These Highlights Do Not Include All The Information Needed To Use Ranolazine Extended-release Tablets Safely And Effectively. See Full Prescribing Information For Ranolazine Extended-release Tablets.
0cf58732-c242-49b0-8e3f-248778b4458d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ranolazine extended-release tablets are indicated for the treatment of chronic angina. Ranolazine extended-release tablets may be used with beta-blockers, nitrates, calcium channel blockers, anti-platelet therapy, lipid-lowering therapy, ACE inhibitors, and angiotensin receptor blockers.
Indications and Usage
Ranolazine extended-release tablets are indicated for the treatment of chronic angina. Ranolazine extended-release tablets may be used with beta-blockers, nitrates, calcium channel blockers, anti-platelet therapy, lipid-lowering therapy, ACE inhibitors, and angiotensin receptor blockers.
Dosage and Administration
500 mg twice daily and increase to 1,000 mg twice daily, based on clinical symptoms ( 2.1 )
Warnings and Precautions
• QT interval prolongation: Can occur with ranolazine. Little data available on high doses, long exposure, use with QT interval-prolonging drugs, potassium channel variants causing prolonged QT interval, in patients with a family history of (or congenital) long QT syndrome, or in patients with known acquired QT interval prolongation. ( 5.1 ) • Renal failure: Monitor renal function after initiation and periodically in patients with moderate to severe renal impairment (CrCL<60 mL/min). If acute renal failure develops, discontinue ranolazine extended-release tablets. ( 5.2 )
Contraindications
Ranolazine is contraindicated in patients: • Taking strong inhibitors of CYP3A [see Drug Interactions (7.1) ] • Taking inducers of CYP3A [see Drug Interactions (7.1) ] • With liver cirrhosis [see Use in Specific Populations (8.6) ]
Adverse Reactions
Most common adverse reactions (>4% and more common than with placebo) are dizziness, headache, constipation, nausea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Glenmark Pharmaceuticals Inc., USA at 1 (888) 721-7115 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
• Moderate CYP3A inhibitors (e.g., diltiazem, verapamil, erythromycin): Limit ranolazine extended-release tablets to 500 mg twice daily. ( 7.1 ) • P-gp inhibitors (e.g., cyclosporine): Ranolazine exposure increased. Titrate ranolazine based on clinical response. ( 7.1 ) • CYP3A substrates: Limit simvastatin to 20 mg when used with ranolazine. Doses of other sensitive CYP3A substrates (e.g., lovastatin) and CYP3A substrates with narrow therapeutic range (e.g., cyclosporine, tacrolimus, sirolimus) may need to be reduced with ranolazine. ( 7.2 ) • OCT2 substrates: Limit the dose of metformin to 1,700 mg daily when used with ranolazine 1000 mg twice daily. Doses of other OCT2 substrates may require adjusted doses. ( 7.2 ) • Drugs transported by P-gp (e.g., digoxin), or drugs metabolized by CYP2D6 (e.g., tricyclic antidepressants) may need reduced doses when used with ranolazine. ( 7.2 )
Storage and Handling
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths: • 500 mg tablets are pink, with “G” on one side and “319” on the other side • 1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side Ranolazine extended-release tablets are available in: Strength NDC Unit-of-Use Bottle (60 Tablets) 500 mg 68462-319-60 Bottle of 500 500 mg 68462-319-05 Unit-of-Use Bottle (60 Tablets) 1000 mg 68462-320-60 Bottle of 500 1000 mg 68462-320-05
How Supplied
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths: • 500 mg tablets are pink, with “G” on one side and “319” on the other side • 1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side Ranolazine extended-release tablets are available in: Strength NDC Unit-of-Use Bottle (60 Tablets) 500 mg 68462-319-60 Bottle of 500 500 mg 68462-319-05 Unit-of-Use Bottle (60 Tablets) 1000 mg 68462-320-60 Bottle of 500 1000 mg 68462-320-05
Medication Information
Warnings and Precautions
• QT interval prolongation: Can occur with ranolazine. Little data available on high doses, long exposure, use with QT interval-prolonging drugs, potassium channel variants causing prolonged QT interval, in patients with a family history of (or congenital) long QT syndrome, or in patients with known acquired QT interval prolongation. ( 5.1 ) • Renal failure: Monitor renal function after initiation and periodically in patients with moderate to severe renal impairment (CrCL<60 mL/min). If acute renal failure develops, discontinue ranolazine extended-release tablets. ( 5.2 )
Indications and Usage
Ranolazine extended-release tablets are indicated for the treatment of chronic angina. Ranolazine extended-release tablets may be used with beta-blockers, nitrates, calcium channel blockers, anti-platelet therapy, lipid-lowering therapy, ACE inhibitors, and angiotensin receptor blockers.
Dosage and Administration
500 mg twice daily and increase to 1,000 mg twice daily, based on clinical symptoms ( 2.1 )
Contraindications
Ranolazine is contraindicated in patients: • Taking strong inhibitors of CYP3A [see Drug Interactions (7.1) ] • Taking inducers of CYP3A [see Drug Interactions (7.1) ] • With liver cirrhosis [see Use in Specific Populations (8.6) ]
Adverse Reactions
Most common adverse reactions (>4% and more common than with placebo) are dizziness, headache, constipation, nausea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Glenmark Pharmaceuticals Inc., USA at 1 (888) 721-7115 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
• Moderate CYP3A inhibitors (e.g., diltiazem, verapamil, erythromycin): Limit ranolazine extended-release tablets to 500 mg twice daily. ( 7.1 ) • P-gp inhibitors (e.g., cyclosporine): Ranolazine exposure increased. Titrate ranolazine based on clinical response. ( 7.1 ) • CYP3A substrates: Limit simvastatin to 20 mg when used with ranolazine. Doses of other sensitive CYP3A substrates (e.g., lovastatin) and CYP3A substrates with narrow therapeutic range (e.g., cyclosporine, tacrolimus, sirolimus) may need to be reduced with ranolazine. ( 7.2 ) • OCT2 substrates: Limit the dose of metformin to 1,700 mg daily when used with ranolazine 1000 mg twice daily. Doses of other OCT2 substrates may require adjusted doses. ( 7.2 ) • Drugs transported by P-gp (e.g., digoxin), or drugs metabolized by CYP2D6 (e.g., tricyclic antidepressants) may need reduced doses when used with ranolazine. ( 7.2 )
Storage and Handling
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths: • 500 mg tablets are pink, with “G” on one side and “319” on the other side • 1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side Ranolazine extended-release tablets are available in: Strength NDC Unit-of-Use Bottle (60 Tablets) 500 mg 68462-319-60 Bottle of 500 500 mg 68462-319-05 Unit-of-Use Bottle (60 Tablets) 1000 mg 68462-320-60 Bottle of 500 1000 mg 68462-320-05
How Supplied
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths: • 500 mg tablets are pink, with “G” on one side and “319” on the other side • 1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side Ranolazine extended-release tablets are available in: Strength NDC Unit-of-Use Bottle (60 Tablets) 500 mg 68462-319-60 Bottle of 500 500 mg 68462-319-05 Unit-of-Use Bottle (60 Tablets) 1000 mg 68462-320-60 Bottle of 500 1000 mg 68462-320-05
Description
Ranolazine extended-release tablets are indicated for the treatment of chronic angina. Ranolazine extended-release tablets may be used with beta-blockers, nitrates, calcium channel blockers, anti-platelet therapy, lipid-lowering therapy, ACE inhibitors, and angiotensin receptor blockers.
