Fluticasone Propionate Cream Usp, 0.05%
0e20d1cc-5c7a-4e0d-810b-b94c4d6dd1c0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rx Only FOR DERMATOLOGIC USE ONLY NOT FOR OPHTHALMIC USE
Indications and Usage
Fluticasone propionate cream, 0.05% is a medium potency corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. Fluticasone propionate cream, 0.05% may be used with caution in pediatric patients 3 months of age or older. The safety and efficacy of drug use for longer than 4 weeks in this population have not been established. The safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Dosage and Administration
Fluticasone propionate cream, 0.05% may be used in adult and pediatric patients 3 months of age or older. Safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients for more than 4 weeks of use have not been established (see PRECAUTIONS: Pediatric Use ). The safety and efficacy of Fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Contraindications
Fluticasone propionate cream, 0.05% is contraindicated in those patients with a history of hypersensitivity to any of the components in the preparation.
Adverse Reactions
In controlled clinical trials of twice daily administration, the total incidence of adverse reactions associated with the use of fluticasone propionate cream, 0.05% was approximately 4%. These adverse reactions were usually mild, self-limiting; and consisted primarily of pruritus, dryness, numbness of fingers, and burning. These events occurred in 2.9%, 1.2%, 1.0%, and 0.6% of patients, respectively. Two clinical studies compared once to twice-daily administration of fluticasone propionate cream, 0.05% for the treatment of moderate to severe eczema. The local drug-related adverse events for the 491 patients enrolled in both studies are shown in Table 1 . In the study enrolling both adult and pediatric patients, the incidence of local adverse events in the 119 pediatric patients ages 1 to 12 years was comparable to the 140 patients ages 13 to 62 years. Fifty-one pediatric patients ages 3 months to 5 years, with moderate to severe eczema, were enrolled in an open-label HPA axis safety study. Fluticasone propionate cream, 0.05% was applied twice daily for 3 to 4 weeks over and arithmetic mean body surface area of 64% (range 35% to 95%). The mean morning cortisol levels with standard deviations before treatment (pre-stimulation mean value =13.76±6.94 mcg/dL, post-stimulation mean value =30.53±7.23 mcg/dL) and at end treatment (pre-stimulation mean value =12.32±6.92 mcg/dL, poststimulation mean value =28.84±7.16 mcg/dL) showed little change. In 2 of 43 (4.7%) patients with end-treatment results, peak cortisol levels following cosyntropin stimulation testing were ≤18 mcg/dL indicating adrenal suppression. Follow-up testing after treatment discontinuation, available for 1 of the 2 subjects, demonstrated a normally responsive HPA axis. Local drug-related adverse events were (see Table 2 ): transient burning, resolving the same day it was reported; transient urticaria, resolving the same day it was reported; erythematous rash; dusky erythema, resolving within one month after cessation of fluticasone propionate cream, 0.05%; and telangiectasia resolving within 3 months after stopping fluticasone propionate cream, 0.05%. Table 1: Drug-Related Adverse Events—Skin Adverse Events Fluticasone Once Daily (n=210) Fluticasone Twice Daily (n=203) Vehicle Twice Daily (n=78) Skin infection 1 (0.5%) 0 0 Infected eczema 1 (0.5%) 2 (1.0%) 0 Viral warts 0 1 (0.5%) 0 Herpes simplex 0 1 (0.5%) 0 Impetigo 1 (0.5%) 0 0 Atopic dermatitis 1 (0.5%) 0 0 Eczema 1 (0.5%) 0 0 Exacerbation of eczema 4 (1.9%) 1 (0.5%) 1 (1.3%) Erythema 0 2 (1.0%) 0 Burning 2 (1.0%) 2 (1.0%) 2 (2.6%) Stinging 0 2 (1.0%) 1 (1.3%) Skin irritation 6 (2.9%) 2 (1.0%) 0 Pruritus 2 (1.0%) 4 (1.9%) 4 (5.1%) Exacerbation of Pruritus 4 (1.9%) 1 (0.5%) 1 (1.3%) Folliculitis 1 (0.5%) 1 (0.5%) 0 Blisters 0 1 (0.5%) 0 Dryness of skin 3 (1.4%) 1 (0.5%) 0 Table 2: Adverse Events* from Pediatric Open-label Trial (n=51) *See text for additional detail. † n=41 Adverse Events Fluticasone Twice Daily Burning 1 (2.0%) Dusky Erythema 1 (2.0%) Erythematous Rash 1 (2.0%) Facial Telangiectasia † 2 (4.9%) Non Facial Telangiectasia 1 (2.0%) Urticaria 1 (2.0%) The following local adverse reactions have been reported infrequently with topical corticosteroids and they may occur more frequently with the use of occlusive dressings and higher potency corticosteroids. These reactions are listed in an approximately decreasing order of occurrence: irritation, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, skin atrophy, striae, hypertrichosis and miliaria. Also, there are reports of the development of pustular psoriasis from chronic plaque psoriasis following reduction or discontinuation of potent topical corticosteroid products.
