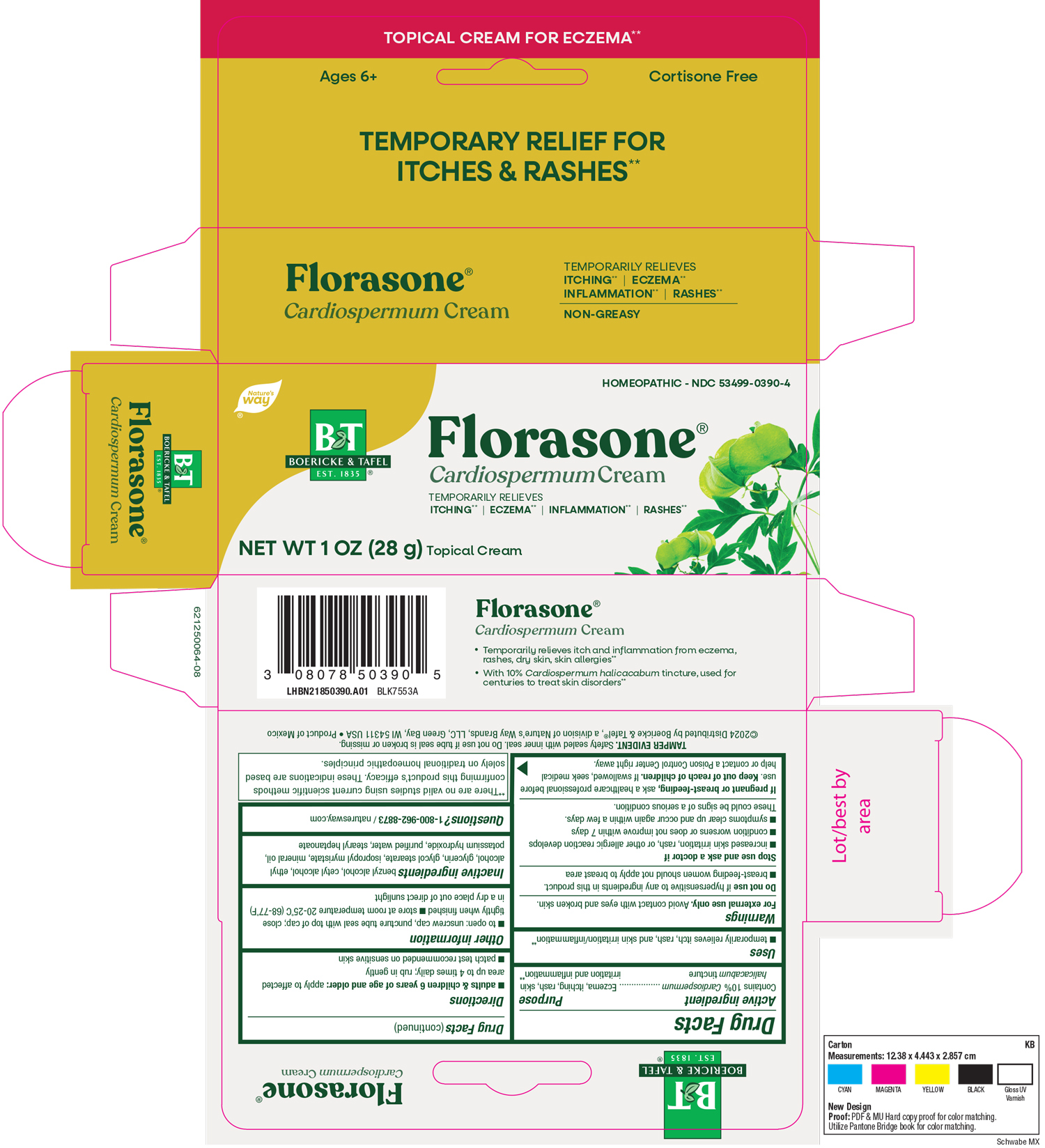
Florasone
0ea5879c-5955-427d-8604-a0677a4bb5cd
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Contains 10% Cardiospermum halicacabum tincture
Indications and Usage
Temporarily relieves itch, rash, and skin irritation/inflammation.
Purpose
Temporarily relieves itch, rash, and skin irritation/inflammation.
Medication Information
Indications and Usage
Temporarily relieves itch, rash, and skin irritation/inflammation.
Purpose
Temporarily relieves itch, rash, and skin irritation/inflammation.
Description
Contains 10% Cardiospermum halicacabum tincture
Section 51945-4
Overdose
If swallowed, seek medical help or contact a Poison Control Center right away.
Warnings
For external use only.
Avoid contact with eyes and broken skin.
Do Not Use
Do not use if hypersensitive to any ingredients in this product.
Breast-feeding woman should not apply to breast area.
Stop Use
Stop use and ask a doctor if increased skin irritation, rash or other allergic reaction develops, condition worsens or does not improve within 7 days, symptoms clear up and occur again within a few days.
These could be signs of a serious condition.
Inactive Ingredient
Benzyl Alcohol
Cetyl Alcohol
Ethyl Alcohol
Glycerin
Glycol Stearate
Isopropyl Myristate
Mineral Oil
Potassium Hydroxide
Purified Water
Stearyl Heptanoate
active Ingredient
Contains 10% Cardiospermum halicacabum tincture
Dosage & Administration
Directions
Adults & children 6 years of age and older: apply to affected area up to 4 times daily; r ub in gently.
Patch test recommended on sensitive skin.
Pregnancy Or Breast Feeding
If pregnant or breast-feeding, ask a healthcare professional before use.
Keep Out of Reach of Children
Keep out of reach of children.
Structured Label Content
Section 51945-4 (51945-4)
Purpose
Temporarily relieves itch, rash, and skin irritation/inflammation.
Overdose
If swallowed, seek medical help or contact a Poison Control Center right away.
Warnings
For external use only.
Avoid contact with eyes and broken skin.
Do Not Use
Do not use if hypersensitive to any ingredients in this product.
Breast-feeding woman should not apply to breast area.
Stop Use
Stop use and ask a doctor if increased skin irritation, rash or other allergic reaction develops, condition worsens or does not improve within 7 days, symptoms clear up and occur again within a few days.
These could be signs of a serious condition.
Inactive Ingredient
Benzyl Alcohol
Cetyl Alcohol
Ethyl Alcohol
Glycerin
Glycol Stearate
Isopropyl Myristate
Mineral Oil
Potassium Hydroxide
Purified Water
Stearyl Heptanoate
active Ingredient (Active Ingredient)
Contains 10% Cardiospermum halicacabum tincture
Indications and Usage
Temporarily relieves itch, rash, and skin irritation/inflammation.
Dosage & Administration
Directions
Adults & children 6 years of age and older: apply to affected area up to 4 times daily; r ub in gently.
Patch test recommended on sensitive skin.
Pregnancy Or Breast Feeding (Pregnancy or Breast-feeding)
If pregnant or breast-feeding, ask a healthcare professional before use.
Keep Out of Reach of Children (Keep out of reach of children)
Keep out of reach of children.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:07.579353 · Updated: 2026-03-14T23:17:30.983574