Methotrexate Injection
0d63ba29-b692-41b4-87e8-351265c8273f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Methotrexate Injection is a folate analog metabolic inhibitor indicated for: • The following neoplastic diseases for the: o Treatment of adult and pediatric patients with acute lymphoblastic leukemia as part of a combination chemotherapy regimen. ( 1.1 ) o Prophylaxis and treatment of adult and pediatric patients with meningeal leukemia. ( 1.2 ) o Treatment of adult and pediatric patients with non-Hodgkin lymphoma. ( 1.3 ) o Treatment of adult and pediatric patients with osteosarcoma as part of a combination chemotherapy regimen. ( 1.4 ) o Treatment of adults with breast cancer as part of a combination chemotherapy regimen. ( 1.5 ) o Treatment of adults with squamous cell carcinoma of the head and neck as a single agent. ( 1.6 ) o Treatment of adults with gestational trophoblastic neoplasia as part of a combination chemotherapy regimen. ( 1.7 ) • Treatment of adults with rheumatoid arthritis (RA). ( 1.8 ) • Treatment of pediatric patients with polyarticular juvenile idiopathic arthritis (pJIA). ( 1.9 ) • Treatment of adults with severe psoriasis. ( 1.10 )
Dosage and Administration
• Verify pregnancy status in females of reproductive potential before starting Methotrexate Injection. ( 2.1 , 4 , 5.1 ) • Neoplastic diseases: Refer to the prescribing information for disease specific dosing recommendations. Follow guidelines for high-dose regimens. ( 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 , 2.8 , 2.9 ) • RA: Recommended starting dosage of 7.5 mg once weekly intramuscularly; adjust dose to achieve an optimal response. ( 2.10 ) • pJIA: Recommended starting dosage of 10 mg/m 2 once weekly subcutaneously or intramuscularly; adjust dose to achieve an optimal response. ( 2.11 ) • Psoriasis: Recommended dosage of 10 mg to 25 mg once weekly intramuscularly or intravenously; adjust dose to achieve optimal response. Once achieved, reduce to lowest possible dosage. ( 2.12 )
Contraindications
Methotrexate Injection is contraindicated in: • Patients with history of severe hypersensitivity to methotrexate [see Warnings and Precautions (5.2) ] . • Pregnancy in patients with non-neoplastic diseases [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1) ] .
Warnings and Precautions
• Secondary malignancies can occur. ( 5.13 ) • Tumor lysis syndrome can occur in patients with rapidly growing tumors. ( 5.14 ) • Immunizations and Risks associated with Live Vaccines: Immunizations may be ineffective. Live vaccines are not recommended due to risk of disseminated infection. ( 5.15 ) • Infertility: Can cause impairment of fertility, oligospermia, and menstrual dysfunction. ( 5.16 , 8.3 )
Adverse Reactions
Formulations with benzyl alcohol can cause severe central nervous toxicity or metabolic acidosis, if used in neonates or low birth weight infants, intrathecally, or in high-dose regimens. Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required, and preservative-free formulations are not available. The preservative benzyl alcohol can cross the placenta; when possible, use the preservative-free formulation when Methotrexate Injection is needed during pregnancy to treat a neoplastic disease [see Use in Specific Populations (8.1) ] .
Drug Interactions
Refer to full prescribing information for drug interactions with Methotrexate Injection. ( 7 )
Description
Warnings and Precautions ( 5.11 ) 05/2025
Medication Information
Warnings and Precautions
• Secondary malignancies can occur. ( 5.13 ) • Tumor lysis syndrome can occur in patients with rapidly growing tumors. ( 5.14 ) • Immunizations and Risks associated with Live Vaccines: Immunizations may be ineffective. Live vaccines are not recommended due to risk of disseminated infection. ( 5.15 ) • Infertility: Can cause impairment of fertility, oligospermia, and menstrual dysfunction. ( 5.16 , 8.3 )
Indications and Usage
Methotrexate Injection is a folate analog metabolic inhibitor indicated for: • The following neoplastic diseases for the: o Treatment of adult and pediatric patients with acute lymphoblastic leukemia as part of a combination chemotherapy regimen. ( 1.1 ) o Prophylaxis and treatment of adult and pediatric patients with meningeal leukemia. ( 1.2 ) o Treatment of adult and pediatric patients with non-Hodgkin lymphoma. ( 1.3 ) o Treatment of adult and pediatric patients with osteosarcoma as part of a combination chemotherapy regimen. ( 1.4 ) o Treatment of adults with breast cancer as part of a combination chemotherapy regimen. ( 1.5 ) o Treatment of adults with squamous cell carcinoma of the head and neck as a single agent. ( 1.6 ) o Treatment of adults with gestational trophoblastic neoplasia as part of a combination chemotherapy regimen. ( 1.7 ) • Treatment of adults with rheumatoid arthritis (RA). ( 1.8 ) • Treatment of pediatric patients with polyarticular juvenile idiopathic arthritis (pJIA). ( 1.9 ) • Treatment of adults with severe psoriasis. ( 1.10 )
Dosage and Administration
• Verify pregnancy status in females of reproductive potential before starting Methotrexate Injection. ( 2.1 , 4 , 5.1 ) • Neoplastic diseases: Refer to the prescribing information for disease specific dosing recommendations. Follow guidelines for high-dose regimens. ( 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 , 2.8 , 2.9 ) • RA: Recommended starting dosage of 7.5 mg once weekly intramuscularly; adjust dose to achieve an optimal response. ( 2.10 ) • pJIA: Recommended starting dosage of 10 mg/m 2 once weekly subcutaneously or intramuscularly; adjust dose to achieve an optimal response. ( 2.11 ) • Psoriasis: Recommended dosage of 10 mg to 25 mg once weekly intramuscularly or intravenously; adjust dose to achieve optimal response. Once achieved, reduce to lowest possible dosage. ( 2.12 )
Contraindications
Methotrexate Injection is contraindicated in: • Patients with history of severe hypersensitivity to methotrexate [see Warnings and Precautions (5.2) ] . • Pregnancy in patients with non-neoplastic diseases [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1) ] .
Adverse Reactions
Formulations with benzyl alcohol can cause severe central nervous toxicity or metabolic acidosis, if used in neonates or low birth weight infants, intrathecally, or in high-dose regimens. Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required, and preservative-free formulations are not available. The preservative benzyl alcohol can cross the placenta; when possible, use the preservative-free formulation when Methotrexate Injection is needed during pregnancy to treat a neoplastic disease [see Use in Specific Populations (8.1) ] .
Drug Interactions
Refer to full prescribing information for drug interactions with Methotrexate Injection. ( 7 )
Description
Warnings and Precautions ( 5.11 ) 05/2025
Section 42229-5
With Preservative (Multiple-Dose Vial)
-
•Methotrexate Injection formulation containing benzyl alcohol as a preservative may be administered by intramuscular, intravenous, or subcutaneous injection [see Dosage and Administration (2.1)]. Methotrexate Injection with preservative may be further diluted with 0.9% Sodium Chloride Injection, USP. Diluted product should be used within 4 hours when stored at room temperature (20°C to 25°C) or 24 hours under refrigeration (2°C to 8°C).
-
•Visually inspect product for particulate matter and discoloration prior to administration. Discard if particulate matter or discoloration is observed.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 05/2025 | |||
|
Patient Information
|
|||
|
What is the most important information I should know about Methotrexate Injection?
Males with female partners who are able to become pregnant:
Severe allergic reactions. Severe allergic reactions can happen with Methotrexate Injection.
|
|||
|
|
||
|
Decreased blood cell counts. Methotrexate Injection can affect your bone marrow and cause decreased red blood cell counts, white blood cell counts, and platelet counts, and a condition where your bone marrow cannot produce these blood cells (aplastic anemia). These decreased blood cell counts can be severe and may lead to a serious infection, the need for blood transfusions, treatment in a hospital, and can be life-threatening. Your healthcare provider will check your blood cell counts before you start and during treatment with Methotrexate Injection. Your healthcare provider will watch you closely for infections during treatment with Methotrexate Injection. |
|||
|
|
|
|
|
Your healthcare provider may give you medicines to support your blood counts or give you transfusions if needed, and change your dose or stop your treatment with Methotrexate Injection if needed. |
|||
|
|
||
|
Your healthcare provider will closely watch you for signs and symptoms of infection during treatment with Methotrexate Injection. Your healthcare provider may hold or stop your treatment with Methotrexate Injection if you develop a serious infection.
Tell your healthcare provider if you develop any signs or symptoms of liver problems during treatment with Methotrexate Injection, including: |
|||
|
|
||
|
Brain and spinal cord (nervous system) problems. Methotrexate Injection can cause nervous system problems that can be severe and last for a short time or last for a long time. These nervous system problems can get progressively worse, may not get better (possibly irreversible), and can cause death.
Call your healthcare provider right away if you or your child develop any new signs or symptoms of a nervous system problem during treatment with Methotrexate Injection, including: |
|||
|
|
||
|
Severe stomach and intestine (gastrointestinal) problems.
Call your healthcare provider right away if you develop: |
|||
|
|
||
|
Lung problems. Lung problems can happen suddenly (acute) with Methotrexate Injection or they can develop over a long period-of-time (chronic). Lung problems may not get better (possibly irreversible) and can cause death in anyone taking Methotrexate Injection. Your healthcare provider will monitor you for lung problems during treatment with Methotrexate Injection. Your healthcare provider may hold or stop your treatment with Methotrexate Injection, if needed.
Limit sunlight exposure during treatment with Methotrexate Injection. Use a broad-spectrum ultraviolet sunscreen and lip balm with a Sun Protection Factor (SPF) of 30 or greater and wear a hat and protective clothing when you will be exposed to sunlight during treatment with Methotrexate Injection. Call your healthcare provider right away if you develop a new or worsening skin rash during treatment with Methotrexate Injection. See “What are the possible side effects of Methotrexate Injection?” for more information about side effects. |
|||
|
What is Methotrexate Injection?
in adults:
Methotrexate Injection is a prescription medicine used:
|
|||
|
Do not receive Methotrexate Injection if you:
|
|||
|
Before you receive Methotrexate Injection, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking certain medicines can affect the amount of methotrexate in your blood and can increase your risk for serious side effects. |
|||
|
How will I receive or take Methotrexate Injection?
|
|||
|
|
|
|
|
|||
|
If you are receiving Methotrexate Injection to treat your cancer:
If you are receiving Methotrexate Injection for treatment of severe psoriasis, rheumatoid arthritis, or polyarticular juvenile idiopathic arthritis:
In all patients receiving Methotrexate Injection:
|
|||
|
What are the possible side effects of Methotrexate Injection?
