73288-004
0c901f64-6890-45c0-9ca7-ee9de87903c4
34390-5
HUMAN OTC DRUG LABEL
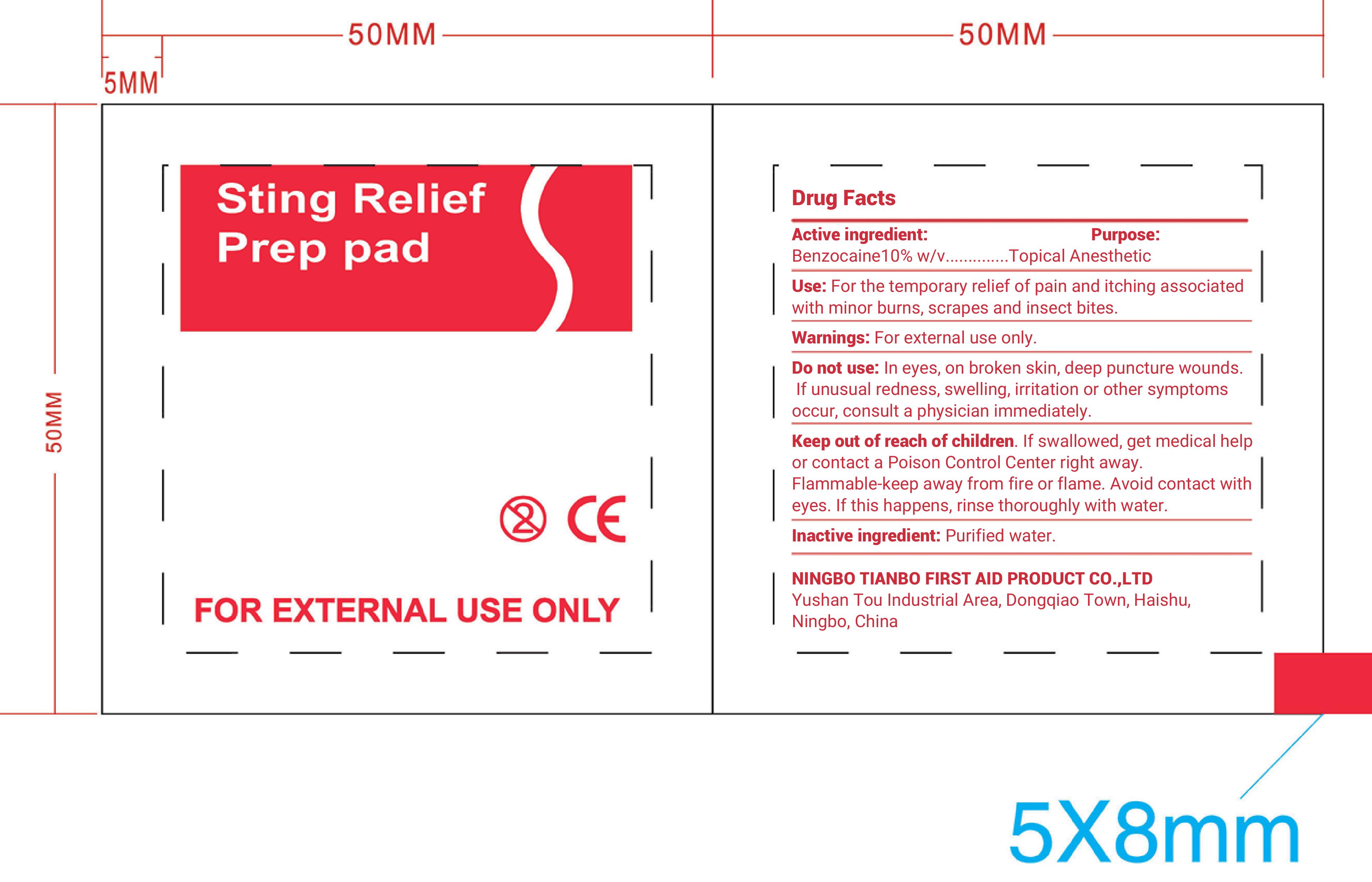
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient: Benzocaine 10% w/v
Medication Information
Warnings and Precautions
Warnings:
For external use only
Do not use: In the eyes, on broken skin, deep puncture wounds. I f unusal redness, swelling, irritation, or other symptoms occur, consult a physician immediately.
Indications and Usage
Uses: For temporary relief of pain and itching associated with minor burns, scrapes, and insect bites
Dosage and Administration
Directions: Wipe injection site vigorously and discard
Description
Active Ingredient: Benzocaine 10% w/v
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away
Flammable. Keep away from fire or flame. Avoid contact with eyes. If this happens, rinse thoroughly with water.
Section 51727-6
Inactive Ingredient
Purified Water
Section 51945-4
Section 55105-1
Purpose: Topical Anesthetic
Section 55106-9
Active Ingredient: Benzocaine 10% w/v
Structured Label Content
Indications and Usage (34067-9)
Uses: For temporary relief of pain and itching associated with minor burns, scrapes, and insect bites
Dosage and Administration (34068-7)
Directions: Wipe injection site vigorously and discard
Warnings and Precautions (34071-1)
Warnings:
For external use only
Do not use: In the eyes, on broken skin, deep puncture wounds. I f unusal redness, swelling, irritation, or other symptoms occur, consult a physician immediately.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away
Flammable. Keep away from fire or flame. Avoid contact with eyes. If this happens, rinse thoroughly with water.
Section 51727-6 (51727-6)
Inactive Ingredient
Purified Water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose: Topical Anesthetic
Section 55106-9 (55106-9)
Active Ingredient: Benzocaine 10% w/v
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:10.784947 · Updated: 2026-03-14T23:12:20.443473