How Supplied
Fluticasone propionate cream USP, 0.05% is supplied in: 15 g tubes NDC 0168-0332-15 30 g tubes NDC 0168-0332-30 60 g tubes NDC 0168-0332-60 Store between 2° and 30° C (36° and 86° F). E. FOUGERA & CO. A division of Fougera PHARMACEUTICALS INC. Melville, NY 11747 46233142A R07/18 #91
Medication Information
Indications and Usage
Fluticasone propionate cream, 0.05% is a medium potency corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. Fluticasone propionate cream, 0.05% may be used with caution in pediatric patients 3 months of age or older. The safety and efficacy of drug use for longer than 4 weeks in this population have not been established. The safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Dosage and Administration
Fluticasone propionate cream, 0.05% may be used in adult and pediatric patients 3 months of age or older. Safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients for more than 4 weeks of use have not been established (see PRECAUTIONS: Pediatric Use ). The safety and efficacy of Fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Contraindications
Fluticasone propionate cream, 0.05% is contraindicated in those patients with a history of hypersensitivity to any of the components in the preparation.
Adverse Reactions
In controlled clinical trials of twice daily administration, the total incidence of adverse reactions associated with the use of fluticasone propionate cream, 0.05% was approximately 4%. These adverse reactions were usually mild, self-limiting; and consisted primarily of pruritus, dryness, numbness of fingers, and burning. These events occurred in 2.9%, 1.2%, 1.0%, and 0.6% of patients, respectively.
Two clinical studies compared once to twice-daily administration of fluticasone propionate cream, 0.05% for the treatment of moderate to severe eczema. The local drug-related adverse events for the 491 patients enrolled in both studies are shown in Table 1 . In the study enrolling both adult and pediatric patients, the incidence of local adverse events in the 119 pediatric patients ages 1 to 12 years was comparable to the 140 patients ages 13 to 62 years.
Fifty-one pediatric patients ages 3 months to 5 years, with moderate to severe eczema, were enrolled in an open-label HPA axis safety study. Fluticasone propionate cream, 0.05% was applied twice daily for 3 to 4 weeks over and arithmetic mean body surface area of 64% (range 35% to 95%).
The mean morning cortisol levels with standard deviations before treatment (pre-stimulation mean value =13.76±6.94 mcg/dL, post-stimulation mean value =30.53±7.23 mcg/dL) and at end treatment (pre-stimulation mean value =12.32±6.92 mcg/dL, poststimulation mean value =28.84±7.16 mcg/dL) showed little change. In 2 of 43 (4.7%) patients with end-treatment results, peak cortisol levels following cosyntropin stimulation testing were ≤18 mcg/dL indicating adrenal suppression. Follow-up testing after treatment discontinuation, available for 1 of the 2 subjects, demonstrated a normally responsive HPA axis. Local drug-related adverse events were (see Table 2 ): transient burning, resolving the same day it was reported; transient urticaria, resolving the same day it was reported; erythematous rash; dusky erythema, resolving within one month after cessation of fluticasone propionate cream, 0.05%; and telangiectasia resolving within 3 months after stopping fluticasone propionate cream, 0.05%.
