Hte - 1019 - 2025-0520
0ce0cb28-5ea1-43e5-8da1-02128b9989a5
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
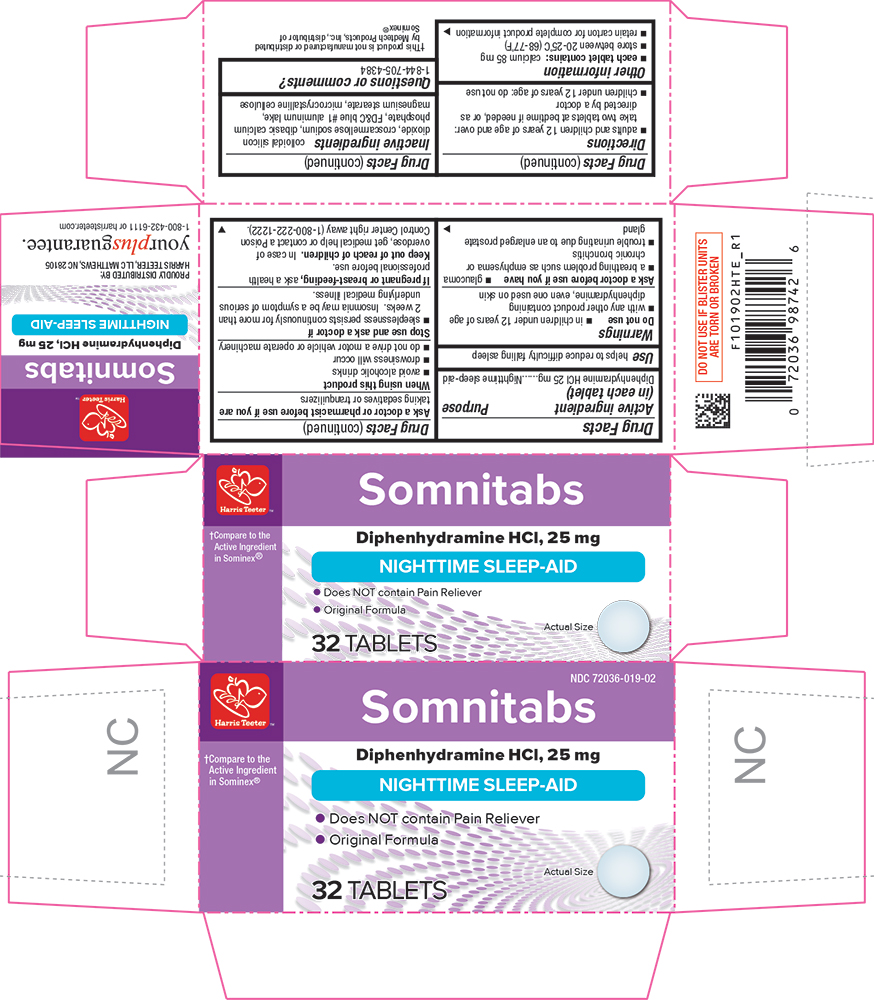
Purpose
Nighttime sleep-aid
Medication Information
Purpose
Nighttime sleep-aid
Description
Drug Facts
Use
helps to reduce difficulty falling asleep
Section 42229-5
Drug Facts
Section 50566-9
Stop use and ask a doctor ifsleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of serious underlying medical illness.
Section 50567-7
When using this productavoid alcoholic drinks
Section 50568-5
Ask a doctor or pharmacist before use if you aretaking sedatives or tranquilizers
Section 53414-9
If pregnant or breast-feeding,ask a health professional before use.
Directions
- adults and children 12 years of age and over: take 2 tablets at bedtime if needed, or as directed by a doctor
Do Not Use
- in children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Other Information
- each tablet contains: calcium 85 mg
- store at room temperature 15°-30°C (59°-86°F)
- retain carton for complete product information
Inactive Ingredients
colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate, FD&C blue #1, magnesium stearate, microcrystalline cellulose
Questions Or Comments?
1-844-705-4384
Principal Display Panel
Harris Teeter™
Somnitabs
Compare to the Active Ingredient in Sominex®
Diphenhydramine HCl, 25 mg
NIGHTTIME SLEEP-AID
•Does NOT contain Pain Reliever
• Original Formula
32 TABLETS
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet)
Diphenhydramine HCl 25 mg
Ask A Doctor Before Use If You Have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Structured Label Content
Use
helps to reduce difficulty falling asleep
Section 42229-5 (42229-5)
Drug Facts
Section 50566-9 (50566-9)
Stop use and ask a doctor ifsleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of serious underlying medical illness.
Section 50567-7 (50567-7)
When using this productavoid alcoholic drinks
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you aretaking sedatives or tranquilizers
Section 53414-9 (53414-9)
If pregnant or breast-feeding,ask a health professional before use.
Purpose
Nighttime sleep-aid
Directions
- adults and children 12 years of age and over: take 2 tablets at bedtime if needed, or as directed by a doctor
Do Not Use (Do not use)
- in children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Other Information (Other information)
- each tablet contains: calcium 85 mg
- store at room temperature 15°-30°C (59°-86°F)
- retain carton for complete product information
Inactive Ingredients (Inactive ingredients)
colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate, FD&C blue #1, magnesium stearate, microcrystalline cellulose
Questions Or Comments? (Questions or comments?)
1-844-705-4384
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Harris Teeter™
Somnitabs
Compare to the Active Ingredient in Sominex®
Diphenhydramine HCl, 25 mg
NIGHTTIME SLEEP-AID
•Does NOT contain Pain Reliever
• Original Formula
32 TABLETS
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Diphenhydramine HCl 25 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:09.360491 · Updated: 2026-03-14T23:08:46.104923