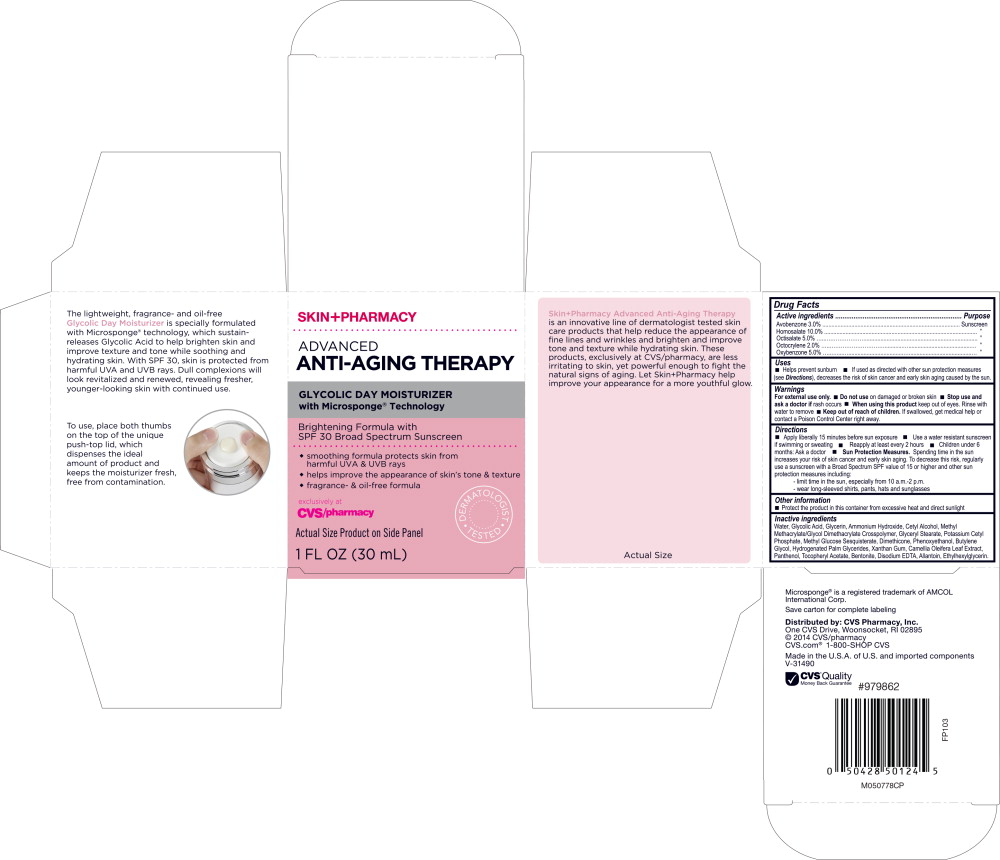
Drug Facts
0d36712d-9b27-4c11-b1e7-a810aae922da
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Avobenzone 3.0% Homosalate 10.0% Octisalate 5.0% Octocrylene 2.0% Oxybenzone 5.0%
Purpose
Sunscreen ” ” ” ”
Medication Information
Purpose
Sunscreen
”
”
”
”
Description
Avobenzone 3.0% Homosalate 10.0% Octisalate 5.0% Octocrylene 2.0% Oxybenzone 5.0%
Uses
- Helps prevent sunburn
- If used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging cause by the sun.
Section 50565-1
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
- Stop use and ask a doctor if rash occurs
Section 50567-7
- When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1
- Do not use on damaged or broken skin
Warnings
For external use only.
Directions
- Apply liberally 15 minutes before sun exposure
- Use a water resistant sunscreen if swimming or sweating
- Reapply at least every 2 hours
- Children under 6 months: Ask a doctor
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
-
-limit time in the sun, especially from 10 a.m.-2 p.m.
-
-wear long-sleeved shirts, pants, hats and sunglasses
-
Other Information
- Protect the product in this container from excessive heat and direct sunlight
Active Ingredients
Avobenzone 3.0%
Homosalate 10.0%
Octisalate 5.0%
Octocrylene 2.0%
Oxybenzone 5.0%
Inactive Ingredients
Water, Glycolic Acid, Glycerin, Ammonium Hydroxide, Cetyl Alcohol, Methyl Methacrylate/Glycol Dimethacrylate Crosspolymer, Glyceryl Stearate, Potassium Cetyl Phosphate, Methyl Glucose Sesquistearate, Dimethicone, Phenoxyethanol, Butylene Glycol, Hydrogenated Palm Glycerides, Xanthan Gum, Camellia Oleifera Leaf Extract, Panthenol, Tocopheryl Acetate, Bentonite, Disodium EDTA, Allantoin, Ethylhexylglycerin.