|
Adverse Events |
Fluticasone |
Fluticasone |
Vehicle |
|
Skin infection |
1 (0.5%) |
0 |
0 |
|
Infected eczema |
1 (0.5%) |
2 (1.0%) |
0 |
|
Viral warts |
0 |
1 (0.5%) |
0 |
|
Herpes simplex |
0 |
1 (0.5%) |
0 |
|
Impetigo |
1 (0.5%) |
0 |
0 |
|
Atopic dermatitis |
1 (0.5%) |
0 |
0 |
|
Eczema |
1 (0.5%) |
0 |
0 |
|
Exacerbation of eczema |
4 (1.9%) |
1 (0.5%) |
1 (1.3%) |
|
Erythema |
0 |
2 (1.0%) |
0 |
|
Burning |
2 (1.0%) |
2 (1.0%) |
2 (2.6%) |
|
Stinging |
0 |
2 (1.0%) |
1 (1.3%) |
|
Skin irritation |
6 (2.9%) |
2 (1.0%) |
0 |
|
Pruritus |
2 (1.0%) |
4 (1.9%) |
4 (5.1%) |
|
Exacerbation of Pruritus |
4 (1.9%) |
1 (0.5%) |
1 (1.3%) |
|
Folliculitis |
1 (0.5%) |
1 (0.5%) |
0 |
|
Blisters |
0 |
1 (0.5%) |
0 |
|
Dryness of skin |
3 (1.4%) |
1 (0.5%) |
0 |
| *See text for additional detail. | |
| † n=41 | |
|
Adverse Events |
Fluticasone Twice Daily |
|
Burning |
1 (2.0%) |
|
Dusky Erythema |
1 (2.0%) |
|
Erythematous Rash |
1 (2.0%) |
|
Facial Telangiectasia† |
2 (4.9%) |
|
Non Facial Telangiectasia |
1 (2.0%) |
|
Urticaria |
1 (2.0%) |
The following local adverse reactions have been reported infrequently with topical corticosteroids and they may occur more frequently with the use of occlusive dressings and higher potency corticosteroids. These reactions are listed in an approximately decreasing order of occurrence: irritation, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, skin atrophy, striae, hypertrichosis and miliaria. Also, there are reports of the development of pustular psoriasis from chronic plaque psoriasis following reduction or discontinuation of potent topical corticosteroid products.
How Supplied
Fluticasone propionate cream USP, 0.05% is supplied in:
15 g tubes NDC 0168-0332-15
30 g tubes NDC 0168-0332-30
60 g tubes NDC 0168-0332-60
Store between 2° and 30° C (36° and 86° F).
E. FOUGERA & CO.
A division of
Fougera
PHARMACEUTICALS INC.
Melville, NY 11747
46233142A
R07/18
#91
Description
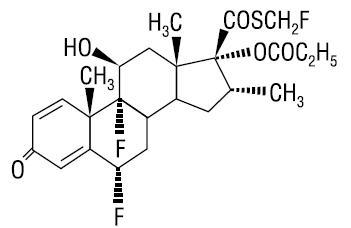
Fluticasone propionate cream USP, 0.05% contains fluticasone propionate USP [(6α,11β,16α,17α)-6,9-difluoro-11-hydroxy-16-methyl-3-oxo-17-(1-oxopropoxy)androsta-1,4-diene-17-carbothioic acid S-fluoromethyl ester], a synthetic fluorinated corticosteroid, for topical dermatologic use. The topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and antipruritic agents.
Chemically, fluticasone propionate is C25H31F3O5S. It has the following structural formula:
Fluticasone propionate USP has a molecular weight of 500.6. It is a white to off-white powder and is insoluble in water.
Each gram of fluticasone propionate cream USP, 0.05% contains fluticasone propionate USP 0.5 mg in a base of propylene glycol, mineral oil, cetostearyl alcohol, Ceteth-20, isopropyl myristate, dibasic sodium phosphate, citric acid, purified water and imidurea as preservative.
Section 34072-9
General
Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency after withdrawal from treatment. Manifestations of Cushing's syndrome, hyperglycemia and glucosuria can also be produced in some patients by systemic absorption of topical corticosteroids while on treatment.
Patients applying a potent topical steroid to a large surface area or to areas under occlusion should be evaluated periodically for evidence of HPA axis suppression. This may be done by using the ACTH stimulation, A.M. plasma cortisol, and urinary free cortisol tests.
If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Recovery of HPA axis function is generally prompt upon discontinuation of topical corticosteroids. Infrequently, signs and symptoms of glucocorticosteroid insufficiency may occur requiring supplemental systemic corticosteroids. For information on systemic supplementation, see prescribing information for those products.
Fluticasone propionate cream, 0.05% caused depression of A.M. plasma cortisol levels in 1 of 6 adult patients when used daily for 7 days in patients with psoriasis or eczema involving at least 30% of the body surface. After 2 days of treatment, this patient developed a 60% decrease from pretreatment values in the A.M. plasma cortisol level. There was some evidence of corresponding decrease in the 24-hour urinary free cortisol levels. The A.M. plasma cortisol level remained slightly depressed for 48 hours but recovered by day 6 of treatment.
Fluticasone propionate cream, 0.05%, caused HPA axis suppression in 2 of 43 pediatric patients, ages 2 and 5 years old, who were treated for 4 weeks covering at least 35% of the body surface area. Follow-up testing 12 days after treatment discontinuation, available for 1 of the 2 subjects, demonstrated a normally responsive HPA axis (see PRECAUTIONS: Pediatric Use ).
