tork_essity d17.002/d17ab
0f72f0cb-9e8e-43bb-862e-0024771ece1b
34390-5
HUMAN OTC DRUG LABEL
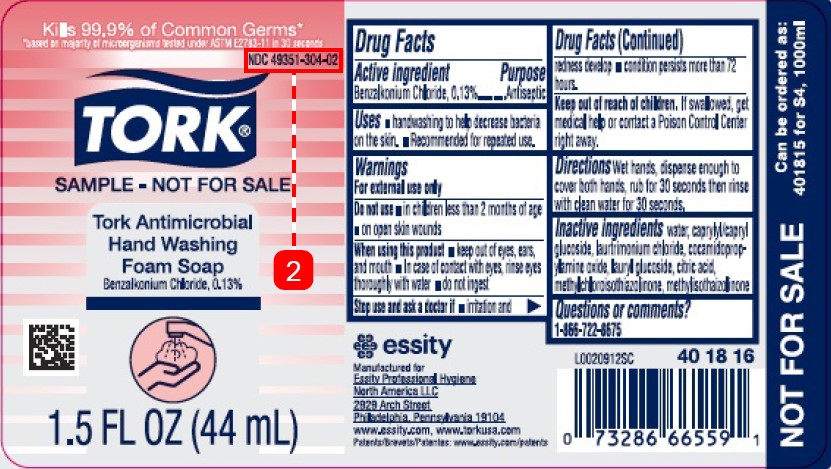
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antiseptic
Description
Benzalkonium Chloride 0.13%
Medication Information
Warnings
For external use only
Uses
- handwashing to help decrease bacteria on the skin
- recommended for repeated use
Purpose
Antiseptic
Directions
- wet hands, dispense enough to cover both hands, rub for 30 seconds then rinse with clean water for 30 seconds
Description
Benzalkonium Chloride 0.13%
Disclaimer
NOT FOR SALE
Can be ordered as:
401815 for S4, 1000ml
Do Not Use
- in children less than 2 months of age
- on open skin wounds
Adverse Reaction
essity
Manufactured for
Essity Professional Hygiene North America LLC
2929 Arch Street
Philadelphia, Pennsylvania 19104
www.essity.com, www.torkusa.com
Patents: www.essity.com/patents
Active Ingredient
Benzalkonium Chloride 0.13%
Inactive Ingredients
water, caprylyl/capryl glucoside, laurtrimonium chloride, cocamidopropylamine oxide, lauryl glucoside, citric acid, methylcholorisothiazonlinone, methylisothiazolinone
Questions Or Comments?
1-866-722-8675
Principal Panel Display
Kills 99.9% of Common Germs*
*based on majority of microorganisms tested under ASTM E2783-11 in 30 seconds
NDC 49351-304-02
TORK ®
SAMPLE - NOT FOR SALE
Tork Antimicrobial Hand Washing Foam Soap
Benzalkonium Chloride 0.13%
1.5 FL OZ (44 mL)
When Using This Product
- keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
- do not ingest
Stop Use and Ask A Doctor If
- irritation and redness develop
- condition persists for more then 72 hours
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Warnings
For external use only
Uses
- handwashing to help decrease bacteria on the skin
- recommended for repeated use
Purpose
Antiseptic
Directions
- wet hands, dispense enough to cover both hands, rub for 30 seconds then rinse with clean water for 30 seconds
Disclaimer
NOT FOR SALE
Can be ordered as:
401815 for S4, 1000ml
Do Not Use (Do not use)
- in children less than 2 months of age
- on open skin wounds
Adverse Reaction (Adverse reaction)
essity
Manufactured for
Essity Professional Hygiene North America LLC
2929 Arch Street
Philadelphia, Pennsylvania 19104
www.essity.com, www.torkusa.com
Patents: www.essity.com/patents
Active Ingredient (Active ingredient)
Benzalkonium Chloride 0.13%
Inactive Ingredients (Inactive ingredients )
water, caprylyl/capryl glucoside, laurtrimonium chloride, cocamidopropylamine oxide, lauryl glucoside, citric acid, methylcholorisothiazonlinone, methylisothiazolinone
Questions Or Comments? (Questions or comments?)
1-866-722-8675
Principal Panel Display
Kills 99.9% of Common Germs*
*based on majority of microorganisms tested under ASTM E2783-11 in 30 seconds
NDC 49351-304-02
TORK ®
SAMPLE - NOT FOR SALE
Tork Antimicrobial Hand Washing Foam Soap
Benzalkonium Chloride 0.13%
1.5 FL OZ (44 mL)
When Using This Product (When using this product)
- keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
- do not ingest
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- irritation and redness develop
- condition persists for more then 72 hours
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:31.471926 · Updated: 2026-03-14T23:12:46.294226