Section 42229-5
Laboratory Abnormalities:
Ranolazine produces elevations of serum creatinine by 0.1 mg/dL, regardless of previous renal function, likely because of inhibition of creatinine’s tubular secretion. In general, the elevation has a rapid onset, shows no signs of progression during long-term therapy, is reversible after discontinuation of ranolazine, and is not accompanied by changes in BUN. In healthy volunteers, ranolazine extended-release tablets 1000 mg twice daily had no effect upon the glomerular filtration rate. More marked and progressive increases in serum creatinine, associated with increases in BUN or potassium, indicating acute renal failure, have been reported after initiation of ranolazine in patients with severe renal impairment [see Warning and Precautions (5.2), Use in Specific Population (8.7)].
Section 42230-3
Patient Information
Ranolazine (ra NOE la zeen)
extended-release tablets
Read this Patient Information before you start taking ranolazine extended-release tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment.
What are ranolazine extended-release tablets?
Ranolazine extended-release tablets are a prescription medicine used to treat angina that keeps coming back (chronic angina).
Ranolazine extended-release tablets may be used with other medicines that are used for heart problems and blood pressure control.
It is not known if ranolazine extended-release tablets are safe and effective in children.
Who should not take ranolazine extended-release tablets?
Do not take ranolazine extended-release tablets if:
-
•you take any of the following medicines:
-
ofor fungus infection: ketoconazole (Nizoral®), itraconazole (Sporanox®, Onmel™)
-
ofor infection: clarithromycin (Biaxin®)
-
ofor depression: nefazodone
-
ofor HIV: nelfinavir (Viracept®), ritonavir (Norvir®), lopinavir and ritonavir (Kaletra®), indinavir (Crixivan®), saquinavir (Invirase®)
-
ofor tuberculosis (TB): rifampin (Rifadin®), rifabutin (Mycobutin®), rifapentine (Priftin®)
-
ofor seizures: phenobarbital, phenytoin (Phenytek®, Dilantin®, Dilantin-125®), carbamazepine (Tegretol®)
-
oSt. John's wort (Hypericum perforatum)
-
-
•you have scarring (cirrhosis) of your liver
What should I tell my doctor before taking ranolazine extended-release tablets?
Before you take ranolazine extended-release tablets, tell your doctor if you:
-
•have or have a family history of a heart problem, called ‘QT prolongation’ or ‘long QT syndrome’.
-
•have liver problems.
-
•have kidney problems.
-
•are pregnant or plan to become pregnant. It is not known if ranolazine will harm your unborn baby.
-
•are breastfeeding or plan to breastfeed. It is not known if ranolazine passes into your breast milk. You and your doctor should decide if you will breastfeed.
Tell your doctor about all the medicines you take, including all prescription and nonprescription medicines, vitamins, and herbal supplements. Ranolazine extended-release tablets may affect the way other medicines work and other medicines may affect how ranolazine extended-release tablets work.
Tell your doctor if you take medicines:
-
•for your heart
-
•for cholesterol
-
•for diabetes
-
•for infection
-
•for fungus
-
•for transplant
-
•for nausea and vomiting because of cancer treatments
-
•for mental problems
Know the medicines you take. Keep a list of them to show your doctor or pharmacist when you get a new medicine.
How should I take ranolazine extended-release tablets?
-
•Take ranolazine extended-release tablets exactly as your doctor tells you.
-
•Your doctor will tell you how many ranolazine extended-release tablets to take and when to take them.
-
•Do not change your dose unless your doctor tells you to.
-
•Tell your doctor if you still have symptoms of angina after starting ranolazine extended-release tablets.
-
•Take ranolazine extended-release tablets by mouth, with or without food.
-
•Swallow the ranolazine extended-release tablets whole. Do not crush, break, or chew ranolazine extended-release tablets before swallowing.
-
•If you miss a dose of ranolazine extended-release tablets, wait to take the next dose of ranolazine extended-release tablets at your regular time. Do not make up for the missed dose. Do not take more than 1 dose at a time.
-
•If you take too many ranolazine extended-release tablets, call your doctor, or go to the nearest emergency room right away.
What should I avoid while taking ranolazine extended-release tablets?
-
•Grapefruit and grapefruit juice. Limit products that have grapefruit in them. They can cause your blood levels of ranolazine extended-release tablets to increase.
-
•Ranolazine extended-release tablets can cause dizziness, lightheadedness, or fainting. If you have these symptoms, do not drive a car, use machinery, or do anything that needs you to be alert.
What are the possible side effects of ranolazine extended-release tablets?
Ranolazine extended-release tablets may cause serious side effects, including:
-
•changes in the electrical activity of your heart called QT prolongation. Your doctor may check the electrical activity of your heart with an ECG. Tell your doctor right away if you feel faint, lightheaded, or feel your heart beating irregularly or fast while taking ranolazine extended-release tablets. These may be symptoms related to QT prolongation.
-
•kidney failure in people who already have severe kidney problems. Your doctor may need to do tests to check how your kidneys are working.
The most common side effects of ranolazine extended-release tablets include:
-
•dizziness
-
•headache
-
•constipation
-
•nausea
Tell your doctor if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of ranolazine extended-release tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ranolazine extended-release tablets?
Store ranolazine extended-release tablets at room temperature between 59°F to 86°F (15°C to 30°C).
Keep ranolazine extended-release tablets and all medicines out of the reach of children.
General information about ranolazine extended-release tablets.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not use ranolazine extended-release tablets for a condition for which they were not prescribed. Do not give ranolazine extended-release tablets to other people, even if they have the same condition you have. They may harm them.
The Patient Information summarizes the most important information about ranolazine extended-release tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about ranolazine extended-release tablets that is written for health professionals.
For more information, call Glenmark Pharmaceuticals Inc., USA at 1 (888) 721-7115.
What is chronic angina?
Chronic angina means pain or discomfort in the chest, jaw, shoulder, back, or arm that keeps coming back. There are other possible signs and symptoms of angina including shortness of breath. Angina usually comes on when you are active or under stress. Chronic angina is a symptom of a heart problem called coronary heart disease (CHD), also known as coronary artery disease (CAD). When you have CHD, the blood vessels in your heart become stiff and narrow. Oxygen-rich blood cannot reach your heart muscle easily. Angina comes on when too little oxygen reaches your heart muscle.
What are the ingredients in ranolazine extended-release tablets?
Active ingredient: ranolazine
Inactive ingredients:
500 mg tablet: carmine, copolymer based ethyl acrylate and methacrylic acid, D&C Yellow #10 aluminum lake, FD&C Yellow #6/sunset yellow FCF aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium hydroxide, sodium lauryl sulfate, titanium dioxide and triacetin.
1000 mg tablet: carmine, copolymer based ethyl acrylate and methacrylic acid, FD&C Red #40/allura red ac aluminum lake, FD&C Yellow #6/sunset yellow FCF aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium hydroxide, sodium lauryl sulfate, titanium dioxide and triacetin.
Trademarks are the property of their respective owners.
Distributed by:
Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
Questions? 1 (888) 721-7115
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: August 2025
Section 44425-7
Store ranolazine extended-release tablets at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage
Hypotension, QT prolongation, bradycardia, myoclonic activity, severe tremor, unsteady gait/incoordination, dizziness, nausea, vomiting, dysphasia, and hallucinations have been seen in cases of oral overdose of ranolazine extended-release. In cases of extreme overdose of ranolazine extended-release fatal outcomes have been reported. In clinical studies, high intravenous exposure resulted in diplopia, paresthesia, confusion, and syncope.
In addition to general supportive measures, continuous ECG monitoring may be warranted in the event of overdose.
Since ranolazine is about 62% bound to plasma proteins, hemodialysis is unlikely to be effective in clearing ranolazine.
15 References
M.A. Suckow et al. The anti-ischemia agent ranolazine promotes the development of intestinal tumors in APC (min/+) mice. Cancer Letters 209(2004):165−9.
11 Description
Ranolazine extended-release tablets are available as film-coated, oblong shaped, extended-release tablets for oral administration.
