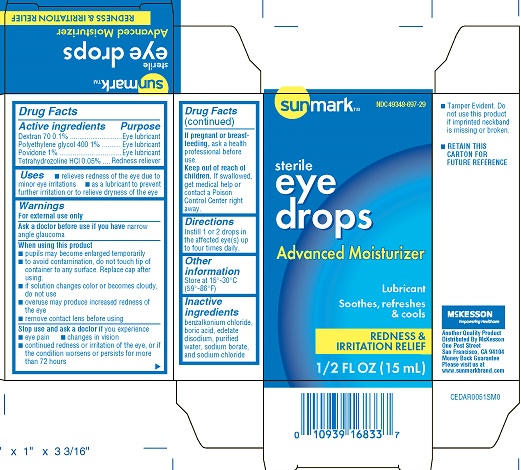
sunmark eye drops advanced moisturizer/lubricant (pld)
0daa4e39-0447-44b6-a7d7-370b12bf9451
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Eye Lubricant Redness reliever
Description
Dextran 70 0.1% povidone 1% Polyethylene glycol 400 1% Tetrahydrozoline HCL 0.05%
Medication Information
Warnings
For external use only
Uses
- relieves redness of the eye due to minor eye irritations
- as a lubricant to prevent further irritation or to relieve dryness of the eye
Purpose
Eye Lubricant
Redness reliever
Directions
Instill 1 or 2 drops in the affected eye(s) up to four times daily.
Other Information
Store at 15 o- 30 oC (59 o- 86 oF)
Inactive Ingredients
benzalkonium chloride, boric acid, edetate disodium, purified water, sodium borate, and sodium chloride
Description
Dextran 70 0.1% povidone 1% Polyethylene glycol 400 1% Tetrahydrozoline HCL 0.05%
Section 51945-4
Active Ingredients
Dextran 70 0.1%
povidone 1%
Polyethylene glycol 400 1%
Tetrahydrozoline HCL 0.05%
When Using This Product
- pupils may become enlarged temporarily
- to avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- if solution changes color or become cloudy, do not use
- overuse may produce increased redness of the eye
- remove contact lens before using
If Pregnant Or Breast Feeding
ask a healthcare professional before use
Keep Out of Reach of Children
If accidentally swallowed, get medical help or contact a Poison Control Center immediately
Ask A Doctor Before Use If You Have
narrow angle glucoma
Stop Use and Ask A Doctor If You Experience
- eye pain
- changes in vision
- continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours
Structured Label Content
Warnings
For external use only
Uses
- relieves redness of the eye due to minor eye irritations
- as a lubricant to prevent further irritation or to relieve dryness of the eye
Purpose
Eye Lubricant
Redness reliever
Directions
Instill 1 or 2 drops in the affected eye(s) up to four times daily.
Other Information (Other information)
Store at 15 o- 30 oC (59 o- 86 oF)
Inactive Ingredients
benzalkonium chloride, boric acid, edetate disodium, purified water, sodium borate, and sodium chloride
Section 51945-4 (51945-4)
Active Ingredients
Dextran 70 0.1%
povidone 1%
Polyethylene glycol 400 1%
Tetrahydrozoline HCL 0.05%
When Using This Product (When using this product)
- pupils may become enlarged temporarily
- to avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- if solution changes color or become cloudy, do not use
- overuse may produce increased redness of the eye
- remove contact lens before using
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
ask a healthcare professional before use
Keep Out of Reach of Children (Keep out of reach of children)
If accidentally swallowed, get medical help or contact a Poison Control Center immediately
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
narrow angle glucoma
Stop Use and Ask A Doctor If You Experience (Stop use and ask a doctor if you experience)
- eye pain
- changes in vision
- continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:38.858799 · Updated: 2026-03-14T23:02:19.584479