diphenhydramine hcl tablets 25 mg pink
0f140474-06ef-4cff-a290-3c1dae1ac65f
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antihistamine
Description
Drug Facts
Medication Information
Active Ingredient
Diphenhydramine HCl 25 mg
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Purpose
Antihistamine
Directions
- take every 4 to 6 hours
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 tablets |
| children 6 to under 12 years | 1 tablet |
| children under 6 years | do not use this product in children under 6 years of age |
Description
Drug Facts
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
Section 50567-7
When using this product
- marked drowsiness may occur
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- avoid alcoholic drinks
- excitability may occur, especially in children
Section 50568-5
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
Section 50569-3
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Section 50570-1
Do not use with any other product containing diphenhydramine, even one used on skin
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Principal Display Panel 25 Mg Label
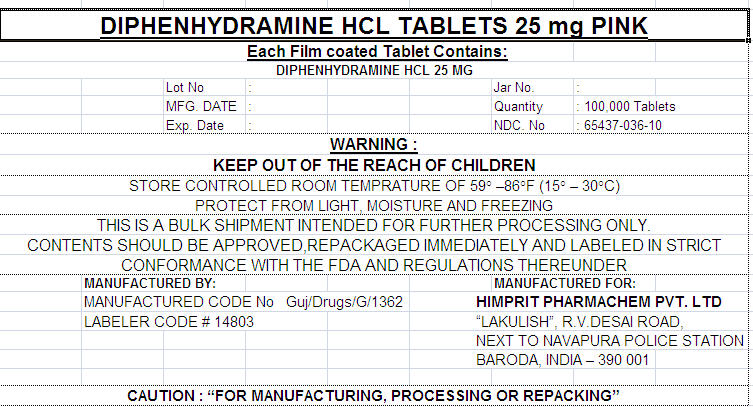
DIPHENHYDRAMINE HCL TABLETS 25 mg PINK
Each Film coated Tablet Contains:
DIPHENHYDRAMINE HCL 25 MG
| Lot No | : | Jar No. | : | ||
| MFG. DATE | : | Quantity | : 100,000 Tablets | ||
| Exp. Date | : | NDC. No | : 65437-036-10 |
WARNING :
KEEP OUT OF THE REACH OF CHILDREN
STORE CONTROLLED ROOM TEMPRATURE OF 59° –86°F (15° – 30°C)
PROTECT FROM LIGHT, MOISTURE AND FREEZING
THIS IS A BULK SHIPMENT INTENDED FOR FURTHER PROCESSING ONLY.
CONTENTS SHOULD BE APPROVED,REPACKAGED IMMEDIATELY AND LABELED IN STRICT
CONFORMANCE WITH THE FDA AND REGULATIONS THEREUNDER
MANUFACTURED BY:
MANUFACTURED CODE No Guj/Drugs/G/1362
LABELER CODE # 14803
MANUFACTURED FOR:
HIMPRIT PHARMACHEM PVT. LTD
"LAKULISH", R.V.DESAI ROAD,
NEXT TO NAVAPURA POLICE STATION
BARODA, INDIA – 390 001
CAUTION : "FOR MANUFACTURING, PROCESSING OR REPACKING"
Structured Label Content
Active Ingredient (Active ingredient (in each tablet))
Diphenhydramine HCl 25 mg
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Purpose
Antihistamine
Directions
- take every 4 to 6 hours
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 tablets |
| children 6 to under 12 years | 1 tablet |
| children under 6 years | do not use this product in children under 6 years of age |
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50567-7 (50567-7)
When using this product
- marked drowsiness may occur
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- avoid alcoholic drinks
- excitability may occur, especially in children
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Section 50570-1 (50570-1)
Do not use with any other product containing diphenhydramine, even one used on skin
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Principal Display Panel 25 Mg Label (PRINCIPAL DISPLAY PANEL - 25 mg Label)
DIPHENHYDRAMINE HCL TABLETS 25 mg PINK
Each Film coated Tablet Contains:
DIPHENHYDRAMINE HCL 25 MG
| Lot No | : | Jar No. | : | ||
| MFG. DATE | : | Quantity | : 100,000 Tablets | ||
| Exp. Date | : | NDC. No | : 65437-036-10 |
WARNING :
KEEP OUT OF THE REACH OF CHILDREN
STORE CONTROLLED ROOM TEMPRATURE OF 59° –86°F (15° – 30°C)
PROTECT FROM LIGHT, MOISTURE AND FREEZING
THIS IS A BULK SHIPMENT INTENDED FOR FURTHER PROCESSING ONLY.
CONTENTS SHOULD BE APPROVED,REPACKAGED IMMEDIATELY AND LABELED IN STRICT
CONFORMANCE WITH THE FDA AND REGULATIONS THEREUNDER
MANUFACTURED BY:
MANUFACTURED CODE No Guj/Drugs/G/1362
LABELER CODE # 14803
MANUFACTURED FOR:
HIMPRIT PHARMACHEM PVT. LTD
"LAKULISH", R.V.DESAI ROAD,
NEXT TO NAVAPURA POLICE STATION
BARODA, INDIA – 390 001
CAUTION : "FOR MANUFACTURING, PROCESSING OR REPACKING"
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:00.913829 · Updated: 2026-03-14T22:52:19.062647