Pediatric patients may be more susceptible to systemic toxicity from equivalent doses due to their larger skin surface to body mass ratios (see PRECAUTIONS: Pediatric Use ).
Fluticasone propionate cream, 0.05%, may cause local cutaneous adverse reactions (see ADVERSE REACTIONS ).
-
Fluticasone propionate cream, 0.05% contains the excipient imidurea which releases traces of formaldehyde as a breakdown product. Formaldehyde may cause allergic sensitization or irritation upon contact with the skin.
If irritation develops, fluticasone propionate cream, 0.05% should be discontinued and appropriate therapy instituted. Allergic contact dermatitis with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation as with most topical products not containing corticosteroids. Such an observation should be corroborated with appropriate diagnostic patch testing.
If concomitant skin infections are present or develop, an appropriate antifungal or antibacterial agent should be used. If a favorable response does not occur promptly, use of fluticasone propionate cream, 0.05% should be discontinued until the infection has been adequately controlled.
Fluticasone propionate cream, 0.05% should not be used in the presence of preexisting skin atrophy and should not be used where infection is present at the treatment site. Fluticasone propionate cream, 0.05% should not be used in the treatment of rosacea and perioral dermatitis.
Section 34075-2
Laboratory Tests
The following tests may be helpful in evaluating patients for HPA axis suppression:
-
•ACTH stimulation test
-
•A.M. plasma cortisol test
-
•Urinary free cortisol test
Section 34076-0
Information for Patients
Patients using topical corticosteroids should receive the following information and instructions:
-
1.This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
-
2.This medication should not be used for any disorder other than that for which it was prescribed.
-
3.The treated skin area should not be bandaged or otherwise covered or wrapped so as to be occlusive unless directed by the physician.
-
4.Patients should report to their physicians any signs of local adverse reactions.
-
5.Parents of pediatric patients should be advised not to use this medication in the treatment of diaper dermatitis. Fluticasone propionate cream, 0.05% should not be applied in the diaper areas as diapers or plastic pants may constitute occlusive dressing (see DOSAGE AND ADMINISTRATION ).
-
6.This medication should not be used on the face, underarms, or groin areas unless directed by a physician.
-
7.As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician.
Section 34077-8
Teratogenic Effects
Pregnancy Category C. Corticosteroids have been shown to be teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Some corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. Teratology studies in the mouse demonstrated fluticasone propionate to be teratogenic (cleft palate) when administered subcutaneously in doses of 45 mcg/kg per day and 150 mcg/kg per day. This dose is approximately 14 and 45 times, respectively, the human topical dose of fluticasone propionate cream, 0.05%. There are no adequate and well-controlled studies in pregnant women. Fluticasone propionate cream, 0.05% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 34080-2
Nursing Mothers
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Because many drugs are excreted in human milk, caution should be exercised when fluticasone propionate cream, 0.05% is administered to a nursing woman.
Section 34081-0
Pediatric Use
Fluticasone propionate cream, 0.05% may be used with caution in pediatric patients as young as 3 months of age. The safety and efficacy of drug use for longer than 4 weeks in this population have not been established. The safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Fluticasone propionate cream, 0.05%, caused HPA axis suppression in 2 of 43 pediatric patients, ages 2 and 5 years old, who were treated for 4 weeks covering at least 35% of the body surface area. Follow-up testing 12 days after treatment discontinuation, available for 1 of the 2 subjects, demonstrated a normally responsive HPA axis (see ADVERSE REACTIONS ). Adverse effects including striae have been reported with use of topical corticosteroids in pediatric patients.
HPA axis suppression, Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients receiving topical corticosteroids. Manifestations of adrenal suppression in pediatric patients include low plasma cortisol levels to an absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
Section 34082-8
Geriatric Use
A limited number of patients above 65 years of age (n=126) have been treated with fluticasone propionate cream in US and non-US clinical trials. While the number of patients is too small to permit separate analysis of efficacy and safety, the adverse reactions reported in this population were similar to those reported by younger patients. Based on available data, no adjustment of dosage of fluticasone propionate cream in geriatric patients warranted.
Section 34083-6
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Two 18-month studies were performed in mice to evaluate the carcinogenic potential of fluticasone propionate when given topically (as a 0.05% ointment) and orally. No evidence of carcinogenicity was found in either study.
Fluticasone propionate was not mutagenic in the standard Ames test, E. coli fluctuation test, S. cerevisiae gene conversion test or Chinese Hamster ovarian cell assay. It was not clastogenic in mouse micronucleus or cultured human lymphocyte tests.