The most common side effects of Methotrexate Injection include:
Possible fertility problems (infertility) in males and females. Methotrexate Injection can cause fertility problems in males and females, and cause sperm production to stop in males, and menstrual problems in females. In females, your periods (menstrual cycle) may be irregular or completely stop when you receive Methotrexate Injection. Your periods may or may not return to normal following treatment. It is not known if your fertility will return after treatment. Talk to your healthcare provider about your risk for infertility if this is a concern for you. |
|||
|
General information about the safe and effective use of Methotrexate Injection.
|
|||
|
What are the ingredients in Methotrexate Injection? For more information, go to www.pfizer.com or call 1-800-438-1985. |
Section 43683-2
|
Warnings and Precautions (5.11) |
05/2025 |
Section 44425-7
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from light.
After first puncture, store multiple-dose vials at 2°C to 8°C, and use within 30 days.
Methotrexate Injection is a hazardous drug. Follow applicable special handling and disposal procedures.1
15 References
-
1."OSHA Hazardous Drugs." OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html.
1.10 Psoriasis
Methotrexate Injection is indicated for the treatment of adults with severe psoriasis.
11 Description
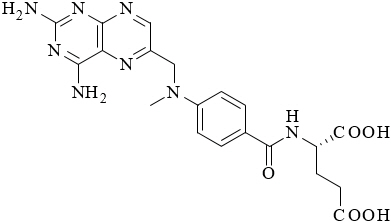
Methotrexate is a folate analog metabolic inhibitor with the chemical name of N-[4-[[(2,4-diamino-6-pteridinyl) methyl]methylamino]benzoyl]-L-glutamic acid and a molecular weight of 454.44. The molecular formula is C20H22N8O5 , and the structural formula is shown below:
Methotrexate Injection with preservative is supplied in sterile multiple-dose vials for intravenous, intramuscular, or subcutaneous use.
-
•Each 25 mg/mL, 2 mL vial contains 50 mg methotrexate equivalent to 54.8 mg of methotrexate sodium, 18.8 mg of benzyl alcohol as a preservative and Sodium chloride 5.2 mg. May contain sodium hydroxide and/or hydrochloric acid to adjust the pH to 8.5.
Preservative-free Methotrexate Injection is supplied in sterile single-dose vials for intravenous, intramuscular, subcutaneous, or intrathecal use.
-
•Each 25 mg/mL, 40 mL vial contains 1000 mg methotrexate equivalent to 1096.7 mg of methotrexate sodium, and the following inactive ingredients: Sodium chloride 196 mg. May contain sodium hydroxide and/or hydrochloric acid to adjust pH to 8.5.
1.4 Osteosarcoma
Methotrexate Injection is indicated for the treatment of adults and pediatric patients with osteosarcoma as part of a combination chemotherapy regimen.
5.16 Infertility
Based on published reports, methotrexate can cause impairment of fertility, oligospermia, and menstrual dysfunction. It is not known if the infertility may be reversible in affected patients. Discuss the risk of effects on reproduction with female and male patients of reproductive potential [see Use in Specific Populations (8.3)].
1.5 Breast Cancer
Methotrexate Injection is indicated for the treatment of adults with breast cancer as part of a combination chemotherapy regimen.
5.8 Neurotoxicity
Methotrexate can cause severe acute and chronic neurotoxicity which can be progressive, irreversible, and fatal. Serious neurotoxicity, including generalized and focal seizures, have occurred in pediatric patients [see Use in Specific Populations (8.4)]. Monitor patients for signs of neurotoxicity and withhold or discontinue Methotrexate Injection when appropriate.
8.4 Pediatric Use
The safety and effectiveness of Methotrexate Injection in pediatric patients have been established for ALL, meningeal leukemia prophylaxis and treatment, non-Hodgkin lymphoma, osteosarcoma and in pJIA. Clinical studies evaluating the use of methotrexate in pediatric patients with pJIA demonstrated safety comparable to that observed in adults with RA [see Adverse Reactions (6.1)]. The safety and effectiveness of Methotrexate Injection have not been established in pediatric patients for the treatment of breast cancer, squamous cell carcinoma of the head and neck, gestational trophoblastic neoplasia, rheumatoid arthritis, and psoriasis. Additional risk information is described below.
8.5 Geriatric Use
Clinical studies of methotrexate did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
5.6 Renal Toxicity
Methotrexate can cause renal toxicity including irreversible acute renal failure. Monitor renal function and withhold or discontinue Methotrexate Injection as needed for severe renal toxicity.
For patients receiving high-dose regimens, follow recommendations to decrease the risk of renal injury and mitigate renal toxicity [see Dosage and Administration (2.2)].
Patients with impaired renal function are at increased risk for methotrexate toxicity [see Use in Specific Populations (8.6)].
Consider administration of glucarpidase in patients with toxic plasma methotrexate concentrations (>1 micromole per liter) and delayed clearance due to impaired renal function. [see Dosage and Administration (2.2)].
5.7 Hepatotoxicity
Methotrexate can cause severe and potentially irreversible hepatotoxicity including fibrosis, cirrhosis, and fatal liver failure [see Adverse Reactions (6.1, 6.2)].
In patients with psoriasis, fibrosis or cirrhosis may occur in the absence of symptoms or abnormal liver function tests. In patients with psoriasis, the risk of hepatotoxicity appears to increase with total cumulative dose and generally occurs after receipt of a total cumulative dose of 1.5 g or more.
The safety of Methotrexate Injection in patients with liver disease is unknown. Avoid use of Methotrexate Injection in patients with chronic liver disease, unless benefits clearly outweigh the risks. The risk of hepatotoxicity is increased with heavy alcohol consumption.
Assess liver function prior to initiating Methotrexate Injection and monitor liver function tests during treatment. Withhold or discontinue Methotrexate Injection as appropriate.
4 Contraindications
Methotrexate Injection is contraindicated in:
-
•Patients with history of severe hypersensitivity to methotrexate [see Warnings and Precautions (5.2)].
-
•Pregnancy in patients with non-neoplastic diseases [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
6 Adverse Reactions
The following adverse reactions are described, or described in greater detail, in other sections:
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
-
•Myelosuppression [see Warnings and Precautions (5.4)]
-
•Serious Infections [see Warnings and Precautions (5.5)]
-
•Renal Toxicity [see Warnings and Precautions (5.6)]
-
•Hepatotoxicity [see Warnings and Precautions (5.7)]
-
•Neurotoxicity [see Warnings and Precautions (5.8)]
-
•Gastrointestinal Toxicity [see Warnings and Precautions (5.9)]
-
•Pulmonary Toxicity [see Warnings and Precautions (5.10)]
-
•Dermatologic Reactions [see Warnings and Precautions (5.11)]
-
•Secondary Malignancies [see Warnings and Precautions (5.13)]
-
•Tumor Lysis Syndrome [see Warnings and Precautions (5.14)]
-
•Increased Risk of Adverse Reactions due to Third-Space Accumulation [see Warnings and Precautions (5.17)]
7 Drug Interactions
Refer to full prescribing information for drug interactions with Methotrexate Injection. (7)
5.4 Myelosuppression
Methotrexate suppresses hematopoiesis and can cause severe and life-threatening pancytopenia, anemia, aplastic anemia, leukopenia, neutropenia, and thrombocytopenia [see Adverse Reactions (6.1)].
Obtain blood counts at baseline and periodically during treatment. Monitor patients for possible clinical complications of myelosuppression. Provide supportive care and withhold, reduce dose, or discontinue Methotrexate Injection as needed.
8.6 Renal Impairment
Methotrexate elimination is reduced in patients with renal impairment [creatinine clearance (CLcr) less than 90 mL/min, calculated using Cockcroft-Gault] [see Clinical Pharmacology (12.3)]. Patients with renal impairment are at increased risk for methotrexate adverse reactions.
Follow recommendations to promote methotrexate elimination and decrease risk of acute kidney injury and other methotrexate toxicities in patients who are receiving intermediate- or high-dose regimens [see Dosage and Administration (2.2) and Warnings and Precautions (5.6)]. Consider reducing the dose or discontinuing Methotrexate Injection in patients with renal impairment as appropriate.
5.5 Serious Infections
Patients treated with methotrexate are at increased risk for developing life-threatening or fatal bacterial, fungal, or viral infections including opportunistic infections such as Pneumocystis jiroveci pneumonia, invasive fungal infections, hepatitis B reactivation, tuberculosis primary infection or reactivation, and disseminated Herpes zoster and cytomegalovirus infections.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with Methotrexate Injection. Withhold or discontinue Methotrexate Injection in patients who develop serious infections.
8.7 Hepatic Impairment
The pharmacokinetics and safety of methotrexate in patients with hepatic impairment is unknown. Patients with hepatic impairment may be at increased risk for methotrexate adverse reaction based on elimination characteristics of methotrexate [see Clinical Pharmacology (12.3)]. Consider reducing the dose or discontinuing Methotrexate Injection in patients with hepatic impairment as appropriate [see Warnings and Precautions (5.7)].
