal Impairment)
In adult patients with liver or renal impairment, a starting dose of one 5 mg tablet every other day is recommended based on pharmacokinetic data. Dosing recommendation for children with liver or renal impairment cannot be made due to lack of data [see Clinical Pharmacology (12.3) ].
8.3 Females and Males of Reproductive Potential
Infertility
There are no data available on human infertility associated with desloratadine.
There were no clinically relevant effects of desloratadine on female fertility in rats. A male specific decrease in fertility occurred at an oral desloratadine dose of 12 mg/kg or greater in rats (approximately 65 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). Male fertility was unaffected at a desloratadine dose of 3 mg/kg (approximately 10 times the summed AUC-based exposure of desloratadine and its metabolite at the RHD). [See Nonclinical Toxicology (13.1) .]
2.1 Adults and Adolescents 12 Years of Age and Over
The recommended dose of Desloratadine Tablets is one 5-mg tablet once daily.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:08.981654 · Updated: 2026-03-14T22:29:06.951873