In a fertility and general reproductive performance study in rats, fluticasone propionate administered subcutaneously to females at up to 50 mcg/kg per day and to males at up to 100 mcg/kg per day (later reduced to 50 mcg/kg per day) had no effect upon mating performance or fertility. These doses are approximately 15 and 30 times respectively the human systemic exposure following use of the recommended human topical dose of fluticasone propionate cream, 0.05%, assuming human percutaneous absorption of approximately 3% and the use in a 70-kg person of 15 g/day.
Section 42229-5
Rx Only
FOR DERMATOLOGIC USE ONLY
NOT FOR OPHTHALMIC USE
Overdosage
Topically applied fluticasone propionate cream, 0.05% can be absorbed in sufficient amounts to produce systemic effects (see PRECAUTIONS ).
Precautions
-
Fluticasone propionate cream, 0.05% contains the excipient imidurea which releases formaldehyde as a breakdown product. Formaldehyde may cause allergic sensitization or irritation upon contact with the skin. Fluticasone propionate cream, 0.05% should not be used in individuals with hypersensitivity to formaldehyde as it may prevent healing or worsen dermatitis.
Clinical Pharmacology
Like other topical corticosteroids, fluticasone propionate has anti-inflammatory, antipruritic and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids, in general, is unclear. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor, arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
Fluticasone propionate is lipophilic and has a strong affinity for the glucocorticoid receptor. It has weak affinity for the progesterone receptor, and virtually no affinity for the mineralocorticoid, estrogen, or androgen receptors. The therapeutic potency of glucocorticoids is related to the half-life of the glucocorticoid-receptor complex. The half-life of the fluticasone propionate-glucocorticoid receptor complex is approximately 10 hours.
Studies performed with fluticasone propionate cream, 0.05% indicate that it is in the medium range of potency as compared with other topical corticosteroids.
Package Label – Principal Display Panel – 15 G Carton
NDC 0168-0332-15
Fougera®
FLUTICASONE PROPIONATE
CREAM, 0.05%
Rx only
-
FOR DERMATOLOGIC USE ONLY
NOT FOR OPHTHALMIC USE
WARNING: Keep out of
reach of children.
NET WT 15 grams
Package Label – Principal Display Panel – 15 G Container
NDC 0168-0332-15
Rx only
Fougera ®
FLUTICASONE PROPIONATE
CREAM, 0.05%
FOR DERMATOLOGIC USE ONLY
NOT FOR OPHTHALMIC USE
WARNING: Keep out of
reach of children.
NET WT 15 grams
Structured Label Content
Section 34072-9 (34072-9)
General
Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency after withdrawal from treatment. Manifestations of Cushing's syndrome, hyperglycemia and glucosuria can also be produced in some patients by systemic absorption of topical corticosteroids while on treatment.
Patients applying a potent topical steroid to a large surface area or to areas under occlusion should be evaluated periodically for evidence of HPA axis suppression. This may be done by using the ACTH stimulation, A.M. plasma cortisol, and urinary free cortisol tests.
If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Recovery of HPA axis function is generally prompt upon discontinuation of topical corticosteroids. Infrequently, signs and symptoms of glucocorticosteroid insufficiency may occur requiring supplemental systemic corticosteroids. For information on systemic supplementation, see prescribing information for those products.
Fluticasone propionate cream, 0.05% caused depression of A.M. plasma cortisol levels in 1 of 6 adult patients when used daily for 7 days in patients with psoriasis or eczema involving at least 30% of the body surface. After 2 days of treatment, this patient developed a 60% decrease from pretreatment values in the A.M. plasma cortisol level. There was some evidence of corresponding decrease in the 24-hour urinary free cortisol levels. The A.M. plasma cortisol level remained slightly depressed for 48 hours but recovered by day 6 of treatment.
Fluticasone propionate cream, 0.05%, caused HPA axis suppression in 2 of 43 pediatric patients, ages 2 and 5 years old, who were treated for 4 weeks covering at least 35% of the body surface area. Follow-up testing 12 days after treatment discontinuation, available for 1 of the 2 subjects, demonstrated a normally responsive HPA axis (see PRECAUTIONS: Pediatric Use ).
Pediatric patients may be more susceptible to systemic toxicity from equivalent doses due to their larger skin surface to body mass ratios (see PRECAUTIONS: Pediatric Use ).
Fluticasone propionate cream, 0.05%, may cause local cutaneous adverse reactions (see ADVERSE REACTIONS ).