1 Indications and Usage
Methotrexate Injection is a folate analog metabolic inhibitor indicated for:
-
•The following neoplastic diseases for the:
-
oTreatment of adult and pediatric patients with acute lymphoblastic leukemia as part of a combination chemotherapy regimen. (1.1)
-
oProphylaxis and treatment of adult and pediatric patients with meningeal leukemia. (1.2)
-
oTreatment of adult and pediatric patients with non-Hodgkin lymphoma. (1.3)
-
oTreatment of adult and pediatric patients with osteosarcoma as part of a combination chemotherapy regimen. (1.4)
-
oTreatment of adults with breast cancer as part of a combination chemotherapy regimen. (1.5)
-
oTreatment of adults with squamous cell carcinoma of the head and neck as a single agent. (1.6)
-
oTreatment of adults with gestational trophoblastic neoplasia as part of a combination chemotherapy regimen. (1.7)
-
-
•Treatment of adults with rheumatoid arthritis (RA). (1.8)
-
•Treatment of pediatric patients with polyarticular juvenile idiopathic arthritis (pJIA). (1.9)
-
•Treatment of adults with severe psoriasis. (1.10)
5.10 Pulmonary Toxicity
Methotrexate-induced pulmonary toxicity including acute or chronic interstitial pneumonitis and irreversible or fatal cases can occur at all dose levels. Monitor patients for signs of pulmonary toxicity and withhold or discontinue Methotrexate Injection as appropriate.
1.3 Non Hodgkin Lymphoma
Methotrexate Injection is indicated for the treatment of adults and pediatric patients with non-Hodgkin lymphoma.
1.8 Rheumatoid Arthritis
Methotrexate Injection is indicated for the treatment of adults with rheumatoid arthritis (RA).
12.1 Mechanism of Action
Methotrexate inhibits dihydrofolic acid reductase. Dihydrofolates must be reduced to tetrahydrofolates by this enzyme before they can be utilized as carriers of one-carbon groups in the synthesis of purine nucleotides and thymidylate. Therefore, methotrexate interferes with DNA synthesis, repair, and cellular replication. Actively proliferating tissues such as malignant cells, bone marrow, fetal cells, buccal and intestinal mucosa, and cells of the urinary bladder are in general more sensitive to this effect of methotrexate.
The mechanism of action in rheumatoid arthritis, pJIA, and in psoriasis is unknown.
5.1 Embryo Fetal Toxicity
Based on published reports and its mechanism of action, methotrexate can cause embryo-fetal toxicity, including fetal death when administered to a pregnant woman.
Methotrexate Injection is contraindicated for use in pregnant women with non-neoplastic diseases. Advise pregnant women with neoplastic diseases of the potential risk to a fetus. The preservative benzyl alcohol can cross the placenta; when possible, use the preservative-free formulation when Methotrexate Injection is needed during pregnancy to treat a neoplastic disease [see Warnings and Precautions (5.3)].
Advise females of reproductive potential to use effective contraception during Methotrexate Injection treatment and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during Methotrexate Injection treatment and for 3 months after the last dose [see Contraindications (4) and Use in Specific Populations (8.1, 8.3, 8.4)].
5.14 Tumor Lysis Syndrome
Methotrexate can induce tumor lysis syndrome in patients with rapidly growing tumors. Institute appropriate treatment for prevention and management of tumor lysis syndrome.
5 Warnings and Precautions
-
•Secondary malignancies can occur. (5.13)
-
•Tumor lysis syndrome can occur in patients with rapidly growing tumors. (5.14)
-
•Immunizations and Risks associated with Live Vaccines: Immunizations may be ineffective. Live vaccines are not recommended due to risk of disseminated infection. (5.15)
-
•Infertility: Can cause impairment of fertility, oligospermia, and menstrual dysfunction. (5.16, 8.3)
2 Dosage and Administration
-
•Verify pregnancy status in females of reproductive potential before starting Methotrexate Injection. (2.1, 4, 5.1)
-
•Neoplastic diseases: Refer to the prescribing information for disease specific dosing recommendations. Follow guidelines for high-dose regimens. (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9)
-
•RA: Recommended starting dosage of 7.5 mg once weekly intramuscularly; adjust dose to achieve an optimal response. (2.10)
-
•pJIA: Recommended starting dosage of 10 mg/m2 once weekly subcutaneously or intramuscularly; adjust dose to achieve an optimal response. (2.11)
-
•Psoriasis: Recommended dosage of 10 mg to 25 mg once weekly intramuscularly or intravenously; adjust dose to achieve optimal response. Once achieved, reduce to lowest possible dosage. (2.12)
5.11 Dermatologic Reactions
Severe, including fatal, dermatologic reactions, such as toxic epidermal necrolysis, Stevens-Johnson syndrome, exfoliative dermatitis, skin necrosis, and erythema multiforme, can occur with methotrexate [see Adverse Reactions (6.1, 6.2)].
Psoriasis may be aggravated by concomitant exposure to ultraviolet radiation.
Methotrexate can cause radiation recall, photodermatitis (sunburn) reactivation, photosensitivity, and severe sunburn reactions. Advise patients to limit sun exposure while taking Methotrexate Injection. Advise patients when outdoors to wear a hat and protective clothing and use a broad-spectrum Ultraviolet A (UVA)/Ultraviolet B (UVB) sunscreen and lip balm (SPF ≥30) to help protect against sunburn.
Monitor patients for signs of dermatologic toxicity and withhold or permanently discontinue Methotrexate Injection for severe dermatologic adverse reactions.
5.13 Secondary Malignancies
Secondary malignancies can occur at all dose levels of methotrexate. In some cases, lymphoproliferative disease that occurred during therapy with low-dose methotrexate regressed completely following withdrawal of methotrexate. If lymphoproliferative disease occurs, discontinue Methotrexate Injection and institute appropriate treatment if lymphoma does not regress.
3 Dosage Forms and Strengths
Injection: Methotrexate Injection is a clear, yellow solution and is supplied in single-dose vials (preservative-free) and multiple-dose vials (with preservative) in the following strengths:
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of methotrexate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: Aplastic anemia, lymphadenopathy, hypogammaglobulinemia
Cardiovascular disorders: Thromboembolic events (including arterial thrombosis, cerebral thrombosis, deep vein thrombosis, retinal vein thrombosis, thrombophlebitis, and pulmonary embolus), pericarditis, pericardial effusion, hypotension, sudden death
Endocrine: Diabetes
Eye disorders: Optic neuropathy, blurred vision, ocular irritation, conjunctivitis, xerophthalmia
Gastrointestinal disorders: Hemorrhagic enteritis, intestinal perforation, gingivitis, pancreatitis, pharyngitis, hematemesis, melena, gastrointestinal ulceration and bleeding
Hepatobiliary disorders: Acute hepatitis, decreased serum albumin, fibrosis, cirrhosis, liver failure
Immune system disorders: Anaphylaxis, anaphylactoid reactions, vasculitis
Metabolism: Hyperglycemia
Musculoskeletal disorders: Stress fracture, soft tissue necrosis, arthralgia, myalgia, osteoporosis
Nervous system disorders: Headaches, drowsiness, blurred vision, speech impairment (including dysarthria and aphasia), transient cognitive dysfunction, mood alteration, unusual cranial sensations, paresis, encephalopathy, leukoencephalopathy, and convulsions. Also, spinal radiculopathy with intrathecal use
Renal disorders: Severe renal toxicity including renal failure, azotemia, hematuria, proteinuria, cystitis
Reproductive disorders: Defective oogenesis or spermatogenesis, loss of libido, impotence, gynecomastia, menstrual dysfunction
Respiratory disorders: Pulmonary fibrosis, respiratory failure, chronic interstitial obstructive pulmonary disease, pleuritic pain and thickening, alveolitis
Skin disorders: Toxic epidermal necrolysis, Stevens-Johnson syndrome, exfoliative dermatitis, skin necrosis, and erythema multiforme, erythematous rashes, pruritus, alopecia, skin ulceration, accelerated nodulosis, urticaria, pigmentary changes, ecchymosis, telangiectasia, photosensitivity, acne, furunculosis
General disorders and administration site conditions: Injection site necrosis, injection site reaction
5.9 Gastrointestinal Toxicity
Methotrexate can cause diarrhea, vomiting, stomatitis, hemorrhagic enteritis and fatal intestinal perforation [see Adverse Reactions (6.1)]. Patients with peptic ulcer disease or ulcerative colitis are at a greater risk of developing severe gastrointestinal adverse reactions.
Withhold or discontinue Methotrexate Injection for severe gastrointestinal toxicity, and institute appropriate supportive care as needed.
8 Use in Specific Populations
5.2 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis, can occur with methotrexate [see Adverse Reactions (6.1)]. If signs or symptoms of anaphylaxis or any other serious hypersensitivity reaction occurs, immediately discontinue Methotrexate Injection and institute appropriate therapy [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials and other studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Commonly reported adverse reactions include ulcerative stomatitis, leukopenia, nausea, and abdominal distress. Other frequently reported adverse reactions are infection, malaise, fatigue, chills, fever, and dizziness.
1.1 Acute Lymphoblastic Leukemia
Methotrexate Injection is indicated for the treatment of adult and pediatric patients with acute lymphoblastic leukemia (ALL) as part of a combination chemotherapy regimen.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.12 Recommended Dosage for Psoriasis
The recommended dosage of Methotrexate Injection is 10 mg to 25 mg intramuscularly or intravenously once weekly until adequate response is achieved.
Adjust the dose gradually to achieve optimal clinical response; do not exceed 25 mg per week. Once optimal clinical response has been achieved, reduce the dosage to the lowest possible dosing regimen.
Administer folic acid or folinic acid to reduce the risk of methotrexate adverse reactions [see Warnings and Precautions (5.12)].
1.7 Gestational Trophoblastic Neoplasia
Methotrexate Injection is indicated for the treatment of adults with gestational trophoblastic neoplasia (GTN) as part of a combination chemotherapy regimen.
2.6 Recommended Dosage for Osteosarcoma
The recommended dosage of Methotrexate Injection is typically 12 g/m2 (maximum 20 g/dose) as an intravenous infusion over 4 hours administered as a component of a combination chemotherapy regimen. Administer leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)]. Subsequent doses may need to be adjusted based on observed peak serum methotrexate concentrations. Dosage and schedule may vary based upon factors such as patient comorbidities, disease state, and prior treatments.
2.7 Recommended Dosage for Breast Cancer
A recommended dosage of Methotrexate Injection is 40 mg/m2 intravenously as a component of a cyclophosphamide- and fluorouracil-based multi-drug regimen.