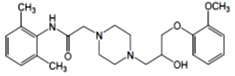
Ranolazine is a racemic mixture, chemically described as 1-piperazineacetamide, N-(2,6-dimethylphenyl)-4-[2-hydroxy-3-(2-methoxyphenoxy)propyl]-, (±)-. It has an empirical formula of C24H33N3O4, a molecular weight of 427.54 g/mol, and the following structural formula:
Ranolazine is a white to off-white powder. Ranolazine is soluble in methanol; sparingly soluble in acetonitrile; slightly soluble in isopropyl alcohol and very slightly soluble in water.
Ranolazine extended-release tablets contain 500 mg or 1000 mg of ranolazine and the following inactive ingredients: carmine, copolymer based ethyl acrylate and methacrylic acid, FD&C Yellow #6/sunset yellow FCF aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium hydroxide, sodium lauryl sulfate, titanium dioxide and triacetin. Additional inactive ingredients for the 500 mg tablet include D&C Yellow #10 aluminum lake; additional inactive ingredients for the 1000 mg tablet include FD&C Red #40/allura red ac aluminum lake.
5.2 Renal Failure
Acute renal failure has been observed in some patients with severe renal impairment (creatinine clearance [CrCL] <30 mL/min) while taking ranolazine. If acute renal failure develops (e.g., marked increase in serum creatinine associated with an increase in blood urea nitrogen [BUN]), discontinue ranolazine and treat appropriately [see Use in Specific Populations (8.7)].
Monitor renal function after initiation and periodically in patients with moderate to severe renal impairment (CrCL <60 mL/min) for increases in serum creatinine accompanied by an increase in BUN.
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
8.5 Geriatric Use
Of the chronic angina patients treated with ranolazine in controlled studies, 496 (48%) were ≥65 years of age, and 114 (11%) were ≥75 years of age. No overall differences in efficacy were observed between older and younger patients. There were no differences in safety for patients ≥65 years compared to younger patients, but patients ≥75 years of age on ranolazine, compared to placebo, had a higher incidence of adverse events, serious adverse events, and drug discontinuations due to adverse events. In general, dose selection for an elderly patient should usually start at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease, or other drug therapy.
4 Contraindications
Ranolazine is contraindicated in patients:
-
•Taking strong inhibitors of CYP3A [see Drug Interactions (7.1)]
-
•Taking inducers of CYP3A [see Drug Interactions (7.1)]
-
•With liver cirrhosis [see Use in Specific Populations (8.6)]
6 Adverse Reactions
Most common adverse reactions (>4% and more common than with placebo) are dizziness, headache, constipation, nausea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Glenmark Pharmaceuticals Inc., USA at 1 (888) 721-7115 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
-
•Moderate CYP3A inhibitors (e.g., diltiazem, verapamil, erythromycin): Limit ranolazine extended-release tablets to 500 mg twice daily. (7.1)
-
•P-gp inhibitors (e.g., cyclosporine): Ranolazine exposure increased. Titrate ranolazine based on clinical response. (7.1)
-
•CYP3A substrates: Limit simvastatin to 20 mg when used with ranolazine. Doses of other sensitive CYP3A substrates (e.g., lovastatin) and CYP3A substrates with narrow therapeutic range (e.g., cyclosporine, tacrolimus, sirolimus) may need to be reduced with ranolazine. (7.2)
-
•OCT2 substrates: Limit the dose of metformin to 1,700 mg daily when used with ranolazine 1000 mg twice daily. Doses of other OCT2 substrates may require adjusted doses. (7.2)
-
•Drugs transported by P-gp (e.g., digoxin), or drugs metabolized by CYP2D6 (e.g., tricyclic antidepressants) may need reduced doses when used with ranolazine. (7.2)
12.3 Pharmacokinetics
Ranolazine is extensively metabolized in the gut and liver and its absorption is highly variable. For example, at a dose of 1000 mg twice daily, the mean steady-state Cmax was 2,600 ng/mL with 95% confidence limits of 400 and 6,100 ng/mL. The pharmacokinetics of the (+) R- and (-) S-enantiomers of ranolazine are similar in healthy volunteers. The apparent terminal half-life of ranolazine is 7 hours. Steady state is generally achieved within 3 days of twice-daily dosing with ranolazine. At steady state over the dose range of 500 to 1000 mg twice daily, Cmax and AUC0–τ increase slightly more than proportionally to dose, 2.2- and 2.4-fold, respectively. With twice-daily dosing, the trough:peak ratio of the ranolazine plasma concentration is 0.3 to 0.6. The pharmacokinetics of ranolazine is unaffected by age, gender, or food.
2.2 Dose Modification
Dose adjustments may be needed when ranolazine extended-release tablets are taken in combination with certain other drugs [see Drug Interactions (7.1)]. Limit the maximum dose of ranolazine extended-release tablets to 500 mg twice daily in patients on moderate CYP3A inhibitors such as diltiazem, verapamil, and erythromycin. Use of ranolazine with strong CYP3A inhibitors is contraindicated [see Contraindications (4), Drug Interactions (7.1)]. Use of P-gp inhibitors, such as cyclosporine, may increase exposure to ranolazine. Titrate ranolazine based on clinical response [see Drug Interactions (7.1)].
2.1 Dosing Information
Initiate ranolazine extended-release tablets dosing at 500 mg twice daily and increase to 1000 mg twice daily, as needed, based on clinical symptoms. Take ranolazine extended-release tablets with or without meals. Swallow ranolazine extended-release tablets whole; do not crush, break, or chew.
The maximum recommended daily dose of ranolazine extended-release tablets is 1000 mg twice daily.
If a dose of ranolazine extended-release tablets is missed, take the prescribed dose at the next scheduled time; do not double the next dose.
1 Indications and Usage
Ranolazine extended-release tablets are indicated for the treatment of chronic angina.
Ranolazine extended-release tablets may be used with beta-blockers, nitrates, calcium channel blockers, anti-platelet therapy, lipid-lowering therapy, ACE inhibitors, and angiotensin receptor blockers.
Principle Display Panel
Ranolazine extended-release tablets, 500 mg
NDC 68462-319-60
60 tablets
12.1 Mechanism of Action
The mechanism of action of ranolazine’s antianginal effects has not been determined. Ranolazine has anti-ischemic and antianginal effects that do not depend upon reductions in heart rate or blood pressure. It does not affect the rate-pressure product, a measure of myocardial work, at maximal exercise. Ranolazine at therapeutic levels can inhibit the cardiac late sodium current (INa). However, the relationship of this inhibition to angina symptoms is uncertain.
The QT prolongation effect of ranolazine on the surface electrocardiogram is the result of inhibition of IKr, which prolongs the ventricular action potential.
14.1 Chronic Stable Angina
CARISA (Combination Assessment of Ranolazine In Stable Angina) was a study in 823 chronic angina patients randomized to receive 12 weeks of treatment with twice-daily ranolazine 750 mg, 1000 mg, or placebo, who also continued on daily doses of atenolol 50 mg, amlodipine 5 mg, or diltiazem CD 180 mg. Sublingual nitrates were used in this study as needed.