-
Fluticasone propionate cream, 0.05% contains the excipient imidurea which releases traces of formaldehyde as a breakdown product. Formaldehyde may cause allergic sensitization or irritation upon contact with the skin.
If irritation develops, fluticasone propionate cream, 0.05% should be discontinued and appropriate therapy instituted. Allergic contact dermatitis with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation as with most topical products not containing corticosteroids. Such an observation should be corroborated with appropriate diagnostic patch testing.
If concomitant skin infections are present or develop, an appropriate antifungal or antibacterial agent should be used. If a favorable response does not occur promptly, use of fluticasone propionate cream, 0.05% should be discontinued until the infection has been adequately controlled.
Fluticasone propionate cream, 0.05% should not be used in the presence of preexisting skin atrophy and should not be used where infection is present at the treatment site. Fluticasone propionate cream, 0.05% should not be used in the treatment of rosacea and perioral dermatitis.
Section 34075-2 (34075-2)
Laboratory Tests
The following tests may be helpful in evaluating patients for HPA axis suppression:
-
•ACTH stimulation test
-
•A.M. plasma cortisol test
-
•Urinary free cortisol test
Section 34076-0 (34076-0)
Information for Patients
Patients using topical corticosteroids should receive the following information and instructions:
-
1.This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
-
2.This medication should not be used for any disorder other than that for which it was prescribed.
-
3.The treated skin area should not be bandaged or otherwise covered or wrapped so as to be occlusive unless directed by the physician.
-
4.Patients should report to their physicians any signs of local adverse reactions.
-
5.Parents of pediatric patients should be advised not to use this medication in the treatment of diaper dermatitis. Fluticasone propionate cream, 0.05% should not be applied in the diaper areas as diapers or plastic pants may constitute occlusive dressing (see DOSAGE AND ADMINISTRATION ).
-
6.This medication should not be used on the face, underarms, or groin areas unless directed by a physician.
-
7.As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician.
Section 34077-8 (34077-8)
Teratogenic Effects
Pregnancy Category C. Corticosteroids have been shown to be teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Some corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. Teratology studies in the mouse demonstrated fluticasone propionate to be teratogenic (cleft palate) when administered subcutaneously in doses of 45 mcg/kg per day and 150 mcg/kg per day. This dose is approximately 14 and 45 times, respectively, the human topical dose of fluticasone propionate cream, 0.05%. There are no adequate and well-controlled studies in pregnant women. Fluticasone propionate cream, 0.05% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 34080-2 (34080-2)
Nursing Mothers
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Because many drugs are excreted in human milk, caution should be exercised when fluticasone propionate cream, 0.05% is administered to a nursing woman.
Section 34081-0 (34081-0)
Pediatric Use
Fluticasone propionate cream, 0.05% may be used with caution in pediatric patients as young as 3 months of age. The safety and efficacy of drug use for longer than 4 weeks in this population have not been established. The safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Fluticasone propionate cream, 0.05%, caused HPA axis suppression in 2 of 43 pediatric patients, ages 2 and 5 years old, who were treated for 4 weeks covering at least 35% of the body surface area. Follow-up testing 12 days after treatment discontinuation, available for 1 of the 2 subjects, demonstrated a normally responsive HPA axis (see ADVERSE REACTIONS ). Adverse effects including striae have been reported with use of topical corticosteroids in pediatric patients.
HPA axis suppression, Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients receiving topical corticosteroids. Manifestations of adrenal suppression in pediatric patients include low plasma cortisol levels to an absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
Section 34082-8 (34082-8)
Geriatric Use
A limited number of patients above 65 years of age (n=126) have been treated with fluticasone propionate cream in US and non-US clinical trials. While the number of patients is too small to permit separate analysis of efficacy and safety, the adverse reactions reported in this population were similar to those reported by younger patients. Based on available data, no adjustment of dosage of fluticasone propionate cream in geriatric patients warranted.
Section 34083-6 (34083-6)
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Two 18-month studies were performed in mice to evaluate the carcinogenic potential of fluticasone propionate when given topically (as a 0.05% ointment) and orally. No evidence of carcinogenicity was found in either study.
Fluticasone propionate was not mutagenic in the standard Ames test, E. coli fluctuation test, S. cerevisiae gene conversion test or Chinese Hamster ovarian cell assay. It was not clastogenic in mouse micronucleus or cultured human lymphocyte tests.
In a fertility and general reproductive performance study in rats, fluticasone propionate administered subcutaneously to females at up to 50 mcg/kg per day and to males at up to 100 mcg/kg per day (later reduced to 50 mcg/kg per day) had no effect upon mating performance or fertility. These doses are approximately 15 and 30 times respectively the human systemic exposure following use of the recommended human topical dose of fluticasone propionate cream, 0.05%, assuming human percutaneous absorption of approximately 3% and the use in a 70-kg person of 15 g/day.