Principal Display Panel 2 Ml Vial Label
Sterile
2 mL Multiple-dose Vial
NDC 61703-350-37
Rx only
Methotrexate Injection, USP
50 mg/2 mL
Isotonic Liquid
(25 mg/mL)
Contains preservative Hazardous Drug
For Intravenous, Intramuscular and Subcutaneous Use Only
Principal Display Panel 2 Ml Vial Carton
VIAL
Hospira
5 x 2 mL Multiple-dose Vial
Sterile
NDC 61703-350-38
Rx only
Methotrexate Injection, USP
Isotonic Liquid
For Intravenous, Intramuscular and
Subcutaneous Use Only
CONTAINS PRESERVATIVE
NOT FOR INTRATHECAL USE
50 mg/ 2 mL
(25 mg/mL)
Hazardous Drug
Principal Display Panel 40 Ml Vial Label
Sterile
40 mL Single-dose vial. Discard unused portion.
NDC 61703-408-41
Rx only
Methotrexate Injection, USP
1 g/40 mL
(25 mg/mL)
For Intravenous, Intramuscular, Subcutaneous and Intrathecal Use Only
Hazardous Drug
Preservative Free
2.1 Important Dosage and Safety Information
-
•Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required and preservative-free formulations are not available [see Warnings and Precautions (5.3) and Use in Specific Populations (8.4)].
-
•Verify pregnancy status in females of reproductive potential before starting Methotrexate Injection [see Contraindications (4) and Warnings and Precautions (5.1)].
-
•For patients switching between a methotrexate product administered orally and Methotrexate Injection, consider potential differences in bioavailability.
Principal Display Panel 40 Ml Vial Carton
VIAL
Hospira
1 x 40 mL Vial
Sterile
NDC 61703-408-41
Rx only
Methotrexate
Injection, USP
Preservative Free
1 g/ 40 mL
(25 mg/mL)
For Intravenous, Intramuscular,
Subcutaneous and Intrathecal Use Only
Single-dose vial. Discard unused portion.
Hazardous Drug
2.14 Administration and Handling Information
Methotrexate Injection is a hazardous drug. Follow applicable special handling and disposable procedures.1
1.9 Polyarticular Juvenile Idiopathic Arthritis
Methotrexate Injection is indicated for the treatment of pediatric patients with polyarticular Juvenile Idiopathic Arthritis (pJIA).
2.13 Dosage Modifications for Adverse Reactions
Discontinue Methotrexate Injection for:
-
•Anaphylaxis or other severe hypersensitivity reactions [see Warnings and Precautions (5.2)]
-
•Lymphoproliferative disease [see Warnings and Precautions (5.13)]
Withhold, dose reduce or discontinue Methotrexate Injection as appropriate for:
-
•Myelosuppression [see Warnings and Precautions (5.4)]
Withhold or discontinue Methotrexate Injection as appropriate for:
-
•Serious infections [see Warnings and Precautions (5.5)]
-
•Renal toxicity [see Warnings and Precautions (5.6)]
-
•Hepatotoxicity [see Warnings and Precautions (5.7)]
-
•Neurotoxicity [see Warnings and Precautions (5.8)]
-
•Gastrointestinal toxicity [see Warnings and Precautions (5.9)]
-
•Pulmonary toxicity [see Warnings and Precautions (5.10)]
-
•Dermatologic reactions [see Warnings and Precautions (5.11)]
2.5 Recommended Dosage for Non Hodgkin Lymphoma
The recommended dosage of Methotrexate Injection varies. When used in combination, recommended dosages range from 10 mg/m2 to 8000 mg/m2 intravenously. When used as a single agent, recommended dosages include 8000 mg/m2 intravenously for central nervous system-directed therapy or 5 to 75 mg intravenously for cutaneous forms of non-Hodgkin lymphoma.
As part of a combination chemotherapy regimen, a recommended dosage of Methotrexate Injection is 1000 mg/m2 or 3000 mg/m2 as an intravenous infusion over 24 hours followed by leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)].
For central nervous system-directed therapy, a recommended dosage of Methotrexate Injection is 8000 mg/m2 as an intravenous infusion over 4 hours as a single agent or in combination with immunochemotherapy at doses ranging from 3000 mg/m2 to 8000 mg/m2 followed by leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)].
For intrathecal Methotrexate Injection (preservative-free), the recommended dose is based on age [see Dosage and Administration (2.4)]. The frequency of administration varies based on whether it is being used for treatment or prophylaxis, and other factors.
8.3 Females and Males of Reproductive Potential
Methotrexate can cause malformations and fetal death at doses less than or equal to the recommended clinical doses [see Use in Specific Populations (8.1)].
1.6 Squamous Cell Carcinoma of the Head and Neck
Methotrexate Injection is indicated for the treatment of adults with squamous cell carcinoma of the head and neck as a single agent.
2.10 Recommended Dosage for Rheumatoid Arthritis
The recommended starting dosage of Methotrexate Injection is 7.5 mg once weekly, administered intramuscularly with escalation to achieve optimal response. Dosages of more than 20 mg once weekly result in an increased risk of serious adverse reactions, including myelosuppression.
When responses are observed, the majority occurred between 3 and 6 weeks from initiation of treatment; however, responses have occurred up to 12 weeks after treatment initiation.
Administer folic acid or folinic acid to reduce the risk of methotrexate adverse reactions [see Warnings and Precautions (5.12)].
1.2 Meningeal Leukemia: Prophylaxis and Treatment
Methotrexate Injection is indicated for the prophylaxis and treatment of meningeal leukemia in adult and pediatric patients.
2.3 Recommended Dosage for Acute Lymphoblastic Leukemia
Methotrexate Injection is used as part of a multi-drug regimen. The recommended dosage varies from 10 to 5000 mg/m2 intravenously. For high-dose Methotrexate Injection regimens, use leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)]. Lower doses (e.g., 20 to 30 mg/m2 per week) may be used intramuscularly. Individualize the dose and schedule of Methotrexate Injection based on disease state, patient risk category, concurrent drugs used, phase of treatment, and response to treatment.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Methotrexate has been evaluated in a number of animal studies for carcinogenic potential with inconclusive results. There is evidence that methotrexate causes chromosomal damage to animal somatic cells and human bone marrow cells [see Use in Specific Populations (8.1, 8.2, 8.3)].
5.15 Immunization and Risks Associated With Live Vaccines
Immunization during Methotrexate Injection treatment may be ineffective.
Disseminated infections following administration of live vaccines have been reported.
Update immunizations according to immunization guidelines prior to initiating Methotrexate Injection. Immunization with live vaccines is not recommended during treatment. The interval between live vaccinations and initiation of Methotrexate Injection should be in accordance with current vaccination guidelines for patients on immunosuppressive therapies.
5.19 Risk of Serious Adverse Reactions With Medication Errors
Serious adverse reactions, including death, have occurred due to medication errors. Most commonly, these errors occurred in patients who were taking methotrexate daily when a weekly dosing regimen was prescribed. Ensure that patients receive the recommended dosage, because medication errors have led to death.
2.9 Recommended Dosage for Gestational Trophoblastic Neoplasia
For patients with low-risk gestational trophoblastic neoplasia (GTN) a recommended dosage for Methotrexate Injection is 30 mg/m2 to 200 mg/m2 or 0.4 mg/kg to1 mg/kg intravenously or intramuscularly.
For patients with high-risk GTN, a recommended dosage for Methotrexate Injection is 300 mg/m2 over 12 hours as an intravenous infusion as a component of a multi-drug regimen.
2.8 Recommended Dosage for Squamous Cell Carcinoma of Head and Neck
The recommended dosage of Methotrexate Injection ranges from 40 to 60 mg/m2 intravenously once weekly.
2.11 Recommended Dosage for Polyarticular Juvenile Idiopathic Arthritis
The recommended starting dosage of Methotrexate Injection is 10 mg/m2 once weekly administered subcutaneously or intramuscularly, with escalation to achieve optimal response. Dosages over 30 mg/m2 per week may result in an increased risk of serious adverse reactions, including myelosuppression. When responses are observed, the majority occurred between 3 and 6 weeks from initiation of treatment; however, responses have occurred up to 12 weeks after treatment initiation.
Administer folic acid or folinic acid to reduce the risk of methotrexate adverse reactions [see Warnings and Precautions (5.12)].
2.4 Recommended Dosage for Meningeal Leukemia: Prophylaxis and Treatment
Use only preservative-free Methotrexate Injection for intrathecal use.
Prior to administration, dilute preservative-free Methotrexate Injection to a concentration of 1 mg/mL in preservative-free 0.9% Sodium Chloride Injection, USP.
The recommended intrathecal dose of Methotrexate Injection (preservative-free) is based on age:
-
•less than 1 year: 6 mg
-
•1 to less than 2 years: 8 mg
-
•2 to less than 3 years: 10 mg
-
•3 to less than 9 years: 12 mg
-
•greater than or equal to 9 years: 12 to15 mg
For treatment of meningeal leukemia, intrathecal methotrexate may be given at intervals of 2 or more days up to twice weekly; however, administration at intervals of less than 1 week may result in increased subacute toxicity. For meningeal leukemia prophylaxis, Methotrexate Injection is administered no more than once weekly.
For patients with Down Syndrome, administer leucovorin rescue with intrathecal Methotrexate Injection.
5.17 Increased Risk of Adverse Reactions Due to Third Space Accumulation
Methotrexate can exit slowly from third-space accumulations resulting in prolonged terminal plasma half-life and toxicity. Evacuate significant third-space accumulations prior to Methotrexate Injection administration [see Clinical Pharmacology (12.3)].
5.3 Risks of Serious Adverse Reactions Due to Benzyl Alcohol Preservative
Formulations with benzyl alcohol can cause severe central nervous toxicity or metabolic acidosis, if used in neonates or low birth weight infants, intrathecally, or in high-dose regimens. Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required, and preservative-free formulations are not available. The preservative benzyl alcohol can cross the placenta; when possible, use the preservative-free formulation when Methotrexate Injection is needed during pregnancy to treat a neoplastic disease [see Use in Specific Populations (8.1)].