In this trial, statistically significant (p <0.05) increases in modified Bruce treadmill exercise duration and time to angina were observed for each ranolazine dose versus placebo, at both trough (12 hours after dosing) and peak (4 hours after dosing) plasma levels, with minimal effects on blood pressure and heart rate. The changes versus placebo in exercise parameters are presented in Table 1. Exercise treadmill results showed no increase in effect on exercise at the 1000 mg dose compared to the 750 mg dose.
| Mean Difference from Placebo (sec) | ||
|---|---|---|
| Study | CARISA (N=791) | |
| Ranolazine Twice-daily Dose | 750 mg | 1000 mg |
|
Exercise Duration |
||
|
Trough |
24a |
24 a |
|
Peak |
34 b |
26 a |
|
Time to Angina |
||
|
Trough |
30 a |
26 a |
|
Peak |
38 b |
38 b |
|
Time to 1 mm ST-Segment Depression |
||
|
Trough |
20 |
21 |
|
Peak |
41 b |
35 b |
a p-value ≤0.05
b p-value ≤0.05
The effects of ranolazine on angina frequency and nitroglycerin use are shown in Table 2.
| Placebo |
Ranolazine Extended-
Release Tablets 750 mg a |
Ranolazine Extended-
Release Tablets 1,000 mg |
||
|---|---|---|---|---|
|
Angina Frequency
|
N |
258 |
272 |
261 |
|
Mean |
3.3 |
2.5 |
2.1 |
|
|
P-value vs placebo |
— |
0.006 |
<0.001 |
|
|
Nitroglycerin Use
|
N |
252 |
262 |
244 |
|
Mean |
3.1 |
2.1 |
1.8 |
|
|
P-value vs placebo |
— |
0.016 |
<0.001 |
a Twice daily
Tolerance to ranolazine did not develop after 12 weeks of therapy. Rebound increases in angina, as measured by exercise duration, have not been observed following abrupt discontinuation of ranolazine extended-release tablets.
Ranolazine has been evaluated in patients with chronic angina who remained symptomatic despite treatment with the maximum dose of an antianginal agent. In the ERICA (Efficacy of Ranolazine In Chronic Angina) trial, 565 patients were randomized to receive an initial dose of ranolazine extended-release tablets 500 mg twice daily or placebo for 1 week, followed by 6 weeks of treatment with ranolazine extended-release tablets 1000 mg twice daily or placebo, in addition to concomitant treatment with amlodipine 10 mg once daily. In addition, 45% of the study population also received long-acting nitrates. Sublingual nitrates were used as needed to treat angina episodes. Results are shown in Table 3. Statistically significant decreases in angina attack frequency (p=0.028) and nitroglycerin use (p=0.014) were observed with ranolazine compared to placebo. These treatment effects appeared consistent across age and use of long-acting nitrates.
| Placebo |
Ranolazine Extended-
Release Tablets a |
||
|---|---|---|---|
|
Angina Frequency
|
N |
281 |
277 |
|
Mean |
4.3 |
3.3 |
|
|
Median |
2.4 |
2.2 |
|
|
Nitroglycerin Use
|
N |
281 |
277 |
|
Mean |
3.6 |
2.7 |
|
|
Median |
1.7 |
1.3 |
a 1,000 mg twice daily
5 Warnings and Precautions
-
•QT interval prolongation: Can occur with ranolazine. Little data available on high doses, long exposure, use with QT interval-prolonging drugs, potassium channel variants causing prolonged QT interval, in patients with a family history of (or congenital) long QT syndrome, or in patients with known acquired QT interval prolongation. (5.1)
-
•Renal failure: Monitor renal function after initiation and periodically in patients with moderate to severe renal impairment (CrCL<60 mL/min). If acute renal failure develops, discontinue ranolazine extended-release tablets. (5.2)
2 Dosage and Administration
500 mg twice daily and increase to 1,000 mg twice daily, based on clinical symptoms (2.1)
3 Dosage Forms and Strengths
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths:
-
•500 mg tablets are pink, with “G” on one side and “319” on the other side
-
•1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side
5.1 Qt Interval Prolongation
Ranolazine blocks IKr and prolongs the QTc interval in a dose-related manner.
Clinical experience in an acute coronary syndrome population did not show an increased risk of proarrhythmia or sudden death [see Clinical Studies (14.2)]. However, there is little experience with high doses (>1,000 mg twice daily) or exposure, other QT-prolonging drugs, potassium channel variants resulting in a long QT interval, in patients with a family history of (or congenital) long QT syndrome, or in patients with known acquired QT interval prolongation.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ranolazine extended-release tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Nervous System Disorders – Abnormal coordination, myoclonus, paresthesia, tremor, and other serious neurologic adverse events have been reported to occur, sometimes concurrently, in patients taking ranolazine. The onset of events was often associated with an increase in ranolazine dose or exposure. Many patients reported symptom resolution following drug discontinuation or dose decrease.
Metabolism and Nutrition Disorders – Cases of hypoglycemia have been reported in diabetic patients on antidiabetic medication.
Psychiatric Disorders – hallucination
Renal and Urinary Disorders – dysuria, urinary retention
Skin and Subcutaneous Tissue Disorders – angioedema, pruritus, rash
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 2,018 patients with chronic angina were treated with ranolazine in controlled clinical trials. Of the patients treated with ranolazine, 1,026 were enrolled in three double-blind, placebo-controlled, randomized studies (CARISA, ERICA, MARISA) of up to 12 weeks’ duration. In addition, upon study completion, 1,251 patients received treatment with ranolazine in open-label, long-term studies; 1,227 patients were exposed to ranolazine for more than 1 year, 613 patients for more than 2 years, 531 patients for more than 3 years, and 326 patients for more than 4 years.
At recommended doses, about 6% of patients discontinued treatment with ranolazine because of an adverse event in controlled studies in angina patients compared to about 3% on placebo. The most common adverse events that led to discontinuation more frequently on ranolazine than placebo were dizziness (1.3% versus 0.1%), nausea (1% versus 0%), asthenia, constipation, and headache (each about 0.5% versus 0%). Doses above 1000 mg twice daily are poorly tolerated.
In controlled clinical trials of angina patients, the most frequently reported treatment-emergent adverse reactions (>4% and more common on ranolazine than on placebo) were dizziness (6.2%), headache (5.5%), constipation (4.5%), and nausea (4.4%). Dizziness may be dose-related. In open-label, long-term treatment studies, a similar adverse reaction profile was observed.
The following additional adverse reactions occurred at an incidence of 0.5 to 4.0% in patients treated with ranolazine and were more frequent than the incidence observed in placebo-treated patients:
Cardiac Disorders – bradycardia, palpitations
Ear and Labyrinth Disorders – tinnitus, vertigo
Eye Disorders – blurred vision
Gastrointestinal Disorders – abdominal pain, dry mouth, vomiting, dyspepsia
General Disorders and Administrative Site Adverse Events – asthenia, peripheral edema
Metabolism and Nutrition Disorders – anorexia
Nervous System Disorders – syncope (vasovagal)
Psychiatric Disorders – confusional state
Renal and Urinary Disorders – hematuria
Respiratory, Thoracic, and Mediastinal Disorders – dyspnea
Skin and Subcutaneous Tissue Disorders – hyperhidrosis
Vascular Disorders – hypotension, orthostatic hypotension
Other (<0.5%) but potentially medically important adverse reactions observed more frequently with ranolazine than placebo treatment in all controlled studies included: angioedema, renal failure, eosinophilia, chromaturia, blood urea increased, hypoesthesia, paresthesia, tremor, pulmonary fibrosis, thrombocytopenia, leukopenia, and pancytopenia.
A large clinical trial in acute coronary syndrome patients was unsuccessful in demonstrating a benefit for ranolazine, but there was no apparent proarrhythmic effect in these high-risk patients [see Clinical Studies (14.2)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Inform patients that ranolazine extended-release tablets will not abate an acute angina episode.
16 How Supplied/storage and Handling
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths:
-
•500 mg tablets are pink, with “G” on one side and “319” on the other side
-
•1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side
Ranolazine extended-release tablets are available in:
| Strength | NDC | |
|---|---|---|
|
Unit-of-Use Bottle (60 Tablets) |
500 mg |
68462-319-60 |
|
Bottle of 500 |
500 mg |
68462-319-05 |
|
Unit-of-Use Bottle (60 Tablets) |
1000 mg |
68462-320-60 |
|
Bottle of 500 |
1000 mg |
68462-320-05 |
8.8 Use in Patients With Heart Failure
Heart failure (NYHA Class I to IV) had no significant effect on ranolazine pharmacokinetics. Ranolazine had minimal effects on heart rate and blood pressure in patients with angina and heart failure NYHA Class I to IV. No dose adjustment of ranolazine is required in patients with heart failure.
