Drug Facts
0f820ef2-88a1-4099-9b71-547bf0788269
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Witch Hazel 50%
Purpose
Astringent
Medication Information
Warnings and Precautions
Purpose
Astringent
Description
Witch Hazel 50%
Use
Do Not
Section 42229-5
Section 50567-7
Section 50568-5
Section 50569-3
Section 53413-1
Section 53414-9
Warnings
Avoid contact with eyes.
Directions
- When practical, cleanse the affected area with mild soap and warm water and rinse thoroughly.
- Gently dry by patting or blotting with toilet tissue or soft cloth before each application of this product. Gently apply to the affected area by patting and then discard. Apply to the affected area up to 6 times daily or after each bowel movement.
- Children under 12 years of age: consult a doctor.
Active Ingredient
Witch Hazel 50%
Other Information
Package Information
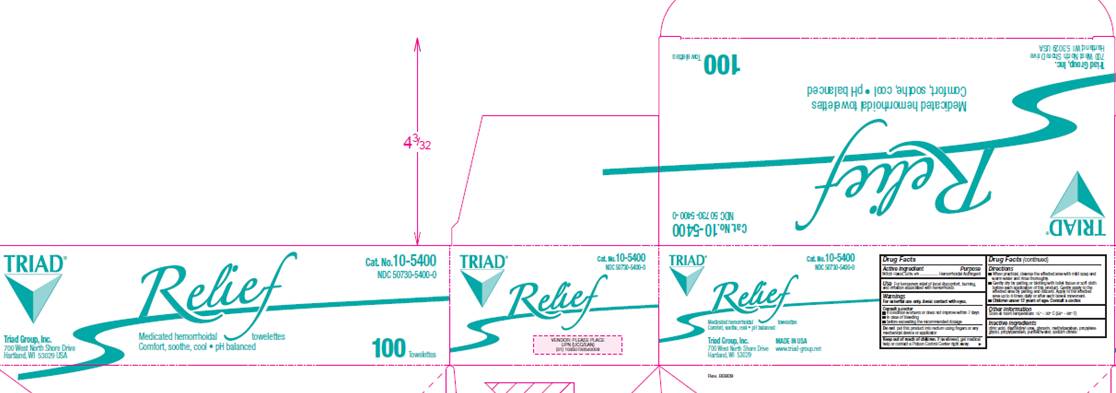
NDC 50730-5400-0
Relief
Medicated hemorrhoidal towelettes
Comfort, soothe, cool - pH balanced
Triad Group, Inc.
700 West North Shore Drive
Hartland, WI 53029 USA
Made in USA
www.triad-group.net
100 Towelettes
Inactive Ingredients
Stop Use and Ask A Doctor
- if condition worsens or does not improve within 7 days
- in case of bleeding
- before exceeding the recommended dosage.
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use
Do Not (Do not)
Warnings and Precautions (34071-1)
Section 42229-5 (42229-5)
Section 50567-7 (50567-7)
Section 50568-5 (50568-5)
Section 50569-3 (50569-3)
Section 53413-1 (53413-1)
Section 53414-9 (53414-9)
Purpose
Astringent
Warnings
Avoid contact with eyes.
Directions
- When practical, cleanse the affected area with mild soap and warm water and rinse thoroughly.
- Gently dry by patting or blotting with toilet tissue or soft cloth before each application of this product. Gently apply to the affected area by patting and then discard. Apply to the affected area up to 6 times daily or after each bowel movement.
- Children under 12 years of age: consult a doctor.
Active Ingredient (Active ingredient)
Witch Hazel 50%
Other Information (Other information)
Package Information
NDC 50730-5400-0
Relief
Medicated hemorrhoidal towelettes
Comfort, soothe, cool - pH balanced
Triad Group, Inc.
700 West North Shore Drive
Hartland, WI 53029 USA
Made in USA
www.triad-group.net
100 Towelettes
Inactive Ingredients
Stop Use and Ask A Doctor (Stop use and ask a doctor)
- if condition worsens or does not improve within 7 days
- in case of bleeding
- before exceeding the recommended dosage.
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:54:58.343083 · Updated: 2026-03-14T22:52:13.708968