5.18 Increased Risk of Soft Tissue and Bone Toxicity With Concomitant Radiotherapy
Concomitant radiation therapy increases the risk of soft tissue necrosis and osteonecrosis associated with methotrexate.
2.2 Recommended Monitoring and Concomitant Therapies for Intermediate and High Dose Regimens
To decrease the risk of severe adverse reactions [see Warnings and Precautions (5)]:
-
•Administer leucovorin rescue in patients receiving Methotrexate Injection doses of 500 mg/m2 or greater (e.g., high-dose).
-
•Consider leucovorin rescue for patients receiving Methotrexate Injection doses between 100 mg/m2 to less than 500 mg/m2 (e.g., intermediate-dose).
Refer to the leucovorin prescribing information for additional information.
-
•For high-dose Methotrexate Injection regimens, follow the supportive care and monitoring instructions below. Also consider for patients receiving intermediate-dose Methotrexate Injection regimens.
-
-Monitor serum creatinine, electrolytes, at baseline and at least daily during therapy
-
-Administer intravenous fluids starting before the first dose and continuing throughout treatment to maintain adequate hydration and urine output
-
-Alkalinize urine starting before the first dose and continuing throughout treatment to maintain a urinary pH of 7 or higher
-
-Monitor methotrexate concentrations at least daily and adjust hydration and leucovorin dosing as needed
-
-
•Administer glucarpidase in patients who have toxic plasma methotrexate concentrations (>1 micromole per liter) and delayed methotrexate clearance due to impaired renal function (refer to the glucarpidase prescribing information for additional information).
Warning: Embryo Fetal Toxicity, Hypersensitivity Reactions, Benzyl Alcohol Toxicity, and Other Serious Adverse Reactions
-
•Methotrexate Injection can cause embryo-fetal toxicity, including fetal death. For non-neoplastic diseases, Methotrexate Injection is contraindicated in pregnancy. Advise females and males of reproductive potential to use effective contraception [see Contraindications (4), Warnings and Precautions (5.1), and Use in Specific Populations (8.1, 8.3)].
-
•Methotrexate Injection is contraindicated in patients with a history of severe hypersensitivity reactions to methotrexate, including anaphylaxis [see Contraindications (4) and Warnings and Precautions (5.2)].
-
•Formulations with benzyl alcohol can cause severe central nervous toxicity or metabolic acidosis. Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required and preservative-free formulations are not available [see Dosage and Administration (2.1) and Warnings and Precautions (5.3)].
-
•Other serious adverse reactions, including death, have been reported with methotrexate. Closely monitor for infections and adverse reactions of the bone marrow, kidneys, liver, nervous system, gastrointestinal tract, lungs, and skin. Withhold or discontinue Methotrexate Injection as appropriate [see Warnings and Precautions (5.4, 5.5, 5.6, 5.7, 5.8, 5.9, 5.10, 5.11)].
Structured Label Content
Section 42229-5 (42229-5)
With Preservative (Multiple-Dose Vial)
-
•Methotrexate Injection formulation containing benzyl alcohol as a preservative may be administered by intramuscular, intravenous, or subcutaneous injection [see Dosage and Administration (2.1)]. Methotrexate Injection with preservative may be further diluted with 0.9% Sodium Chloride Injection, USP. Diluted product should be used within 4 hours when stored at room temperature (20°C to 25°C) or 24 hours under refrigeration (2°C to 8°C).
-
•Visually inspect product for particulate matter and discoloration prior to administration. Discard if particulate matter or discoloration is observed.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 05/2025 | |||
|
Patient Information
|
|||
|
What is the most important information I should know about Methotrexate Injection?
Males with female partners who are able to become pregnant:
Severe allergic reactions. Severe allergic reactions can happen with Methotrexate Injection.
|
|||
|
|
||
|
Decreased blood cell counts. Methotrexate Injection can affect your bone marrow and cause decreased red blood cell counts, white blood cell counts, and platelet counts, and a condition where your bone marrow cannot produce these blood cells (aplastic anemia). These decreased blood cell counts can be severe and may lead to a serious infection, the need for blood transfusions, treatment in a hospital, and can be life-threatening. Your healthcare provider will check your blood cell counts before you start and during treatment with Methotrexate Injection. Your healthcare provider will watch you closely for infections during treatment with Methotrexate Injection. |
|||
|
|
|
|
|
Your healthcare provider may give you medicines to support your blood counts or give you transfusions if needed, and change your dose or stop your treatment with Methotrexate Injection if needed. |
|||
|
|
||
|
Your healthcare provider will closely watch you for signs and symptoms of infection during treatment with Methotrexate Injection. Your healthcare provider may hold or stop your treatment with Methotrexate Injection if you develop a serious infection.
Tell your healthcare provider if you develop any signs or symptoms of liver problems during treatment with Methotrexate Injection, including: |
|||
|
|
||
|
Brain and spinal cord (nervous system) problems. Methotrexate Injection can cause nervous system problems that can be severe and last for a short time or last for a long time. These nervous system problems can get progressively worse, may not get better (possibly irreversible), and can cause death.
Call your healthcare provider right away if you or your child develop any new signs or symptoms of a nervous system problem during treatment with Methotrexate Injection, including: |
|||
|
|
||
|
Severe stomach and intestine (gastrointestinal) problems.
Call your healthcare provider right away if you develop: |
|||
|
|
||
|
Lung problems. Lung problems can happen suddenly (acute) with Methotrexate Injection or they can develop over a long period-of-time (chronic). Lung problems may not get better (possibly irreversible) and can cause death in anyone taking Methotrexate Injection. Your healthcare provider will monitor you for lung problems during treatment with Methotrexate Injection. Your healthcare provider may hold or stop your treatment with Methotrexate Injection, if needed.
Limit sunlight exposure during treatment with Methotrexate Injection. Use a broad-spectrum ultraviolet sunscreen and lip balm with a Sun Protection Factor (SPF) of 30 or greater and wear a hat and protective clothing when you will be exposed to sunlight during treatment with Methotrexate Injection. Call your healthcare provider right away if you develop a new or worsening skin rash during treatment with Methotrexate Injection. See “What are the possible side effects of Methotrexate Injection?” for more information about side effects. |
|||
|
What is Methotrexate Injection?
in adults:
Methotrexate Injection is a prescription medicine used:
|
|||
|
Do not receive Methotrexate Injection if you:
|
|||
|
Before you receive Methotrexate Injection, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking certain medicines can affect the amount of methotrexate in your blood and can increase your risk for serious side effects. |
|||
|
How will I receive or take Methotrexate Injection?
|
|||
|
|
|
|
|
|||
|
If you are receiving Methotrexate Injection to treat your cancer:
If you are receiving Methotrexate Injection for treatment of severe psoriasis, rheumatoid arthritis, or polyarticular juvenile idiopathic arthritis:
In all patients receiving Methotrexate Injection:
|
|||
|
What are the possible side effects of Methotrexate Injection?
The most common side effects of Methotrexate Injection include:
Possible fertility problems (infertility) in males and females. Methotrexate Injection can cause fertility problems in males and females, and cause sperm production to stop in males, and menstrual problems in females. In females, your periods (menstrual cycle) may be irregular or completely stop when you receive Methotrexate Injection. Your periods may or may not return to normal following treatment. It is not known if your fertility will return after treatment. Talk to your healthcare provider about your risk for infertility if this is a concern for you. |
|||
|
General information about the safe and effective use of Methotrexate Injection.
|
|||
|
What are the ingredients in Methotrexate Injection? For more information, go to www.pfizer.com or call 1-800-438-1985. |
Section 43683-2 (43683-2)
|
Warnings and Precautions (5.11) |
05/2025 |
Section 44425-7 (44425-7)
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from light.
After first puncture, store multiple-dose vials at 2°C to 8°C, and use within 30 days.
Methotrexate Injection is a hazardous drug. Follow applicable special handling and disposal procedures.1
15 References (15 REFERENCES)
-
1."OSHA Hazardous Drugs." OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html.
1.10 Psoriasis
Methotrexate Injection is indicated for the treatment of adults with severe psoriasis.
11 Description (11 DESCRIPTION)
Methotrexate is a folate analog metabolic inhibitor with the chemical name of N-[4-[[(2,4-diamino-6-pteridinyl) methyl]methylamino]benzoyl]-L-glutamic acid and a molecular weight of 454.44. The molecular formula is C20H22N8O5 , and the structural formula is shown below:
Methotrexate Injection with preservative is supplied in sterile multiple-dose vials for intravenous, intramuscular, or subcutaneous use.
-
•Each 25 mg/mL, 2 mL vial contains 50 mg methotrexate equivalent to 54.8 mg of methotrexate sodium, 18.8 mg of benzyl alcohol as a preservative and Sodium chloride 5.2 mg. May contain sodium hydroxide and/or hydrochloric acid to adjust the pH to 8.5.
Preservative-free Methotrexate Injection is supplied in sterile single-dose vials for intravenous, intramuscular, subcutaneous, or intrathecal use.
-
•Each 25 mg/mL, 40 mL vial contains 1000 mg methotrexate equivalent to 1096.7 mg of methotrexate sodium, and the following inactive ingredients: Sodium chloride 196 mg. May contain sodium hydroxide and/or hydrochloric acid to adjust pH to 8.5.
1.4 Osteosarcoma
Methotrexate Injection is indicated for the treatment of adults and pediatric patients with osteosarcoma as part of a combination chemotherapy regimen.
5.16 Infertility
Based on published reports, methotrexate can cause impairment of fertility, oligospermia, and menstrual dysfunction. It is not known if the infertility may be reversible in affected patients. Discuss the risk of effects on reproduction with female and male patients of reproductive potential [see Use in Specific Populations (8.3)].
1.5 Breast Cancer
Methotrexate Injection is indicated for the treatment of adults with breast cancer as part of a combination chemotherapy regimen.
5.8 Neurotoxicity
Methotrexate can cause severe acute and chronic neurotoxicity which can be progressive, irreversible, and fatal. Serious neurotoxicity, including generalized and focal seizures, have occurred in pediatric patients [see Use in Specific Populations (8.4)]. Monitor patients for signs of neurotoxicity and withhold or discontinue Methotrexate Injection when appropriate.