Microsponge® is a registered trademark of AMCOL International Corp.
Save carton for complete labeling.
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2014 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
Made in the U.S.A. of U.S. and imported components V-31490
CVS ® Quality
Money Back Guarantee
#979862
M050778CP
FP103
Principal Display Panel Jar Label
SKIN+PHARMACY
ADVANCED
ANTI-AGING THERAPY
GLYCOLIC DAY MOISTURIZER
with Microsponge
®
Technology
Brightening Formula with
SPF 30 Broad Spectrum Sunscreen
Principal Display Panel Carton Label
SKIN+PHARMACY
ADVANCED
ANTI-AGING THERAPY
GLYCOLIC DAY MOISTURIZER
with Microsponge
®
Technology
Brightening Formula with
SPF 30 Broad Spectrum Screen
- smoothing formula protects skin from harmful UVA & UVB rays
- helps improve the appearance of skin's tone & texture
- fragrance- & oil-free formula
exclusively at
CVS/pharmacy
DERMATOLOGIST TESTED
Actual Size Product on Side Panel
1 FL OZ (30 mL)
Structured Label Content
Uses
- Helps prevent sunburn
- If used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging cause by the sun.
Section 50565-1 (50565-1)
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
- Stop use and ask a doctor if rash occurs
Section 50567-7 (50567-7)
- When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1 (50570-1)
- Do not use on damaged or broken skin
Purpose
Sunscreen
”
”
”
”
Warnings
For external use only.
Directions
- Apply liberally 15 minutes before sun exposure
- Use a water resistant sunscreen if swimming or sweating
- Reapply at least every 2 hours
- Children under 6 months: Ask a doctor
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
-
-limit time in the sun, especially from 10 a.m.-2 p.m.
-
-wear long-sleeved shirts, pants, hats and sunglasses
-
Other Information (Other information)
- Protect the product in this container from excessive heat and direct sunlight
Active Ingredients (Active ingredients)
Avobenzone 3.0%
Homosalate 10.0%
Octisalate 5.0%
Octocrylene 2.0%
Oxybenzone 5.0%
Inactive Ingredients (Inactive ingredients)
Water, Glycolic Acid, Glycerin, Ammonium Hydroxide, Cetyl Alcohol, Methyl Methacrylate/Glycol Dimethacrylate Crosspolymer, Glyceryl Stearate, Potassium Cetyl Phosphate, Methyl Glucose Sesquistearate, Dimethicone, Phenoxyethanol, Butylene Glycol, Hydrogenated Palm Glycerides, Xanthan Gum, Camellia Oleifera Leaf Extract, Panthenol, Tocopheryl Acetate, Bentonite, Disodium EDTA, Allantoin, Ethylhexylglycerin.
Microsponge® is a registered trademark of AMCOL International Corp.
Save carton for complete labeling.
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2014 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
Made in the U.S.A. of U.S. and imported components V-31490
CVS ® Quality
Money Back Guarantee
#979862
M050778CP
FP103
Principal Display Panel Jar Label (Principal Display Panel - Jar Label)
SKIN+PHARMACY
ADVANCED
ANTI-AGING THERAPY
GLYCOLIC DAY MOISTURIZER
with Microsponge
®
Technology
Brightening Formula with
SPF 30 Broad Spectrum Sunscreen
Principal Display Panel Carton Label (Principal Display Panel - Carton Label)
SKIN+PHARMACY
ADVANCED
ANTI-AGING THERAPY
GLYCOLIC DAY MOISTURIZER
with Microsponge
®
Technology
Brightening Formula with
SPF 30 Broad Spectrum Screen
- smoothing formula protects skin from harmful UVA & UVB rays
- helps improve the appearance of skin's tone & texture
- fragrance- & oil-free formula
exclusively at
CVS/pharmacy
DERMATOLOGIST TESTED
Actual Size Product on Side Panel
1 FL OZ (30 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:17.309937 · Updated: 2026-03-14T22:54:40.661795