Livergen
0e72f898-9685-47c9-b617-00145254bf8c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
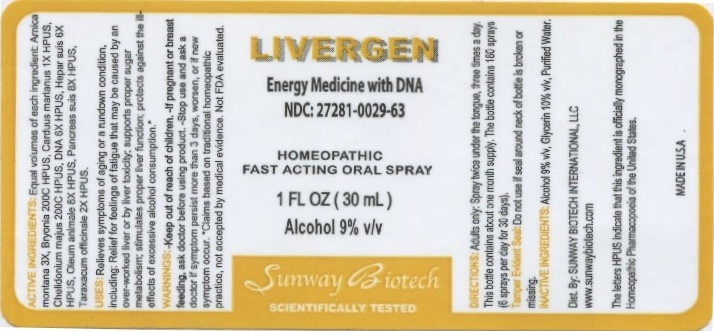
ACTIVE INGREDIENTS: Equal volumes of each ingredient: Arnica montana 3X, Bryonia 200C HPUS, Carduus marianus 1X HPUS, Chelidonium majus 200C HPUS, DNA 6X HPUS, Hepar suis 6X HPUS, Oleum animale 8X HPUS, Pancreas suis 8X HPUS, Taraxacum officinale 2X HPUS. The letters HPUS indicate that this ingredient is officially monographed in the Homeopathic Pharmacopeia of the United States.
Medication Information
Warnings and Precautions
WARNINGS: If pregnant or breast feeding, ask a doctor before using product. Stop use and ask a doctor if symptoms persist more than 3 days, worsen, or if new symptoms occur.
Indications and Usage
USES: Relieves symptoms of aging or a rundown condition, including: Relief for feelings of fatigue that may be caused by an over-worked liver or by liver toxixity; supports proper sugar metabolism; stimulates proper liver function; protects against the ill-effects of excessive alcohol consumption.*
*Claims based on traditional homeopathic practice, not accepted by medical evidence. Not FDA evaluated.
Dosage and Administration
DIRECTIONS: Adults only: Spray twice under the tongue, three times a day. This bottle contains about a 1 month supply. The bottle contains 180 sprays (6 sprays per day for 30 days).
Description
ACTIVE INGREDIENTS: Equal volumes of each ingredient: Arnica montana 3X, Bryonia 200C HPUS, Carduus marianus 1X HPUS, Chelidonium majus 200C HPUS, DNA 6X HPUS, Hepar suis 6X HPUS, Oleum animale 8X HPUS, Pancreas suis 8X HPUS, Taraxacum officinale 2X HPUS. The letters HPUS indicate that this ingredient is officially monographed in the Homeopathic Pharmacopeia of the United States.
Section 50565-1
- Keep out of reach of children.
Section 51727-6
INACTIVE INGREDIENTS: Alcohol 8% v/v, Glycerin 10% v/v, Purified Water.
Section 51945-4
LIVERGEN
Energy medicine with DNA
NDC: 27281-029-63
HOMEOPATHIC FAST ACTING ORAL SPRAY
1 FL OZ (30 mL)
Alcohol 9% v/v
Sunway Biotech
SCIENTIFICALLY TESTED
Section 53413-1
Dist. By: SUNWAY BIOTECH INTERNATIONAL, LLC
www.sunwaybiotech.com
Section 55105-1
Energy medicine with DNA
Section 55106-9
ACTIVE INGREDIENTS: Equal volumes of each ingredient: Arnica montana 3X, Bryonia 200C HPUS, Carduus marianus 1X HPUS, Chelidonium majus 200C HPUS, DNA 6X HPUS, Hepar suis 6X HPUS, Oleum animale 8X HPUS, Pancreas suis 8X HPUS, Taraxacum officinale 2X HPUS.
The letters HPUS indicate that this ingredient is officially monographed in the Homeopathic Pharmacopeia of the United States.
Section 60561-8
Tamper Evident Seal: Do not use if seal around neck of bottle is broken or missing.
Structured Label Content
Indications and Usage (34067-9)
USES: Relieves symptoms of aging or a rundown condition, including: Relief for feelings of fatigue that may be caused by an over-worked liver or by liver toxixity; supports proper sugar metabolism; stimulates proper liver function; protects against the ill-effects of excessive alcohol consumption.*
*Claims based on traditional homeopathic practice, not accepted by medical evidence. Not FDA evaluated.
Dosage and Administration (34068-7)
DIRECTIONS: Adults only: Spray twice under the tongue, three times a day. This bottle contains about a 1 month supply. The bottle contains 180 sprays (6 sprays per day for 30 days).
Warnings and Precautions (34071-1)
WARNINGS: If pregnant or breast feeding, ask a doctor before using product. Stop use and ask a doctor if symptoms persist more than 3 days, worsen, or if new symptoms occur.
Section 50565-1 (50565-1)
- Keep out of reach of children.
Section 51727-6 (51727-6)
INACTIVE INGREDIENTS: Alcohol 8% v/v, Glycerin 10% v/v, Purified Water.
Section 51945-4 (51945-4)
LIVERGEN
Energy medicine with DNA
NDC: 27281-029-63
HOMEOPATHIC FAST ACTING ORAL SPRAY
1 FL OZ (30 mL)
Alcohol 9% v/v
Sunway Biotech
SCIENTIFICALLY TESTED
Section 53413-1 (53413-1)
Dist. By: SUNWAY BIOTECH INTERNATIONAL, LLC
www.sunwaybiotech.com
Section 55105-1 (55105-1)
Energy medicine with DNA
Section 55106-9 (55106-9)
ACTIVE INGREDIENTS: Equal volumes of each ingredient: Arnica montana 3X, Bryonia 200C HPUS, Carduus marianus 1X HPUS, Chelidonium majus 200C HPUS, DNA 6X HPUS, Hepar suis 6X HPUS, Oleum animale 8X HPUS, Pancreas suis 8X HPUS, Taraxacum officinale 2X HPUS.
The letters HPUS indicate that this ingredient is officially monographed in the Homeopathic Pharmacopeia of the United States.
Section 60561-8 (60561-8)
Tamper Evident Seal: Do not use if seal around neck of bottle is broken or missing.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:10:04.615830 · Updated: 2026-03-14T23:15:14.298659