8.4 Pediatric Use
The safety and effectiveness of Methotrexate Injection in pediatric patients have been established for ALL, meningeal leukemia prophylaxis and treatment, non-Hodgkin lymphoma, osteosarcoma and in pJIA. Clinical studies evaluating the use of methotrexate in pediatric patients with pJIA demonstrated safety comparable to that observed in adults with RA [see Adverse Reactions (6.1)]. The safety and effectiveness of Methotrexate Injection have not been established in pediatric patients for the treatment of breast cancer, squamous cell carcinoma of the head and neck, gestational trophoblastic neoplasia, rheumatoid arthritis, and psoriasis. Additional risk information is described below.
8.5 Geriatric Use
Clinical studies of methotrexate did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
5.6 Renal Toxicity
Methotrexate can cause renal toxicity including irreversible acute renal failure. Monitor renal function and withhold or discontinue Methotrexate Injection as needed for severe renal toxicity.
For patients receiving high-dose regimens, follow recommendations to decrease the risk of renal injury and mitigate renal toxicity [see Dosage and Administration (2.2)].
Patients with impaired renal function are at increased risk for methotrexate toxicity [see Use in Specific Populations (8.6)].
Consider administration of glucarpidase in patients with toxic plasma methotrexate concentrations (>1 micromole per liter) and delayed clearance due to impaired renal function. [see Dosage and Administration (2.2)].
5.7 Hepatotoxicity
Methotrexate can cause severe and potentially irreversible hepatotoxicity including fibrosis, cirrhosis, and fatal liver failure [see Adverse Reactions (6.1, 6.2)].
In patients with psoriasis, fibrosis or cirrhosis may occur in the absence of symptoms or abnormal liver function tests. In patients with psoriasis, the risk of hepatotoxicity appears to increase with total cumulative dose and generally occurs after receipt of a total cumulative dose of 1.5 g or more.
The safety of Methotrexate Injection in patients with liver disease is unknown. Avoid use of Methotrexate Injection in patients with chronic liver disease, unless benefits clearly outweigh the risks. The risk of hepatotoxicity is increased with heavy alcohol consumption.
Assess liver function prior to initiating Methotrexate Injection and monitor liver function tests during treatment. Withhold or discontinue Methotrexate Injection as appropriate.
4 Contraindications (4 CONTRAINDICATIONS)
Methotrexate Injection is contraindicated in:
-
•Patients with history of severe hypersensitivity to methotrexate [see Warnings and Precautions (5.2)].
-
•Pregnancy in patients with non-neoplastic diseases [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described, or described in greater detail, in other sections:
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
-
•Myelosuppression [see Warnings and Precautions (5.4)]
-
•Serious Infections [see Warnings and Precautions (5.5)]
-
•Renal Toxicity [see Warnings and Precautions (5.6)]
-
•Hepatotoxicity [see Warnings and Precautions (5.7)]
-
•Neurotoxicity [see Warnings and Precautions (5.8)]
-
•Gastrointestinal Toxicity [see Warnings and Precautions (5.9)]
-
•Pulmonary Toxicity [see Warnings and Precautions (5.10)]
-
•Dermatologic Reactions [see Warnings and Precautions (5.11)]
-
•Secondary Malignancies [see Warnings and Precautions (5.13)]
-
•Tumor Lysis Syndrome [see Warnings and Precautions (5.14)]
-
•Increased Risk of Adverse Reactions due to Third-Space Accumulation [see Warnings and Precautions (5.17)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Refer to full prescribing information for drug interactions with Methotrexate Injection. (7)
5.4 Myelosuppression
Methotrexate suppresses hematopoiesis and can cause severe and life-threatening pancytopenia, anemia, aplastic anemia, leukopenia, neutropenia, and thrombocytopenia [see Adverse Reactions (6.1)].
Obtain blood counts at baseline and periodically during treatment. Monitor patients for possible clinical complications of myelosuppression. Provide supportive care and withhold, reduce dose, or discontinue Methotrexate Injection as needed.
8.6 Renal Impairment
Methotrexate elimination is reduced in patients with renal impairment [creatinine clearance (CLcr) less than 90 mL/min, calculated using Cockcroft-Gault] [see Clinical Pharmacology (12.3)]. Patients with renal impairment are at increased risk for methotrexate adverse reactions.
Follow recommendations to promote methotrexate elimination and decrease risk of acute kidney injury and other methotrexate toxicities in patients who are receiving intermediate- or high-dose regimens [see Dosage and Administration (2.2) and Warnings and Precautions (5.6)]. Consider reducing the dose or discontinuing Methotrexate Injection in patients with renal impairment as appropriate.
5.5 Serious Infections
Patients treated with methotrexate are at increased risk for developing life-threatening or fatal bacterial, fungal, or viral infections including opportunistic infections such as Pneumocystis jiroveci pneumonia, invasive fungal infections, hepatitis B reactivation, tuberculosis primary infection or reactivation, and disseminated Herpes zoster and cytomegalovirus infections.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with Methotrexate Injection. Withhold or discontinue Methotrexate Injection in patients who develop serious infections.
8.7 Hepatic Impairment
The pharmacokinetics and safety of methotrexate in patients with hepatic impairment is unknown. Patients with hepatic impairment may be at increased risk for methotrexate adverse reaction based on elimination characteristics of methotrexate [see Clinical Pharmacology (12.3)]. Consider reducing the dose or discontinuing Methotrexate Injection in patients with hepatic impairment as appropriate [see Warnings and Precautions (5.7)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Methotrexate Injection is a folate analog metabolic inhibitor indicated for:
-
•The following neoplastic diseases for the:
-
oTreatment of adult and pediatric patients with acute lymphoblastic leukemia as part of a combination chemotherapy regimen. (1.1)
-
oProphylaxis and treatment of adult and pediatric patients with meningeal leukemia. (1.2)
-
oTreatment of adult and pediatric patients with non-Hodgkin lymphoma. (1.3)
-
oTreatment of adult and pediatric patients with osteosarcoma as part of a combination chemotherapy regimen. (1.4)
-
oTreatment of adults with breast cancer as part of a combination chemotherapy regimen. (1.5)
-
oTreatment of adults with squamous cell carcinoma of the head and neck as a single agent. (1.6)
-
oTreatment of adults with gestational trophoblastic neoplasia as part of a combination chemotherapy regimen. (1.7)
-
-
•Treatment of adults with rheumatoid arthritis (RA). (1.8)
-
•Treatment of pediatric patients with polyarticular juvenile idiopathic arthritis (pJIA). (1.9)
-
•Treatment of adults with severe psoriasis. (1.10)
5.10 Pulmonary Toxicity
Methotrexate-induced pulmonary toxicity including acute or chronic interstitial pneumonitis and irreversible or fatal cases can occur at all dose levels. Monitor patients for signs of pulmonary toxicity and withhold or discontinue Methotrexate Injection as appropriate.
1.3 Non Hodgkin Lymphoma (1.3 Non-Hodgkin Lymphoma)
Methotrexate Injection is indicated for the treatment of adults and pediatric patients with non-Hodgkin lymphoma.
1.8 Rheumatoid Arthritis
Methotrexate Injection is indicated for the treatment of adults with rheumatoid arthritis (RA).
12.1 Mechanism of Action
Methotrexate inhibits dihydrofolic acid reductase. Dihydrofolates must be reduced to tetrahydrofolates by this enzyme before they can be utilized as carriers of one-carbon groups in the synthesis of purine nucleotides and thymidylate. Therefore, methotrexate interferes with DNA synthesis, repair, and cellular replication. Actively proliferating tissues such as malignant cells, bone marrow, fetal cells, buccal and intestinal mucosa, and cells of the urinary bladder are in general more sensitive to this effect of methotrexate.
The mechanism of action in rheumatoid arthritis, pJIA, and in psoriasis is unknown.
5.1 Embryo Fetal Toxicity (5.1 Embryo-Fetal Toxicity)
Based on published reports and its mechanism of action, methotrexate can cause embryo-fetal toxicity, including fetal death when administered to a pregnant woman.
Methotrexate Injection is contraindicated for use in pregnant women with non-neoplastic diseases. Advise pregnant women with neoplastic diseases of the potential risk to a fetus. The preservative benzyl alcohol can cross the placenta; when possible, use the preservative-free formulation when Methotrexate Injection is needed during pregnancy to treat a neoplastic disease [see Warnings and Precautions (5.3)].
Advise females of reproductive potential to use effective contraception during Methotrexate Injection treatment and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during Methotrexate Injection treatment and for 3 months after the last dose [see Contraindications (4) and Use in Specific Populations (8.1, 8.3, 8.4)].
5.14 Tumor Lysis Syndrome
Methotrexate can induce tumor lysis syndrome in patients with rapidly growing tumors. Institute appropriate treatment for prevention and management of tumor lysis syndrome.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Secondary malignancies can occur. (5.13)
-
•Tumor lysis syndrome can occur in patients with rapidly growing tumors. (5.14)
-
•Immunizations and Risks associated with Live Vaccines: Immunizations may be ineffective. Live vaccines are not recommended due to risk of disseminated infection. (5.15)
-
•Infertility: Can cause impairment of fertility, oligospermia, and menstrual dysfunction. (5.16, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Verify pregnancy status in females of reproductive potential before starting Methotrexate Injection. (2.1, 4, 5.1)
-
•Neoplastic diseases: Refer to the prescribing information for disease specific dosing recommendations. Follow guidelines for high-dose regimens. (2.2, 2.3, 2.4, 2.5, 2.6, 2.7, 2.8, 2.9)
-
•RA: Recommended starting dosage of 7.5 mg once weekly intramuscularly; adjust dose to achieve an optimal response. (2.10)
-
•pJIA: Recommended starting dosage of 10 mg/m2 once weekly subcutaneously or intramuscularly; adjust dose to achieve an optimal response. (2.11)
-
•Psoriasis: Recommended dosage of 10 mg to 25 mg once weekly intramuscularly or intravenously; adjust dose to achieve optimal response. Once achieved, reduce to lowest possible dosage. (2.12)
5.11 Dermatologic Reactions
Severe, including fatal, dermatologic reactions, such as toxic epidermal necrolysis, Stevens-Johnson syndrome, exfoliative dermatitis, skin necrosis, and erythema multiforme, can occur with methotrexate [see Adverse Reactions (6.1, 6.2)].