8.7 Use in Patients With Renal Impairment
A pharmacokinetic study of ranolazine in subjects with severe renal impairment (CrCL <30 mL/min) was stopped when 2 of 4 subjects developed acute renal failure after receiving ranolazine extended-release tablets 500 mg twice daily for 5 days (lead-in phase) followed by 1000 mg twice a day (1 dose in one subject and 11 doses in the other). Increases in creatinine, BUN, and potassium were observed in 3 subjects during the 500 mg lead-in phase. One subject required hemodialysis, while the other 2 subjects improved upon drug discontinuation [see Warning and precautions (5.2) . ]. Monitor renal function periodically in patients with moderate to severe renal impairment. Discontinue ranolazine extended-release tablets if acute renal failure develops.
In a separate study, Cmax was increased between 40% and 50% in patients with mild, moderate, or severe renal impairment compared to patients with no renal impairment, suggesting a similar increase in exposure in patients with renal failure independent of the degree of impairment. The pharmacokinetics of ranolazine has not been assessed in patients on dialysis.
8.9 Use in Patients With Diabetes Mellitus
A population pharmacokinetic evaluation of data from angina patients and healthy subjects showed no effect of diabetes on ranolazine pharmacokinetics. No dose adjustment is required in patients with diabetes.
Ranolazine produces small reductions in HbA1c in patients with diabetes, the clinical significance of which is unknown. Ranolazine should not be considered a treatment for diabetes.
8.6 Use in Patients With Hepatic Impairment
Ranolazine is contraindicated in patients with liver cirrhosis. In a study of cirrhotic patients, the Cmax of ranolazine was increased 30% in cirrhotic patients with mild (Child-Pugh Class A) hepatic impairment, but increased 80% in cirrhotic patients with moderate (Child-Pugh Class B) hepatic impairment compared to patients without hepatic impairment. This increase was not enough to account for the 3-fold increase in QT prolongation seen in cirrhotic patients with mild to moderate hepatic impairment [see Clinical Pharmacology (12.2)].
14.2 Lack of Benefit in Acute Coronary Syndrome
In a large (n=6560) placebo-controlled trial (MERLIN-TIMI 36) in patients with acute coronary syndrome, there was no benefit shown on outcome measures. However, the study is somewhat reassuring regarding proarrhythmic risks, as ventricular arrhythmias were less common on ranolazine [see Clinical Pharmacology (12.2)], and there was no difference between ranolazine and placebo in the risk of all-cause mortality (relative risk ranolazine:placebo 0.99 with an upper 95% confidence limit of 1.22).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Ranolazine tested negative for genotoxic potential in the following assays: Ames bacterial mutation assay, Saccharomyces assay for mitotic gene conversion, chromosomal aberrations assay in Chinese hamster ovary (CHO) cells, mammalian CHO/HGPRT gene mutation assay, and mouse and rat bone marrow micronucleus assays.
There was no evidence of carcinogenic potential in mice or rats. The highest oral doses used in the carcinogenicity studies were 150 mg/kg/day for 21 months in rats (900 mg/m2/day) and 50 mg/kg/day for 24 months in mice (150 mg/m2/day). These maximally tolerated doses are 0.8 and 0.1 times, respectively, the daily maximum recommended human dose (MRHD) of 2,000 mg on a surface area basis. A published study reported that ranolazine promoted tumor formation and progression to malignancy when given to transgenic APC (min/+) mice at a dose of 30 mg/kg twice daily [see References (15)]. The clinical significance of this finding is unclear.
In male and female rats, oral administration of ranolazine that produced exposures (AUC) approximately 3-fold or 5-fold higher, respectively, than the MRHD had no effect on fertility.
Structured Label Content
Section 42229-5 (42229-5)
Laboratory Abnormalities:
Ranolazine produces elevations of serum creatinine by 0.1 mg/dL, regardless of previous renal function, likely because of inhibition of creatinine’s tubular secretion. In general, the elevation has a rapid onset, shows no signs of progression during long-term therapy, is reversible after discontinuation of ranolazine, and is not accompanied by changes in BUN. In healthy volunteers, ranolazine extended-release tablets 1000 mg twice daily had no effect upon the glomerular filtration rate. More marked and progressive increases in serum creatinine, associated with increases in BUN or potassium, indicating acute renal failure, have been reported after initiation of ranolazine in patients with severe renal impairment [see Warning and Precautions (5.2), Use in Specific Population (8.7)].
Section 42230-3 (42230-3)
Patient Information
Ranolazine (ra NOE la zeen)
extended-release tablets
Read this Patient Information before you start taking ranolazine extended-release tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment.
What are ranolazine extended-release tablets?
Ranolazine extended-release tablets are a prescription medicine used to treat angina that keeps coming back (chronic angina).
Ranolazine extended-release tablets may be used with other medicines that are used for heart problems and blood pressure control.
It is not known if ranolazine extended-release tablets are safe and effective in children.
Who should not take ranolazine extended-release tablets?
Do not take ranolazine extended-release tablets if:
-
•you take any of the following medicines:
-
ofor fungus infection: ketoconazole (Nizoral®), itraconazole (Sporanox®, Onmel™)
-
ofor infection: clarithromycin (Biaxin®)
-
ofor depression: nefazodone
-
ofor HIV: nelfinavir (Viracept®), ritonavir (Norvir®), lopinavir and ritonavir (Kaletra®), indinavir (Crixivan®), saquinavir (Invirase®)
-
ofor tuberculosis (TB): rifampin (Rifadin®), rifabutin (Mycobutin®), rifapentine (Priftin®)
-
ofor seizures: phenobarbital, phenytoin (Phenytek®, Dilantin®, Dilantin-125®), carbamazepine (Tegretol®)
-
oSt. John's wort (Hypericum perforatum)
-
-
•you have scarring (cirrhosis) of your liver
What should I tell my doctor before taking ranolazine extended-release tablets?
Before you take ranolazine extended-release tablets, tell your doctor if you:
-
•have or have a family history of a heart problem, called ‘QT prolongation’ or ‘long QT syndrome’.
-
•have liver problems.
-
•have kidney problems.
-
•are pregnant or plan to become pregnant. It is not known if ranolazine will harm your unborn baby.
-
•are breastfeeding or plan to breastfeed. It is not known if ranolazine passes into your breast milk. You and your doctor should decide if you will breastfeed.
Tell your doctor about all the medicines you take, including all prescription and nonprescription medicines, vitamins, and herbal supplements. Ranolazine extended-release tablets may affect the way other medicines work and other medicines may affect how ranolazine extended-release tablets work.
Tell your doctor if you take medicines:
-
•for your heart
-
•for cholesterol
-
•for diabetes
-
•for infection
-
•for fungus
-
•for transplant
-
•for nausea and vomiting because of cancer treatments
-
•for mental problems
Know the medicines you take. Keep a list of them to show your doctor or pharmacist when you get a new medicine.
How should I take ranolazine extended-release tablets?
-
•Take ranolazine extended-release tablets exactly as your doctor tells you.
-
•Your doctor will tell you how many ranolazine extended-release tablets to take and when to take them.
-
•Do not change your dose unless your doctor tells you to.
-
•Tell your doctor if you still have symptoms of angina after starting ranolazine extended-release tablets.
-
•Take ranolazine extended-release tablets by mouth, with or without food.
-
•Swallow the ranolazine extended-release tablets whole. Do not crush, break, or chew ranolazine extended-release tablets before swallowing.
-
•If you miss a dose of ranolazine extended-release tablets, wait to take the next dose of ranolazine extended-release tablets at your regular time. Do not make up for the missed dose. Do not take more than 1 dose at a time.
-
•If you take too many ranolazine extended-release tablets, call your doctor, or go to the nearest emergency room right away.
What should I avoid while taking ranolazine extended-release tablets?