Section 42229-5 (42229-5)
Rx Only
FOR DERMATOLOGIC USE ONLY
NOT FOR OPHTHALMIC USE
Overdosage (OVERDOSAGE)
Topically applied fluticasone propionate cream, 0.05% can be absorbed in sufficient amounts to produce systemic effects (see PRECAUTIONS ).
Description (DESCRIPTION)
Fluticasone propionate cream USP, 0.05% contains fluticasone propionate USP [(6α,11β,16α,17α)-6,9-difluoro-11-hydroxy-16-methyl-3-oxo-17-(1-oxopropoxy)androsta-1,4-diene-17-carbothioic acid S-fluoromethyl ester], a synthetic fluorinated corticosteroid, for topical dermatologic use. The topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and antipruritic agents.
Chemically, fluticasone propionate is C25H31F3O5S. It has the following structural formula:
Fluticasone propionate USP has a molecular weight of 500.6. It is a white to off-white powder and is insoluble in water.
Each gram of fluticasone propionate cream USP, 0.05% contains fluticasone propionate USP 0.5 mg in a base of propylene glycol, mineral oil, cetostearyl alcohol, Ceteth-20, isopropyl myristate, dibasic sodium phosphate, citric acid, purified water and imidurea as preservative.
Precautions (PRECAUTIONS)
-
Fluticasone propionate cream, 0.05% contains the excipient imidurea which releases formaldehyde as a breakdown product. Formaldehyde may cause allergic sensitization or irritation upon contact with the skin. Fluticasone propionate cream, 0.05% should not be used in individuals with hypersensitivity to formaldehyde as it may prevent healing or worsen dermatitis.
How Supplied (HOW SUPPLIED)
Fluticasone propionate cream USP, 0.05% is supplied in:
15 g tubes NDC 0168-0332-15
30 g tubes NDC 0168-0332-30
60 g tubes NDC 0168-0332-60
Store between 2° and 30° C (36° and 86° F).
E. FOUGERA & CO.
A division of
Fougera
PHARMACEUTICALS INC.
Melville, NY 11747
46233142A
R07/18
#91
Adverse Reactions (ADVERSE REACTIONS)
In controlled clinical trials of twice daily administration, the total incidence of adverse reactions associated with the use of fluticasone propionate cream, 0.05% was approximately 4%. These adverse reactions were usually mild, self-limiting; and consisted primarily of pruritus, dryness, numbness of fingers, and burning. These events occurred in 2.9%, 1.2%, 1.0%, and 0.6% of patients, respectively.
Two clinical studies compared once to twice-daily administration of fluticasone propionate cream, 0.05% for the treatment of moderate to severe eczema. The local drug-related adverse events for the 491 patients enrolled in both studies are shown in Table 1 . In the study enrolling both adult and pediatric patients, the incidence of local adverse events in the 119 pediatric patients ages 1 to 12 years was comparable to the 140 patients ages 13 to 62 years.
Fifty-one pediatric patients ages 3 months to 5 years, with moderate to severe eczema, were enrolled in an open-label HPA axis safety study. Fluticasone propionate cream, 0.05% was applied twice daily for 3 to 4 weeks over and arithmetic mean body surface area of 64% (range 35% to 95%).
The mean morning cortisol levels with standard deviations before treatment (pre-stimulation mean value =13.76±6.94 mcg/dL, post-stimulation mean value =30.53±7.23 mcg/dL) and at end treatment (pre-stimulation mean value =12.32±6.92 mcg/dL, poststimulation mean value =28.84±7.16 mcg/dL) showed little change. In 2 of 43 (4.7%) patients with end-treatment results, peak cortisol levels following cosyntropin stimulation testing were ≤18 mcg/dL indicating adrenal suppression. Follow-up testing after treatment discontinuation, available for 1 of the 2 subjects, demonstrated a normally responsive HPA axis. Local drug-related adverse events were (see Table 2 ): transient burning, resolving the same day it was reported; transient urticaria, resolving the same day it was reported; erythematous rash; dusky erythema, resolving within one month after cessation of fluticasone propionate cream, 0.05%; and telangiectasia resolving within 3 months after stopping fluticasone propionate cream, 0.05%.