Psoriasis may be aggravated by concomitant exposure to ultraviolet radiation.
Methotrexate can cause radiation recall, photodermatitis (sunburn) reactivation, photosensitivity, and severe sunburn reactions. Advise patients to limit sun exposure while taking Methotrexate Injection. Advise patients when outdoors to wear a hat and protective clothing and use a broad-spectrum Ultraviolet A (UVA)/Ultraviolet B (UVB) sunscreen and lip balm (SPF ≥30) to help protect against sunburn.
Monitor patients for signs of dermatologic toxicity and withhold or permanently discontinue Methotrexate Injection for severe dermatologic adverse reactions.
5.13 Secondary Malignancies
Secondary malignancies can occur at all dose levels of methotrexate. In some cases, lymphoproliferative disease that occurred during therapy with low-dose methotrexate regressed completely following withdrawal of methotrexate. If lymphoproliferative disease occurs, discontinue Methotrexate Injection and institute appropriate treatment if lymphoma does not regress.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: Methotrexate Injection is a clear, yellow solution and is supplied in single-dose vials (preservative-free) and multiple-dose vials (with preservative) in the following strengths:
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of methotrexate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: Aplastic anemia, lymphadenopathy, hypogammaglobulinemia
Cardiovascular disorders: Thromboembolic events (including arterial thrombosis, cerebral thrombosis, deep vein thrombosis, retinal vein thrombosis, thrombophlebitis, and pulmonary embolus), pericarditis, pericardial effusion, hypotension, sudden death
Endocrine: Diabetes
Eye disorders: Optic neuropathy, blurred vision, ocular irritation, conjunctivitis, xerophthalmia
Gastrointestinal disorders: Hemorrhagic enteritis, intestinal perforation, gingivitis, pancreatitis, pharyngitis, hematemesis, melena, gastrointestinal ulceration and bleeding
Hepatobiliary disorders: Acute hepatitis, decreased serum albumin, fibrosis, cirrhosis, liver failure
Immune system disorders: Anaphylaxis, anaphylactoid reactions, vasculitis
Metabolism: Hyperglycemia
Musculoskeletal disorders: Stress fracture, soft tissue necrosis, arthralgia, myalgia, osteoporosis
Nervous system disorders: Headaches, drowsiness, blurred vision, speech impairment (including dysarthria and aphasia), transient cognitive dysfunction, mood alteration, unusual cranial sensations, paresis, encephalopathy, leukoencephalopathy, and convulsions. Also, spinal radiculopathy with intrathecal use
Renal disorders: Severe renal toxicity including renal failure, azotemia, hematuria, proteinuria, cystitis
Reproductive disorders: Defective oogenesis or spermatogenesis, loss of libido, impotence, gynecomastia, menstrual dysfunction
Respiratory disorders: Pulmonary fibrosis, respiratory failure, chronic interstitial obstructive pulmonary disease, pleuritic pain and thickening, alveolitis
Skin disorders: Toxic epidermal necrolysis, Stevens-Johnson syndrome, exfoliative dermatitis, skin necrosis, and erythema multiforme, erythematous rashes, pruritus, alopecia, skin ulceration, accelerated nodulosis, urticaria, pigmentary changes, ecchymosis, telangiectasia, photosensitivity, acne, furunculosis
General disorders and administration site conditions: Injection site necrosis, injection site reaction
5.9 Gastrointestinal Toxicity
Methotrexate can cause diarrhea, vomiting, stomatitis, hemorrhagic enteritis and fatal intestinal perforation [see Adverse Reactions (6.1)]. Patients with peptic ulcer disease or ulcerative colitis are at a greater risk of developing severe gastrointestinal adverse reactions.
Withhold or discontinue Methotrexate Injection for severe gastrointestinal toxicity, and institute appropriate supportive care as needed.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.2 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis, can occur with methotrexate [see Adverse Reactions (6.1)]. If signs or symptoms of anaphylaxis or any other serious hypersensitivity reaction occurs, immediately discontinue Methotrexate Injection and institute appropriate therapy [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials and other studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Commonly reported adverse reactions include ulcerative stomatitis, leukopenia, nausea, and abdominal distress. Other frequently reported adverse reactions are infection, malaise, fatigue, chills, fever, and dizziness.
1.1 Acute Lymphoblastic Leukemia
Methotrexate Injection is indicated for the treatment of adult and pediatric patients with acute lymphoblastic leukemia (ALL) as part of a combination chemotherapy regimen.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.12 Recommended Dosage for Psoriasis
The recommended dosage of Methotrexate Injection is 10 mg to 25 mg intramuscularly or intravenously once weekly until adequate response is achieved.
Adjust the dose gradually to achieve optimal clinical response; do not exceed 25 mg per week. Once optimal clinical response has been achieved, reduce the dosage to the lowest possible dosing regimen.
Administer folic acid or folinic acid to reduce the risk of methotrexate adverse reactions [see Warnings and Precautions (5.12)].
1.7 Gestational Trophoblastic Neoplasia
Methotrexate Injection is indicated for the treatment of adults with gestational trophoblastic neoplasia (GTN) as part of a combination chemotherapy regimen.
2.6 Recommended Dosage for Osteosarcoma
The recommended dosage of Methotrexate Injection is typically 12 g/m2 (maximum 20 g/dose) as an intravenous infusion over 4 hours administered as a component of a combination chemotherapy regimen. Administer leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)]. Subsequent doses may need to be adjusted based on observed peak serum methotrexate concentrations. Dosage and schedule may vary based upon factors such as patient comorbidities, disease state, and prior treatments.
2.7 Recommended Dosage for Breast Cancer
A recommended dosage of Methotrexate Injection is 40 mg/m2 intravenously as a component of a cyclophosphamide- and fluorouracil-based multi-drug regimen.
Principal Display Panel 2 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 2 mL Vial Label)
Sterile
2 mL Multiple-dose Vial
NDC 61703-350-37
Rx only
Methotrexate Injection, USP
50 mg/2 mL
Isotonic Liquid
(25 mg/mL)
Contains preservative Hazardous Drug
For Intravenous, Intramuscular and Subcutaneous Use Only
Principal Display Panel 2 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 2 mL Vial Carton)
VIAL
Hospira
5 x 2 mL Multiple-dose Vial
Sterile
NDC 61703-350-38
Rx only
Methotrexate Injection, USP
Isotonic Liquid
For Intravenous, Intramuscular and
Subcutaneous Use Only
CONTAINS PRESERVATIVE
NOT FOR INTRATHECAL USE
50 mg/ 2 mL
(25 mg/mL)
Hazardous Drug
Principal Display Panel 40 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 40 mL Vial Label)
Sterile
40 mL Single-dose vial. Discard unused portion.
NDC 61703-408-41
Rx only
Methotrexate Injection, USP
1 g/40 mL
(25 mg/mL)
For Intravenous, Intramuscular, Subcutaneous and Intrathecal Use Only
Hazardous Drug
Preservative Free
2.1 Important Dosage and Safety Information
-
•Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required and preservative-free formulations are not available [see Warnings and Precautions (5.3) and Use in Specific Populations (8.4)].
-
•Verify pregnancy status in females of reproductive potential before starting Methotrexate Injection [see Contraindications (4) and Warnings and Precautions (5.1)].
-
•For patients switching between a methotrexate product administered orally and Methotrexate Injection, consider potential differences in bioavailability.
Principal Display Panel 40 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 40 mL Vial Carton)
VIAL
Hospira
1 x 40 mL Vial
Sterile
NDC 61703-408-41
Rx only
Methotrexate
Injection, USP
Preservative Free
1 g/ 40 mL
(25 mg/mL)
For Intravenous, Intramuscular,
Subcutaneous and Intrathecal Use Only
Single-dose vial. Discard unused portion.
Hazardous Drug
2.14 Administration and Handling Information
Methotrexate Injection is a hazardous drug. Follow applicable special handling and disposable procedures.1
1.9 Polyarticular Juvenile Idiopathic Arthritis
Methotrexate Injection is indicated for the treatment of pediatric patients with polyarticular Juvenile Idiopathic Arthritis (pJIA).
2.13 Dosage Modifications for Adverse Reactions
Discontinue Methotrexate Injection for:
-
•Anaphylaxis or other severe hypersensitivity reactions [see Warnings and Precautions (5.2)]
-
•Lymphoproliferative disease [see Warnings and Precautions (5.13)]
Withhold, dose reduce or discontinue Methotrexate Injection as appropriate for:
-
•Myelosuppression [see Warnings and Precautions (5.4)]
Withhold or discontinue Methotrexate Injection as appropriate for:
-
•Serious infections [see Warnings and Precautions (5.5)]
-
•Renal toxicity [see Warnings and Precautions (5.6)]
-
•Hepatotoxicity [see Warnings and Precautions (5.7)]
-
•Neurotoxicity [see Warnings and Precautions (5.8)]
-
•Gastrointestinal toxicity [see Warnings and Precautions (5.9)]
-
•Pulmonary toxicity [see Warnings and Precautions (5.10)]
-
•Dermatologic reactions [see Warnings and Precautions (5.11)]
2.5 Recommended Dosage for Non Hodgkin Lymphoma (2.5 Recommended Dosage for Non-Hodgkin Lymphoma)
The recommended dosage of Methotrexate Injection varies. When used in combination, recommended dosages range from 10 mg/m2 to 8000 mg/m2 intravenously. When used as a single agent, recommended dosages include 8000 mg/m2 intravenously for central nervous system-directed therapy or 5 to 75 mg intravenously for cutaneous forms of non-Hodgkin lymphoma.
As part of a combination chemotherapy regimen, a recommended dosage of Methotrexate Injection is 1000 mg/m2 or 3000 mg/m2 as an intravenous infusion over 24 hours followed by leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)].