-
•Grapefruit and grapefruit juice. Limit products that have grapefruit in them. They can cause your blood levels of ranolazine extended-release tablets to increase.
-
•Ranolazine extended-release tablets can cause dizziness, lightheadedness, or fainting. If you have these symptoms, do not drive a car, use machinery, or do anything that needs you to be alert.
What are the possible side effects of ranolazine extended-release tablets?
Ranolazine extended-release tablets may cause serious side effects, including:
-
•changes in the electrical activity of your heart called QT prolongation. Your doctor may check the electrical activity of your heart with an ECG. Tell your doctor right away if you feel faint, lightheaded, or feel your heart beating irregularly or fast while taking ranolazine extended-release tablets. These may be symptoms related to QT prolongation.
-
•kidney failure in people who already have severe kidney problems. Your doctor may need to do tests to check how your kidneys are working.
The most common side effects of ranolazine extended-release tablets include:
-
•dizziness
-
•headache
-
•constipation
-
•nausea
Tell your doctor if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of ranolazine extended-release tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ranolazine extended-release tablets?
Store ranolazine extended-release tablets at room temperature between 59°F to 86°F (15°C to 30°C).
Keep ranolazine extended-release tablets and all medicines out of the reach of children.
General information about ranolazine extended-release tablets.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not use ranolazine extended-release tablets for a condition for which they were not prescribed. Do not give ranolazine extended-release tablets to other people, even if they have the same condition you have. They may harm them.
The Patient Information summarizes the most important information about ranolazine extended-release tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about ranolazine extended-release tablets that is written for health professionals.
For more information, call Glenmark Pharmaceuticals Inc., USA at 1 (888) 721-7115.
What is chronic angina?
Chronic angina means pain or discomfort in the chest, jaw, shoulder, back, or arm that keeps coming back. There are other possible signs and symptoms of angina including shortness of breath. Angina usually comes on when you are active or under stress. Chronic angina is a symptom of a heart problem called coronary heart disease (CHD), also known as coronary artery disease (CAD). When you have CHD, the blood vessels in your heart become stiff and narrow. Oxygen-rich blood cannot reach your heart muscle easily. Angina comes on when too little oxygen reaches your heart muscle.
What are the ingredients in ranolazine extended-release tablets?
Active ingredient: ranolazine
Inactive ingredients:
500 mg tablet: carmine, copolymer based ethyl acrylate and methacrylic acid, D&C Yellow #10 aluminum lake, FD&C Yellow #6/sunset yellow FCF aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium hydroxide, sodium lauryl sulfate, titanium dioxide and triacetin.
1000 mg tablet: carmine, copolymer based ethyl acrylate and methacrylic acid, FD&C Red #40/allura red ac aluminum lake, FD&C Yellow #6/sunset yellow FCF aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium hydroxide, sodium lauryl sulfate, titanium dioxide and triacetin.
Trademarks are the property of their respective owners.
Distributed by:
Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
Questions? 1 (888) 721-7115
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: August 2025
Section 44425-7 (44425-7)
Store ranolazine extended-release tablets at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Hypotension, QT prolongation, bradycardia, myoclonic activity, severe tremor, unsteady gait/incoordination, dizziness, nausea, vomiting, dysphasia, and hallucinations have been seen in cases of oral overdose of ranolazine extended-release. In cases of extreme overdose of ranolazine extended-release fatal outcomes have been reported. In clinical studies, high intravenous exposure resulted in diplopia, paresthesia, confusion, and syncope.
In addition to general supportive measures, continuous ECG monitoring may be warranted in the event of overdose.
Since ranolazine is about 62% bound to plasma proteins, hemodialysis is unlikely to be effective in clearing ranolazine.
15 References (15 REFERENCES)
M.A. Suckow et al. The anti-ischemia agent ranolazine promotes the development of intestinal tumors in APC (min/+) mice. Cancer Letters 209(2004):165−9.
11 Description (11 DESCRIPTION)
Ranolazine extended-release tablets are available as film-coated, oblong shaped, extended-release tablets for oral administration.
Ranolazine is a racemic mixture, chemically described as 1-piperazineacetamide, N-(2,6-dimethylphenyl)-4-[2-hydroxy-3-(2-methoxyphenoxy)propyl]-, (±)-. It has an empirical formula of C24H33N3O4, a molecular weight of 427.54 g/mol, and the following structural formula:
Ranolazine is a white to off-white powder. Ranolazine is soluble in methanol; sparingly soluble in acetonitrile; slightly soluble in isopropyl alcohol and very slightly soluble in water.
Ranolazine extended-release tablets contain 500 mg or 1000 mg of ranolazine and the following inactive ingredients: carmine, copolymer based ethyl acrylate and methacrylic acid, FD&C Yellow #6/sunset yellow FCF aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium hydroxide, sodium lauryl sulfate, titanium dioxide and triacetin. Additional inactive ingredients for the 500 mg tablet include D&C Yellow #10 aluminum lake; additional inactive ingredients for the 1000 mg tablet include FD&C Red #40/allura red ac aluminum lake.
5.2 Renal Failure
Acute renal failure has been observed in some patients with severe renal impairment (creatinine clearance [CrCL] <30 mL/min) while taking ranolazine. If acute renal failure develops (e.g., marked increase in serum creatinine associated with an increase in blood urea nitrogen [BUN]), discontinue ranolazine and treat appropriately [see Use in Specific Populations (8.7)].
Monitor renal function after initiation and periodically in patients with moderate to severe renal impairment (CrCL <60 mL/min) for increases in serum creatinine accompanied by an increase in BUN.
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
8.5 Geriatric Use
Of the chronic angina patients treated with ranolazine in controlled studies, 496 (48%) were ≥65 years of age, and 114 (11%) were ≥75 years of age. No overall differences in efficacy were observed between older and younger patients. There were no differences in safety for patients ≥65 years compared to younger patients, but patients ≥75 years of age on ranolazine, compared to placebo, had a higher incidence of adverse events, serious adverse events, and drug discontinuations due to adverse events. In general, dose selection for an elderly patient should usually start at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease, or other drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
Ranolazine is contraindicated in patients:
-
•Taking strong inhibitors of CYP3A [see Drug Interactions (7.1)]
-
•Taking inducers of CYP3A [see Drug Interactions (7.1)]
-
•With liver cirrhosis [see Use in Specific Populations (8.6)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (>4% and more common than with placebo) are dizziness, headache, constipation, nausea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Glenmark Pharmaceuticals Inc., USA at 1 (888) 721-7115 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
-
•Moderate CYP3A inhibitors (e.g., diltiazem, verapamil, erythromycin): Limit ranolazine extended-release tablets to 500 mg twice daily. (7.1)
-
•P-gp inhibitors (e.g., cyclosporine): Ranolazine exposure increased. Titrate ranolazine based on clinical response. (7.1)
-
•CYP3A substrates: Limit simvastatin to 20 mg when used with ranolazine. Doses of other sensitive CYP3A substrates (e.g., lovastatin) and CYP3A substrates with narrow therapeutic range (e.g., cyclosporine, tacrolimus, sirolimus) may need to be reduced with ranolazine. (7.2)
-
•OCT2 substrates: Limit the dose of metformin to 1,700 mg daily when used with ranolazine 1000 mg twice daily. Doses of other OCT2 substrates may require adjusted doses. (7.2)
-
•Drugs transported by P-gp (e.g., digoxin), or drugs metabolized by CYP2D6 (e.g., tricyclic antidepressants) may need reduced doses when used with ranolazine. (7.2)
12.3 Pharmacokinetics
Ranolazine is extensively metabolized in the gut and liver and its absorption is highly variable. For example, at a dose of 1000 mg twice daily, the mean steady-state Cmax was 2,600 ng/mL with 95% confidence limits of 400 and 6,100 ng/mL. The pharmacokinetics of the (+) R- and (-) S-enantiomers of ranolazine are similar in healthy volunteers. The apparent terminal half-life of ranolazine is 7 hours. Steady state is generally achieved within 3 days of twice-daily dosing with ranolazine. At steady state over the dose range of 500 to 1000 mg twice daily, Cmax and AUC0–τ increase slightly more than proportionally to dose, 2.2- and 2.4-fold, respectively. With twice-daily dosing, the trough:peak ratio of the ranolazine plasma concentration is 0.3 to 0.6. The pharmacokinetics of ranolazine is unaffected by age, gender, or food.