|
Adverse Events |
Fluticasone |
Fluticasone |
Vehicle |
|
Skin infection |
1 (0.5%) |
0 |
0 |
|
Infected eczema |
1 (0.5%) |
2 (1.0%) |
0 |
|
Viral warts |
0 |
1 (0.5%) |
0 |
|
Herpes simplex |
0 |
1 (0.5%) |
0 |
|
Impetigo |
1 (0.5%) |
0 |
0 |
|
Atopic dermatitis |
1 (0.5%) |
0 |
0 |
|
Eczema |
1 (0.5%) |
0 |
0 |
|
Exacerbation of eczema |
4 (1.9%) |
1 (0.5%) |
1 (1.3%) |
|
Erythema |
0 |
2 (1.0%) |
0 |
|
Burning |
2 (1.0%) |
2 (1.0%) |
2 (2.6%) |
|
Stinging |
0 |
2 (1.0%) |
1 (1.3%) |
|
Skin irritation |
6 (2.9%) |
2 (1.0%) |
0 |
|
Pruritus |
2 (1.0%) |
4 (1.9%) |
4 (5.1%) |
|
Exacerbation of Pruritus |
4 (1.9%) |
1 (0.5%) |
1 (1.3%) |
|
Folliculitis |
1 (0.5%) |
1 (0.5%) |
0 |
|
Blisters |
0 |
1 (0.5%) |
0 |
|
Dryness of skin |
3 (1.4%) |
1 (0.5%) |
0 |
| *See text for additional detail. | |
| † n=41 | |
|
Adverse Events |
Fluticasone Twice Daily |
|
Burning |
1 (2.0%) |
|
Dusky Erythema |
1 (2.0%) |
|
Erythematous Rash |
1 (2.0%) |
|
Facial Telangiectasia† |
2 (4.9%) |
|
Non Facial Telangiectasia |
1 (2.0%) |
|
Urticaria |
1 (2.0%) |
The following local adverse reactions have been reported infrequently with topical corticosteroids and they may occur more frequently with the use of occlusive dressings and higher potency corticosteroids. These reactions are listed in an approximately decreasing order of occurrence: irritation, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, skin atrophy, striae, hypertrichosis and miliaria. Also, there are reports of the development of pustular psoriasis from chronic plaque psoriasis following reduction or discontinuation of potent topical corticosteroid products.
Contraindications (CONTRAINDICATIONS)
Fluticasone propionate cream, 0.05% is contraindicated in those patients with a history of hypersensitivity to any of the components in the preparation.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Like other topical corticosteroids, fluticasone propionate has anti-inflammatory, antipruritic and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids, in general, is unclear. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor, arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
Fluticasone propionate is lipophilic and has a strong affinity for the glucocorticoid receptor. It has weak affinity for the progesterone receptor, and virtually no affinity for the mineralocorticoid, estrogen, or androgen receptors. The therapeutic potency of glucocorticoids is related to the half-life of the glucocorticoid-receptor complex. The half-life of the fluticasone propionate-glucocorticoid receptor complex is approximately 10 hours.
Studies performed with fluticasone propionate cream, 0.05% indicate that it is in the medium range of potency as compared with other topical corticosteroids.
Indications and Usage (INDICATIONS AND USAGE)
Fluticasone propionate cream, 0.05% is a medium potency corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. Fluticasone propionate cream, 0.05% may be used with caution in pediatric patients 3 months of age or older. The safety and efficacy of drug use for longer than 4 weeks in this population have not been established. The safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Fluticasone propionate cream, 0.05% may be used in adult and pediatric patients 3 months of age or older. Safety and efficacy of fluticasone propionate cream, 0.05% in pediatric patients for more than 4 weeks of use have not been established (see PRECAUTIONS: Pediatric Use ). The safety and efficacy of Fluticasone propionate cream, 0.05% in pediatric patients below 3 months of age have not been established.
Package Label – Principal Display Panel – 15 G Carton (PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – 15 G CARTON)
NDC 0168-0332-15
Fougera®
FLUTICASONE PROPIONATE
CREAM, 0.05%
Rx only
-
FOR DERMATOLOGIC USE ONLY
NOT FOR OPHTHALMIC USE
WARNING: Keep out of
reach of children.
NET WT 15 grams
Package Label – Principal Display Panel – 15 G Container (PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – 15 G CONTAINER)
NDC 0168-0332-15
Rx only
Fougera ®
FLUTICASONE PROPIONATE
CREAM, 0.05%
FOR DERMATOLOGIC USE ONLY
NOT FOR OPHTHALMIC USE
WARNING: Keep out of
reach of children.
NET WT 15 grams
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:46.856952 · Updated: 2026-03-14T22:30:51.485924