For central nervous system-directed therapy, a recommended dosage of Methotrexate Injection is 8000 mg/m2 as an intravenous infusion over 4 hours as a single agent or in combination with immunochemotherapy at doses ranging from 3000 mg/m2 to 8000 mg/m2 followed by leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)].
For intrathecal Methotrexate Injection (preservative-free), the recommended dose is based on age [see Dosage and Administration (2.4)]. The frequency of administration varies based on whether it is being used for treatment or prophylaxis, and other factors.
8.3 Females and Males of Reproductive Potential
Methotrexate can cause malformations and fetal death at doses less than or equal to the recommended clinical doses [see Use in Specific Populations (8.1)].
1.6 Squamous Cell Carcinoma of the Head and Neck
Methotrexate Injection is indicated for the treatment of adults with squamous cell carcinoma of the head and neck as a single agent.
2.10 Recommended Dosage for Rheumatoid Arthritis
The recommended starting dosage of Methotrexate Injection is 7.5 mg once weekly, administered intramuscularly with escalation to achieve optimal response. Dosages of more than 20 mg once weekly result in an increased risk of serious adverse reactions, including myelosuppression.
When responses are observed, the majority occurred between 3 and 6 weeks from initiation of treatment; however, responses have occurred up to 12 weeks after treatment initiation.
Administer folic acid or folinic acid to reduce the risk of methotrexate adverse reactions [see Warnings and Precautions (5.12)].
1.2 Meningeal Leukemia: Prophylaxis and Treatment
Methotrexate Injection is indicated for the prophylaxis and treatment of meningeal leukemia in adult and pediatric patients.
2.3 Recommended Dosage for Acute Lymphoblastic Leukemia
Methotrexate Injection is used as part of a multi-drug regimen. The recommended dosage varies from 10 to 5000 mg/m2 intravenously. For high-dose Methotrexate Injection regimens, use leucovorin rescue in accordance with high-dose methotrexate regimen guidelines [see Dosage and Administration (2.2)]. Lower doses (e.g., 20 to 30 mg/m2 per week) may be used intramuscularly. Individualize the dose and schedule of Methotrexate Injection based on disease state, patient risk category, concurrent drugs used, phase of treatment, and response to treatment.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Methotrexate has been evaluated in a number of animal studies for carcinogenic potential with inconclusive results. There is evidence that methotrexate causes chromosomal damage to animal somatic cells and human bone marrow cells [see Use in Specific Populations (8.1, 8.2, 8.3)].
5.15 Immunization and Risks Associated With Live Vaccines (5.15 Immunization and Risks Associated with Live Vaccines)
Immunization during Methotrexate Injection treatment may be ineffective.
Disseminated infections following administration of live vaccines have been reported.
Update immunizations according to immunization guidelines prior to initiating Methotrexate Injection. Immunization with live vaccines is not recommended during treatment. The interval between live vaccinations and initiation of Methotrexate Injection should be in accordance with current vaccination guidelines for patients on immunosuppressive therapies.
5.19 Risk of Serious Adverse Reactions With Medication Errors (5.19 Risk of Serious Adverse Reactions with Medication Errors)
Serious adverse reactions, including death, have occurred due to medication errors. Most commonly, these errors occurred in patients who were taking methotrexate daily when a weekly dosing regimen was prescribed. Ensure that patients receive the recommended dosage, because medication errors have led to death.
2.9 Recommended Dosage for Gestational Trophoblastic Neoplasia
For patients with low-risk gestational trophoblastic neoplasia (GTN) a recommended dosage for Methotrexate Injection is 30 mg/m2 to 200 mg/m2 or 0.4 mg/kg to1 mg/kg intravenously or intramuscularly.
For patients with high-risk GTN, a recommended dosage for Methotrexate Injection is 300 mg/m2 over 12 hours as an intravenous infusion as a component of a multi-drug regimen.
2.8 Recommended Dosage for Squamous Cell Carcinoma of Head and Neck
The recommended dosage of Methotrexate Injection ranges from 40 to 60 mg/m2 intravenously once weekly.
2.11 Recommended Dosage for Polyarticular Juvenile Idiopathic Arthritis
The recommended starting dosage of Methotrexate Injection is 10 mg/m2 once weekly administered subcutaneously or intramuscularly, with escalation to achieve optimal response. Dosages over 30 mg/m2 per week may result in an increased risk of serious adverse reactions, including myelosuppression. When responses are observed, the majority occurred between 3 and 6 weeks from initiation of treatment; however, responses have occurred up to 12 weeks after treatment initiation.
Administer folic acid or folinic acid to reduce the risk of methotrexate adverse reactions [see Warnings and Precautions (5.12)].
2.4 Recommended Dosage for Meningeal Leukemia: Prophylaxis and Treatment
Use only preservative-free Methotrexate Injection for intrathecal use.
Prior to administration, dilute preservative-free Methotrexate Injection to a concentration of 1 mg/mL in preservative-free 0.9% Sodium Chloride Injection, USP.
The recommended intrathecal dose of Methotrexate Injection (preservative-free) is based on age:
-
•less than 1 year: 6 mg
-
•1 to less than 2 years: 8 mg
-
•2 to less than 3 years: 10 mg
-
•3 to less than 9 years: 12 mg
-
•greater than or equal to 9 years: 12 to15 mg
For treatment of meningeal leukemia, intrathecal methotrexate may be given at intervals of 2 or more days up to twice weekly; however, administration at intervals of less than 1 week may result in increased subacute toxicity. For meningeal leukemia prophylaxis, Methotrexate Injection is administered no more than once weekly.
For patients with Down Syndrome, administer leucovorin rescue with intrathecal Methotrexate Injection.
5.17 Increased Risk of Adverse Reactions Due to Third Space Accumulation (5.17 Increased Risk of Adverse Reactions Due to Third-Space Accumulation)
Methotrexate can exit slowly from third-space accumulations resulting in prolonged terminal plasma half-life and toxicity. Evacuate significant third-space accumulations prior to Methotrexate Injection administration [see Clinical Pharmacology (12.3)].
5.3 Risks of Serious Adverse Reactions Due to Benzyl Alcohol Preservative (5.3 Risks of Serious Adverse Reactions due to Benzyl Alcohol-Preservative)
Formulations with benzyl alcohol can cause severe central nervous toxicity or metabolic acidosis, if used in neonates or low birth weight infants, intrathecally, or in high-dose regimens. Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required, and preservative-free formulations are not available. The preservative benzyl alcohol can cross the placenta; when possible, use the preservative-free formulation when Methotrexate Injection is needed during pregnancy to treat a neoplastic disease [see Use in Specific Populations (8.1)].
5.18 Increased Risk of Soft Tissue and Bone Toxicity With Concomitant Radiotherapy (5.18 Increased Risk of Soft Tissue and Bone Toxicity with Concomitant Radiotherapy)
Concomitant radiation therapy increases the risk of soft tissue necrosis and osteonecrosis associated with methotrexate.
2.2 Recommended Monitoring and Concomitant Therapies for Intermediate and High Dose Regimens (2.2 Recommended Monitoring and Concomitant Therapies for Intermediate- and High-Dose Regimens)
To decrease the risk of severe adverse reactions [see Warnings and Precautions (5)]:
-
•Administer leucovorin rescue in patients receiving Methotrexate Injection doses of 500 mg/m2 or greater (e.g., high-dose).
-
•Consider leucovorin rescue for patients receiving Methotrexate Injection doses between 100 mg/m2 to less than 500 mg/m2 (e.g., intermediate-dose).
Refer to the leucovorin prescribing information for additional information.
-
•For high-dose Methotrexate Injection regimens, follow the supportive care and monitoring instructions below. Also consider for patients receiving intermediate-dose Methotrexate Injection regimens.
-
-Monitor serum creatinine, electrolytes, at baseline and at least daily during therapy
-
-Administer intravenous fluids starting before the first dose and continuing throughout treatment to maintain adequate hydration and urine output
-
-Alkalinize urine starting before the first dose and continuing throughout treatment to maintain a urinary pH of 7 or higher
-
-Monitor methotrexate concentrations at least daily and adjust hydration and leucovorin dosing as needed
-
-
•Administer glucarpidase in patients who have toxic plasma methotrexate concentrations (>1 micromole per liter) and delayed methotrexate clearance due to impaired renal function (refer to the glucarpidase prescribing information for additional information).
Warning: Embryo Fetal Toxicity, Hypersensitivity Reactions, Benzyl Alcohol Toxicity, and Other Serious Adverse Reactions (WARNING: EMBRYO-FETAL TOXICITY, HYPERSENSITIVITY REACTIONS, BENZYL ALCOHOL TOXICITY, and OTHER SERIOUS ADVERSE REACTIONS)
-
•Methotrexate Injection can cause embryo-fetal toxicity, including fetal death. For non-neoplastic diseases, Methotrexate Injection is contraindicated in pregnancy. Advise females and males of reproductive potential to use effective contraception [see Contraindications (4), Warnings and Precautions (5.1), and Use in Specific Populations (8.1, 8.3)].
-
•Methotrexate Injection is contraindicated in patients with a history of severe hypersensitivity reactions to methotrexate, including anaphylaxis [see Contraindications (4) and Warnings and Precautions (5.2)].
-
•Formulations with benzyl alcohol can cause severe central nervous toxicity or metabolic acidosis. Use only preservative-free Methotrexate Injection for treatment of neonates or low birth weight infants and for intrathecal use. Do not use benzyl alcohol-containing formulations for high-dose regimens unless immediate treatment is required and preservative-free formulations are not available [see Dosage and Administration (2.1) and Warnings and Precautions (5.3)].
-
•Other serious adverse reactions, including death, have been reported with methotrexate. Closely monitor for infections and adverse reactions of the bone marrow, kidneys, liver, nervous system, gastrointestinal tract, lungs, and skin. Withhold or discontinue Methotrexate Injection as appropriate [see Warnings and Precautions (5.4, 5.5, 5.6, 5.7, 5.8, 5.9, 5.10, 5.11)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:34.275509 · Updated: 2026-03-14T22:27:35.794501