2.2 Dose Modification
Dose adjustments may be needed when ranolazine extended-release tablets are taken in combination with certain other drugs [see Drug Interactions (7.1)]. Limit the maximum dose of ranolazine extended-release tablets to 500 mg twice daily in patients on moderate CYP3A inhibitors such as diltiazem, verapamil, and erythromycin. Use of ranolazine with strong CYP3A inhibitors is contraindicated [see Contraindications (4), Drug Interactions (7.1)]. Use of P-gp inhibitors, such as cyclosporine, may increase exposure to ranolazine. Titrate ranolazine based on clinical response [see Drug Interactions (7.1)].
2.1 Dosing Information
Initiate ranolazine extended-release tablets dosing at 500 mg twice daily and increase to 1000 mg twice daily, as needed, based on clinical symptoms. Take ranolazine extended-release tablets with or without meals. Swallow ranolazine extended-release tablets whole; do not crush, break, or chew.
The maximum recommended daily dose of ranolazine extended-release tablets is 1000 mg twice daily.
If a dose of ranolazine extended-release tablets is missed, take the prescribed dose at the next scheduled time; do not double the next dose.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Ranolazine extended-release tablets are indicated for the treatment of chronic angina.
Ranolazine extended-release tablets may be used with beta-blockers, nitrates, calcium channel blockers, anti-platelet therapy, lipid-lowering therapy, ACE inhibitors, and angiotensin receptor blockers.
Principle Display Panel
Ranolazine extended-release tablets, 500 mg
NDC 68462-319-60
60 tablets
12.1 Mechanism of Action
The mechanism of action of ranolazine’s antianginal effects has not been determined. Ranolazine has anti-ischemic and antianginal effects that do not depend upon reductions in heart rate or blood pressure. It does not affect the rate-pressure product, a measure of myocardial work, at maximal exercise. Ranolazine at therapeutic levels can inhibit the cardiac late sodium current (INa). However, the relationship of this inhibition to angina symptoms is uncertain.
The QT prolongation effect of ranolazine on the surface electrocardiogram is the result of inhibition of IKr, which prolongs the ventricular action potential.
14.1 Chronic Stable Angina
CARISA (Combination Assessment of Ranolazine In Stable Angina) was a study in 823 chronic angina patients randomized to receive 12 weeks of treatment with twice-daily ranolazine 750 mg, 1000 mg, or placebo, who also continued on daily doses of atenolol 50 mg, amlodipine 5 mg, or diltiazem CD 180 mg. Sublingual nitrates were used in this study as needed.
In this trial, statistically significant (p <0.05) increases in modified Bruce treadmill exercise duration and time to angina were observed for each ranolazine dose versus placebo, at both trough (12 hours after dosing) and peak (4 hours after dosing) plasma levels, with minimal effects on blood pressure and heart rate. The changes versus placebo in exercise parameters are presented in Table 1. Exercise treadmill results showed no increase in effect on exercise at the 1000 mg dose compared to the 750 mg dose.
| Mean Difference from Placebo (sec) | ||
|---|---|---|
| Study | CARISA (N=791) | |
| Ranolazine Twice-daily Dose | 750 mg | 1000 mg |
|
Exercise Duration |
||
|
Trough |
24a |
24 a |
|
Peak |
34 b |
26 a |
|
Time to Angina |
||
|
Trough |
30 a |
26 a |
|
Peak |
38 b |
38 b |
|
Time to 1 mm ST-Segment Depression |
||
|
Trough |
20 |
21 |
|
Peak |
41 b |
35 b |
a p-value ≤0.05
b p-value ≤0.05
The effects of ranolazine on angina frequency and nitroglycerin use are shown in Table 2.
| Placebo |
Ranolazine Extended-
Release Tablets 750 mg a |
Ranolazine Extended-
Release Tablets 1,000 mg |
||
|---|---|---|---|---|
|
Angina Frequency
|
N |
258 |
272 |
261 |
|
Mean |
3.3 |
2.5 |
2.1 |
|
|
P-value vs placebo |
— |
0.006 |
<0.001 |
|
|
Nitroglycerin Use
|
N |
252 |
262 |
244 |
|
Mean |
3.1 |
2.1 |
1.8 |
|
|
P-value vs placebo |
— |
0.016 |
<0.001 |
a Twice daily
Tolerance to ranolazine did not develop after 12 weeks of therapy. Rebound increases in angina, as measured by exercise duration, have not been observed following abrupt discontinuation of ranolazine extended-release tablets.
Ranolazine has been evaluated in patients with chronic angina who remained symptomatic despite treatment with the maximum dose of an antianginal agent. In the ERICA (Efficacy of Ranolazine In Chronic Angina) trial, 565 patients were randomized to receive an initial dose of ranolazine extended-release tablets 500 mg twice daily or placebo for 1 week, followed by 6 weeks of treatment with ranolazine extended-release tablets 1000 mg twice daily or placebo, in addition to concomitant treatment with amlodipine 10 mg once daily. In addition, 45% of the study population also received long-acting nitrates. Sublingual nitrates were used as needed to treat angina episodes. Results are shown in Table 3. Statistically significant decreases in angina attack frequency (p=0.028) and nitroglycerin use (p=0.014) were observed with ranolazine compared to placebo. These treatment effects appeared consistent across age and use of long-acting nitrates.
| Placebo |
Ranolazine Extended-
Release Tablets a |
||
|---|---|---|---|
|
Angina Frequency
|
N |
281 |
277 |
|
Mean |
4.3 |
3.3 |
|
|
Median |
2.4 |
2.2 |
|
|
Nitroglycerin Use
|
N |
281 |
277 |
|
Mean |
3.6 |
2.7 |
|
|
Median |
1.7 |
1.3 |
a 1,000 mg twice daily
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•QT interval prolongation: Can occur with ranolazine. Little data available on high doses, long exposure, use with QT interval-prolonging drugs, potassium channel variants causing prolonged QT interval, in patients with a family history of (or congenital) long QT syndrome, or in patients with known acquired QT interval prolongation. (5.1)
-
•Renal failure: Monitor renal function after initiation and periodically in patients with moderate to severe renal impairment (CrCL<60 mL/min). If acute renal failure develops, discontinue ranolazine extended-release tablets. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
500 mg twice daily and increase to 1,000 mg twice daily, based on clinical symptoms (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths:
-
•500 mg tablets are pink, with “G” on one side and “319” on the other side
-
•1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side
5.1 Qt Interval Prolongation (5.1 QT Interval Prolongation)
Ranolazine blocks IKr and prolongs the QTc interval in a dose-related manner.
Clinical experience in an acute coronary syndrome population did not show an increased risk of proarrhythmia or sudden death [see Clinical Studies (14.2)]. However, there is little experience with high doses (>1,000 mg twice daily) or exposure, other QT-prolonging drugs, potassium channel variants resulting in a long QT interval, in patients with a family history of (or congenital) long QT syndrome, or in patients with known acquired QT interval prolongation.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ranolazine extended-release tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Nervous System Disorders – Abnormal coordination, myoclonus, paresthesia, tremor, and other serious neurologic adverse events have been reported to occur, sometimes concurrently, in patients taking ranolazine. The onset of events was often associated with an increase in ranolazine dose or exposure. Many patients reported symptom resolution following drug discontinuation or dose decrease.
Metabolism and Nutrition Disorders – Cases of hypoglycemia have been reported in diabetic patients on antidiabetic medication.
Psychiatric Disorders – hallucination
Renal and Urinary Disorders – dysuria, urinary retention
Skin and Subcutaneous Tissue Disorders – angioedema, pruritus, rash
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 2,018 patients with chronic angina were treated with ranolazine in controlled clinical trials. Of the patients treated with ranolazine, 1,026 were enrolled in three double-blind, placebo-controlled, randomized studies (CARISA, ERICA, MARISA) of up to 12 weeks’ duration. In addition, upon study completion, 1,251 patients received treatment with ranolazine in open-label, long-term studies; 1,227 patients were exposed to ranolazine for more than 1 year, 613 patients for more than 2 years, 531 patients for more than 3 years, and 326 patients for more than 4 years.
At recommended doses, about 6% of patients discontinued treatment with ranolazine because of an adverse event in controlled studies in angina patients compared to about 3% on placebo. The most common adverse events that led to discontinuation more frequently on ranolazine than placebo were dizziness (1.3% versus 0.1%), nausea (1% versus 0%), asthenia, constipation, and headache (each about 0.5% versus 0%). Doses above 1000 mg twice daily are poorly tolerated.
In controlled clinical trials of angina patients, the most frequently reported treatment-emergent adverse reactions (>4% and more common on ranolazine than on placebo) were dizziness (6.2%), headache (5.5%), constipation (4.5%), and nausea (4.4%). Dizziness may be dose-related. In open-label, long-term treatment studies, a similar adverse reaction profile was observed.
The following additional adverse reactions occurred at an incidence of 0.5 to 4.0% in patients treated with ranolazine and were more frequent than the incidence observed in placebo-treated patients:
Cardiac Disorders – bradycardia, palpitations
Ear and Labyrinth Disorders – tinnitus, vertigo
Eye Disorders – blurred vision
Gastrointestinal Disorders – abdominal pain, dry mouth, vomiting, dyspepsia
General Disorders and Administrative Site Adverse Events – asthenia, peripheral edema
Metabolism and Nutrition Disorders – anorexia
Nervous System Disorders – syncope (vasovagal)
Psychiatric Disorders – confusional state
Renal and Urinary Disorders – hematuria
Respiratory, Thoracic, and Mediastinal Disorders – dyspnea
Skin and Subcutaneous Tissue Disorders – hyperhidrosis
Vascular Disorders – hypotension, orthostatic hypotension
Other (<0.5%) but potentially medically important adverse reactions observed more frequently with ranolazine than placebo treatment in all controlled studies included: angioedema, renal failure, eosinophilia, chromaturia, blood urea increased, hypoesthesia, paresthesia, tremor, pulmonary fibrosis, thrombocytopenia, leukopenia, and pancytopenia.
A large clinical trial in acute coronary syndrome patients was unsuccessful in demonstrating a benefit for ranolazine, but there was no apparent proarrhythmic effect in these high-risk patients [see Clinical Studies (14.2)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Inform patients that ranolazine extended-release tablets will not abate an acute angina episode.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Ranolazine extended-release tablets are supplied as film-coated, oblong shaped, extended-release tablets in the following strengths:
-
•500 mg tablets are pink, with “G” on one side and “319” on the other side
-
•1000 mg tablets are reddish brown, with “G” on one side and “320” on the other side
Ranolazine extended-release tablets are available in:
| Strength | NDC | |
|---|---|---|
|
Unit-of-Use Bottle (60 Tablets) |
500 mg |
68462-319-60 |
|
Bottle of 500 |
500 mg |
68462-319-05 |
|
Unit-of-Use Bottle (60 Tablets) |
1000 mg |
68462-320-60 |
|
Bottle of 500 |
1000 mg |
68462-320-05 |
8.8 Use in Patients With Heart Failure (8.8 Use in Patients with Heart Failure)
Heart failure (NYHA Class I to IV) had no significant effect on ranolazine pharmacokinetics. Ranolazine had minimal effects on heart rate and blood pressure in patients with angina and heart failure NYHA Class I to IV. No dose adjustment of ranolazine is required in patients with heart failure.
8.7 Use in Patients With Renal Impairment (8.7 Use in Patients with Renal Impairment)
A pharmacokinetic study of ranolazine in subjects with severe renal impairment (CrCL <30 mL/min) was stopped when 2 of 4 subjects developed acute renal failure after receiving ranolazine extended-release tablets 500 mg twice daily for 5 days (lead-in phase) followed by 1000 mg twice a day (1 dose in one subject and 11 doses in the other). Increases in creatinine, BUN, and potassium were observed in 3 subjects during the 500 mg lead-in phase. One subject required hemodialysis, while the other 2 subjects improved upon drug discontinuation [see Warning and precautions (5.2) . ]. Monitor renal function periodically in patients with moderate to severe renal impairment. Discontinue ranolazine extended-release tablets if acute renal failure develops.
In a separate study, Cmax was increased between 40% and 50% in patients with mild, moderate, or severe renal impairment compared to patients with no renal impairment, suggesting a similar increase in exposure in patients with renal failure independent of the degree of impairment. The pharmacokinetics of ranolazine has not been assessed in patients on dialysis.
8.9 Use in Patients With Diabetes Mellitus (8.9 Use in Patients with Diabetes Mellitus)
A population pharmacokinetic evaluation of data from angina patients and healthy subjects showed no effect of diabetes on ranolazine pharmacokinetics. No dose adjustment is required in patients with diabetes.
Ranolazine produces small reductions in HbA1c in patients with diabetes, the clinical significance of which is unknown. Ranolazine should not be considered a treatment for diabetes.
8.6 Use in Patients With Hepatic Impairment (8.6 Use in Patients with Hepatic Impairment)
Ranolazine is contraindicated in patients with liver cirrhosis. In a study of cirrhotic patients, the Cmax of ranolazine was increased 30% in cirrhotic patients with mild (Child-Pugh Class A) hepatic impairment, but increased 80% in cirrhotic patients with moderate (Child-Pugh Class B) hepatic impairment compared to patients without hepatic impairment. This increase was not enough to account for the 3-fold increase in QT prolongation seen in cirrhotic patients with mild to moderate hepatic impairment [see Clinical Pharmacology (12.2)].
14.2 Lack of Benefit in Acute Coronary Syndrome
In a large (n=6560) placebo-controlled trial (MERLIN-TIMI 36) in patients with acute coronary syndrome, there was no benefit shown on outcome measures. However, the study is somewhat reassuring regarding proarrhythmic risks, as ventricular arrhythmias were less common on ranolazine [see Clinical Pharmacology (12.2)], and there was no difference between ranolazine and placebo in the risk of all-cause mortality (relative risk ranolazine:placebo 0.99 with an upper 95% confidence limit of 1.22).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Ranolazine tested negative for genotoxic potential in the following assays: Ames bacterial mutation assay, Saccharomyces assay for mitotic gene conversion, chromosomal aberrations assay in Chinese hamster ovary (CHO) cells, mammalian CHO/HGPRT gene mutation assay, and mouse and rat bone marrow micronucleus assays.
There was no evidence of carcinogenic potential in mice or rats. The highest oral doses used in the carcinogenicity studies were 150 mg/kg/day for 21 months in rats (900 mg/m2/day) and 50 mg/kg/day for 24 months in mice (150 mg/m2/day). These maximally tolerated doses are 0.8 and 0.1 times, respectively, the daily maximum recommended human dose (MRHD) of 2,000 mg on a surface area basis. A published study reported that ranolazine promoted tumor formation and progression to malignancy when given to transgenic APC (min/+) mice at a dose of 30 mg/kg twice daily [see References (15)]. The clinical significance of this finding is unclear.
In male and female rats, oral administration of ranolazine that produced exposures (AUC) approximately 3-fold or 5-fold higher, respectively, than the MRHD had no effect on fertility.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:37.159631 · Updated: 2026-03-14T22:44:06.575433